Abstract
Induction of indoleamine 2,3-dioxygenase (IDO1) is an established cellular response to infection with numerous pathogens. Several mechanisms, such as IDO1-mediated tryptophan (Trp) depletion, but also accumulation of Trp catabolites, have been associated with the antimicrobial effects of IDO+ cells. Recent findings of IDO1 as an immunoinhibitory and signaling molecule extended these previous observations. Using infection of professional phagocytes with Listeria monocytogenes (L.m.) as a model, we illustrate that IDO1 induction is a species-specific event observed in human, but not murine myeloid, cells. Knockdown and inhibition experiments indicate that IDO1 enzymatic activity is required for the anti-L.m. effect. Surprisingly, the IDO1-mediated antimicrobial effect is less prominent when Trp is depleted, but can be significantly amplified by tryptophan excess, leading to increased accumulation of catabolites that promote enhanced bactericidal activity. We observed a pathogen-specific pattern with kynurenine and 3-hydroxy-kynurenine being most potent against L.m., but not against other bacteria. Hence, apparent discrepant findings concerning IDO1-mediated antimicrobial mechanisms can be reconciled by a model of species and pathogen-specificity of IDO1 function. Our findings highlight the necessity to consider species- and pathogen-specific aspects of host–pathogen interactions when elucidating the individual role of antimicrobial proteins such as IDO1.
Introduction
Indoleamine 2,3-dioxygenase (IDO1) has been recognized as one of the major response elements during infection of mammalian cells with a large variety of pathogens, 1 including Gram-positive and Gram-negative bacteria,2–8 viruses9–12 and parasites such as Toxoplasma gondii and Leishmania donovani.1,13–15 Both phagocytic and non-phagocytic cells from different tissues and organs, including the immune system,3–6,16–18 have been shown to induce IDO1 expression and enzymatic activity upon infection. In many of these model systems, enzymatic activity of IDO1 was linked to antimicrobial activity against the invading pathogen. So far, no unifying mechanisms could be established for the role of IDO1 in host–pathogen interaction, with various reports praising the depletion of tryptophan (Trp) to be the major mechanism associated with oxygen-independent bactericidal activity,3,4,6,7,13,19,20 while some others propose Trp catabolites to mediate the antimicrobial effect.8,14,15 In some host–pathogen interactions, IDO1 expression is even considered deleterious to the host.21–25 Alternatively, IDO1 might be co-induced in a bystander fashion without exerting a role for the control of infection, as has been recently suggested for Mycobacterium tuberculosis. 21 Thus, there appears to be a significant heterogeneity in potential outcomes of IDO1 induction and function upon infection with different pathogens in different cell types and species. These different and sometimes discrepant outcomes might be best explained by a model of species- and pathogen-specific induction of IDO1 during host–pathogen interactions, and the molecular mechanisms of IDO1 action might be similarly specific. As general rules concerning IDO1 function during host–pathogen interactions have not yet been established we cannot predict the role of IDO1 for other so far uncharacterized host–pathogen interactions.
We have previously shown that IDO1 is among the most strongly induced genes and regulated proteins in human dendritic cells (DC) upon infection with Listeria monocytogenes (L.m.) both in vitro and in situ. 26 In addition, we established a correlation between IDO1 induction in so-called regulatory DC (matured in the presence of TNF-α, TLR2 ligands and prostaglandin E2) and gain of T-cell-suppressive function, as well as bactericidal activity against L.m. 17 However, as these regulatory DCs express numerous other molecules that might account for the observed biological functions, our previous work did not exclude the possibility that IDO1 does not play a major role for the antimicrobial activity. Similarly, although myeloid cells, including macrophages (MΦ) and DCs, have been shown to play a key role in the clearance of L.m. infection in mice, 27 a role for IDO1 has not been established in these studies. Furthermore, it cannot be ruled out, similar to findings for M. tuberculosis, 21 that IDO1 might be non-essential in the murine model of L.m. infection. To elucidate whether these different findings can be explained by species- and pathogen-specific regulation of IDO1 expression and function, we used the infection of myeloid cells with L.m. as a model. Here we present evidence for IDO1-mediated accumulation of Trp catabolites to be an important defense mechanism of human, but not murine, myeloid cells in response to L.m. infection, and show that these catabolites are bactericidal preferentially for this specific pathogen.
Materials and methods
Peripheral blood samples
Blood samples were collected from healthy blood donors at the Institute for Experimental Hematology and Transfusion Medicine of the University Hospital Bonn, Bonn, Germany, after informed written consent was obtained. All experiments were approved by the University of Bonn Institutional Review Board.
Bacterial strains
L.m. EGD-e strain and Listeria innocua (L.i.) 28 were cultured as described before. 29 Streptococcus pyogenes (S.p.) was cultured in BD Trypticase Soy Agar II with 5% sheep blood (BD Biosciences, Heidelberg, Germany).
Generation and maturation of human DC and MΦ
CD14 + monocytes were isolated from PBMCs using CD14 MicroBeads (Miltenyi Biotec, Bergisch Gladbach, Germany) and cultured in vitro to obtain either immature DC (immDC) or MΦ as previously described. 26 For DC maturation into several subtypes, the DC culture medium was supplemented with either 800 IU/ml recombinant (rh)-TNF-α alone (matDC) or in combination with 1 µg/ml prostaglandin E2 (Sigma-Aldrich, Taufkirchen, Germany) and 1 µg/ml Pam3CSK4 (Invivogen, San Francisco, CA, USA) for 72 h.17,26 MΦ were stimulated using 200 IU/ml rh-IFN-γ for 72 h. To evaluate the cytotoxicity of Trp catabolites on IDO-negative immDC (immDCIDO-), as well as IDO-expressing DC (DCIDO+), cells were incubated with 100 µM of different Trp catabolites (Sigma-Aldrich, Taufkirchen, Germany) for 24 h, and viability was assessed by propidium iodide staining (Invitrogen, Karlsruhe, Germany). All cytokines were purchased from Immunotools (Friesoythe, Germany).
Generation of murine DC and MΦ
Murine DC were generated from the bone marrow of 10–16-wk-old C57BL/6 mice as previously described. 30 Cells were cultured for 7 d in Iscove’s modified Dulbecco’s media (PAA, Pasching, Austria) supplemented with 10% FCS (Invitrogen, Karlsruhe, Germany), 100 U/ml penicillin and 0.1 mg/ml streptomycin (PAA, Pasching, Austria), and 10 ng/ml recombinant (rm) granulocyte–macrophage colony-stimulating factor (CSF). For the generation of bone marrow-derived murine MΦ (mMΦ) the same protocol was followed, but the cells were cultured with 10 ng/ml rm-murine-CSF. Non-thioglycollate murine peritoneal, splenic and liver MΦ were isolated and sorted according to previously described procedures,31–33 and further cultured as described above.
L.m. infection
Human MΦ and DCs were infected with wild type FITC-labeled L.m., EGD-e strain as previously described. 26 Cultures with an infection efficiency of more than 60% were used for further experiments. Upon infection, DC and MΦ were cultured for 24 h in CellGro or RPMI supplemented with 50 µg/ml gentamycin (Sigma-Aldrich, Taufkirchen, Germany), respectively. Murine DC and MΦ were cultured 24 h prior to infection with RPMI without antibiotics. L.m. infection was performed in the presence of mouse serum (Sigma-Aldrich, Taufkirchen, Germany) for 30 min using a multiplicity of infection of 5. The infected cell cultures were washed and further incubated for 24 h in RPMI supplemented with 50 µg/ml gentamycin.
Determination of kynurenine and nitrites
The amount of kynurenine (Kyn) in supernatants of DC and MΦ was determined as previously described. 34 The OD was measured with a Medgenix 400 AT microplatereader (BioTek Instruments, Bad Friedrichshall, Germany) at 492 nm. Nitrites (NO2-) determination was performed on supernatants using the Griess reaction system (Promega, Madison, WI, USA).
Assessment of the viability of intracellular L.m.
Infected DC or MΦ were incubated for 6 h at 37℃ in Hank’s balanced salt solution (HBSS) alone (referred as ‘Trp depletion’) or in the presence of 24 and 98 µM Trp, or 25 and 100 µM
IDO1 silencing and activity inhibition
Four different small interfering RNAs (siRNAs) (ON-TARGET plus; Thermo Scientific, Rockford, IL, USA) with the sequences 5′-UCACCAAAUCCACGAUCAU-3′, 5′-UUUCAGUGUUCUUCGCAUA-3′, 5-GUAUGAAGGGUUCUGGGAA-3′ and 5′-GAACGGGACACUUUGCUAA-3′ were used in combination to silence IDO1 gene expression. As control, renilla siRNA was used. Electroporation was performed with a BioRadGenePulserXCell II using a square wave protocol (2 pulses 1000 v, 0.5 ms). The silencing efficiency was assessed by Western blot. To assess L.m. intracellular viability after the inhibition of IDO1 enzymatic activity, DC and MΦ were treated 1 h before infection with 150 µM 1-methyl-
Western blot
IDO1 expression was assessed in human cells by mouse anti-human monoclonal Ab (Oriental Yeast, Tokyo, Japan) and in murine cells by rat anti-mouse monoclonal Ab (Biolegend, San Diego, CA, USA and Merck Millipore, Darmstadt, Germany). For the detection of inducible nitric oxide synthase (iNOS) protein a rabbit polyclonal Ab (Cayman Chemical, Ann Arbor, MI, USA) was used. The signals were detected on the Odissey system (Li-cor Biosciences, Bad Homburg, Germany). β-Actin expression was used as loading control (panactin Ab; Merck Millipore). Band intensity analysis was performed using ImageJ software (http://imagej.nih.gov/ij/)..
Bactericidal activity exerted by tryptophan catabolites
L.m., L.i. and S.p. were cultured in RPMI alone or supplemented with the following Trp catabolites: Kyn, 3-Hydroxykynurenine (3HK), anthranilic acid (AA), hydroxy-anthranilic acid (HAA), picolinic (PA), quinolic acid (QA), kynurenic acid (KyA) (Sigma-Aldrich, Taufkirchen, Germany) in a concentration range from 12 to 100 µM. After 24 h bacteria were plated in BHI (L.m. and L.i.) or blood agar (S.p.) for CFU analysis.
Statistical analysis
Statistical analysis was performed using SigmaPlot 10 (Systat Software, Erkrath, Germany). To calculate statistical significance a two-sample two-tailed t-test was used. Comparisons with a P-Value < 0.05 were statistically significant.
Results
IDO1 is expressed after L.m. infection in human, but not in murine, myeloid cells
We previously demonstrated that following L.m. infection several pro-inflammatory cytokines, such as TNF-α and IFN-γ, are induced at very early time points, followed by induction of IDO1 protein expression at later time points (12 hpi).17,26 These observations triggered important questions. First, is IDO1 expression after 12 h too late to be functionally important? Second, is IDO1 induction by infection dependent on DC maturation? Third, could findings in human cells be extrapolated to the murine system? Fourth, is there co-regulation of IDO1 with iNOS? Utilizing an infrared imaging system, we enhanced the sensitivity of IDO1 protein detection and clearly showed elevated protein expression in immDC at 6 hpi (Figure 1a, upper panel). Quantification analysis revealed a significant increase of IDO1 expression (almost 10-fold) as early as 8 hpi (Figure 1a, lower panel). To determine whether DC maturation has an effect, we infected immDCIDO-, matDC and DCIDO+ and compared IDO1 induction (Figure 1b). While IDO was significantly induced in immDC (18 ± 3.5-fold) and matDC (11 ± 3-fold) IDO1 expression remained constant in DCIDO+ before and after L.m. infection (Figure 1b). Taken together, L.m. infection of DC leads to induction of IDO1 in IDO- cells, but does not alter IDO1 expression once the protein is already expressed. To extrapolate our findings to the murine system we assessed expression of IDO1 and iNOS in murine cells in response to L.m. infection. In contrast to human cells (Figure 1b), infection of murine MΦ or DC resulted in prominent induction of iNOS (Figure 1c), whereas IDO1 was not detected (Figure 1c). To verify whether induction of iNOS, but not of IDO1, is a general response to L.m. infection in murine myeloid cells, we infected macrophages from different tissues, including spleen, liver and peritoneum. Like bone marrow-derived macrophages, dominant induction of iNOS was found after L.m. infection in hepatic, splenic and peritoneal MΦ. However, at 24 hpi IDO1 was not detected in any of the tissue MΦ tested (Supplementary Figure S1). Determining IDO1 activity by evaluating Kyn and NO2- accumulation in supernatants of infected DC and MΦ of both species demonstrated that the activity of both enzymes correlated with protein expression. While human cells produced significant levels of Kyn, but no NO2−, the situation was exactly the opposite in murine cells. These findings clearly indicate that the regulation of IDO and iNOS shows species-specific differences between human and murine myeloid cells, thereby curtailing any further study of IDO1 regulation in murine myeloid cells with respect to L.m. infection.
IDO1, but not iNOS, is highly induced after L.m.infection in human myeloid cells. (a) IDO1 protein expression was assessed by Western blot in human infected immDC at the indicated time points after infection with L.m. IDO1 expression was estimated by analyzing the intensity of each band normalized by β-actin, used as loading control. The results were plotted as relative expression (RE) compared with non-infected immDC (n = 3, mean ± SD). (b) iNOS and IDO1 expression were assessed by Western blot at 24 hpi in human immDC, matDC and DCIDO+. iNOS protein was not detectable; IDO1 RE was calculated as described for (a) (n = 4, mean ± SD). (c) iNOS and IDO1 expression were evaluated in murine DC and MΦ. IDO1 expression was not detectable; iNOS RE was calculated as described for (a) (n = 4, mean ± SD). (D, E) IDO1 and iNOS activity were evaluated in supernatants of infected human DC and MΦ (d) or murine DC and MΦ (E) at 24 hpi via Ehrlich colorimetric method for detection of Kyn (white bars) and Griess reaction for detection of NO2− (grey bars), respectively (n = 3, mean ± SD). *P < 0.05.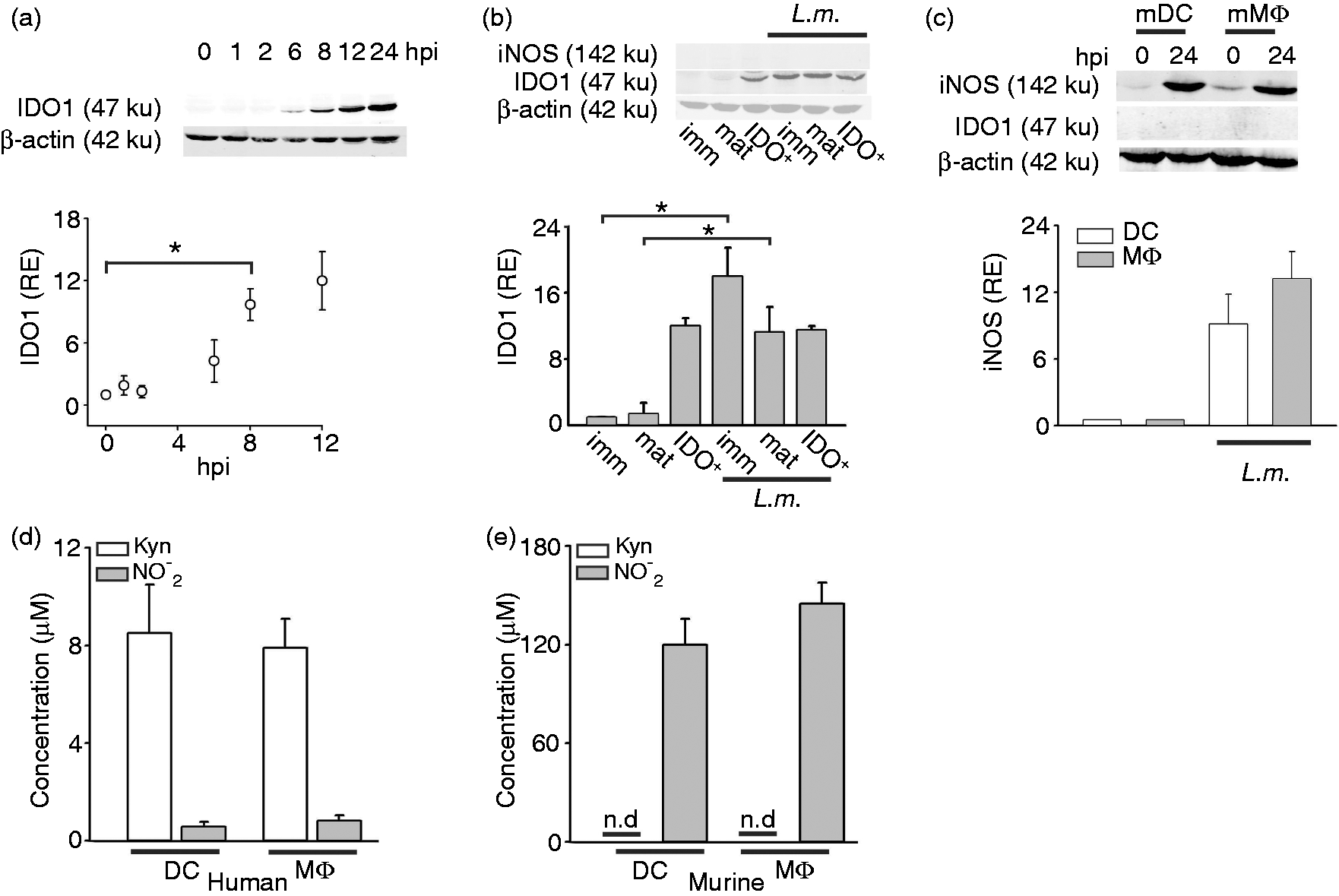
IDO1 activity is important to control L.m. growth in human DC
We previously provided indirect evidence that IDO1 might be involved in restricting growth of L.m. as DCIDO+ were more effective in controlling viable L.m. than immDCIDO− or matDCIDO−.
17
In fact, DCIDO+ showed significantly less bacterial burden than immDC and matDC at 6 hpi (Figure 2a). Furthermore, we extended these findings to human MΦ, demonstrating that MΦIDO+ cells showed significantly (60 ± 14%) less bacterial burden than their IDO1- counterparts (Figure 2b). As the genetic murine model was not informative, we established a very efficient knockdown (96 ± 4%) of IDO1 in human DC, which resulted in the absence of IDO1 even after L.m. infection (Figure 2c) but also no off-target effects in DC phenotype or function (Supplementary Figures S2 and S3). The recently described indoleamine 2, 3-dioxygenase 2 (IDO2)
35
was also elevated in L.m.-infected DC and MΦ (data not shown). However, a complete abrogation of Kyn production after IDO1 silencing (Figure 2d) suggested that IDO1 and not IDO2 is the major Trp catabolizing enzyme in human myeloid cells. More importantly, loss of IDO1 expression and function following knockdown resulted in loss of control of bacterial burden at later time points (6 h, Figure 2e). To our knowledge, these are the first observations of an IDO knockdown in primary human myeloid cells, indicating participation of IDO1 in the control of bacterial burden during L.m. infection.
IDO1 knockdown leads to unrestrained L.m. growth. (a, b) Bactericidal activity of human immDC, matDC and DCIDO+ (A, n = 4, mean ± SD) and of MΦIDO- and MΦIDO+ (B, n = 3, mean ± SD) was evaluated by CFU assay 6 hpi. (c) IDO1 silencing efficiency was evaluated by Western blot in DCIDO+ infected with L.m. One representative experiment out of four is shown. (d) Kyn accumulation in supernatants of DCIDO+ infected by L.m. treated with control or IDO1-specific siRNA was evaluated by Ehrlich colorimetric method (n = 4, mean ± SD). E, Anti-bacterial activity of DCIDO+ untreated or treated with control siRNA and IDO1-specific siRNA was evaluated by CFU analysis 6 hpi (n = 4, mean ± SD). *P < 0.05.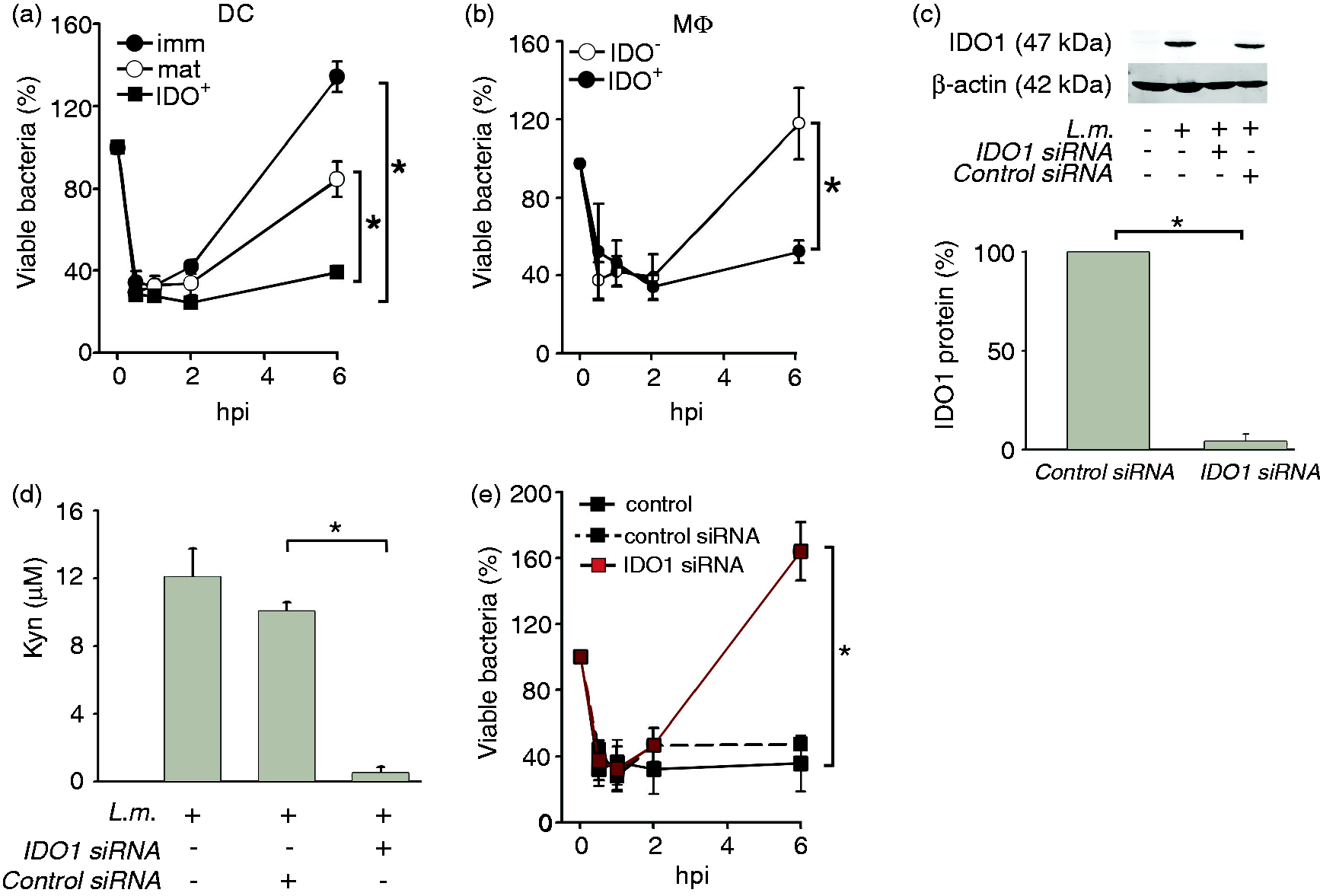
Enzymatic activity of IDO1 is required for the control of L.m.
To further prove that the enzymatic function of IDO1 mediates the control of L.m. in human myeloid cells, we blocked the enzymatic activity using the IDO1 functional inhibitor 1-MTand re-assessed bactericidal activity by determining the bacterial load and viability, as well as Kyn production as surrogate for enzymatic IDO1 activity. DC and MΦ treated with 1-MT showed more than 50% reduction in Kyn levels indicating that IDO1 enzymatic activity was significantly reduced under these conditions (Figure 3a). When assessing bacterial burden in DCIDO+ a significant increase (60 ± 15%) comparable to matDCIDO− (see Figure 2a) was observed in the presence of 1-MT, indicating that the enzymatic activity of IDO1 was necessary for DCIDO+ to keep the bacterial burden in check (Figure 3b). Similarly, 1-MT-treated MΦIDO+ also showed significant augmentation of bacterial burden relative to 1-MT-untreated controls (Figure 3c). Altogether, our data suggest that the enzymatic activity of IDO1 is required to control L.m. growth in human myeloid cells.
Control of L.m. infection in DCIDO+ and MΦIDO+ is dependent on IDO1 enzymatic activity. (A) IDO1 enzymatic activity was assessed in infected DCIDO+ (gray bars) and MΦIDO+ (white bars) 6 hpi after treatment with 150 µM 1-MT. Kyn accumulation was measured by Ehrlich colorimetric method (n = 3, mean ± SD). (b) Anti-bacterial activity of DCIDO+ was evaluated by CFU in the presence and absence of 150 µM IDO1 inhibitor 1-MT (n = 3, mean ± SD). C, Anti-bacterial activity of MΦIDO+ was assessed by CFU assay in presence or absence of 150 µM 1-MT. *P < 0.05.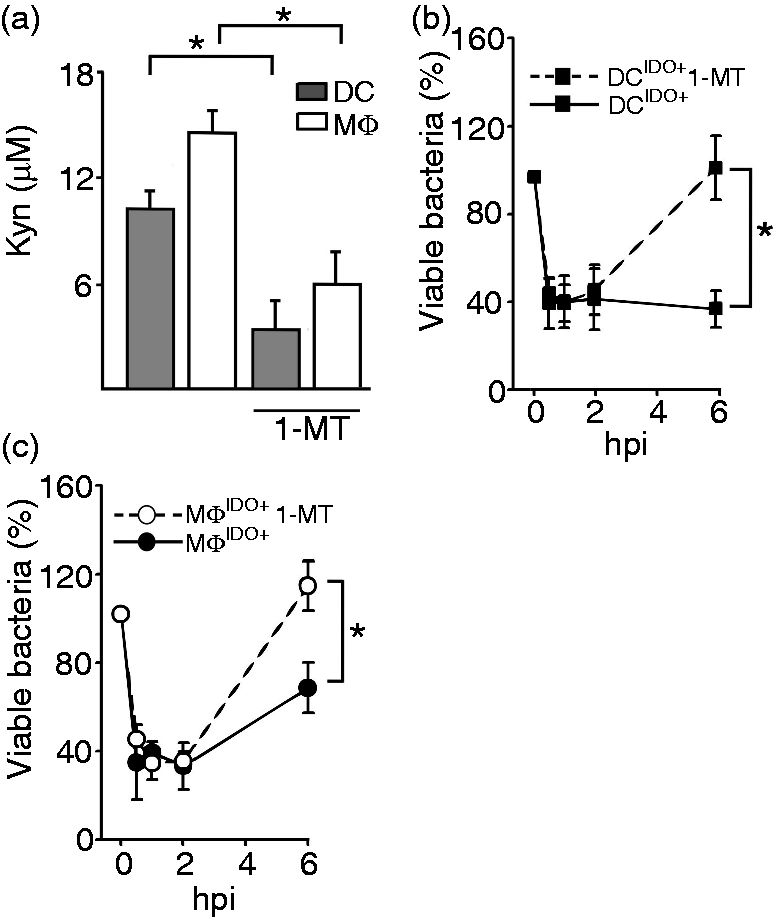
IDO1 exerts its microbicidal activity via kynurenines
Trp depletion, but also induction of toxic downstream catabolites collectively known as Kyns, have been shown to be the major mechanisms of the microbicidal activity of IDO1.3,8,14,20,36 However, the mechanism of IDO1 action in primary myeloid cells as one of the major targets of L.m. infection in humans
26
has not yet been studied. In order to establish whether Trp starvation or Trp catabolites accumulation mediates the bactericidal activity of human DCIDO+ against L.m. the bacterial burden of DCIDO+ and immDCIDO− was compared under Trp depletion or Trp excess. Unexpectedly, Trp excess enabled DCIDO+ to increase their control of L.m. (Figure 4a). Under these conditions approximately 30% of bacteria were viable in IDO+ DC at 6 hpi compared with a significantly higher bacterial burden in infected DCIDO+ cultured under Trp depletion (80% viable L.m.). In contrast, the bacterial burden in immDCIDO− was similarly high (>120%) under both conditions. To rule out that Trp depletion might decrease DCIDO+ viability, infected DCIDO+ cells were incubated in a Trp-free medium during 6 h. The percentage of viable cells was assessed at 0 and 6 hpi. Our results showed that incubation of DCIDO+ in a Trp-free environment did not lead to a significant decrease in viability during this period of time (Figure 4b).The enhanced activity of DCIDO+ in Trp excess was clearly associated with increased Kyn accumulation in culture supernatants, suggesting the latter are responsible for the bactericidal effect (Figure 4c). In supernatants of DCIDO+ cultured in the absence of Trp only background levels of Kyn were detected. Furthermore, at 6 hpi, DCIDO+ cultured under Trp excess produced three times more Kyn than under Trp depletion or immDC, reaching a maximum of 12 ± 3.5 µM (P = 0.02) (Figure 4c). To further investigate the role of Kyn, immDCIDO− were incubated with increasing concentrations of Kyn. After 6 hpi the bacterial burden was significantly reduced in immDC cultures supplemented with exogenous Kyn to levels observed for DCIDO+ (Figure 4d). Moreover, this effect showed dose-dependency with higher levels of Kyn inducing a stronger reduction of the bacterial burden at the 6 hpi time point. Recently, it was described that Trp catabolites have a negative impact on cell viability.
37
To rule out whether direct cytotoxic effects of Kyns can interfere with the antimicrobial activity of IDO-competent myeloid cells, we incubated immDCIDO− and DCIDO+ in the presence of otherwise cytotoxic amounts
37
of different Trp catabolites (Figure 4e). None of the tested catabolites had a deleterious effect on DC viability. We conclude that accumulation of Trp catabolites, but not Trp depletion, restrained intracellular growth of L.m. in human myeloid cells without compromising their cell viability.
Trp starvation does not mediate the anti-bacterial activity of DCIDO+. (a) Bacterial burden of infected immDCIDO- and DCIDO+ cultured under Trp excess (98 µM) or depletion was evaluated in CFU assays 6 h after L.m. infection (n = 4, mean ± SD). (b) Viability of immDC infected with L.m. under Trp depletion conditions at 0 and 6 hpi was assessed via propidium iodide staining. (c) Kyn production was assessed by Ehrlich colorimetric reaction in supernatants of infected immDC and DCIDO+ at 6 hpi under Trp excess or Trp depletion conditions (n = 4, mean ± SD). (d) immDC anti-bacterial activity was assessed by CFU under control conditions (HBSS buffer) or in HBSS in the presence of Kyn 25, 50 and 100 µM (n = 4, mean ± SD). (E) Viability of immDC and DCIDO+ untreated or exposed over 24 h to 100 µM of different Trp catabolites was assessed by propidium iodide staining (n = 3, mean ± SD),*P < 0.05.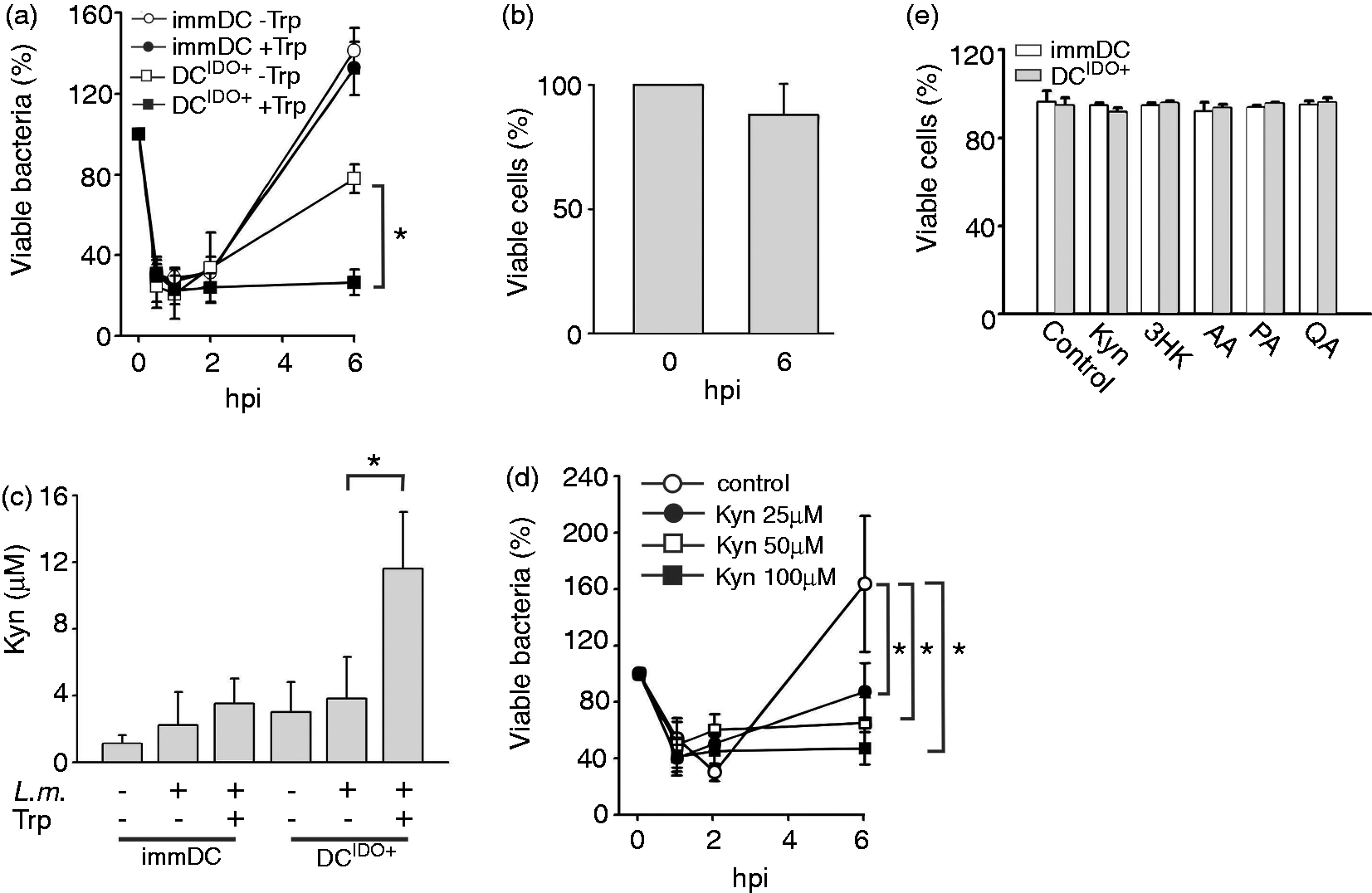
Trp degradation encompasses several enzymatic steps leading to a series of catabolites intermediates (Figure 5a). As all enzymes of the Kyn pathway are expressed in MΦ
38
we explored the possibility that other Trp catabolites might also contribute to the observed bactericidal activity of DCIDO+ and MΦIDO+. To answer this question L.m. were cultured in the presence of several intermediates of the Kyn pathway for 24 h and the bactericidal viability was determined. At high concentrations L.m. was sensitive to all tested Trp catabolites except to KyA, and virtually no viable colonies were detected (Figure 5b). Yet KyA has been suggested to control the growth of bacteria in the digestive system,
39
its function in L.m.-infected human myeloid cells has not been investigated so far. KyA had the lowest impact on L.m. growth, and reduced the bacterial viability by around 20 ± 7% at a concentration of 100 μM. Strikingly, Kyn and its direct degradation product 3HK showed the most prominent effects reducing L.m. viability to 42 ± 7% and 55 ± 6% at 12 μM, similar to the concentration of Kyn detected on supernatants of DCIDO+ (Figure 5b). Similarly, most Trp catabolites were bactericidal for S.p., albeit the pattern was different (Figure 5c). In contrast to L.m. 3HK did not show a significant effect on S.p. Most strikingly, L.i. was practically insensitive to low and intermediate concentrations of the catabolites. Only at high concentrations was a significant reduction of viability observed for Kyn, 3HK, AA and QA (Figure 5c). We conclude that the bactericidal profile of Trp catabolites is species-specific. In the case of L.m., all major intermediates of the Kyn pathway seem to contribute to the antimicrobial effect of DCIDO+ and MΦIDO+, while other Listeria species and other Gram-positive bacteria show a distinct pattern of sensitivity against Trp catabolites. In addition, we tested the effect of Trp catabolites on the Gram-negative bacteria Shigella flexneri. However, under the current experimental setting none of the tested catabolites showed a significant effect on the growth of this pathogenic bacterium (data not shown). In comparison, Narui et al.
8
reported a significant effect of 3HK on the Gram-negative bacterium Escherichia coli at a concentration of approximately 100 μM,
8
suggesting once more the pathogen-specific sensitivity of bacteria to kynurenines.
Trp catabolites have intrinsic anti-bacterial properties. (a) Schematic representation of the kynurenine pathway. (b) L.m. viability was assessed by CFU 24 h after exposure to Trp catabolites in a concentration range from 12 to 100 µM (n = 3 mean ± SD). (c) S.p. viability was evaluated by CFU 24 h after exposure to Trp catabolites in a concentration range from 25 to 100 µM (n = 3 mean ± SD). (d) L.i. viability was evaluated by CFU 24 h after exposure to different Kyn concentration (n = 3 mean ± SD). The following conventions were used for (B), (C) and (D): Kyn
 ; 3HK
; 3HK ; AA
; AA ; PA
; PA ; QA
; QA ; HAA
; HAA , KyA
, KyA , *P < 0.05 (Student’s t-test).
, *P < 0.05 (Student’s t-test).
Discussion
Intracellular mechanisms are an important line of defense against pathogens, and the Trp-catabolizing enzyme IDO1 has been suggested to evoke such mechanisms.2,3,13 Somewhat inconsistent findings from different cellular systems, species and different pathogens make it difficult to reconcile whether IDO1 always plays a role in pathogen defense.22,23,25 We provide evidence that IDO1 is induced upon challenge with pathogenic L.m. in human, but not murine, myeloid cells. A reciprocal observation was observed for iNOS with induction in murine, but not in human, myeloid cells, clearly supporting species-specificity of IDO1 induction upon challenge with the same pathogen. Knockdown and inhibitory experiments of IDO1 in primary human myeloid cells revealed that IDO1 expression conferred bactericidal activity to these cells and strongly suggested that the enzymatic activity of IDO1 is required for this intracellular defense mechanism. In contrast to previous findings reported for other cellular systems and pathogens,3,13,20 we clearly demonstrate that Kyn and 3HK accumulation as a consequence of IDO1 enzymatic activity is the major mechanism of the antimicrobial effect. More striking, allowing human myeloid cells to produce excess amounts of catabolites by exposure to elevated levels of Trp clearly promotes the antimicrobial capacities of these cells. These findings were further supported by conferring antimicrobial activity to IDO1- human DC in the presence of excess amounts of Kyn. The pattern of effectiveness of Trp catabolites, however, showed pathogen-specificity, as Kyn and 3HK are not as potent against phylogenetically related bacteria, such as L.i., or other bacteria, such as S.p. Different responses of myeloid cells to other pathogens, such as Chlamydia, have also been described. For example, DC infected with Chlamydia pneumoniae in the presence of excess Trp showed reduced growth restriction of this pathogen, 16 while it was shown for Chlamydia trachomatis that Trp starvation resulted in reduced anti-bacterial effects.40,41
Lack of IDO1 induction upon infection of murine myeloid cells is in agreement with previous reports demonstrating that NO or peroxynitrite donors can inhibit IDO1 activity via nitration of tyrosine residues42,43 or via post-translational modification promoting degradation. 35 These processes are dependent on iNOS expression, which we and others 44 demonstrate for murine, but not for human, myeloid cells, indicating species-specific IDO1 regulation in myeloid cells. In vivo evidence in murine models demonstrating that IDO1 plays a role during T. gondii, and L. major 45 or L.m. infection during pregnancy 46 has not been linked directly to IDO1 expression in myeloid cells, indicating differential IDO1 regulation among cell types and tissues in mice. In contrast to L.m. infection, iNOS and IDO1 were shown to be induced in murine MΦ infected with Trypanosoma cruzi.14,15 Yet another pattern was described for M. tuberculosis. 21 While mRNA expression of IDO1 was observed in murine MΦ upon infection, infection of IDO-/- mice revealed IDO1 activity not to be essential for control of M. tuberculosis. However, keeping in mind that granuloma formation in mice requires loss of iNOS, 47 the lack of IDO1 function in iNOS wild type mice might not reflect the role of IDO1 that has been proposed for human tuberculosis owing to species-specific differences of the host response towards this human-specific pathogen.
By knockdown of IDO1, inhibition of its enzymatic activity, or replenishment of Trp or its catabolites, we demonstrate that the antimicrobial effect against L.m. in human myeloid cells is due to enrichment of Trp catabolites. This is different to other pathogens for which Trp starvation was determined as the major antimicrobial effect3,6,13,19,48 again supporting a pathogen-specific role of IDO1 induction during infection. In contrast to other pathogens auxotroph for Trp, virulent strains of L.m. can synthetize aromatic amino acids, including Trp.49–52 This can be seen as an adaptation mechanism of L.m. to survive in mammalian cells that react to infection by inducing IDO1 thereby depleting Trp.
From the host perspective, however, Trp depletion cannot be the major host defense mechanism against prototrophic pathogens such as L.m. Interestingly, both human monocyte-derived macrophages and DCs were able to restrain bacterial growth at early time points before IDO1 expression was induced, indicating that other cell-autonomous defense mechanisms must play a role in the control of infection at early time points. A very likely mechanism might be the production of reactive oxygen species (ROS), as they have been identified as key mechanism to restrict L.m. growth within the first 30 min of infection. 53 Indeed, we have preliminary evidence that ROS production in human DC and MΦ reaches a maximum at the early time points between 0 and 2 h after L.m. infection, suggesting that this mechanism might be responsible for the control of bacterial growth during this time frame (data not shown). Furthermore, the harsh environment provided by the phagosome, together with the action of proteases might contribute to restrain L.m. growth before it escapes this compartment within the first hpi. 54 It also cannot be ruled out that important effector mechanisms, including IFNs or ILs, e.g. IL-6, 55 might be involved in anti-bacterial effects of infected human myeloid cells.
We have recently linked strong localized IDO1 expression to human granulomatous diseases.17,26 IDO1 homologues have not been found in invertebrates including Caenorhabditis elegans and Drosophila melanogaster, while in some non-mammalian vertebrates like zebra fish or Xenopus laevis only proto-IDO proteins with low efficiency for Trp degradation were described. 56 L.m. does not induce IDO1 expression in murine myeloid cells and there is no true equivalent to L.m.-associated human chronic granulomatous disease, making it difficult to study the effect of IDO1 on L.m. in myeloid cells directly in vivo. While human in situ and in vitro data support a model of containment for strong IDO1 expression in human myeloid cells in building the ringwall structure of granuloma,17,26,57 recent observations in the zebra fish strongly argue that granulomas primarily function as a repository of infecting bacteria allowing for subsequent spread of disease.58,59 Even though the idea of growth restriction in a potential reservoir, i.e. granuloma, appears to be counterintuitive, we present data herein suggesting that these are complex interactions involving temporal physiological adaptations both in the host and the invading bacterium.
The role of IDO1 in myeloid cells during infection appears to be species- and pathogen-specific. These findings also underline the need to carefully consider the right model organisms and cell types to be studied when addressing the role of IDO1 in a human disease context. Further development of methodologies allowing the analysis of host–pathogen interactions in their natural habitats is required.
Footnotes
Funding
This work was supported, in part, by the German Research Foundation [SFB 670, SFB 704, INST 217/575-1, INST 217/576-1, INST 217/577-1] (to J.L.S) and the NRW Graduate School Chemical Biology (to A.N.-C). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Acknowledgements
We thank Michael Kraut and Laura Bohmann for their technical assistance, and J. Oldenburg for providing us with blood samples from healthy individuals.
Conflict of interest
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
