Abstract
The insulin sensitizing thiazolidinedione drugs, rosiglitazone and pioglitazone are specific peroxisome proliferator-activated receptor-gamma agonists and reduce pro-inflammatory responses in patients with type 2 diabetes and coronary artery disease, and may be beneficial in sepsis. Sepsis was induced in 8–10-wk-old C57BL/6 mice by cecal ligation and puncture (CLP) with a 22 -g double puncture technique. Mice received an i.p. injection of vehicle (DMSO:PBS) or pioglitazone (20 mg/kg) at 1 h and 6 h after CLP, and were sacrificed at various time points. In sepsis, vehicle-treated mice had hypoglycemia, increased lung injury and increased lung neutrophil infiltration. Pro-inflammatory plasma cytokines were increased, but the plasma adipokine, adiponectin, was decreased in vehicle-treated septic mice. This corresponded with inhibitor κB (IκBα) protein degradation and an increase in NF-κB activity in lung. Pioglitazone treatment improved plasma Glc and adiponectin levels, and decreased pro-inflammatory cytokines. Lung IκBα protein expression increased and corresponded with a decrease in NF-κB activity in the lung from pioglitazone-treated mice. Pioglitazone reduces the inflammatory response in polymicrobial sepsis in part through inhibition of NF-κB and may be a novel therapy in sepsis.
Introduction
Sepsis is a potentially life-threatening disorder causing death in an estimated 215,000 patients per yr in the USA. 1 The mortality rate increases with age from 10% in children to 38% in the elderly.1,2 Treating patients with severe sepsis costs nearly $17 billion.3–5 Unfortunately, few clinical studies show a difference in outcomes in patients with sepsis.6–9
The nuclear receptor peroxisome proliferator-activated receptor-gamma (PPARγ) is involved in the regulation of sepsis-induced inflammation. Data from our laboratory and others demonstrate that in animal models of sepsis PPARγ expression is markedly down-regulated in the lung, liver and peripheral blood cells.10–14 This down-regulation is associated with severe inflammation and multiple organ failure. On the contrary, treatment with activators of PPARγ reduces inflammation and improves survival in experimental animals. In a pilot clinical trial in pediatric patients with sepsis, we found that PPARγ expression is decreased in peripheral blood mononuclear cells from children with septic shock compared with controls. 10
The thiazolidinediones (TZDs), pioglitazone and rosiglitazone, are Food and Drug Administration (FDA)-approved medications that activate PPARγ. TZDs are used therapeutically as insulin-sensitizers for patients with type 2 diabetes. In patients with type 2 diabetes TZDs have favorable effects on vascular function and inflammation.15–17 Pioglitazone also decreases inflammatory mediators in patients with type 2 diabetes and coronary artery disease. 15 PPARγ agonists are beneficial in experimental models of infection. In influenza pneumonia, PPAR gamma agonists improve survival.18–20 Additionally, in mice subjected to cecal ligation and puncture (CLP) pioglitazone administration decreased inflammation and improved survivalin apolipoprotein E knockout mice. 21 As a result, TZDs may modulate inflammation and be beneficial in infection-related inflammatory conditions.
Therefore, pioglitazone, through its activation of PPARγ and subsequent anti-inflammatory response, may prove a novel therapeutic in sepsis. However, the mechanisms involved in the anti-inflammatory responses of pioglitazone treatment in sepsis remain unknown. We hypothesized that pioglitazone treatment will decrease the inflammatory response in mice subjected to polymicrobial sepsis.
Materials and methods
Materials
The primary Ab for PPARγ was obtained from Thermo Fisher Scientific (Rockford, IL, USA) and Abs for inhibitor kB (IκBα), ICAM-1 and actin were obtained from Santa Cruz Biotechnology (Dallas, TX, USA). All other chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA). Pioglitazone was obtained from Enzo Life Sciences (Farmingdale, NY, USA).
Mice
The investigation conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health, and was approved by the institutional Animal Care and Use Committee at Cincinnati Children’s Hospital Medical Center. 22 The experimental groups consisted of male C57BL/6 mice at 8–10 wks of age supplied by Charles River Laboratories International (Wilmington, MA, USA). The mice were housed in the animal facility at the Cincinnati Children’s Research Foundation. Food and water were provided ad libitum. Polymicrobial sepsis was induced by CLP, and mice were sacrificed at various time points thereafter. Plasma and lung samples were collected for biochemical studies, as described below.
Mouse model of CLP
CLP was performed as previously described. 23 Mice were anesthetized with isoflurane via nose cone throughout the surgical procedure. After opening the abdomen, the cecum was exteriorized and ligated by a 6.0 silk ligature at its base without obstructing intestinal continuity. The cecum was punctured twice with a 22-gauge needle and returned to the peritoneal cavity. The abdominal incision was closed with silk running sutures and liquid topical adhesive. After the procedure, animals were fluid resuscitated with sterile saline (0.6 ml) injected subcutaneously, but did not receive antibiotics. Sham mice underwent an abdominal incision and exteriorization of the cecum but did not undergo CLP. Mice received i.p. injection of vehicle (DMSO:PBS) or pioglitazone (20 mg/kg, 1:1 DMSO:PBS) at 1 and 6 h after CLP. The dose of pioglitazone was chosen based on published reports and modified as a result of results from our laboratory.24,25
Histology
Lungs were fixed in 4% paraformaldehyde and embedded in paraffin. Sections were stained with hematoxylin and eosin.
Measurement of myeloperoxidase activity
Myeloperoxidase (MPO) activity was determined as an index of neutrophil accumulation in lung as previously described. 23 Tissues were homogenized in a solution containing 0.5% hexa-decyl-trimethyl-ammonium bromide dissolved in 10 mM potassium phosphate buffer (pH 7) and were centrifuged for 30 min at 20,000 g at 4℃. An aliquot of the supernatant was allowed to react with a solution of tetra-methyl-benzidine (1.6 mM) and 0.1 mM H2O2. The rate of change in absorbance was measured by spectrophotometry at 650 nm. MPO activity was defined as the quantity of enzyme degrading 1 µmol H2O2/min at 37℃ and was expressed in units per 100 mg of tissue.
Plasma levels of adipokines and cytokines
Plasma levels of TNF-α, IL-6 and adiponectin (high molecular mass adiponectin hexamers and trimers) were measured by use of the multiplex assay kit (Millipore, Billerica, MA, USA) using the protocol recommended by the manufacturer.
Blood Glc levels
Glc levels were determined by i-STAT measurement at time of tissue harvest.
Plasma levels of 15 d-PGJ2
Plasma samples of 15-deoxy-Δ12,14-prostaglandin J2 (15 d-PGJ2) were measured by an enzyme immunoassay kit (Enzo Life Sciences) using the protocol recommended by the manufacturer.
Subcellular fractionation and nuclear protein extraction
Tissue samples were homogenized in a buffer containing 0.32 M sucrose, 10 mM Tris-HCl, 1 mM ethylene glycol tetraacetic acid (EGTA), 2 mM EDTA, 5 mM NaN3, 10 mM β-mercaptoethanol, 50 mM NaF, 20 µM leupeptin, 0.15 µM pepstatin A, and 0.2 mM phenylmethylsulphonyl fluoride (PMSF), 1 mM sodium orthovanadate, and 0.4 nM microcystin. 11 The homogenates were centrifuged (1000 g at 4℃, 10 min). The supernatant (cytosol + membrane extract) was collected and stored. The pellets were solubilized in Triton buffer [1% Triton X-100, 150 mM NaCl, 10 mM Tris-HCl (pH 7.4), 1 mM EGTA, 1 mM EDTA, 0.2 mM sodium orthovanadate, 20 µM leupeptin A, and 0.2 mM PMSF]. The lysates were centrifuged (15,000 g, at 4℃, 30 min) and the supernatant (nuclear extract) collected and stored at −80℃. The amount of protein was quantified by Bradford assay.
Western blot analysis
The nuclear or cytosol content of PPARγ, IκBα, ICAM-1 and actin in the lung were determined by immunoblot analyses. Extracts were boiled in equal volumes of loading buffer (125 mmol/l Tris-HCl, pH 6.8, 4% SDS, 20% glycerol and 10% 2-mercaptoethanol) and 50 µg of protein was loaded per lane on 8–16% Tris-glycine gradient gel. Proteins were separated electrophoretically and transferred to nitrocellulose membranes. For immunoblotting, membranes were blocked with 5% non-fat dried milk in Tris-buffered saline for 1 h and then incubated with primary Abs against PPARγ, IκBα and ICAM-1. The membranes were washed in Tris-buffered saline with 0.1% Tween 20 and incubated with secondary peroxidase-conjugated Ab. Membranes were re-probed with primary Ab against actin to ensure equal loading. Detection was enhanced by chemiluminescence and exposed to photographic film. Densitometric analysis of blots was performed using ImageQuant (Molecular Dynamics, Sunnyvale, CA, USA).
Determination of NF-κB activity
NF-κB (p65) activation was detected in liver nuclear extracts using an ELISA-based transcription factor assay kit, as described previously. 26 Nuclear protein, 10 µg, was obtained from liver nuclear extracts and added to a 96-well plate to which oligonucleotide containing the NF-κB consensus binding sequence had been immobilized (Active Motif North America, Carlsbad, CA, USA). Ab directed against the p65 subunit was added followed by a secondary HRP conjugated Ab. Developing solution utilizing a colorimetric readout was used. The plate was read by a spectrophotometer at 450 nm with a reference wavelength of 655 nm. A wild type and mutated oligonucleotide were used for NF-κB binding to monitor the specificity of the assay (data not shown).
Statistical analysis
Statistical analysis of plasma cytokines, adiponectin, and 15 d-PGJ2 levels were performed using the Mann–Whitney Rank Sum test, and are expressed in the text as median with interquartile range (IQR). Statistical analyses of lung MPO activity were compared by analysis of variance with Holm-Sidak method. A t-test was used to compare plasma Glc levels, densitometric analysis of lung IκBα and PPARγ, and lung NF-κB p65 activity, and are expressed in the text as mean ± SEM. Three to four animals were used for each group. A value of P ≤ 0.05 was considered significant.
Results
Pioglitazone reduces lung injury and lung neutrophil infiltration after induction of polymicrobial sepsis
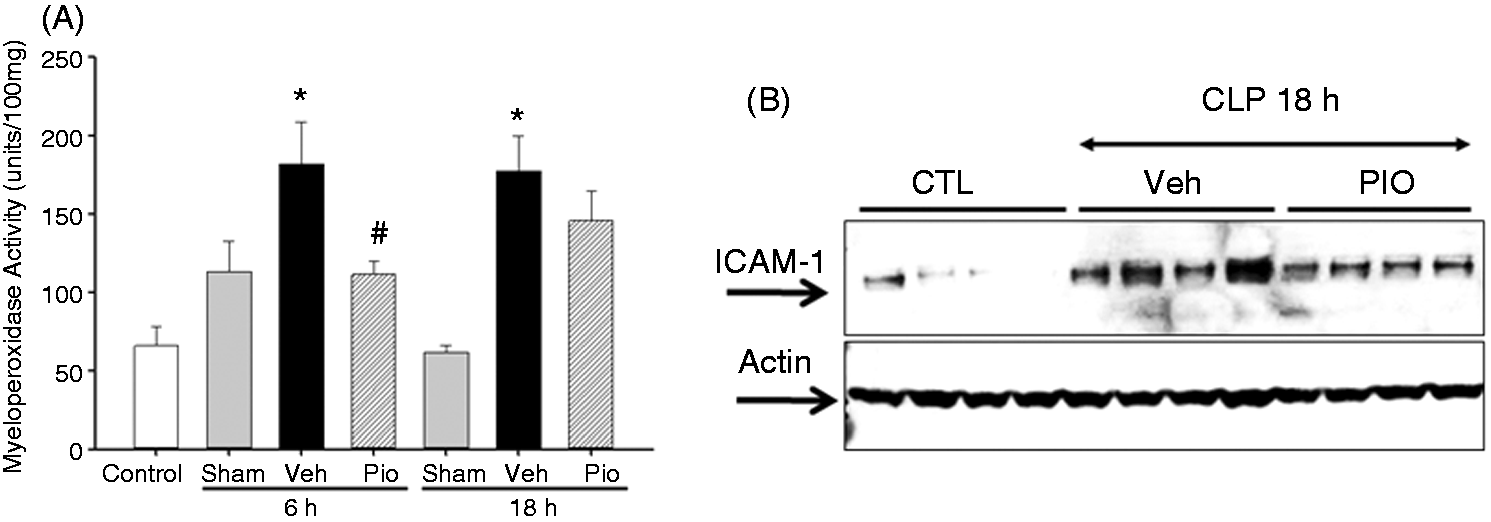
To determine the pioglitazone effects in polymicrobial sepsis, mice were subjected to CLP and sacrificed at various time points. As early as 6 h after CLP, vehicle-treated mice exhibited marked lung injury characterized by extravasation of red cells, alveolar edema and accumulation of inflammatory cells (Figure 1C). This was associated with a significant increase in lung neutrophil infiltration quantified by MPO assay. Specifically, MPO activity was increased 6 and 18 h after CLP in vehicle-treated mice (182 ± 27 and 177 ± 22 U/100 mg tissue, respectively) when compared with sham mice (113 ± 19 and 13 ± 5 U/100 mg tissue, respectively, P < 0.05) (Figure 2A). Pioglitazone treatment revealed a marked reduction of inflammatory cells in the lung with the return to normal lung architecture (Figure 1D). This was associated with a significant reduction in lung neutrophil infiltration compared with vehicle treatment at 6 h after CLP (111 ± 9 U/100 mg tissue, P < 0.05) (Figure 2A). To investigate the mechanism through which neutrophil infiltration is increased after CLP, expression of ICAM-1 was investigated by Western blot analysis. Lung expression of ICAM-1 was increased at 18 h after CLP in vehicle-treated mice compared with control mice (Figure 2B). Treatment with pioglitazone reduced ICAM-1 expression in the lung, although this was not statistically significant (P = 0.1).
Pioglitazone improves lung injury after CLP. (A) Lung from control mice revealing normal architecture. (B) Lung from 6-h sham mice. (C) Lung from vehicle-treated mice showing interstitial hemorrhage, neutrophil infiltration and obliteration of normal architecture. Black arrows denote neutrophil infiltration. (D) Lung from pioglitazone-treated mice reveals reduction of hemorrhage and reduction of neutrophil infiltration and amelioration of lung injury. Black arrows denote neutrophil infiltration. 40 × magnification. Effect of pioglitazone (pio) on lung injury after CLP. (A) MPO activity was determined at 6 and 18 h after CLP. *P < 0.05 versus sham and #P < 0.05 versus vehicle (veh). n = 3–4 mice/group and samples were run in duplicate. (B) Representative Western blot analysis for lung expression of ICAM-1 at 18 h after CLP. n = 4 mice/group. CTL: control.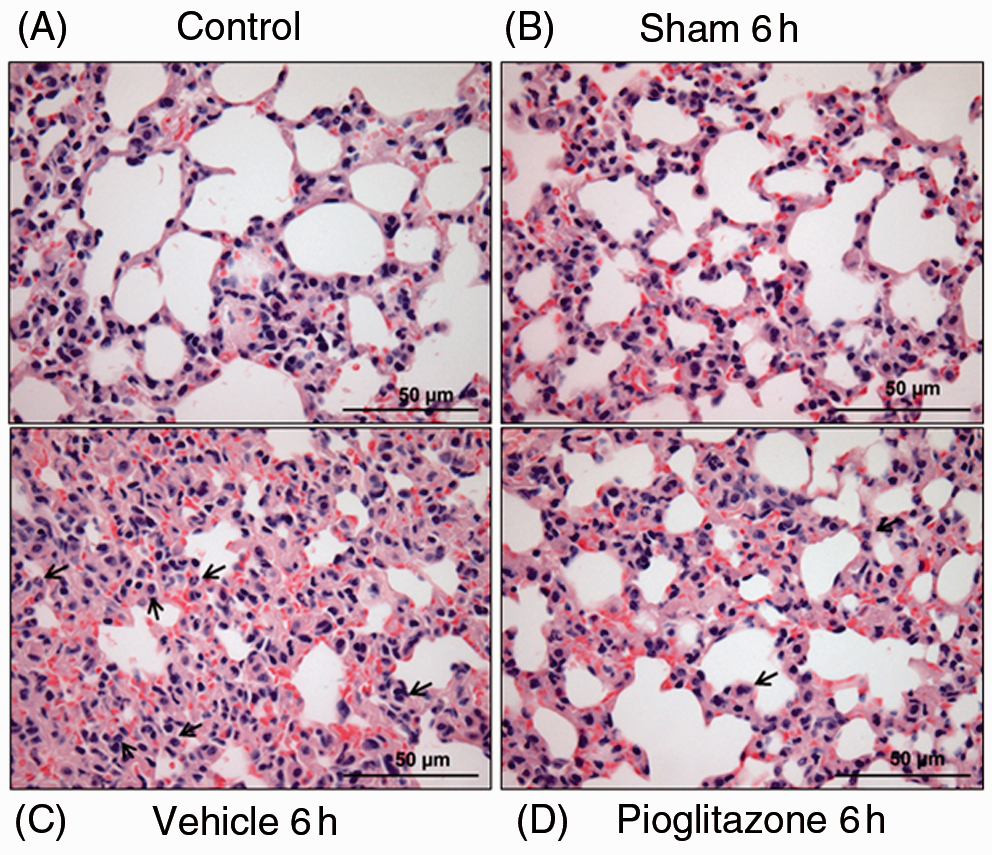
Pioglitazone reduces pro-inflammatory cytokines and increases adiponectin levels
To further investigate the effect of pioglitazone on the systemic inflammatory response we measured plasma cytokine levels. The plasma cytokines TNF-α and IL-6 were increased in vehicle-treated mice at 6 h after CLP [74 pg/ml (IQR 10.3–165.1) and 36,588 pg/ml (IQR 32,891–37,095), respectively] compared with sham mice (10 pg/ml and 144 pg/ml, respectively, P < 0.05) (Figure 3). Pioglitazone treatment reduced TNF-α and IL-6 plasma levels at 6 h after CLP [16 pg/ml (6–38) and 17,866 pg/ml (5001–34,792) respectively, P < 0.05].
Adiponectin, an adipose tissue-derived cytokine, has a peroxisome proliferator response element (PPRE) in its promoter region.
27
Consequently, changes in PPARγ activity may be reflected as changes in adiponectin.
10
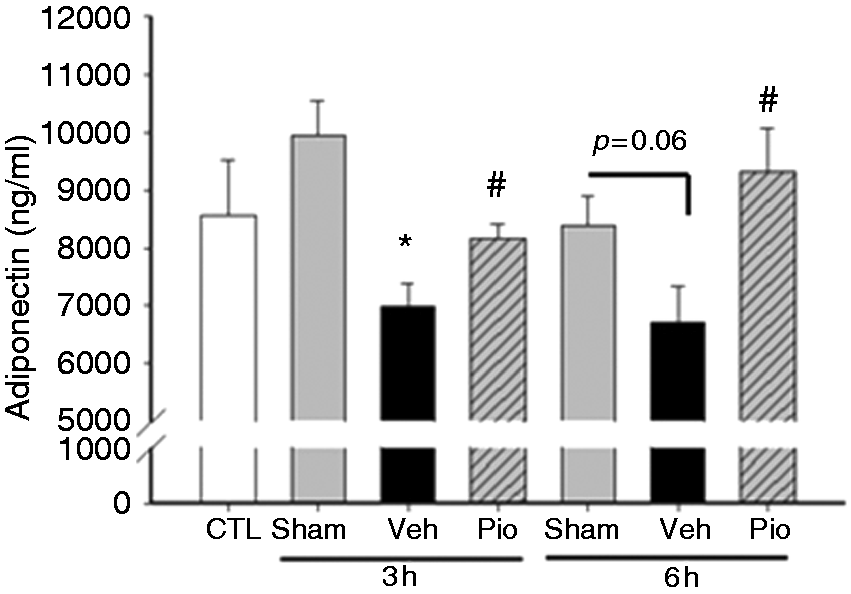
Adiponectin levels were lower in septic vehicle-treated mice at 3 and 6 h after CLP (Figure 4). However, treatment with pioglitazone increased plasma adiponectin levels (Figure 4).
Pioglitazone (Pio) reduces plasma cytokines after CLP. Plasma cytokine levels (A) TNF-α and (B) IL-6 were measured at 6 h after CLP. *P < 0.05 versus control and #P < 0.05 versus vehicle. n = 3–4 mice/group and samples were run in duplicate. Pioglitazone (Pio) increases plasma adiponectin levels after CLP. Plasma adiponectin were measured at 3 and 6 h after CLP. *P < 0.05 versus control (CTL) and **P < 0.05 versus vehicle (Veh). n = 3–4 mice/group and samples were run in duplicate.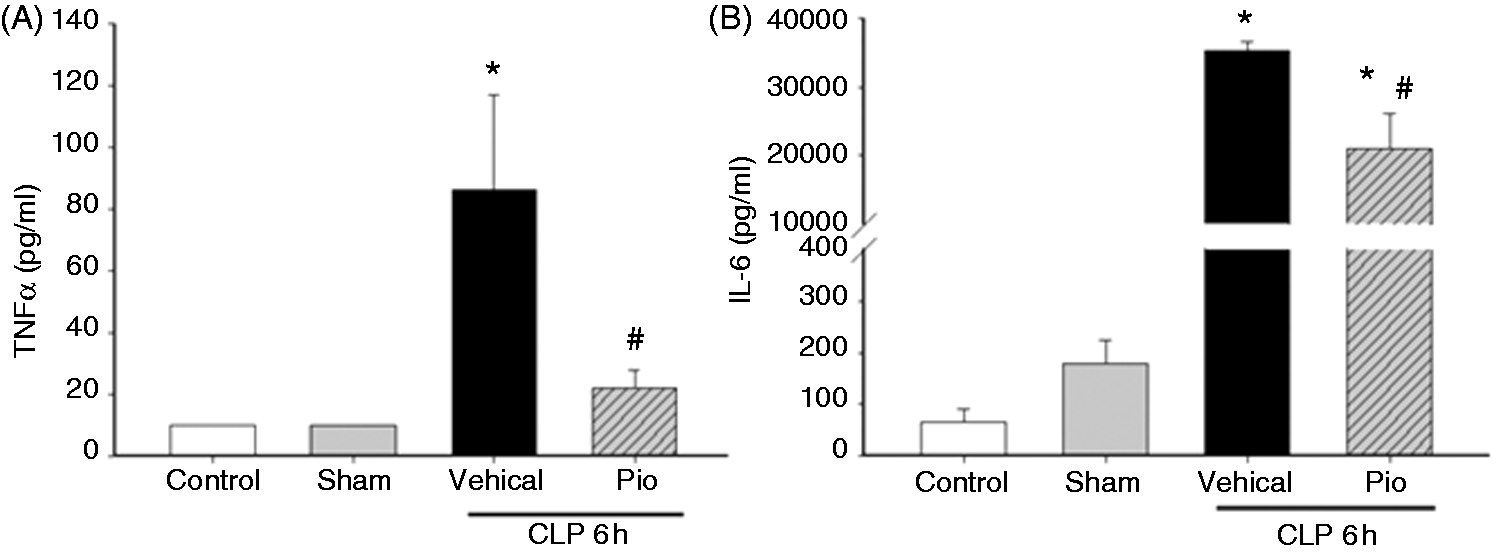
Pioglitazone improves Glc control
Pioglitazone improves Glc control and thus is used clinically as a treatment for diabetes. Therefore, we investigated the glucose effects of pioglitazone following CLP. Vehicle-treated mice had a significant decrease in blood glucose levels at 6 h after CLP compared with sham mice (60 ± 4 mg/dl versus 160 ± 12 mg/dl, respectively, P < 0.01). However, pioglitazone treatment significantly increased Glc levels at 6 h after CLP compared with mice treated with vehicle alone (101 ± 13 mg/dl, P < 0.05).
Pioglitazone increases 15 d-PGJ2 plasma levels
The cyclopentenone prostaglandin, 15 d-PGJ2, is an endogenous ligand for PPARγ so we investigated whether pioglitazone alters plasma 15 d-PGJ2 levels in polymicrobial sepsis. Plasma levels of 15 d-PGJ2 were markedly diminished in vehicle-treated mice at 6 h after CLP when compared with sham [2420 pg/ml (IQR 1975–3348) versus 4011 pg/ml (IQR 3546–5695), P < 0.05] (Figure 5). However, mice treated with pioglitazone had significantly higher plasma levels of 15 d-PGJ2 compared with vehicle-treated mice [4332 pg/ml (IQR 3271–5464), P < 0.05]. These results demonstrate that treatment with synthetic, exogenous ligand of PPARγ also affect the endogenous ligand.
Plasma levels of 15 d-PGJ2 are altered after CLP and restored by pioglitazone (Pio). Plasma levels of 15 d-PGJ2 were evaluated by ELISA at 3 and 6 h after CLP. *P < 0.05 versus control, **P < 0.05 versus sham, ***P < 0.05 versus vehicle (Veh). n = 3–4 mice/group and samples were run in duplicate.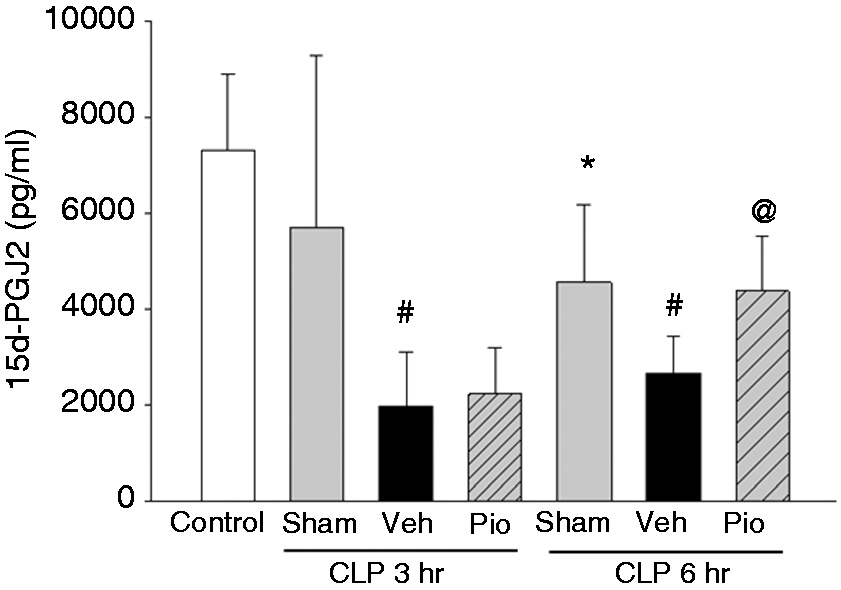
Pioglitazone increases lung PPARγ protein expression
As pioglitazone treatment altered inflammatory cytokine production and improved Glc control we sought to determine whether these effect were associated with changes in PPARγ expression in the lung. At Western blot analysis, expression of PPARγ was decreased in the nuclear compartment in lungs of vehicle-treated mice at 18 h after CLP (Figure 6). Treatment with pioglitazone increased lung PPARγ protein expression after CLP compared with vehicle treated mice.
Effect of pioglitazone (Pio) treatment on nuclear PPARγ expression in the lung. Representative Western blot analysis for lung expression of the two isoforms of PPARγ (PPARγ1 and PPARγ2) and relative densitometric analysis. *P < 0.05 versus sham. n = 3 mice/group. Veh: vehicle.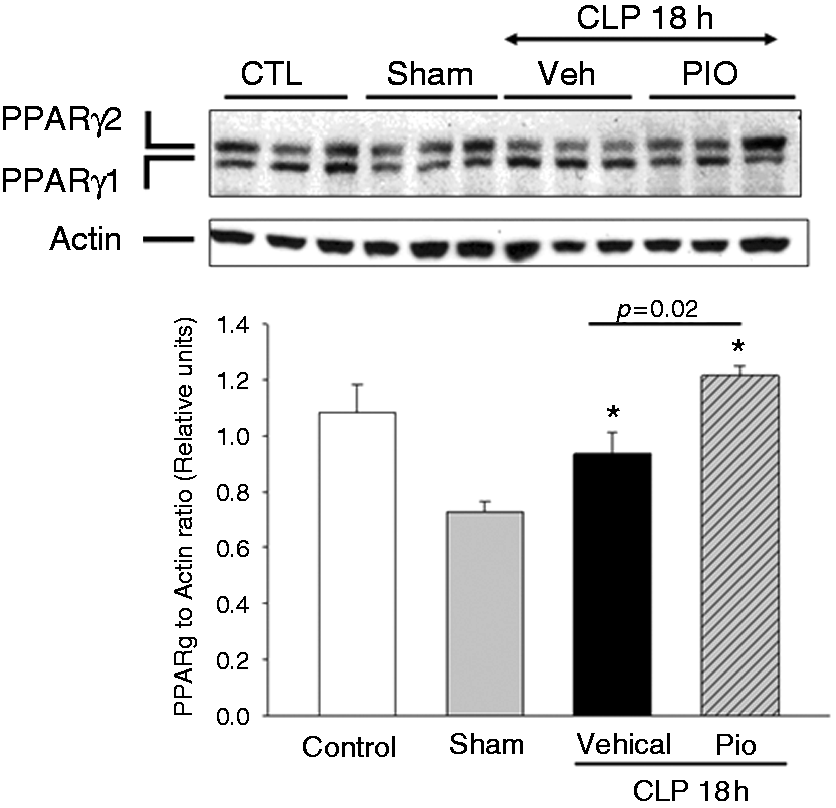
Pioglitazone inhibits NF-κB activity in the lung after CLP
To investigate the mechanism of action of pioglitazone, we evaluated the activation of the NF-κB pathway. Vehicle-treated mice exhibited marked IκBα protein degradation in the lung as measured by Western blot analysis at 18 h after CLP (Figure 7). The reduction in IκBα protein expression was associated with a significant increase in NF-κB p65 DNA binding activity in the lung at 18 h after CLP compared with controls (0.39 relative units ± 0.03 versus 0.21 relative units ± 0.03, P < 0.05). However, treatment with pioglitazone increased lung IκBα protein expression and reduced NF-κB DNA binding at 18 h after CLP versus vehicle treatment (0.16 relative units ± 0.1, P < 0.05).
Effect of pioglitazone (Pio) treatment on IκBα expression in the lung. Representative Western blot analysis for lung expression of cytosol IκBα and relative densitometric analysis. *P < 0.05 versus sham. n = 3 mice/group. Veh: vehicle.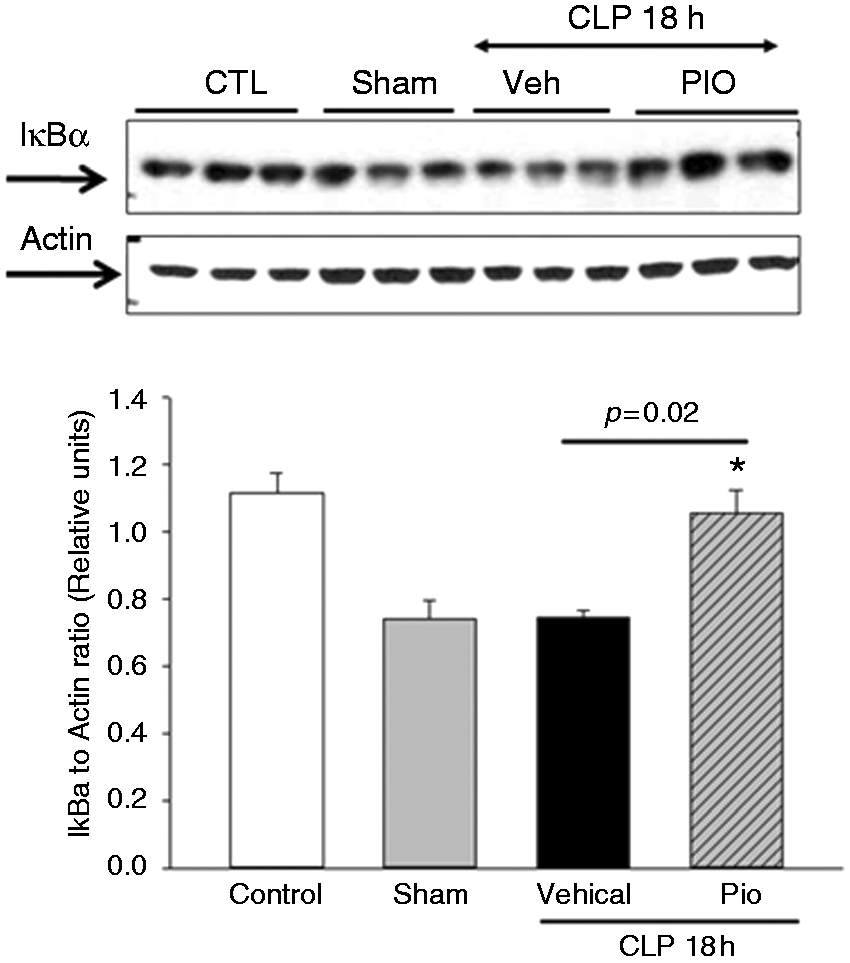
Discussion
There are very few therapies that improve outcomes in patients with sepsis. Therefore, it is imperative that other therapeutic options be explored. Activators of PPARγ may be novel therapeutic agents in sepsis. 28 Preclinical work from our laboratory has demonstrated that PPARγ expression is down-regulated on the endothelium of thoracic aortas and in the lung in polymicrobial sepsis, and that this down-regulation is associated with poor survival.11,12 Furthermore, in animal models of sepsis treatment with the PPARγ ligand, 15 d-PGJ2, decreases neutrophil infiltration and adhesion molecule expression in the lung and small intestine. 11
Pioglitazone and rosiglitazone are FDA-approved medications that are used clinically in diabetes. These medications also activate PPARγ and may prove novel therapies in clinical sepsis. However, concerns have been raised regarding the long-term use of rosiglitazone in adults and its association with heart failure and death. 29 Although there have been important concerns regarding the use of rosiglitazone in adults and its association with heart failure and death, a large meta-analysis demonstrated that pioglitazone is associated with a significantly lower risk of death, myocardial infarction or stroke in patients with type 2 diabetes mellitus compared with placebo. 30 In order to have a translatable model that is clinically relevant and given the concerns regarding rosiglitazone we have used pioglitazone as an activator of PPARγ.
PPARs are ligand-dependent transcription factors and nuclear receptors that influence cellular responses by altering gene expression. Although PPARs were initially described as important in triglyceride and cholesterol homeostasis these receptors are also important in regulating the inflammatory response. 31 PPARγ can trans-activate and trans-repress target genes through ligand-dependent and ligand-independent mechanisms.32–35 The insulin-sensitizing drugs, TZDs, and the cyclopentenone prostaglandin, 15 d-PGJ2, are specific PPARγ agonists.36–38 The endogenous ligand, 15 d-PGJ2, can repress the expression of inflammatory genes in activated macrophages, including TNF-α and COX-2. 39 Data from our laboratory and others demonstrate that 15 d-PGJ2 has anti-inflammatory effects on NF-κB activation. This inhibition occurs through PPARγ-dependent and PPARγ−independent mechanisms.40–43 One mechanism by which 15 d-PGJ2 has effects is through binding of the electrophilic carbon in the cyclopentenone ring to cellular proteins, modifying signaling pathways. 44 This mechanism may account for the direct repression of NF-κB by 15 d-PGJ2. 45 Clinically, 15 d-PGJ2 production may predict PPARγ activation in vivo. 15 d-PGJ2 can be measured in urine, synovial fluid and plasma.46,47 Reduced levels of 15 d-PGJ2 from articular chondrocytes correlate with the degree of inflammation in patients with rheumatoid and osteo-arthritis, and were lower than levels from patients undergoing joint surgery for traumatic fracture. Our data demonstrate that plasma levels of 15 d-PGJ2 are decreased in an experimental sepsis and correlate with an increase in inflammatory mediators. However, treatment with pioglitazone restores 15 d-PGJ2 and decreases inflammation.
The exogenous activators of PPARγ, pioglitazone and rosiglitazone, exert anti-inflammatory effects in experimental models of inflammation. Activation of PPARγ by rosiglitazone inhibited LPS release of high mobility group box 1, a late inflammatory mediator in sepsis, from RAW 264.7 macrophages. 48 In a mouse model of endotoxin, rosiglitazone improved survival and cardiac dysfunction. 49 In experimental polymicrobial sepsis, rosiglitazone decreased leukocyte rolling and adhesion in the brain microvasculature after CLP compared with vehicle-treated mice. 50 Tsujimura et al. 51 demonstrated that pretreatment with pioglitazone for 7 d prior to CLP improved sepsis survival compared with pretreatment with vehicle only. In the current study, pioglitazone decreased pro-inflammatory plasma cytokines and resulted in a reduction in neutrophil infiltration and lung injury following induction of sepsis. Mice treated with pioglitazone had higher plasma 15 d-PGJ2 levels and increased PPARγ protein expression in the lung. Mechanistically, in the lung, these effects were associated with NF-κB inhibition. Our current findings support these data and demonstrate that pioglitazone, even when given after the induction of sepsis, inhibits anti-inflammatory mediators and may exert anti-inflammatory effects by modulating NF-κB activity.
Based on the presence of a direct binding site for PPARγ in its promoter region, adiponectin is a target for PPARγ. 52 Adiponectin is an adipocyte-derived protein that is secreted into human plasma.53–56 The adiponectin protein exists as multimer complexes in plasma: a low-molecular mass trimer, a middle-molecular mass hexamer and a high-molecular mass protein (HMMA). 57 In humans, the HMMA has been proposed as the most potent form mediating the metabolic actions of adiponectin.58,59 In addition to its metabolic actions adiponectin has anti-inflammatory effects, and plays a role in the innate and adaptive immune response. In an animal model of polymicrobial sepsis, plasma adiponectin levels were lower after sepsis and inversely correlated with plasma TNF-α and endotoxin levels. 60 Adiponectin knockout mice had higher inflammatory cytokine production and higher mortality after polymicrobial sepsis compared with wild type mice.61,62 Adiponectin reduces LPS-induced lung injury and when adiponectin is knocked out, mice treated with LPS have increased systemic and lung TNF-α levels and worse lung injury. 63 Data from the current study support the findings that plasma adiponectin is decreased in experimental sepsis, and that this decrease is associated with increased systemic and lung inflammation. In our study, treatment with pioglitazone, however, was able to restore plasma adiponectin levels.
Adiponectin is also altered in patients with sepsis. In separate human studies, healthy volunteers demonstrated no significant change in plasma adiponectin levels after endotoxin; however, the adiponectin receptors, AdipoR1 and AdipoR2, were decreased in monocytes after endotoxemia injection.64,65 Data from our laboratory utilizing a cohort of critically ill children with sepsis demonstrated contrasting findings to these studies but are consistent with published data in other inflammatory conditions. We found that on the first day of hospitalization plasma high molecular mass adiponectin levels are increased in children with septic shock compared with controls. 10 A similar increase in adiponectin was demonstrated in patients with inflammation from arthritis and inflammatory bowel disease.66,67 These findings suggest that adiponectin may play a role in sepsis. Adiponectin has a PPRE in its promoter regions; therefore, changes in PPARγ activity may be reflected in changes in plasma adiponectin. 27 Multiple studies have demonstrated that treatment with thiazolidinediones increase plasma adiponectin levels and increase adiponectin mRNA expression in adipocytes and adipose tissue.68–70
Further studies are needed to determine the exact mechanisms that account for these protective effects and whether pioglitazone protection in sepsis is through alterations in Glc, adiponectin and PPARγ, or through alternative actions. Although not investigated in this study, the anti-inflammatory effects of pioglitazone may be a result of an increase in adiponectin and not directly related to PPARγ effects. Adiponectin inhibits LPS-induced TNF-α production by macrophages. 71 Adiponectin can also directly bind to LPS extracellularly.60,72 In a dose-dependent manner, recombinant adiponectin suppressed Limulus amoebocyte lysate activity through direct binding of LPS. 60 15d-PGJ2 is derived from arachidonic acid via the cyclo-oxygenase (COX) pathway. Therefore, increases in COX expression may result in 15 d-PGJ2 expression. Shibata et al. 73 demonstrated that adiponectin exerts anti-inflammatory protection in myocardial ischemia-reperfusion injury through COX-2-dependent action. 74 Inhibiting COX-2 in cardiac myocytes following myocardial ischemia reperfusion injury resulted in a reduction in adiponectin-stimulated prostaglandin E2 production. Additionally, inhibiting COX-2 reversed the suppressive effect of adiponectin on TNF-α plasma levels. 73 This suggests that adiponectin protects the myocardium in ischemia-reperfusion injury through COX-2 suppression of TNF-α. It is possible, in our current study, that pioglitazone could increase plasma 15 d-PGJ2 through increasing adiponectin and COX-2 expression, and subsequently increasing 15d-PGJ2.
The goal of the current work was to understand whether pioglitazone protected lung injury as a result of the systemic inflammatory response in sepsis. A limitation of our study is the lack of survival data. To be clinically relevant, these studies need to be combined with antibiotic therapy. However, for the current study, to investigate a direct effect of pioglitazone therapy on sepsis-induced acute lung injury, pioglitazone was given as the sole therapy without any other interfering medication. Longer-term therapeutic interventional studies will be needed to investigate whether pioglitazone has a survival benefit.
In conclusion, our study demonstrates that polymicrobial sepsis leads to hypoglycemia, elevated plasma levels of pro-inflammatory cytokines and lung injury. Mechanistically, this corresponds with IκBα protein degradation and an increase in NF-κB activity in the lung. Treatment with pioglitazone, when given after the induction of sepsis, improves glucose levels and reduces pro-inflammatory plasma cytokine levels. Furthermore, pioglitazone reduces lung injury most likely through inhibition of NF-κB activity. These findings suggest that pioglitazone may be a beneficial and novel therapy in clinical sepsis.
Footnotes
Funding
This work was supported, in part, by the National Institutes of Health [K08 GM093135 (JK) and R01 GM067202 (BZ)].
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
