Abstract
Primary responses in sepsis-mediated inflammation are regulated by pro-inflammatory cytokines. Variations in the cytokine genes might modify their transcription or expression, plasma cytokines levels and response to sepsis. Activation protein-1 (AP-1) and NF-κB regulate cytokines gene expression in sepsis. A total of 90 severely septic and 91 non-infected patients were prospectively studied.
Introduction
Septic shock is the most common cause of death in intensive care units (ICU). Despite modern intensive care and antibiotic treatments, the mortality of sepsis still remains high, ranging from 20% to 30% in septic shock.
Primary responses in sepsis-mediated inflammation are regulated by pro-inflammatory cytokines, initially by the primary cytokines TNF-α and IL-1 and later by secondary pro-and anti-inflammatory mediators such as IL-6 and IL-10, respectively. Many cytokines display variations in their gene promoter regions. Polymorphisms in this promoter area might modify their transcription or expression, plasma cytokines levels and response to sepsis although results are controversial.1–19 Promoters contain cis-acting elements that represent short sequence motifs specific for binding of DNA-binding proteins nuclear factors such as the activation protein-1 (
The aims of this study were to investigate whether different cytokines genes SNPs might associate with susceptibility to sepsis or influence the sepsis outcome with/without associated changes in plasma cytokines levels. In order to answer these questions, plasma levels of IL-1β, IL-6, IL-8, IL-10 and TNF-α as well as those of monocyte chemoattractant protein 1 (MCP-1) were measured and different cytokine SNPs:
Patients and methods
Patients
A total of 90 patients with severe sepsis admitted to the ICU of the Hospital Universitario Central de Asturias (HUCA) in Oviedo, Spain, were enrolled in the study. Patients were included as septic if they fulfilled the diagnosis of severe sepsis according to the 1992 International Sepsis Definitions Conference Criteria modified in 2003 and 2016 (Sepsis-3).21–23 In addition, all septic patients had positive blood cultures or a microbiologically demonstrated source of bacterial infection at ICU admission. All the infections were community-acquired. Patients with cancer, HIV infection, transplantation or other causes of immunodepression were excluded. A total of 91 uninfected patients admitted consecutively to the ICU for other non-infective diseases, mostly with severe polytrauma and cerebro-vascular attacks (CVA), were used as controls. APACHE II scores and number of organs that failed were calculated in septic patients and uninfected controls. Patients and controls were members of a homogeneous Caucasian population, and were residents of the same region (Asturias, Northern Spain), which has a small foreign immigrant population (less than 5%). Each participant or their legal representatives gave informed consent for the study, which was approved by the Ethics Committee of the HUCA. Haemodynamic and other organ dysfunction failures were defined according to Marshall. 24 Treatment of organ failures, volume resuscitation and supportive therapy for sepsis were based on currently applied guidelines. 25
Plasma cytokines
Blood (10 ml) was drawn by venipuncture in EDTA-containing tubes within the first 24 h of ICU admission in all the study individuals, and in a subgroup of 14 sepsis patients and 15 controls, chosen at random; samples were also obtained at d 1, 3 and 7 of ICU stay. Tubes were centrifuged for 5 min at 1800
Cytokine SNP genotyping and linkage disequilibrium analysis
DNA was obtained from peripheral blood cells and stored at –20°C before use. The following SNPs of cytokines were genotyped by PCR:
After genotyping the previous SNPs of cytokines, a linkage disequilibrium (LD) analysis of
Other laboratory analysis
A complete haemogram, coagulation and general biochemistry were obtained from patients and controls at d 1 of ICU admission.
Neutrophil isolation
Peripheral blood (10 ml) was drawn from each patient and control in an EDTA-K3E tube (Vacuette, Greiner Bio-One, Kremsmuenster, Austria) and neutrophils were separated with Ficoll-Hypaque (δ = 1.113) (Lymphoprep™, Axis-Shield Poc AS, Oslo, Norway) following the manufacturer’s instructions. Cells were re-suspended in HAM’S F-12 medium (GIBCO, Paisley, UK) and counted using with a Cell-Dyn® 3200 Coulter (Abbott Laboratories, Abbott Park, IL, USA). Neutrophil purity was always > 98%.
Neutrophil mRNA extraction and AP-1 and NF-κB expression analysis
Total RNA from the isolated neutrophils was extracted using the E.Z.N.A. Total RNA Kit (Omega Bio-Tek, Inc., Norcross, GA, USA) according to the manufacturer’s instructions. The quantity of total RNA extracted was measured using spectrophotometry at 260 nm. The RNA was then reverse-transcribed to cDNA using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer’s protocol. Gene transcription levels of
Statistical analysis
Results are expressed as median and inter-quartile range (IQR), or proportions as appropriate. Correlations between the cytokine values and other continuous variables were assessed with the Spearman’s test. The Pearson
Results
Patients clinical and standard laboratory characteristics and outcome
Table 1 shows the demographic, clinical and standard laboratory characteristics and Table 2 shows the plasma cytokine levels and
Demographic and clinical characteristics and standard laboratory measures of ICU septic and uninfected patients.
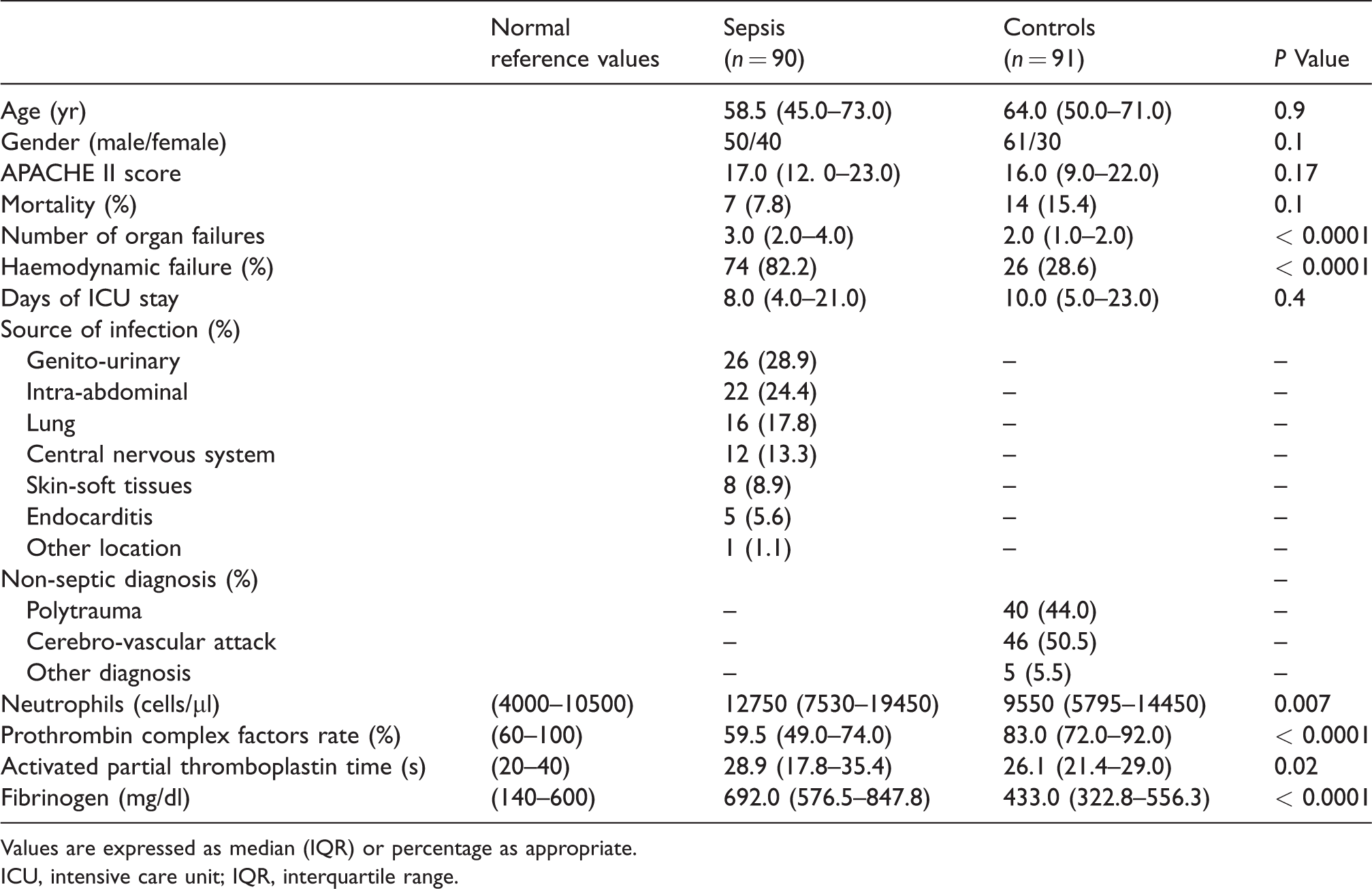
Values are expressed as median (IQR) or percentage as appropriate.
ICU, intensive care unit; IQR, interquartile range.
Plasma cytokines and neutrophils activating transcription factor expression data of ICU septic and uninfected patients.
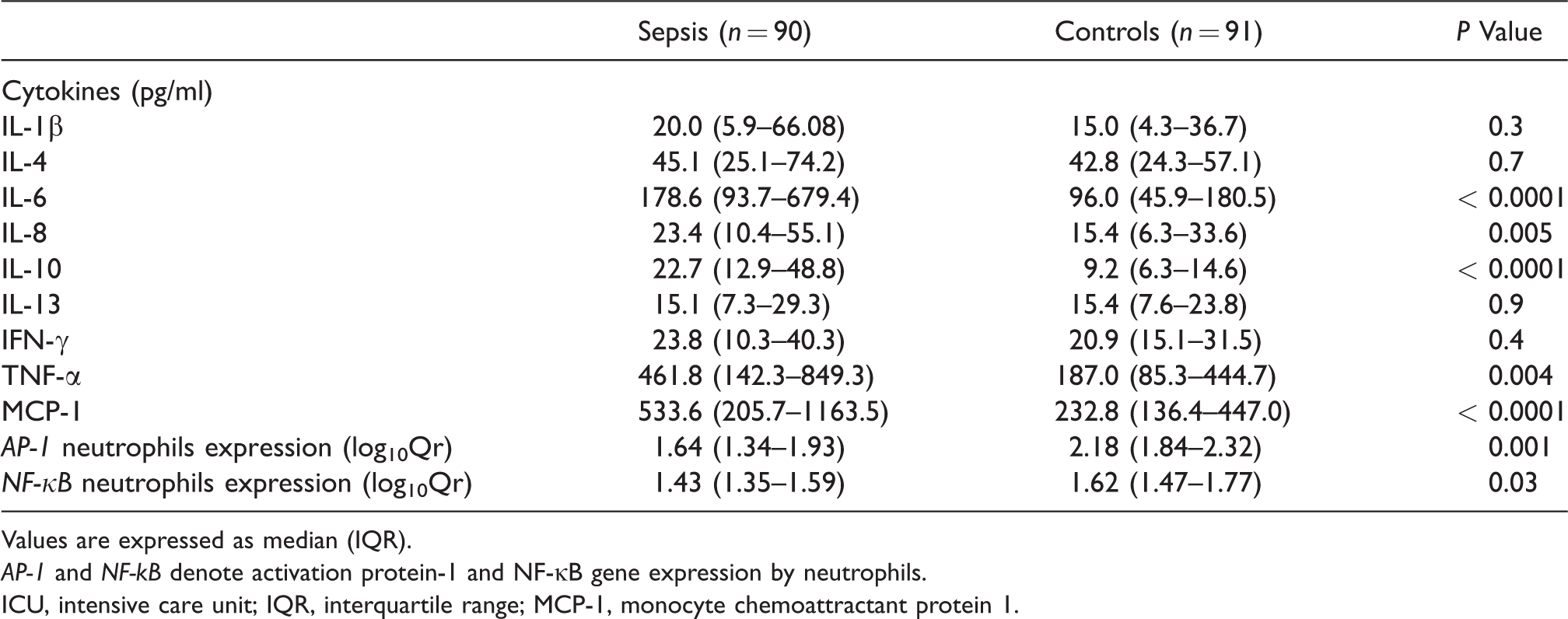
Values are expressed as median (IQR).
ICU, intensive care unit; IQR, interquartile range; MCP-1, monocyte chemoattractant protein 1.
Demography, clinical, laboratory and genotypic features in survivors and non-survivors.
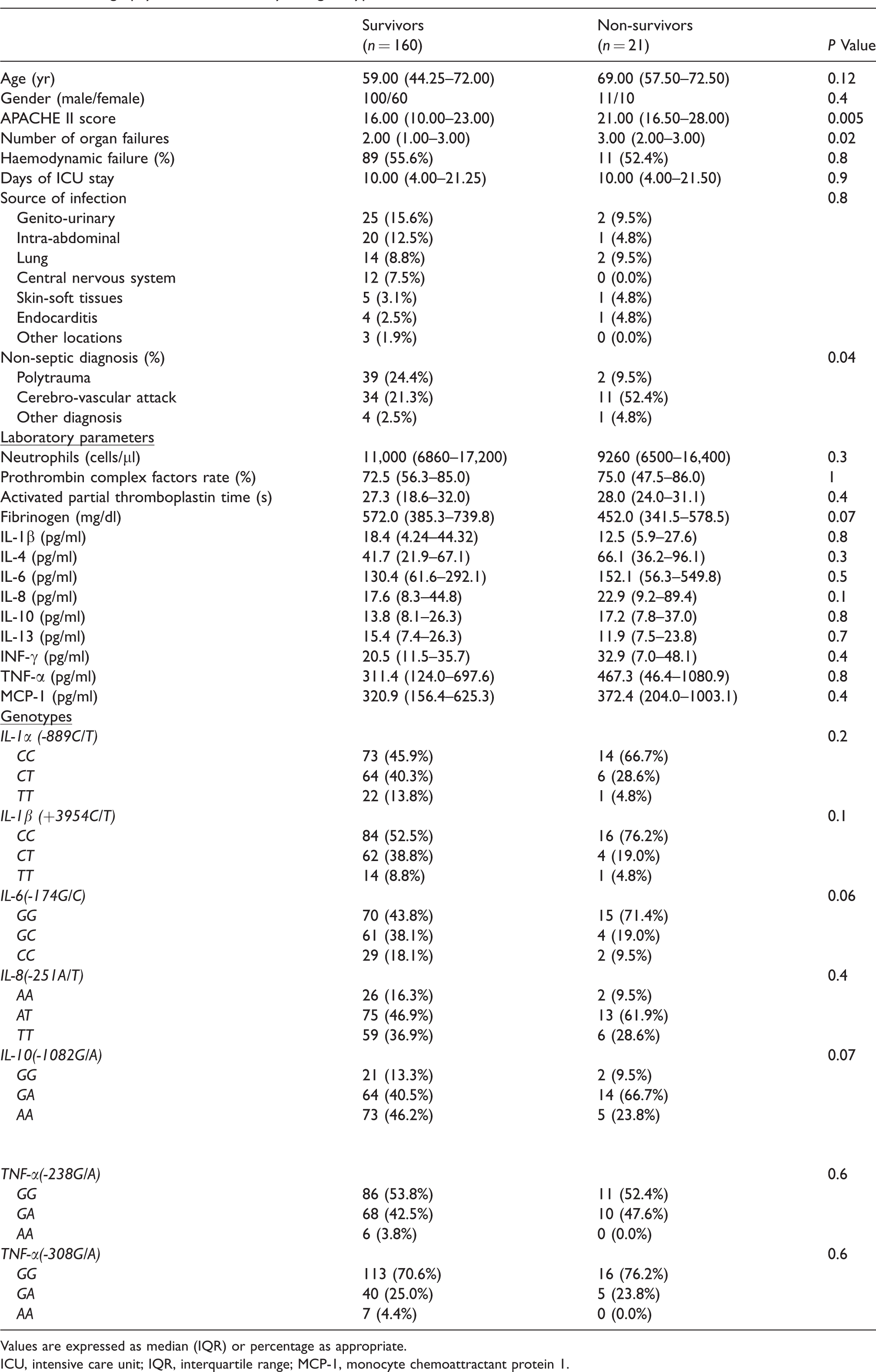
Values are expressed as median (IQR) or percentage as appropriate.
ICU, intensive care unit; IQR, interquartile range; MCP-1, monocyte chemoattractant protein 1.
Septic and uninfected ICU patients were matched in age and sex. There were no differences in APACHE II score. A total of 21 individuals died (11.6%). Although more uninfected controls died, there were no statistically significant differences compared with the septic patients. As expected, the number of organs that failed was significantly higher in septic patients. In addition, the APACHE II score and the number of organ failures were significantly higher in non-survivors (21 patients) compared with survivors (160 patients) (Table 3). Positive blood cultures were obtained in 75 (83.3%) of septic patients; 40 (53.3%) of them grew Gram-negative bacteria, mostly
Plasma cytokine levels and AP-1 and NF-κB neutrophil expression in septic and uninfected controls
We observed significantly higher levels of neutrophils, activated partial thromboplastin time, fibrinogen and plasma IL-6, IL-8, IL-10, TNF-α and MCP-1 in septic patients compared with uninfected controls at d 1 of ICU admission. Prothrombin complex factor rate was decreased in septic compared with uninfected ICU controls. In the septic group, patients who died had significantly higher levels of IL-8 and IL-10 than those who survived (69.3 pg/ml (23.7–938.4) versus 21.4 (10.1–50.3),
Cytokine SNPs and plasma cytokine values
Table 4 shows the genotypic and allelic frequencies of different cytokine SNPs in septic and control patients.
Genotypic and allelic frequencies of different cytokine SNPs in ICU septic patients and uninfected controls, and their association with circulating plasma cytokines.
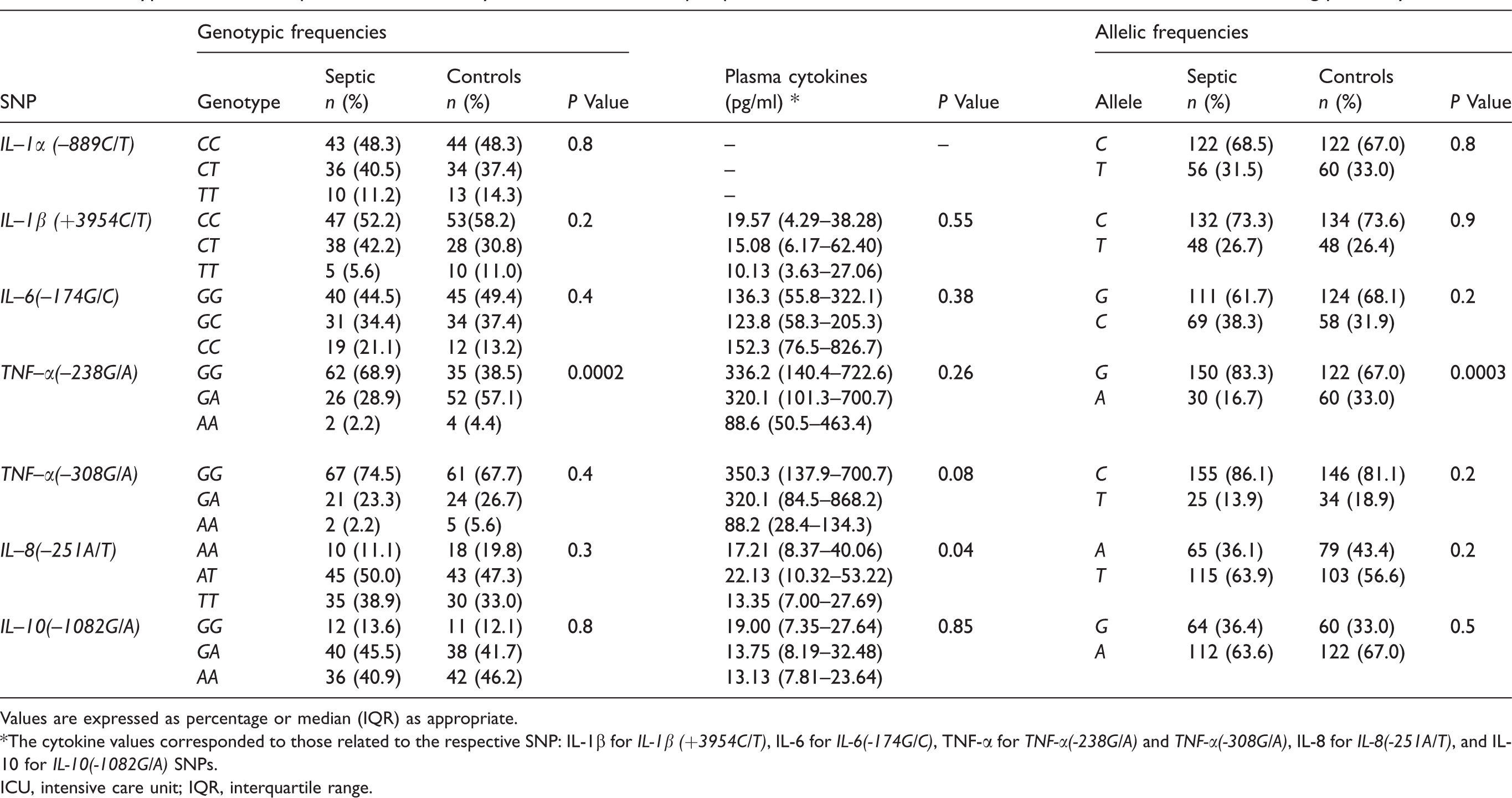
Values are expressed as percentage or median (IQR) as appropriate.
*The cytokine values corresponded to those related to the respective SNP: IL-1β for
ICU, intensive care unit; IQR, interquartile range.
Of all the cytokine SNPs studied, we found that only the variant
The diverse cytokine SNPs were not associated with different concentrations of their respective cytokines in the patients as a whole (
Correlations of the diverse plasma cytokines in sepsis.
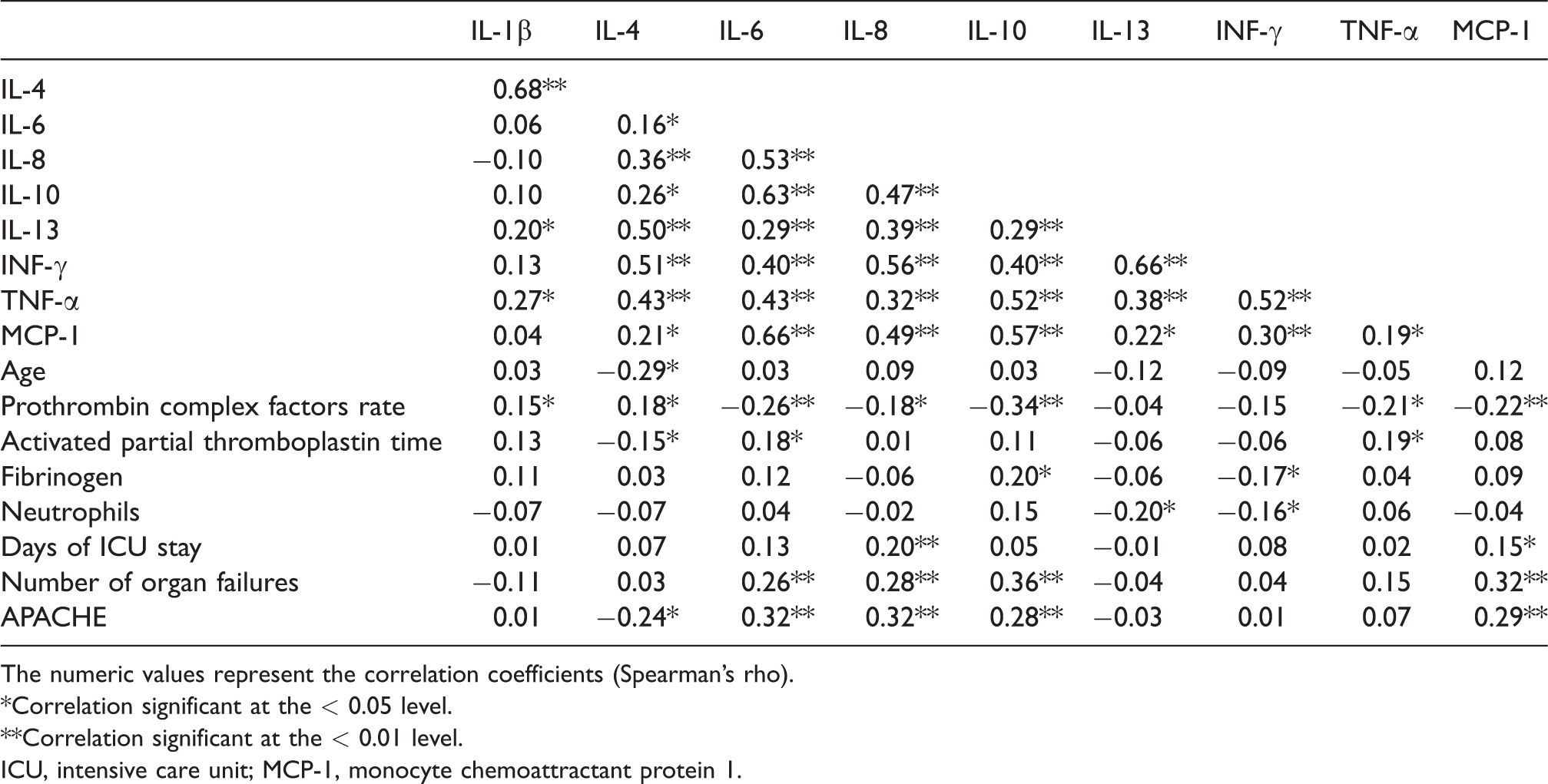
The numeric values represent the correlation coefficients (Spearman's rho).
*Correlation significant at the < 0.05 level.
**Correlation significant at the < 0.01 level.
ICU, intensive care unit; MCP-1, monocyte chemoattractant protein 1.
The number of organs that failed correlated with plasma levels of IL-6, IL-8, IL-10 and MCP-1, while the APACHE II score correlated with plasma levels of IL-6, IL-8, IL-10 and MCP-1 and negatively with plasma levels of IL-4. Similarly, most cytokines correlated among themselves, frequently with correlation coefficients higher than 0.5. No differences in the
LD analysis
Estimates of LD based on D′ and r2 were different for cases and controls. For cases we observed these values: D′ = 0.9503 and r2 = 0.8534. With these values, we can say that the two SNPs,
Plasma cytokine timecourse according to diagnosis
The course of cytokines over time was evaluated in a subgroup of 14 sepsis and 15 control patients, who underwent measurements at d 1, 3 and 7 of ICU admission. Among the septic patients, only IL-10 showed a statistically significant decrease between the d 1 and 7 measurements (
Regarding the comparison between the two groups, the slopes of the IL-10 and TNF-α curves over time were different between septic and control patients (
Multivariate analysis
Variables with a
Variables independently associated with sepsis and death.

*
CI, confidence interval; OR, odds ratio.
Discussion
We report for the first time an association of the
Some attention has been paid so far to the role played by the
Direct functional studies provided contrasting results on the effect of the variant
Changes in DNA-binding proteins nuclear factors expression might be other reasonable hypothesis to explain the protective effect of the
We have no clear explanation for the decreased
Many studies based on few cytokines and chemokines measurements in sepsis have been published. In the present study, we assessed the profiles of nine inflammatory cytokines and chemokines in sepsis using the multiplex Ab array that measures multiple proteins simultaneously within a single sample. The simultaneous assessments of multiple cytokines have the advantage of discovering possible associations among them and with sepsis severity parameters. Most of the cytokines and MCP-1 studied here showed significantly increased plasma levels in septic patients compared with uninfected controls, as previously published.47–63 IL-6, IL-8, IL-10 and MCP-1 plasma levels correlated with a high number that organs that failed and high APACHE II score in our cohort, as reported by others.47–55 We found an independent association of plasma levels of IL-8 with sepsis diagnosis and mortality. IL-8 levels seemed to be a useful tool to predict sepsis mortality in our study. Our data agree with previous reports that showed the predictive value of IL-8 for diagnosis of sepsis after burn injury and of neonatal sepsis.56–62
Regarding the time course of expression of cytokines and chemokines, only IL-10 exhibited a persistent decrease between d 1 and 7 of ICU admission in septic patients. A confounding effect of sepsis therapy cannot be ruled out. However, except for TNF-α, cytokine plasma levels did not change in uninfected ICU controls that did not receive sepsis treatment. We could speculate that increases in IL-10 plasma levels over time might imply sepsis therapy failure or sepsis relapse. Our data agree with other reports from burned and septic patients. In such patients, in addition to having decreasing time course IL-10 slopes, this cytokine was a predictor of ICU mortality.54–56, 63
The main limitations of our study are the relatively small sample size, the low mortality rate and our Caucasian-only population. The relatively small sample size (90 septic cases and 91 controls) might bias the results in SNP-related studies such as the present one. More research in multi-ethnic larger populations of septic patients is needed to confirm our findings.
We report here the differential regulation of various inflammatory proteins that might contribute to sepsis pathophysiology.
Footnotes
Acknowledgements
This work was supported by the Oviedo University research grants (UNIOV-12-MA-03 and SV- PA-13-ECOEMP-57) given to V.A. and by grants from Janssen Pharmaceuticals and Red Temática de Investigación de SIDA (RIS), Instituto de Salud Carlos III (ISCIII), Plan Estatal I+D+i & European Regional Development Fund (ERDF) ‘‘Una manera de hacer Europa’’ (grant RD06/0006/0010).
These results were presented in part at the 54th ICAAC Meeting, ASM, Washington DC, 5–9 September 2014, Abstract E-935.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
