Abstract
In this study, resveratrol (RSV) – a potent sirtuin 1 activator – was found to have beneficial effects on glucolipid metabolism and improve inflammatory mediators and markers of oxidative stress. Diabetic (
Introduction
Diabetes mellitus (DM) is a serious public health problem worldwide. The 2011 National Diabetes Statistics indicate that among US residents aged 65 years and above, 10.9 million (26.9%) suffered from diabetes in 2010, and approximately 1.9 million people aged 20 years or older were newly diagnosed with DM. 1 It is estimated that DM increases the risk of cardiovascular disease (CVD) from two- to fourfold. 2 Moreover, CVD is the most prevalent cause of morbidity and mortality in patients with DM. 3
Both DM and its complications, such as nephropathy, retinopathy, neuropathy, impaired wound healing and accelerated atherosclerosis, have been associated with a range of cellular and subcellular changes. 4 Several pathophysiological conditions are known to be involved in the development of DM, including dyslipidemia, inflammatory cytokine production and oxidative stress.5,6 A chronic inflammatory state in vessels is now recognized as an important link between DM and its vascular complications. 7 During the process of inflammation, macrophages are recruited to sites of vascular injury and roll along the vascular endothelium, where they become activated by surface-bound chemokines. The macrophages adhere strongly to the vascular endothelium and are transferred through the monolayer of endothelial cells (ECs). Certain inflammatory molecules, such as the intercellular adhesion molecule-1 (ICAM-1), endothelial vascular cell adhesion molecule-1 (VCAM-1) and monocyte chemoattractant protein-1 (MCP-1), mediate the adhesion and infiltration of macrophages. 8
Growing evidence indicates that sirtuin 1 (SIRT1), the mammalian homologue of yeast Sirt2, is involved in increased metabolism of glucose and lipids, inflammatory responses and oxidative stress, which together contribute to the development of DM. 9 Moreover, resveratrol (RSV), a potent SIRT1 activator, has been shown to have beneficial effects on glucolipid metabolism and ageing in vivo and in vitro.10–12 These findings suggest that RSV might be a novel useful component for the prevention and treatment of DM and its related complications. Anti-inflammatory properties of RSV, manifesting as inhibition of prostaglandin synthesis via cyclooxygenase-1 (COX-1), have been demonstrated in a rat model.13,14 In a recent study, it was found that SIRT1 could deacetylate RelA/p65 and suppress its binding to DNA in ECs; therefore, RSV was involved in a crucial step in nuclear factor kappa B (NF-κB) signalling activation and in reducing the expression of endothelial adhesion molecules. 15 In addition, some in vitro studies found that RSV could reduce both the stimulated expression of VCAM-1 and monocyte adhesion to human vascular ECs.16,17 This suggests that RSV may act as a rapid molecular signal interfering in the mechanism of adhesion molecule expression and monocyte infiltration. However, few studies have focussed on the effect of RSV on vascular inflammation and macrophage infiltration in diabetic animals. We hypothesize that RSV might attenuate the expression of adhesion molecules (such as VCAM-1, ICAM-1 and MCP-1) in the aorta of diabetic mice, and the inhibition of the NF-κB pathway might explain the beneficial effect of RSV.
Materials and methods
Chemicals and animals
Unless otherwise stated, all chemical reagents were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Male C57BL/KsJ-
Plasma biochemical analysis
The blood samples were obtained from the tail veins of mice after withholding food for 6 h. Plasma samples were acquired on centrifugation of the blood samples at 4°C at 1000 g and were stored at −80°C until analysis. The levels of plasma insulin, glycated haemoglobin (HbA1c), total cholesterol (TC), triglycerides (TGs) and free fatty acids (FFAs) were measured enzymatically with commercial kits from Wako Chemicals USA, Inc. (Richmond, VA, USA) using an auto-analyser (Hitachi 7100; Hitachi Ltd, Tokyo, Japan).
Measurement of plasma ICAM-1, VCAM-1and MCP-1 levels using enzyme-linked immunosorbent assay
The concentrations of plasma-soluble ICAM-1, VCAM-1 and MCP-1 were assessed by mouse ICAM-1, VCAM-1 and MCP-1 Quantikine enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s instructions.
Preparation of aortic rings
After 8 weeks of treatment, mice were anaesthetized by intraperitoneal injection of 3% pentobarbital (30 mg/kg). The mice aortas were carefully dissected and placed into ice-cold Krebs solution (pH 7.4) containing 118 mM NaCl, 4.7 mM KCl, 1.1 mM MgSO4, 1.2 mM KH2PO4, 1.5 mM CaCl2, 25 mM NaHCO3 and 10 mM glucose. Cross sections of the aortic sinus were isolated for immunohistochemical or immunofluorescent staining. The remaining aortic samples were frozen and stored at −80°C for western blot analysis.
Immunohistochemistry and immunofluorescence assays
Isolated aortic sinus tissues were fixed by immersion in 4% paraformaldehyde for 48 h at 4°C and were incubated in 30% sucrose for 2 days. Each aortic sinus was embedded in paraffin. The paraffin-embedded sections (5 µm thick) were placed on poly-
To detect activated macrophages in aortic tissues, antigen retrieval was performed using citrate buffer. The primary antibody was rabbit anti-mac-3 (1:100 dilution; Santa Cruz Biotech Inc.). After incubation with the secondary antibody, P-phycoerythrin (PE) (Beyotime Biotech Inc.) was added to visualize antibody binding. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (Beyotime Biotech Inc.). For the quantitative analysis, the average score of 10–20 randomly selected areas was calculated using National Institutes of Health (NIH) Image Pro Plus 6.0 software.
Protein extraction and western blot analysis
The total and nuclear proteins in mouse aortic vessels were analysed by western blotting. Nuclear NF-κB p65 was extracted from the aortas using a nuclear extraction kit (Beyotime Biotech Inc.). Protein concentrations in the supernatants were measured using the bicinchoninic acid (BCA) protein assay kit (Pierce Chemical Company, Rockford, IL, USA). Proteins (40 µg) were separated by 8% or 10% sodium dodecyl sulphate (SDS)-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride (PVDF) membranes, which were then washed with Tris-buffered saline, blocked with 5% non-fat milk for 1 h and then incubated overnight at 4°C with the appropriate primary antibody at dilutions recommended by the supplier. After blots were washed with Tris-buffered saline with Tween 20 (TBST), they were probed with horseradish peroxidase–conjugated secondary antibody for 1 h at room temperature. The blots were then developed with SuperSignal-enhanced chemiluminescent substrate solution (Pierce Chemical Company). Blots were probed with antibodies against ICAM-1, VCAM-1 and MCP-1 (all from Santa Cruz Biotech Inc.), as well as against phosphorylated IκB kinase (IKK)-α/β, phosphorylated IκB and NF-κB p65 (all from Cell Signaling Technology, Inc., Danvers, MA, USA).
Statistical analysis
Data analysis was conducted with SPSS 16.0 software package for Windows (SPSS Inc., Chicago, IL, USA). Data distributions were examined for normality and homogeneity of variance, and normally distributed values were expressed as mean ± standard deviation (SD). Comparisons among groups were performed using one-way analysis of variance (ANOVA) with the Newman–Keuls post hoc test, followed by the least significance difference test. For non-parametric analysis, we used the Mann–Whitney U-test with post hoc analysis to evaluate differences between groups. Values of
Results
Effects of RSV on characteristics of experimental mice
Table 1 details the characteristics of three groups of mice at the end of the experimental period. The final body weights of mice were significantly higher in
Effects of resveratrol on body weight, blood glucose levels and lipid profiles in treated and untreated diabetic mice and in untreated non-diabetic (control) mice.
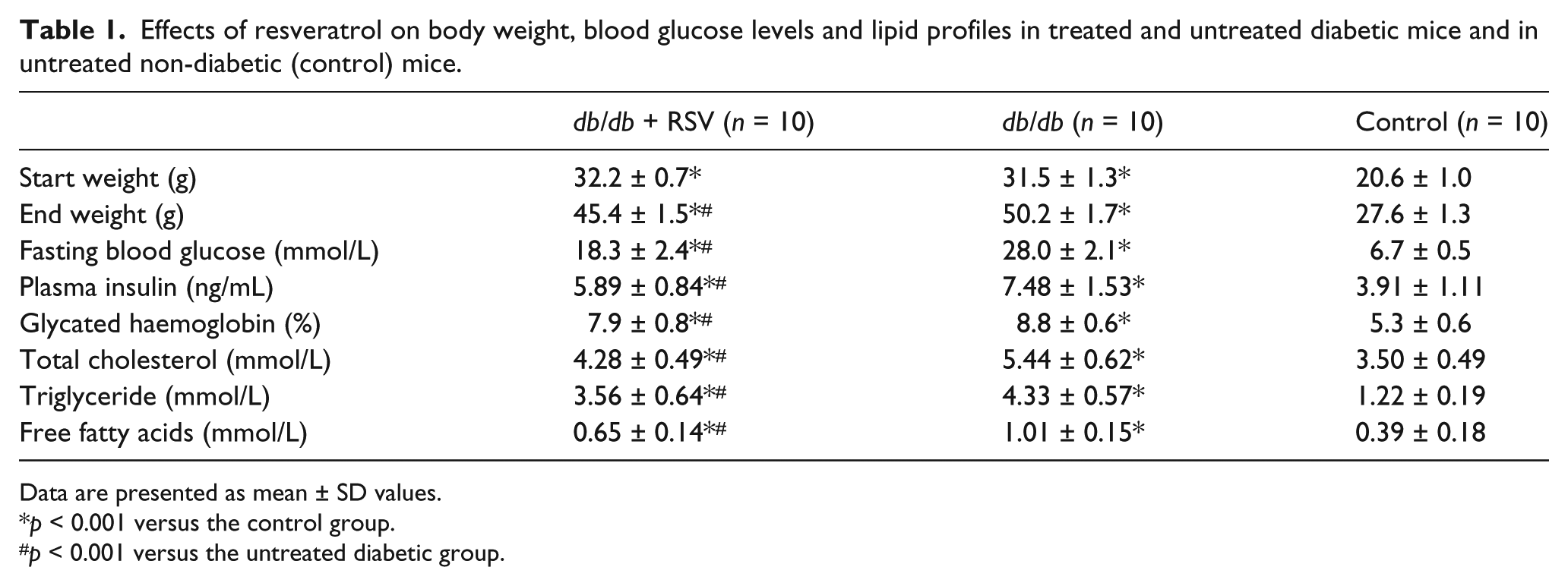
Data are presented as mean ± SD values.
Effects of RSV on glycometabolism and lipid metabolism
Levels of blood glucose, plasma insulin, HbA1c, cholesterol, TG and free fatty acids were significantly lower in diabetic mice treated with RSV than in untreated mice (all
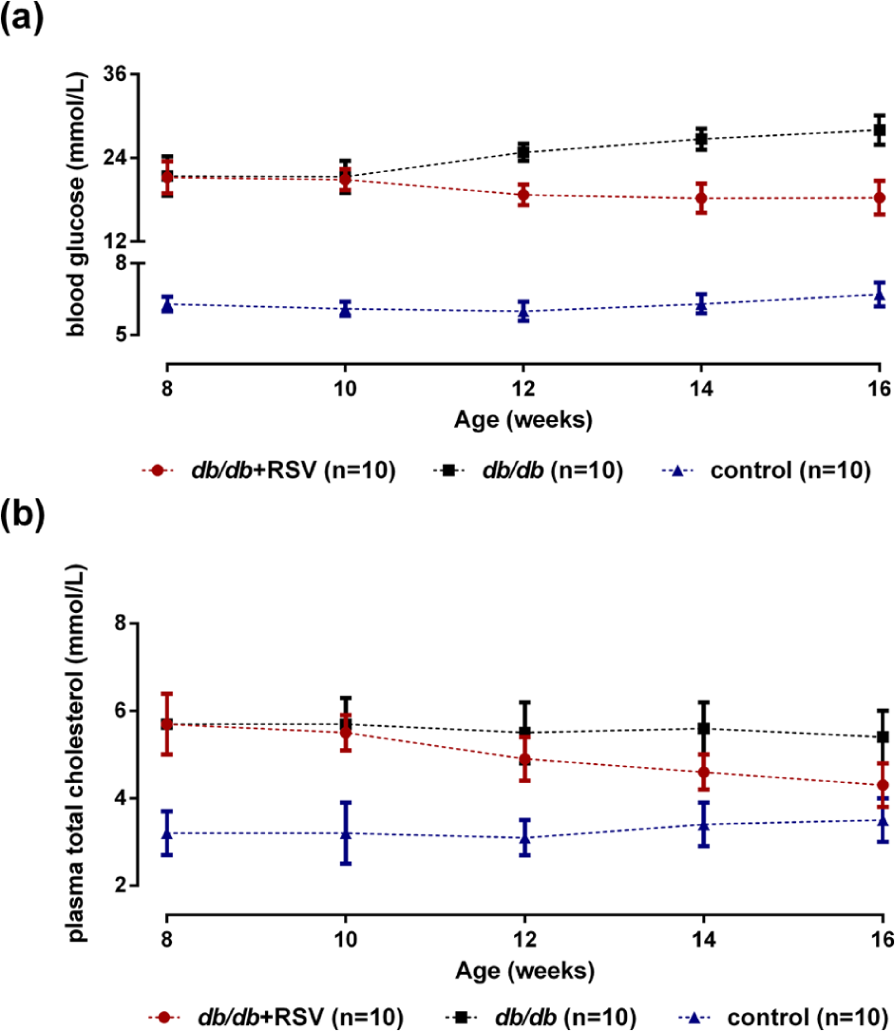
Effect of RSV on blood glucose and plasma total cholesterol levels in
SIRT1 expression in major organs and aortas of experimental mice
Because
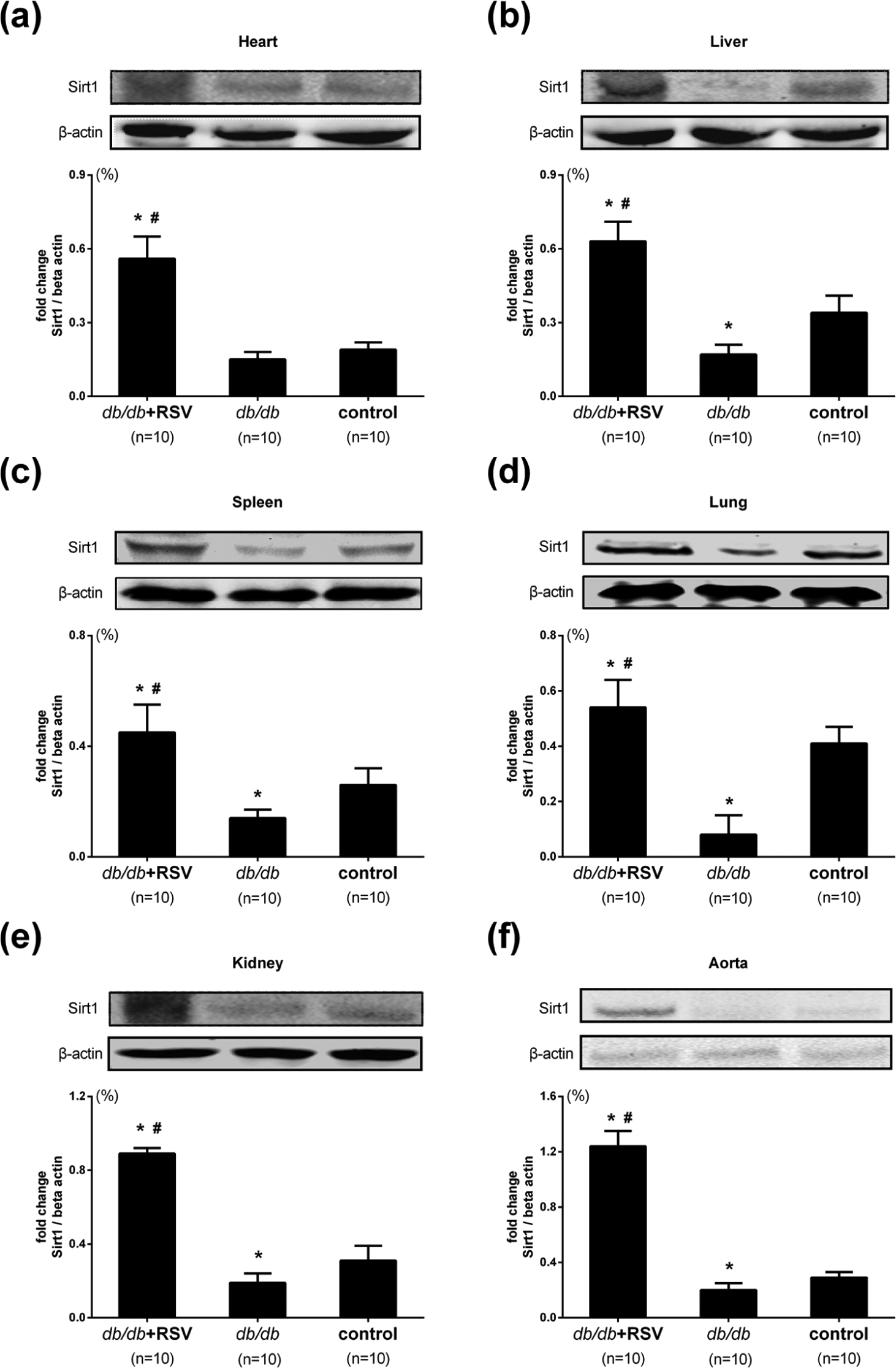
SIRT1 expression in major organs and aortic tissues of treated and untreated
Effect of RSV on the expressions of ICAM-1, VCAM-1 and MCP-1 in aortic tissues
Quantitation of the vascular adhesion molecules ICAM-1 (Figure 3(a) and (b)), VCAM-1 (Figure 3(a) and (c)) and MCP-1 (Figure 3(a) and (d)) in aortic tissue by immunohistochemical staining revealed significantly higher levels of ICAM-1, VCAM-1 and MCP-1 in untreated
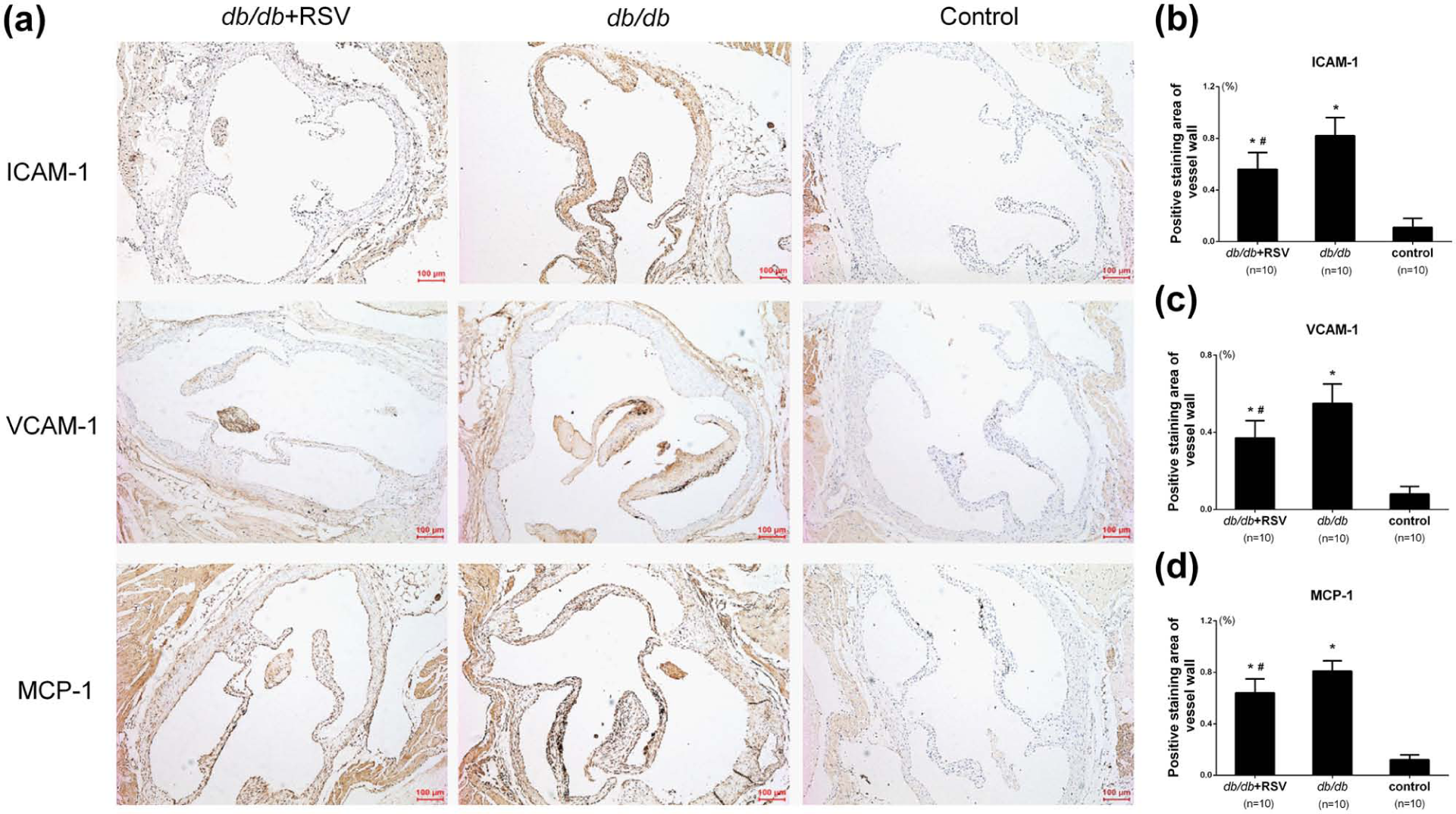
Effect of RSV on the expression of ICAM-1, VCAM-1 and MCP-1 in aortic tissues. (a) Representative immunohistochemical results of ICAM-1, VCAM-1 and MCP-1 in aortic tissues of mice. (b) The levels of ICAM-1 expression were analysed by immunohistochemical staining that involved using a polyclonal antibody to ICAM-1 to quantify its expressions in aorta. (c) The levels of VCAM-1 expression were analysed by immunohistochemical staining that involved using a polyclonal antibody to VCAM-1 to quantify its expressions in aorta. (d) The levels of MCP-1 expression were analysed by immunohistochemical staining that involved using a polyclonal antibody to MCP-1 to quantify its expressions in aorta.
Western blot analysis provided similar results. Whereas levels of ICAM-1, VCAM-1 and MCP-1 were significantly higher in the aortic tissues of untreated
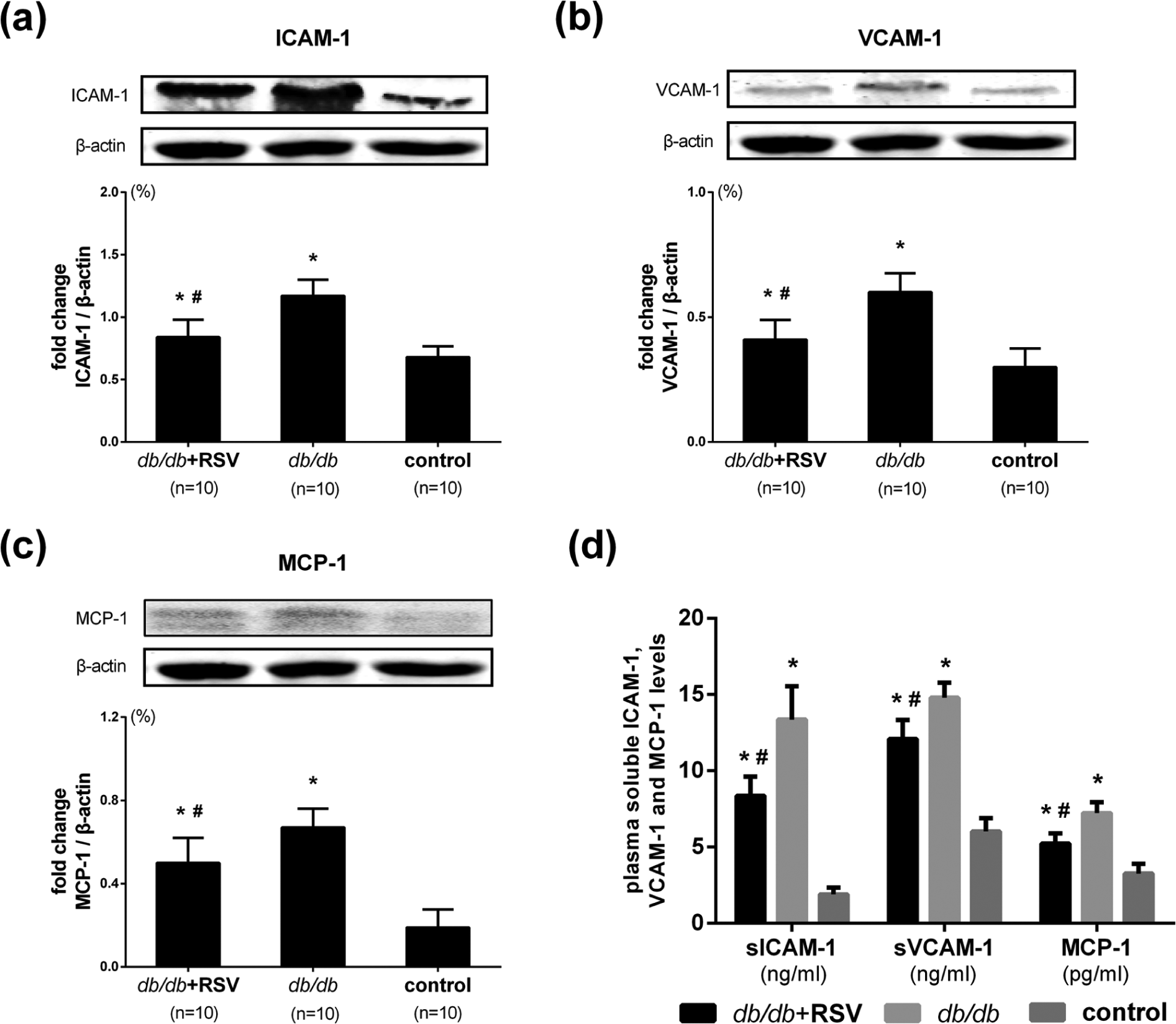
Effect of RSV on ICAM-1, VCAM-1 and MCP-1 expression in the aorta and plasma inflammatory biomarker levels. (a) Levels of ICAM-1 protein expression were analysed by western blotting by using a polyclonal antibody to ICAM-1 to quantify ICAM-1 expression in aortic tissues. (b) Levels of VCAM-1 protein expression were analysed by western blotting by using a polyclonal antibody to VCAM-1 to quantify VCAM-1 expression in aortic tissues. (c) Levels of MCP-1 protein expression were analysed by western blotting by using a polyclonal antibody to MCP-1 to quantify MCP-1 expression in aortic tissues. (d) Levels of plasma-based biomarkers of inflammation were measured in plasma from diabetic treated or untreated mice.
Effect of RSV on plasma inflammatory biomarkers
Levels of the plasma-soluble chemokines ICAM-1, VCAM-1 and MCP-1 were measured at the end of the study. The levels of plasma-soluble ICAM-1 in the RSV-treated diabetic, untreated diabetic and non-diabetic control groups were 8.36 ± 1.26, 13.36 ± 2.19 and 1.89 ± 0.45 ng/mL, respectively. The levels of plasma-soluble VCAM-1 in the RSV-treated diabetic, untreated diabetic and non-diabetic control groups were 12.08 ± 1.25, 14.79 ± 1.00 and 6.02 ± 0.87 ng/mL, respectively. The levels of plasma MCP-1 in the RSV-treated diabetic, untreated diabetic and non-diabetic control groups were 5.22 ± 0.68, 7.23 ± 0.72 and 3.26 ± 0.63 pg/mL, respectively. The results showed that the plasma levels of ICAM-1, VCAM-1 and MCP-1 in the untreated
Effect of RSV on macrophage infiltration in aortic tissues
We next investigated whether RSV treatment could reduce macrophage infiltration in a mouse model of DM. Levels of mac-3–positive macrophages, which provide a measure of the infiltration of activated macrophages, were lower in RSV-treated
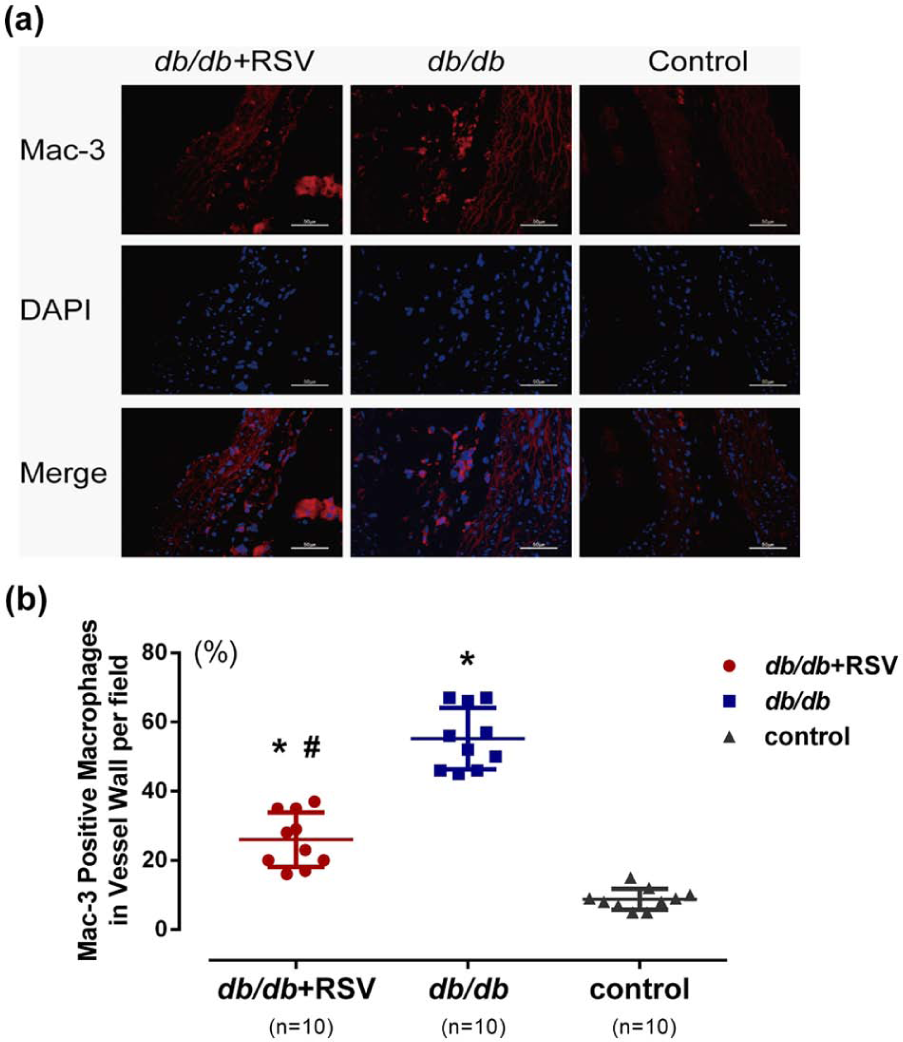
Effect of RSV on macrophage infiltration in the aorta. (a) Representative results of the infiltration of activated macrophages (Mac-3; red) into the vessel wall (staining with DAPI (blue) depicts nuclei; scale bar: 50 µm). (b) There are significant differences among all three groups (
RSV-mediated suppression of NF-κB pathway activity in aortic tissues of db/db mice
Levels of phosphorylated IKK-α/β, phosphorylated IκB, phosphorylated NF-κB p65 and non-phosphorylated NF-κB p65 were significantly greater in the aortas of
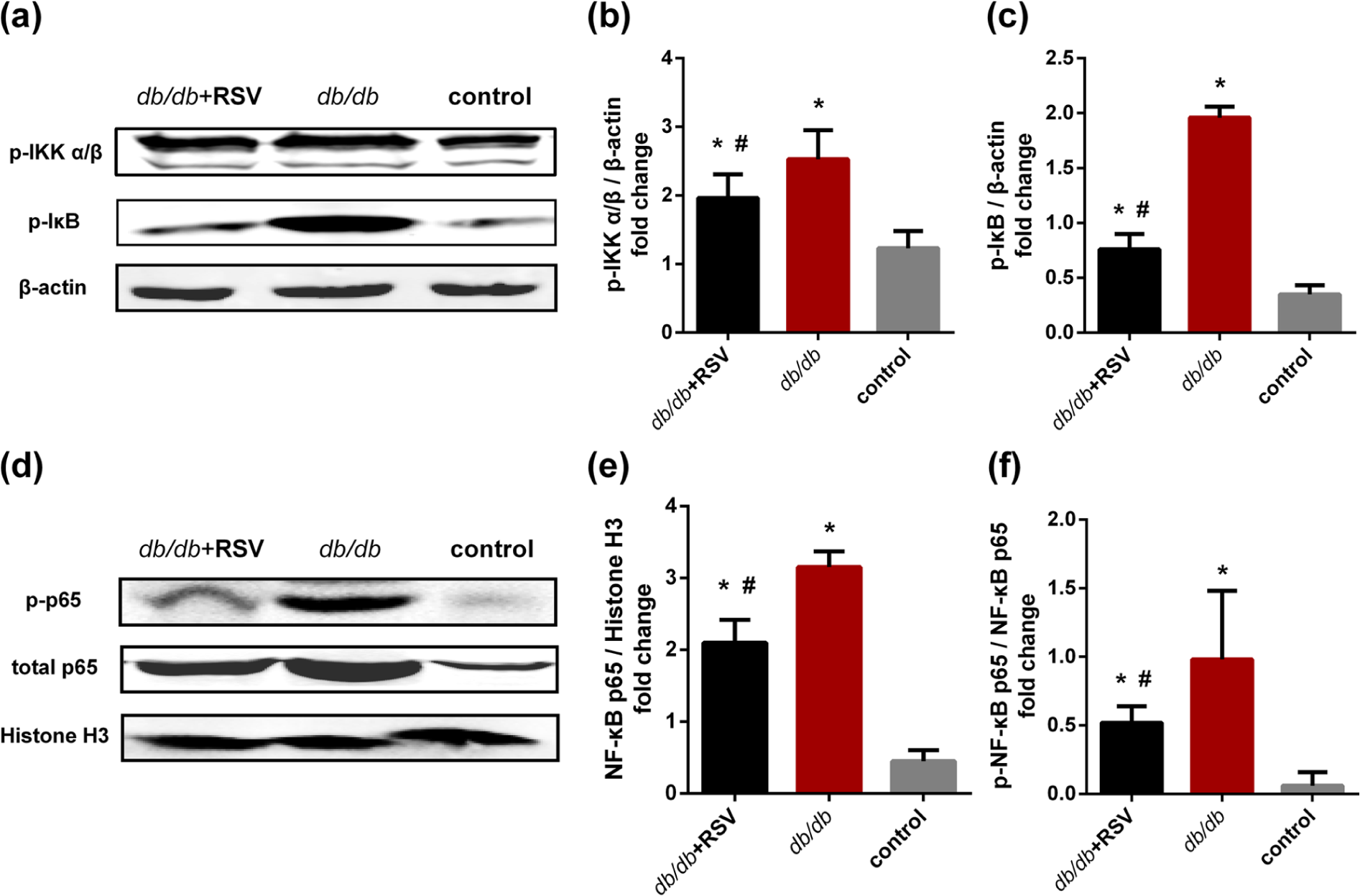
Effect of RSV on NF-κB activity in the aorta. Levels of phosphorylated IKK-α/β, phosphorylated IκB and both phosphorylated and unphosphorylated forms of p65 were detected in aortic tissues by western blot analysis. (a) Representative results of western blotting for phosphorylated IKK-α/β, phosphorylated IκB and beta-actin levels in aortic tissues. (b and c) The levels of phosphorylated IKK-α/β and phosphorylated IκB expression were analysed by western blotting that involved using a polyclonal antibody to phosphorylated IKK-α/β and phosphorylated IκB to quantify the expression in aorta. (d) Representative results of western blot for phosphorylated NF-κB p65, total NF-κB p65 and histone H3 levels in aortic tissues. (e and f) The levels of phosphorylated NF-κB p65 and total p65 protein expression were analysed by western blotting that involved using polyclonal antibodies to phosphorylated NF-κB p65 and total p65 to quantify the expression in aortic tissues.
Discussion
The hyperglycaemia associated with DM seems to play a significant role in atherogenesis, with the associated complications of atherosclerosis making the greatest contributions to the morbidity, mortality and enormous healthcare-related expenditure associated with patients who have DM. 21 Epidemiologic studies have established an association between the inflammatory response and the occurrence of type-2 DM (T2DM) and its associated complications. 22 However, the exact mechanisms of the inflammation in T2DM are still poorly understood. The main finding of this study is that oral RSV treatment can reduce vascular inflammation and macrophage infiltration in diabetic mice. Therefore, our findings suggest that RSV might be an effective supplement or nutrient capable of preventing DM-induced vascular complications.
Endothelial activation and increased monocyte adhesion are the key early events that trigger vascular inflammation as part of the pathophysiology of DM. 23 Some adhesion molecules, such as endothelial selectin (E-selectin), ICAM-1 and VCAM-1, are localized on the membranes of activated platelets and either monocytes or macrophages, as well as on the vascular endothelium. 24 When activated ECs present these molecules and secrete them into the blood stream, blood-circulating monocytes migrate to the sub-endothelial space and further activate into monocyte-derived macrophages and foam cells; this ultimately leads to the development of atherosclerotic lesions. 24
Together with E-selectin, ICAM-1 and VCAM-1, MCP-1 also contributes to the transmigration and infiltration of monocytes and macrophages in vessels, as well as to insulin resistance, by inducing a chronic inflammatory state.25,26 Haubner et al. 27 reported that exposure of ECs isolated from diabetic subjects to high glucose concentrations increased their rates of MCP-1 release by 40%–70%, with an associated 10%–20% increase in the levels of VCAM-1. In addition, a significant negative correlation was reported between the MCP-1 G-2518 genotype and both plasma MCP-1 levels and the prevalence of T2DM. 28 Many investigations confirmed that circulating levels of ICAM-1, VCAM-1 and MCP-1 were significantly higher in patients with T2DM than in otherwise comparable healthy individuals.29–33 Therefore, levels of ICAM-1, VCAM-1 and MCP-1 could be considered as diagnostic markers and therapeutic targets for DM and complications associated with DM.
The activation of inflammatory signalling has been suggested to contribute to the development of diabetic macrovascular complications.25,26,34 The transcriptional regulator NF-κB, which is one of the most intensively studied signalling intermediates, plays a central role in the inflammatory response. Hyperglycaemia accelerates the activation of NF-κB, which then induces the expression of many inflammatory genes that encode several mediators of atherogenesis, including inflammatory chemokines and adhesion molecules. 34 Activation of NF-κB and nuclear localization of p65 have been detected in vascular smooth muscle cells (VSMCs), macrophages and ECs, which are involved in human atherosclerotic lesions. 35 Moreover, NF-κB is also involved in adherence, diapedesis and accumulation of immune cells such as monocytes or lymphocytes in the vessel wall, which play a significant role in atherosclerotic plaque formation. 36 In addition, NF-κB activation has an influence on the proliferation of VSMCs 37 and mediates the expression of a variety of proinflammatory molecules by macrophages and VSMCs. 38 Hence, the NF-κB pathway is strongly linked to the pathogenesis of CVDs and DM.
Bordone et al.
39
found that SIRT1 represses the uncoupling protein (UCP) gene
RSV was also reported to improve dysfunction of vascular endothelium cells and to inhibit the apoptosis of pancreatic β-cells both in vivo and in vitro.47,48 In other studies, RSV administration reduced the levels of inflammatory mediators, as well as of oxidative markers.49,50 It seems that RSV could decrease high glucose–induced superoxide production via upregulation of SIRT1, induction of FOXO3a and inhibition of p47phox in monocytes.
51
Moreover, Liu et al.
52
reported that RSV could not only suppress advanced glycated end product (AGE)-induced COX-2 and production of NO and
However, our results provided no insights into the effects of different doses of RSV on the expression of inflammatory genes or the activity of NF-κB. Furthermore, the possibility cannot be excluded that RSV might have other profound biological effects on organs or tissues. Therefore, further studies will be required to better understand the clinical effects and side effects of RSV in vascular and other DM-related diseases.
Conclusion
In summary, our investigation provided in vivo evidence that RSV could reduce levels of ICAM-1, VCAM-1 and MCP-1 both in plasma and in the aortic tissues of
Footnotes
Acknowledgements
Rong Guo, Baoxin Liu and Ke Wang contributed equally to this article and should be considered co-first authors. The authors also thank Dr Wenhui Peng for his assistance in research design.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by grants from National Natural Science Foundation of China (no. 81070107).
