Abstract
This study was conducted to evaluate whether dietary supplementation with
Introduction
Commercially-reared food animals encounter serial pathogenic and nonpathogenic immunological challenges throughout production. 1 In particular, weaned piglets (approximately 3–5 wks of age) are susceptible to immunological challenge as a result of their less mature immune and digestive systems. 2 The stress response is indicated by growth retardance, decreased growth rate of lean tissue and quantitative changes in nutritional requirements.3,4 Immunological challenge involves the release of a variety of pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6.3,4 Over-release of these pro-inflammatory cytokines has an adverse effect on growth and feed efficiency. In addition, overproduction of pro-inflammatory cytokines can result in the injury of tissues. As the most important metabolic organ, the main detoxification site and the primary defense barrier, the liver is liable to be injured by immunological challenge.5,6
Traditionally, arginine (Arg) is thought of as a nutritionally non-essential amino acid.
7
However, in the last two decades, increasing evidence has shown that Arg plays an important role in various physiological and biological processes.8,9 Of particular interest, Arg is the substrate for the synthesis of NO, a key mediator of various physiological functions,
9
the immune response
10
and neurological function.
11
Arg is known to exist in a number of animal cells, including hepatocytes and Kupffer cells.
12
Hepatocytes take up large amounts of Arg from the hepatic urea cycle and liver failure is associated with high plasma Arg level.
13
Research has shown that Arg exerted a protective effect in many liver injury models, such as hepatic ischemia reperfusion injury in pigs,
14
and acute cholestasis-induced liver damage in rats.
15
In addition, Arg supplementation may be beneficial in offering protection to mice following methicillin-resistant Staphylococcus aureus challenge.
16
Wilcken et al.
13
reported that most studies in animal model have provided evidence for beneficial effects of oral
TLRs play a central role in the activation of the innate system as a result of the recognition of bacterial pathogen-associated molecular patterns. Of them, TLR4, the receptor of LPS, is a major player and triggers the activation of different intracellular signaling cascades, such as activation of NF-κB and the production of reactive oxygen species (ROS). 17 Activated NF-κB stimulates the synthesis of pro-inflammatory cytokines, including TNF-α, IL-1β and IL-6. 17 Consequently, ROS and pro-inflammatory cytokines are involved in the activation of host defense mechanisms and tissue injury. 17
In our current experiment, Escherichia coli LPS was administered as an inflammatory agent to establish the model of liver injury following the model of Masaki et al.
5
Our objective was to evaluate whether dietary supplementation with
Materials and methods
Experimental animals and design
Ingredient composition of the basal diet (as-fed basis).
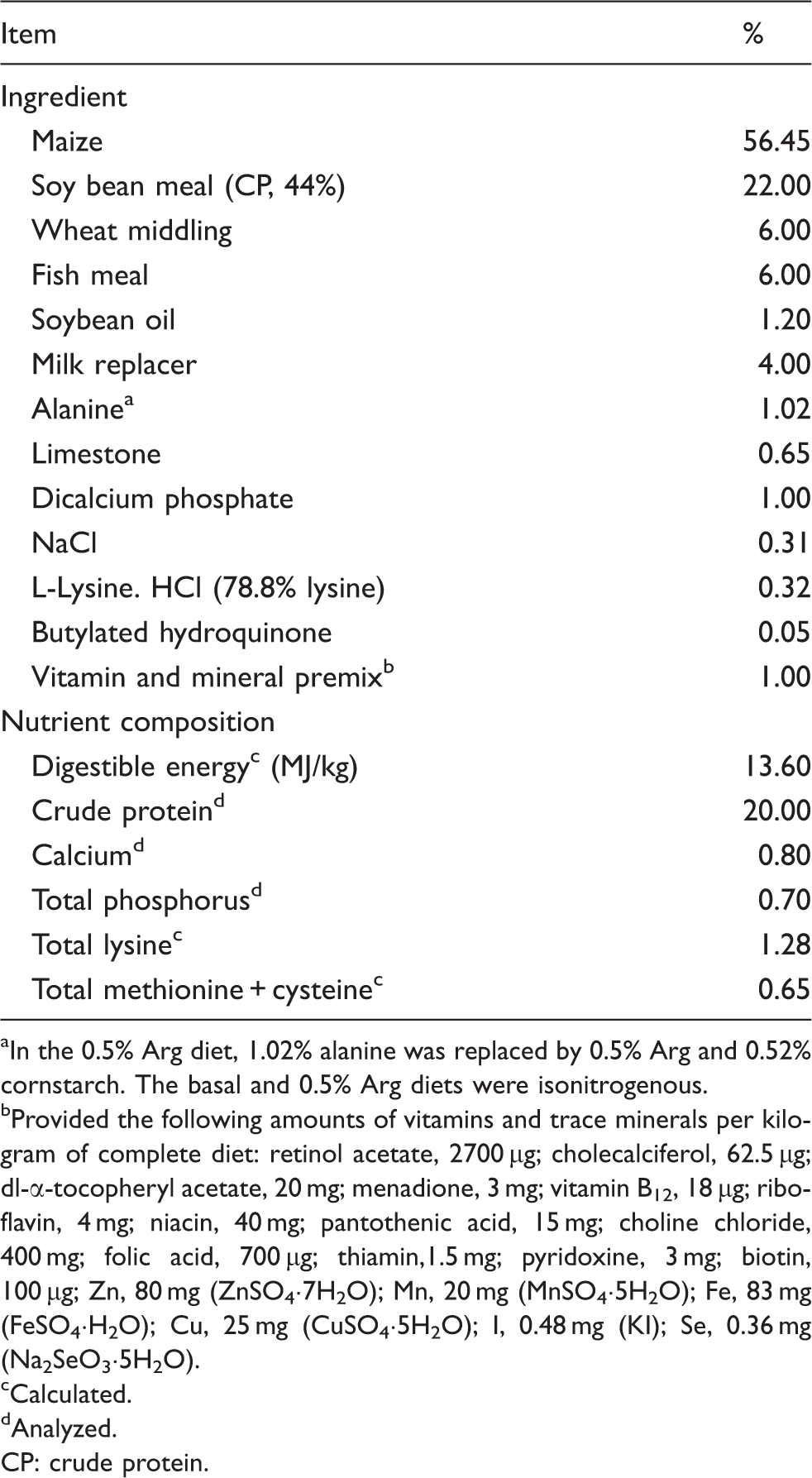
In the 0.5% Arg diet, 1.02% alanine was replaced by 0.5% Arg and 0.52% cornstarch. The basal and 0.5% Arg diets were isonitrogenous.
Provided the following amounts of vitamins and trace minerals per kilogram of complete diet: retinol acetate, 2700 µg; cholecalciferol, 62.5 µg; dl-α-tocopheryl acetate, 20 mg; menadione, 3 mg; vitamin B12, 18 µg; riboflavin, 4 mg; niacin, 40 mg; pantothenic acid, 15 mg; choline chloride, 400 mg; folic acid, 700 µg; thiamin,1.5 mg; pyridoxine, 3 mg; biotin, 100 µg; Zn, 80 mg (ZnSO4·7H2O); Mn, 20 mg (MnSO4·5H2O); Fe, 83 mg (FeSO4·H2O); Cu, 25 mg (CuSO4·5H2O); I, 0.48 mg (KI); Se, 0.36 mg (Na2SeO3·5H2O).
Calculated.
Analyzed.
CP: crude protein.
Treatments included: (1) non-challenged control (CONTR)—pigs were fed a control diet and injected intraperitoneally with 0.9% sterile saline; (2) LPS-challenged control (LPS) —pigs were fed with the same control diet and challenged by intraperitioneal injection with E. coli LPS (E. coli serotype 055: B5; purity >99%; Sigma Chemical Inc., St Louis, MO, USA); (3) LPS + 0.5% Arg treatment—pigs were fed with a 0.5% Arg (
Blood and tissue sample collections
At 4 h post-challenge, blood samples were collected into 10-ml uncoated vacuum tubes (Becton Dickinson Vacutainer System, Franklin Lakes, NJ, USA) and centrifuged (3500 g for 10 min) to separate serum. Sera were stored at −80°C until analysis. Following blood sample collection, all pigs were humanely killed by intravenous injection of sodium pentobarbital (40 mg/kg BM). A midline laparotomy was performed. The abdomen was dissected and the liver was obtained with the cholecyst removal. The 0.5 cm3 segments removed from the liver and flushed with ice-cold PBS (pH 7.4). One fragment of these samples was fixed in fresh 4% paraformaldehyde/PBS and embedded in paraffin; the remaining portions were immediately frozen in liquid nitrogen and then stored at −80°C for further analysis.
The liver samples were fixed for 72 h, dehydrated with gradient concentration of alcohols (70–100%), cleared with xylene and embedded in paraffin. Cross-sections of segments approximately 5-µm thick with a microtome (American Optical Co., Scientific Instrument Div., Buffalo, NY, USA) for liver morphology, Kupffer cell and mast cell counting, and immunohistochemistry analysis.
The frozen liver samples were weighted and homogenized in ice-cold PBS-EDTA(0.05 M Na3PO4, 2.0 M NaCl, 2 × 10−3 M EDTA, pH 7.4) with a ratio at 1 : 10 (wt/vol) and then centrifuged at 900 g for 10 min at 4°C. Protein concentration of liver homogenates was measured by the method of Lowry 22 using a detergent-compatible protein assay (Bio-Rad Laboratories, Hercules, CA, USA) and BSA as standards.
Liver morphology, and Kupffer cell and mast cell counting
Tissue slides (5 µm) for liver morphology were deparaffinized and stained with hematoxylin and eosin (HE). The method of HE was according to Liu et al. 20 The resulting slides were arranged in a random sequence and were examined blindly using a light microscope with a computer-assisted morphometric system (BioScan Optimetric, BioScan Inc., Edmonds, WA, USA) by a pathologist for hepatic injury.
Tissue slides (5 µm) for Kupffer cells counting were deparaffinized and stained with HE and Perls’ Prussian blue according to Tan et al. 23 Tissue slides for mast cell counting were deparaffinized and stained with toluidine blue according to Mangia et al. 24 In each section, 10 fields were examined using a light microscope with a computer-assisted morphometric system. The number of mast cells or Kupffer cells was counted. The percentages of mast cells or Kupffer cells were calculated as the ratio of the number of mast cells or Kupffer cells to the total number of cells.
Measurement of serum enzyme activity
The activity of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (AKP) and lactate dehydrogenase (LDH) were measured by colorimetric method using commercial kits (Nanjing Jiancheng Biological Product, Nanjing, China) according to the manufacturer's recommendations. The activity of glutamyl transpeptidase (GGT) was measured by enzymatic rate method using a commercial kit (Shanghai Fenghui Medical Technology Company, Shanghai, China).
Measurement of TNF-α
The TNF-α level of liver homogenates was assayed using a commercially-available porcine ELISA kit (R&D Systems, Inc., Minneapolis, MN, USA). The minimum detectable dose was 3.7 ng/l. The intra- and inter-assay coefficients of variance (CVs) were 4.9% and 8.9% respectively. The TNF-α level in liver homogenates was expressed as pg/mg protein.
Liver antioxidant enzymes, malondialdehyde, reduced glutathione and oxidized glutathione
The lipid peroxidation product malondialdehyde (MDA) of liver homogenates was measured by the thiobarbituric acid reaction. 25 Superoxide dismutase (SOD) activity was evaluated by inhibition of nitroblue tetrazolium reduction by superoxide anion generated by the xanthine/xanthine oxidase system. 25 Reduced glutathione (GSH) and oxidized glutathione (GSSG) were detected by 5,5'-dithio bis-(2-nitrobenzoic acid) reaction. 26 Catalase (CAT) activity was determined by visible spectrophotometry; 26 glutathione peroxidase (GSH-px) activity was determined by colorimetric method; 26 MDA, SOD, GSH, GSSG, CAT and GSH-px were determined by commercially-available kits (Nanjing Jiancheng Biological Product) according to the manufacturer's recommendations.
Immunohistochemical staining for detection of iNOS, TLR4, and NF-κB
Tissue slides (5 µm) were deparaffinized, rehydrated and hydrated to buffer (PBS, pH 7.4). The slides were treated with 3% H2O2 in distilled water for 10 min to quench endogenous peroxidase activity and heated up in 10 m
Statistical analysis
All data were analyzed by ANOVA using the general linear model procedures of SAS (SAS Inc., Cary, NC, USA) appropriate for randomized complete block design. LPS pigs were compared by pre-planned contrasts with either CONTR pigs to determine the effect of LPS challenge or to LPS + 0.5% pigs to determine the effect of Arg supplementation within challenged pigs. All results were expressed as means ± SD. Differences were considered as significant when P < 0.05.
Results
Liver morphology
Regarding the histopathological structure of the liver, no obvious changes were found in the livers of control pigs (Figure 1A). However, in the LPS group, morphological changes associated with liver injury, such as hepatocyte caryolysis, karyopycnosis, fibroblast proliferation and hepatic cell cords arrangement in disorder, were observed (Figure 1B). Compared with the LPS group, liver injury was significantly attenuated in the LPS + 0.5% Arg group (Figure 1C).
Photomicrographs of liver sections stained with hematoxylin and eosin. (A) CONTR group (pigs fed a control diet and injected with sterile saline). No obvious changes were found. (B) LPS group (pigs fed the same control diet and challenged with E. coli LPS). Significant morphological changes associated with liver injury, such as hepatocyte caryolysis (a), karyopycnosis (b), fibroblast proliferation (c) and hepatic cell cords arrangement in disorder were observed. (C) LPS + 0.5% Arg group (pigs fed a 0.5% Arg diet and challenged with LPS). Liver injury was significantly attenuated. Original magnifications 400×. Scale bars = 125 µm.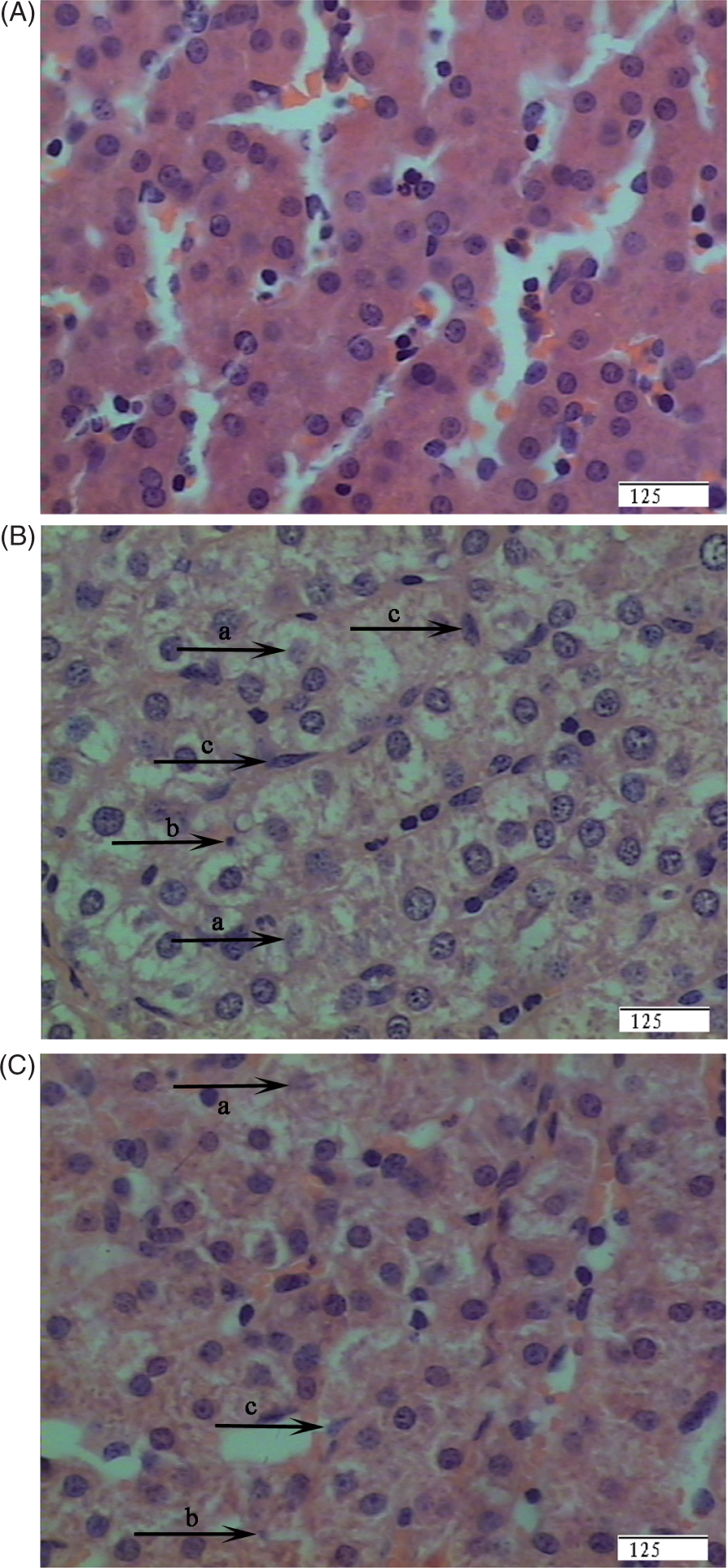
Serum enzyme activity
Effect of Arg supplementation on serum enzyme activity of weaned pigs after 4 h E. coli LPS challenge.
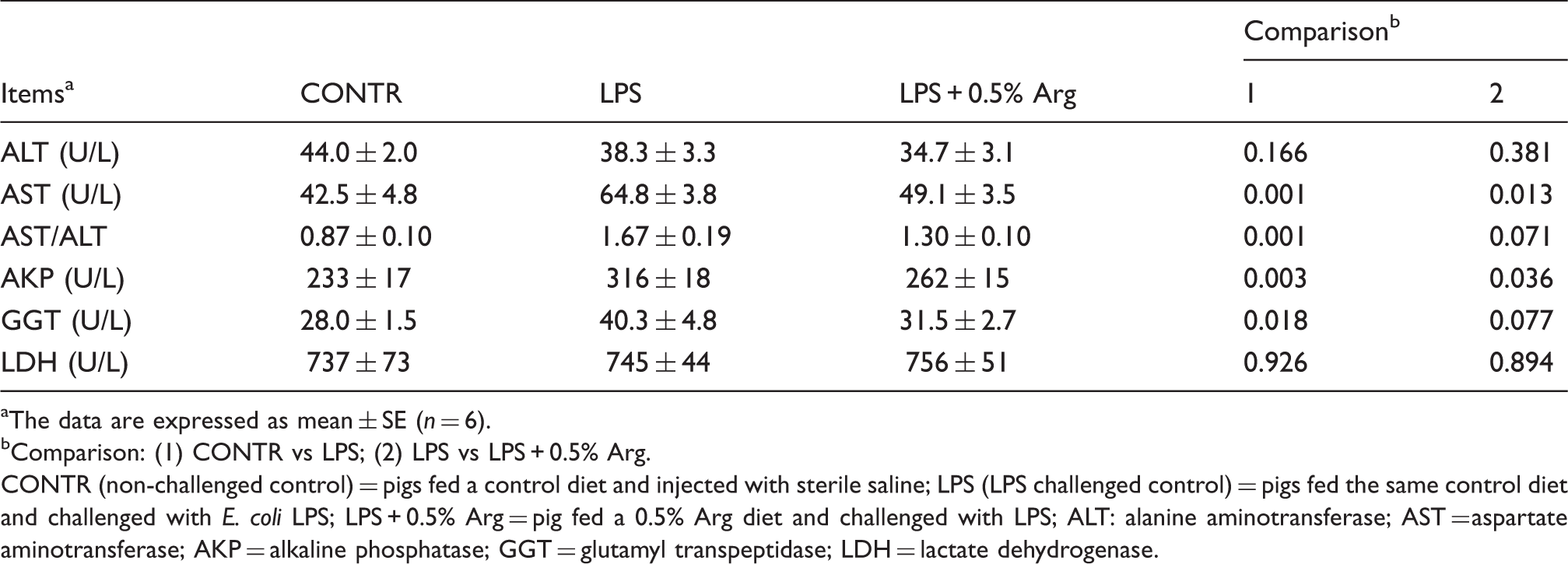
The data are expressed as mean ± SE (n = 6).
Comparison: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg.
CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg = pig fed a 0.5% Arg diet and challenged with LPS; ALT: alanine aminotransferase; AST = aspartate aminotransferase; AKP = alkaline phosphatase; GGT = glutamyl transpeptidase; LDH = lactate dehydrogenase.
Liver TNF-α level and percentage of iNOS-positive cells
Effect of Arg supplementation on liver TNF-α content and iNOS-positive cell percentage of weaned pigs after 4 h E. coli LPS challenge.
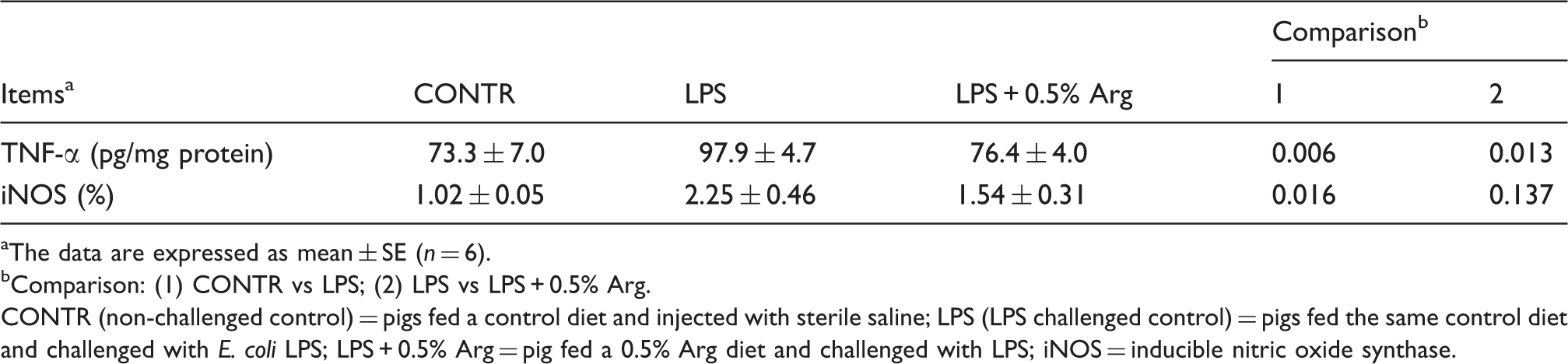
The data are expressed as mean ± SE (n = 6).
Comparison: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg.
CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg = pig fed a 0.5% Arg diet and challenged with LPS; iNOS = inducible nitric oxide synthase.
Mast cell and Kupffer cell
Effect of Arg supplementation on liver mast cells and Kupffer cell count of weaned pigs 4 h after E. coli LPS challenge.
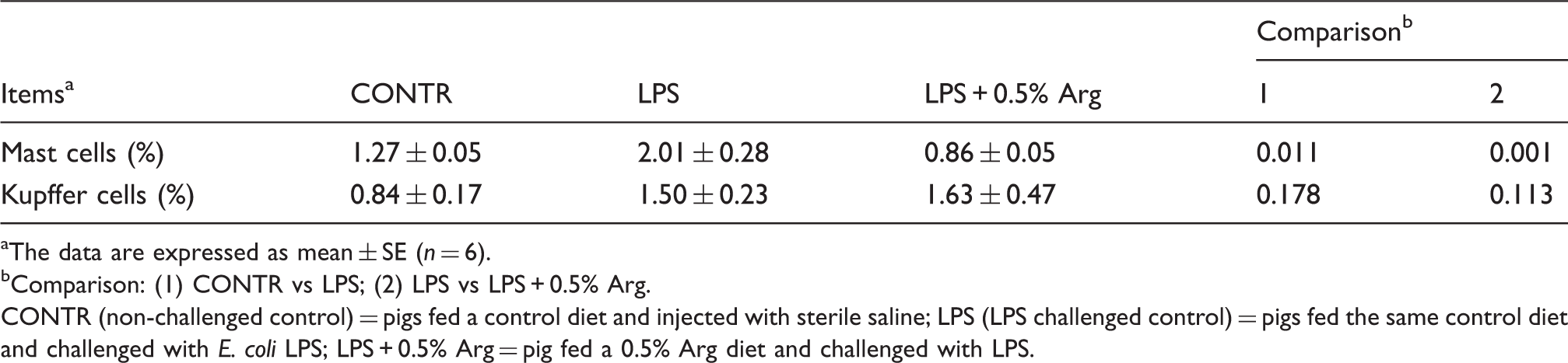
The data are expressed as mean ± SE (n = 6).
Comparison: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg.
CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg = pig fed a 0.5% Arg diet and challenged with LPS.
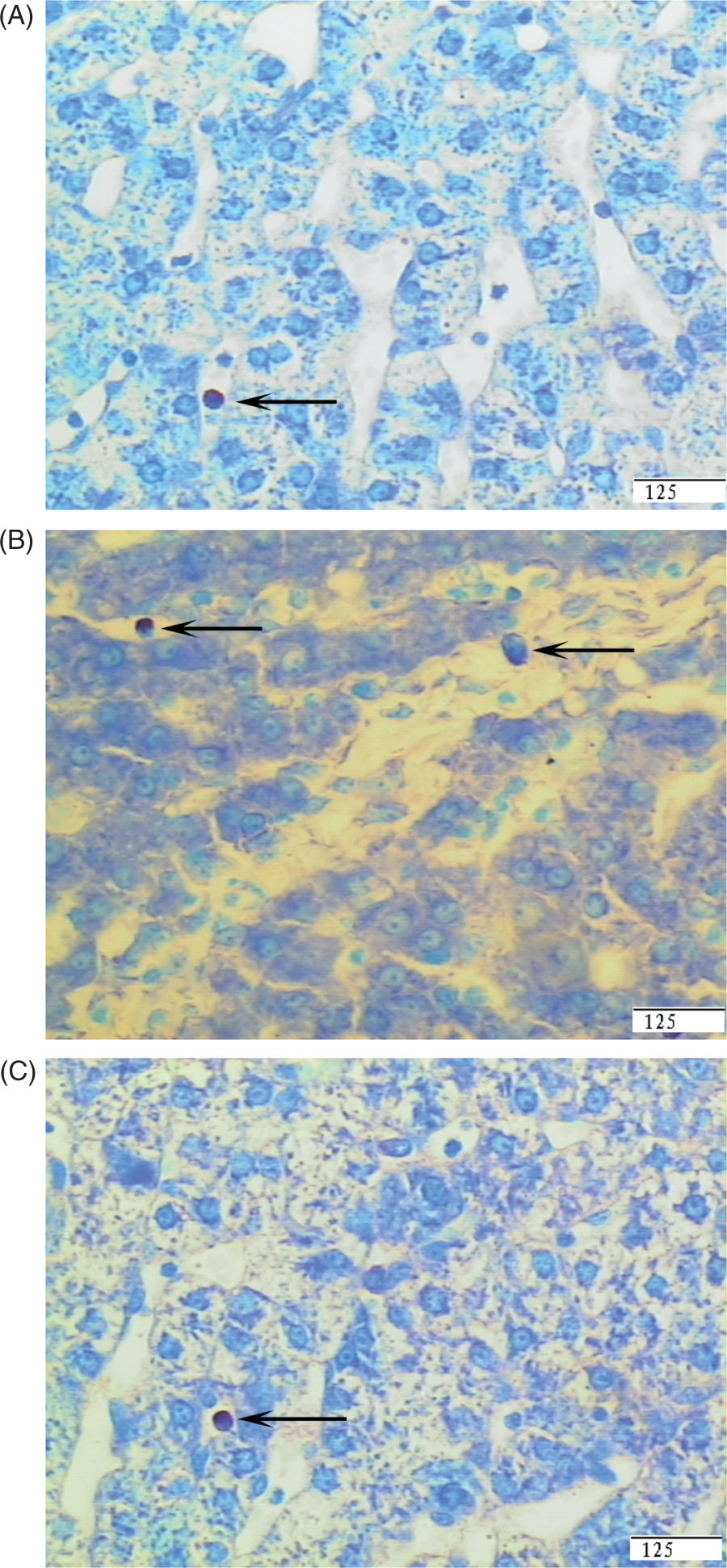
Representative photomicrographs showing mast cells stained with toluidine blue. Liver sections from the CONTR group (pigs fed a control diet and injected with sterile saline) (A), LPS group (pigs fed the same control diet and challenged with E. coli LPS) (B) or LPS + 0.5% Arg group (pigs fed a 0.5% Arg diet and challenged with LPS) (C). An increase in mast cell number was observed in liver in the LPS group compared with the CONTR group. Arg supplementation decreased the mast cell number compared with the LPS group. Original magnifications 400×. Scale bars = 125 µm.
Liver antioxidant enzymes, GSH, GSSG and MDA
Effect of Arg supplementation on liver antioxidant enzymes, GSH, GSSG and MDA of weaned pigs 4 h after E. coli LPS challenge.
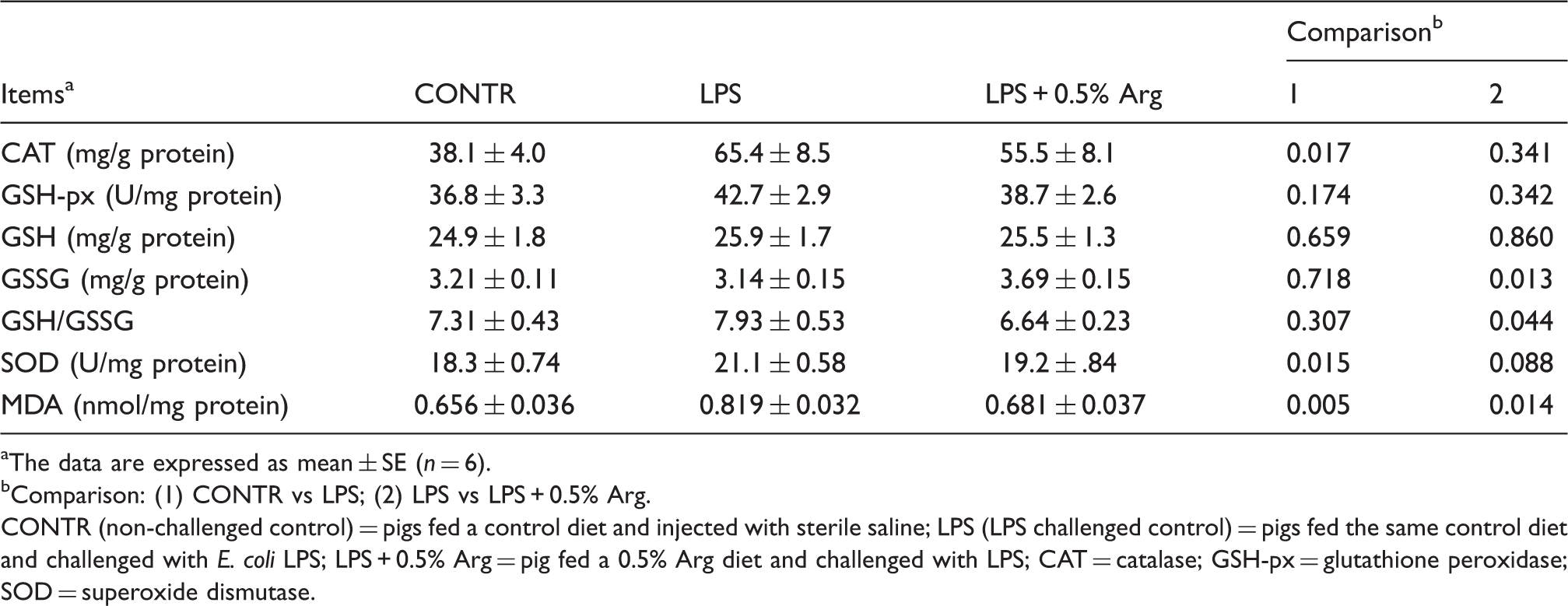
The data are expressed as mean ± SE (n = 6).
Comparison: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg.
CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg = pig fed a 0.5% Arg diet and challenged with LPS; CAT = catalase; GSH-px = glutathione peroxidase; SOD = superoxide dismutase.
NF-κB and TLR4-positive cell percentage
Effect of Arg supplementation on liver TLR4 and NF-κB-positive cell percentage of weaned pigs 4 h after E. coli LPS challenge.
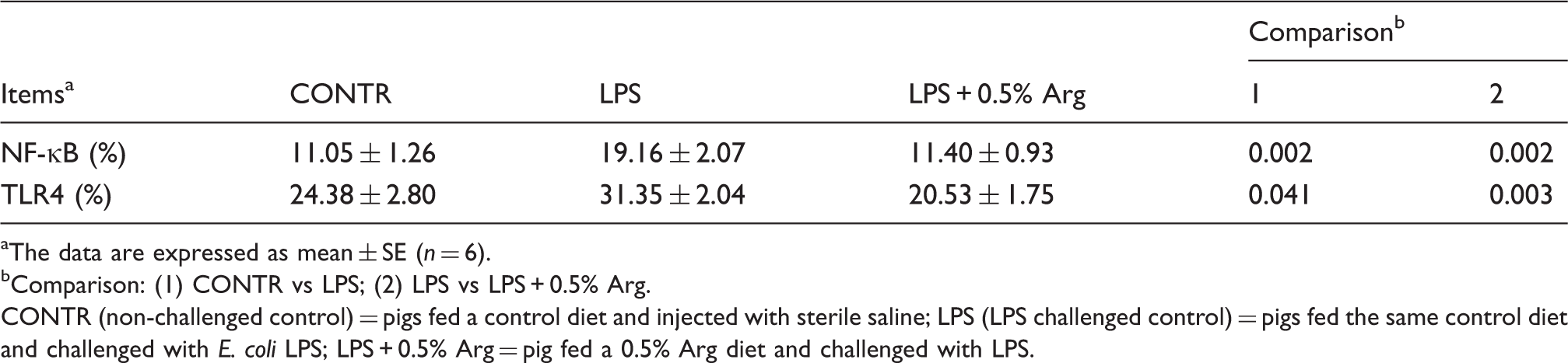
The data are expressed as mean ± SE (n = 6).
Comparison: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg.
CONTR (non-challenged control) = pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control) = pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg = pig fed a 0.5% Arg diet and challenged with LPS.
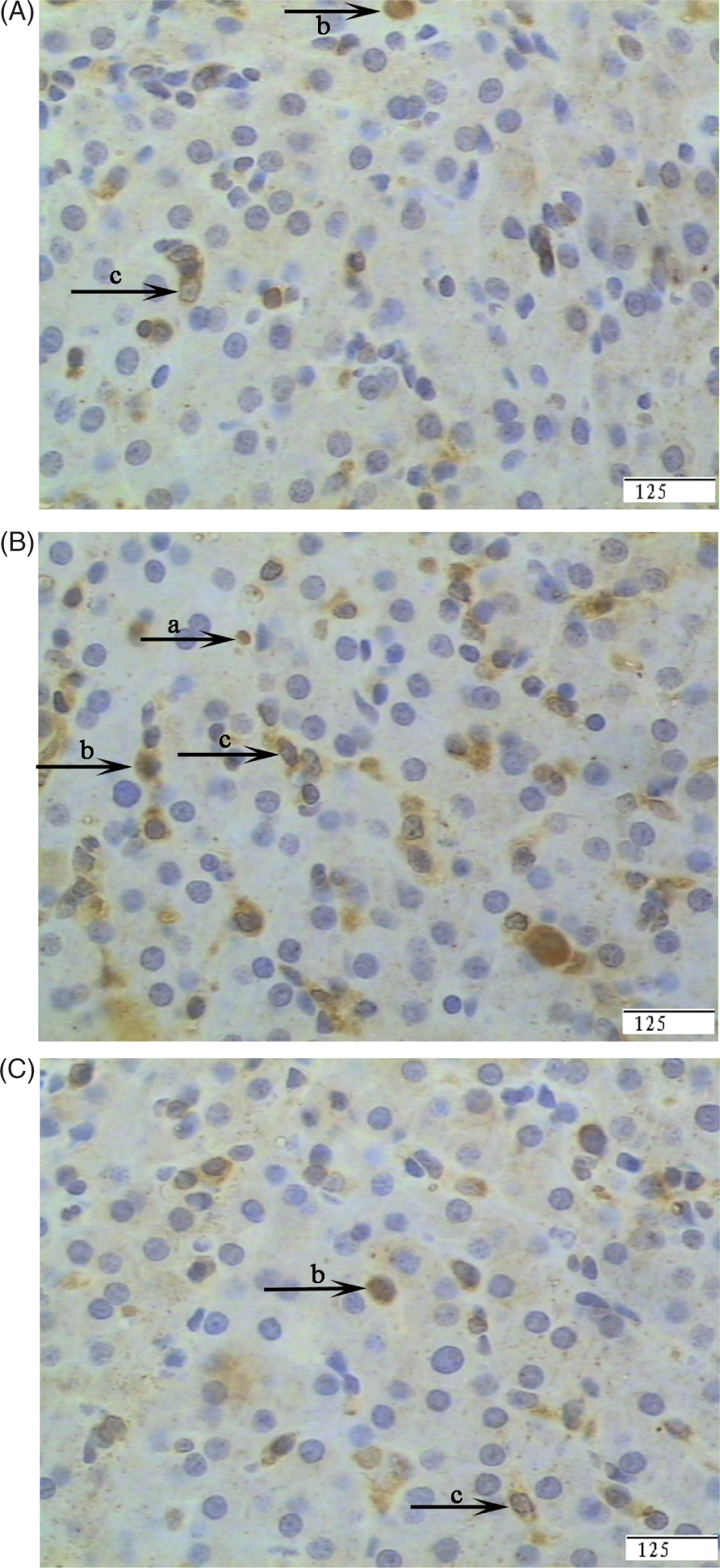
Representative photomicrographs showing immunostaining for NF-κB. Liver sections from the CONTR group (pigs fed a control diet and injected with sterile saline) (A), LPS group (pigs fed the same control diet and challenged with E. coli LPS) (B) or LPS + 0.5% Arg group (pigs fed a 0.5% Arg diet and challenged with LPS) (C). Cells that stained positive for NF-κB were observed mainly in immune cells (a), hepatocytes (b) and fibroblasts (c). The number of NF-κB-positive cells was increased in the LPS group compared with the CONTR group. Arg supplementation decreased positive cell percentage of NF-κB compared with the LPS group. Original magnifications 400×. Scale bars = 125 µm.
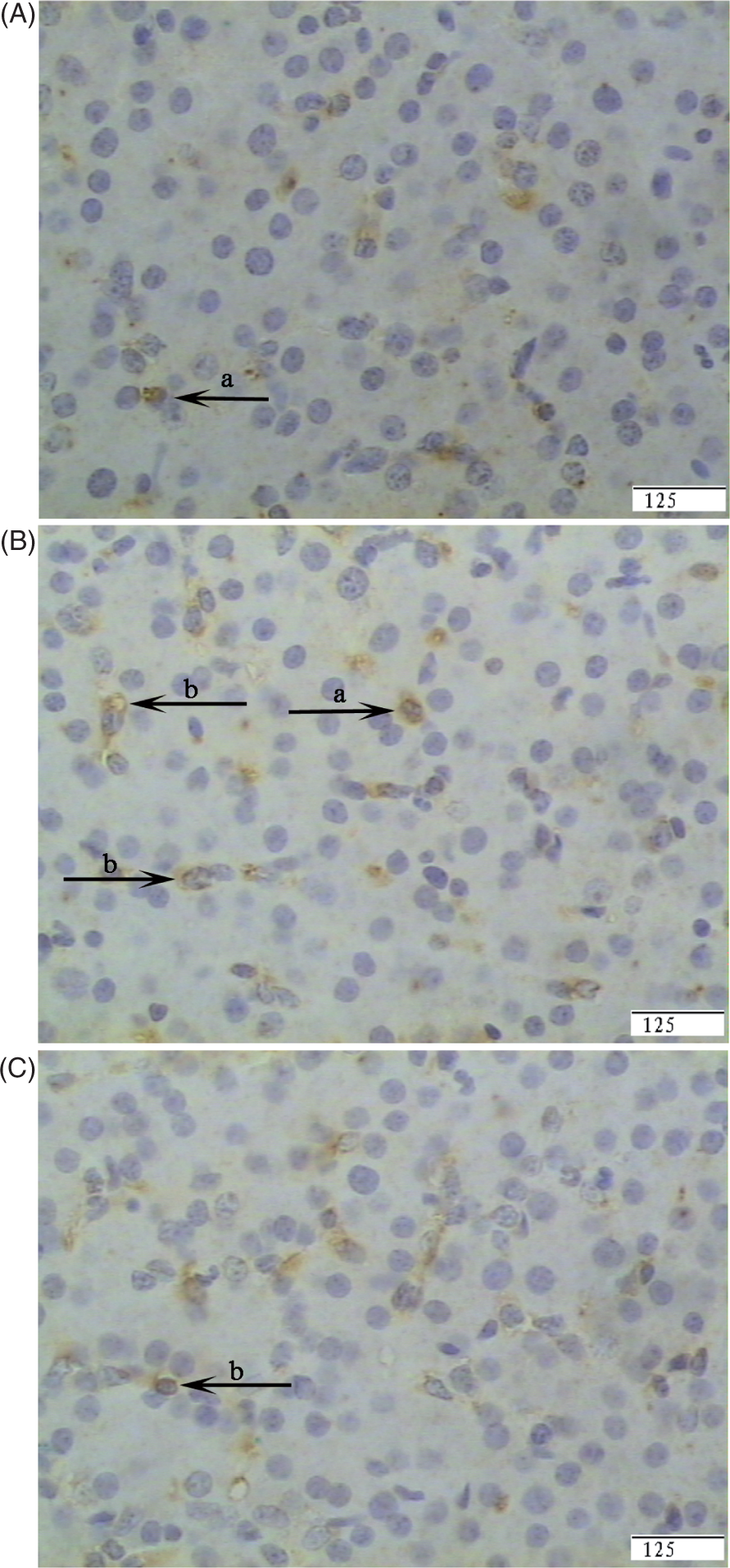
Representative photomicrographs showing immunostaining for TLR4. Liver sections from the CONTR group (pigs fed a control diet and injected with sterile saline) (A), LPS group (pigs fed the same control diet and challenged with E. coli LPS) (B) or the LPS + 0.5% Arg group (pigs fed a 0.5% Arg diet and challenged with LPS) (C). Cells that stained positive for TLR4 were observed mainly in immune cells (a) and to a lesser extent in hepatocytes (c). The number of TLR4-positive cells was increased in the LPS group compared with the CONTR group. Arg supplementation decreased the positive cell percentages of TLR4 compared with the LPS group. Original magnifications 400×. Scale bars = 125 µm.
Discussion
To evaluate whether Arg supplementation could alleviate liver injury through an anti-inflammatory role in weaned pigs, we took advantage of a model for inducing liver injury in pigs by injecting E. coli LPS according to the model of Masaki et al. 5 LPS is the prototypical example of endotoxin, found in the outer membrane of various Gram-negative bacteria, which is an important cause of disease. Multiple lines of evidence show that LPS is a co-factor in liver injury. 27 LPS-induced liver injury has been used commonly as an experimental model in which to analyze the mechanism of endotoxin-induced acute liver injury. 5 A number of inflammatory cytokines, including TNF-α, play pivotal roles in the development of LPS-induced liver injury. 5
Our previous study showed that feeding 0.5% Arg supplementation for 18 d alleviated the mass loss in weaned pigs after LPS challenge, which indicates the importance of exogenous Arg supply in weaning pigs under stress, infection and diseases. 20 In this study, intraperitoneal injection of LPS caused morphologic change in liver, including hepatocyte caryolysis, karyopycnosis and fibroblast proliferation, which is consistent with the report of Xu et al. 28 Compared with LPS pigs, 0.5% Arg supplementation attenuated the liver injury caused by LPS. In agreement with our study, Giovanardi et al. 14 reported that pharmacological preconditioning using intraportal infusion of Arg attenuated the impairment of liver cell morphology and mitochondrial ultrastructure in pigs subjected to hepatic ischemia reperfusion. In addition, Chattopadhyay et al. 29 showed that Arg exerted a protective effect against necrosis and apoptosis caused by experimental ischemic and reperfusion in rat liver.
AST and ALT are hepatic intracellular enzymes. Serum elevation in both of these enzymes reflects leakage from injured hepatocytes and is considered to be a sensitive indicator of liver injury. 30 The high AST/ALT ratio also indicates liver damage. 30 In addition, serum AKP and GGT activities have also been described as valuable parameters of hepatic injury and function. 31 In our experiment, LPS increased the activities of AST, AKP and GGT, and AST/ALT ratio, indicating that LPS induced liver function injury. Interestingly, LPS challenge numerically decreased the activity of serum ALT. In agreement with our findings, Sano et al. 32 reported that pretreatment with 100 µg/kg LPS significantly reduced serum ALT level. In the present study, Arg alleviated the increase of serum AST and AKP activity induced by LPS challenge and tended to decrease serum GGT activity and the AST/ALT ratio, indicating that Arg was effective in improving liver function. In agreement with our findings, Giovanardi et al. 14 reported that Arg significantly suppressed the increase of the serum ALT and AST levels in an ischemia-reperfusion experiment in pigs. Muriel and González 15 showed that treatment with Arg partially prevented the increase in the two enzymes activities induced by 3 d-biliary obstruction in rats.
Induction of iNOS may be responsible for tissue injury via the formation of nitric oxide-dependent nitrating species, such as peroxynitrite. 33 In the current study, we hypothesized that Arg exerted its protective effect on liver morphology and function through attenuating the liver inflammatory response. Consistent with liver morphology and function impairment caused by the LPS challenge, increased liver iNOS expression and TNF-α level were observed. Over-production of these pro-inflammatory parameters can have a negative influence on liver morphology and function.5,34 In the present study, 0.5% Arg supplementation attenuated the inflammatory response by decreasing the TNF-α level in liver compared with LPS pigs. In agreement with our findings, Zhang et al. 35 reported that Arg treatment reduced TNF-α concentration in the liver of acute hemorrhagic, necrotizing pancreatitis rats. In addition, Arg exerted an inhibitory effect on pro-inflammatory cytokine production in many other stress models.20,36,37 Arg down-regulated pro-inflammatory cytokine expression or production in spleen, thymus, lung and liver of burned rats, 37 in serum and lung of immature rats after gut ischemia-reperfusion 36 and in peritoneal lavage fluid of septic rats, 38 thus preventing the development of inflammation. Our previous study also showed that Arg down-regulated the expression of intestinal cytokines, thus alleviating the intestinal impairment caused by LPS challenge in weaned pigs. 20 In our current study, it is possible that feeding pigs dietary Arg attenuated liver morphology and function impairment partially by suppressing pro-inflammatory cytokine production.
Mast cells are strategically placed close to blood vessels and nerves in tissues. Mast cells are the major cell type to store TNF-α and are thus primed to trigger TNF-mediated inflammatory responses most rapidly.
39
Therefore, mast cells play a pivotal role in allergic and inflammatory responses.
40
In our study, 0.5% Arg supplementation attenuated the increase in the number of mast cells and also alleviated the increase in TNF-α level caused by LPS, which indicates that Arg inhibited the mast cells in releasing TNF-α, thus alleviating liver injury.
It is well known that oxidative stress is one of major factors contributing to tissue injury.25,26 SOD, CAT and GSH-px are regarded as the first line of the antioxidant enzyme system against ROS generated during oxidative stress; MDA is an end-product of lipid peroxidation resulting from reactions with free radicals.
25
In the current study, consistent with attenuated injury of liver morphology and function caused by the LPS challenge, a decrease of liver MDA content was also observed after 0.5% Arg supplementation. In agreement with our observations, Calabrese et al.
43
reported that a significant decrease of MDA content in Arg-treated pigs than the control group in ischemia-reperfusion injury. Lin et al.
26
showed that
To explore the molecular mechanism by which Arg attenuated liver inflammatory response and oxidative stress, we examined the role of the TLR4 signaling pathway. TLRs play a central role in the activation of the innate system as a result of the recognition of bacterial pathogen-associated molecular patterns (especially LPS), which triggers the activation of different intracellular signaling cascades, such as activation of NF-κB and the production of ROS. 17 Activated NF-κB stimulates the synthesis of pro-inflammatory cytokines, including TNF-α, IL-1β and IL-6. 17 Consequently, ROS and pro-inflammatory cytokines are closely associated with tissue injury. 17
In the present study, we have observed that TLR4 and NF-κB expression were down-regulated and the release of TNF-α was decreased in Arg-supplemented pigs after LPS challenge. Therefore, it is possible that the protective effects of Arg on liver injury were associated with decreasing the release of liver pro-inflammatory cytokines through inhibiting TLR4 and NF-κB signaling. Similarly, Tan et al. 46 reported that addition of Arg to culture medium reduced the relative levels of TLR4 and phosphorylated levels of NF-κB in LPS-treated IPEC-1 cells. In addition, in a rat model of LPS-induced injury, Arg inhibited the NF-κB DNA binding and stabilized the I-κB complex, both of which may account for the decrease in pro-inflammatory cytokines. 47 Li et al. 48 also showed that administration of Arg protected lung from LPS-induced injury by inhibiting NF-κB activation and subsequently inhibiting the NF-κB-mediated release of inflammatory factors.
Lin et al.
49
reported that ROS generation was mediated through the TLR4/NADPH oxidative pathway. In our study, we found that TLR4 expression was down-regulated and the level of MDA (the end-product of lipid peroxidation resulting from reactions with free radicals)50,51 was decreased in Arg-supplemented pigs after LPS challenge. Zeng et al.
52
reported that
In conclusion, dietary supplementation of 0.5% Arg exerts beneficial effects in alleviating liver injury of LPS-challenged pigs. It is possible that the protective effects of Arg on the liver are associated with decreasing the release of liver pro-inflammatory cytokines and free radicals through the inhibition of TLR4 signaling.
Footnotes
Funding
The authors express their gratitude to the Program for New Century Excellent Talents in University (NCET-10-0158), the National Natural Science Foundation of China (31172222, 30972109), the Key Project of Chinese Ministry of Education (209082), the Project of Natural Science Foundation of Hubei Province (2010CDA050, 2009CDB006) and Wuhan Science and Technology Program (201171034320) for financial support.
Conflict of interest
The authors have no conflicts of interest to declare.
