Abstract
This study evaluated whether fish oil exerted a hepatoprotective effect in a LPS-induced liver injury model via regulation of TLR4 and nucleotide-binding oligomerization domain protein (NOD) signaling pathways. Twenty-four piglets were used in a 2 × 2 factorial design, and the main factors included diet (5% corn oil or 5% fish oil) and immunological challenge (LPS or saline). Fish oil resulted in enrichment of eicosapentaenoic acid, docosahexaenoic acid and total (n-3) polyunsaturated fatty acids in liver. Less severe liver injury was observed in pigs fed fish oil, as evidenced by improved serum biochemical parameters and less severe histological liver damage. In addition, higher expression of liver tight junction proteins, and lower hepatocyte proliferation and higher hepatocyte apoptosis were observed in pigs fed fish oil. The improved liver integrity in pigs fed fish oil was concurrent with reduced hepatic mRNA expression of TLR4, myeloid differentiation factor 88, IL-1 receptor-associated kinase 1 and TNF-α receptor-associated factor 6, and NOD1, NOD2 and receptor-interacting serine/threonine-protein kinase 2, as well as reduced hepatic protein expression of NF-κB p65, leading to reduced hepatic pro-inflammatory mediators. These results indicate that fish oil improves liver integrity partially via inhibition of TLR4 and NOD signaling pathways under an inflammatory condition.
Keywords
Introduction
The liver is the most important metabolic organ and the main detoxification site in the body. 1 However, numerous factors, such as bacterial and viral infection or inflammation, lead to activation of macrophages (Kupffer cells) with an increased production of pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6, which, consequently, leads to parenchymal liver damage and liver dysfunction, such as acute hepatitis. 2 Among these cytokines, TNF-α has been shown to be the terminal mediator of hepatic injury and organ failure in several hepatitis models. 2 Therefore, nutritional modulation of liver inflammatory response, especially the production of pro-inflammatory cytokines may play a beneficial role in attenuating the liver injury.
Growing evidence indicates that long chain n-3 polyunsaturated fatty acids (PUFAs), such as eicosapentaenoic acid [EPA, 20:5(n-3)] and docosahexaenoic acid [DHA, 22:6(n-3)], which are found mainly in fish oil, can reduce the inflammatory responses and exert beneficial effects on some inflammatory diseases, such as inflammatory bowel diseases, rheumatoid arthritis and asthma in animal models and clinical trials.3,4 The beneficial role of n-3 PUFAs may be associated with their inhibitory effects on the over-release of inflammatory mediators, such as pro-inflammatory cytokines. 3 In addition, n-3 PUFAs have been shown to possess hepatoprotective effects in several liver injury models, such as chemically-induced acute hepatitis, 2 cholestatic liver injury, 5 radiation-induced liver injury 6 and liver ischemia-reperfusion injury. 7 However, the mechanism(s) underlying the beneficial effect of n-3 PUFAs on liver remain to be elucidated.
Pattern recognition receptors, such as transmembrane TLRs and cytoplasmic nucleotide-binding oligomerization domain proteins (NODs), are important protein families that recognize pathogen associated molecular patterns (PAMPs), and regulate innate and adaptive immune responses.8,9 Among the TLR family, TLR4 is the best-characterized member; it is responsible for recognizing endotoxin (LPS) from Gram-negative bacteria.10,11 Among the NOD family, NOD1 and NOD2 are the most studied members; they possess the ability to connect with LPS and peptidoglycan, and to transduce a TLR-independent signal. 9 TLR4 has been investigated extensively in pigs,12,13 and molecular cloning and functional characterization of NOD1 and NOD2 have been performed.14,15 Interaction of PAMPs with TLRs or NODs triggers multiple downstream intracellular signals that lead to the activation of NF-κB, 8 which then induces activation of inflammatory genes, such as TNF-α, IL-1β and IL-6. 8 Consequently, the pro-inflammatory cytokines mediate the host’s defense against invading pathogens, but can also elicit collateral host tissue injury.
In the present study, Escherichia coli LPS, a potent endotoxin, was injected to build upon acute liver injury model following the model of Li et al., 1 Masaki et al. 16 and Schmöcker et al. 2 The LPS model is a well-established model for macrophage-dependent liver injury.2,16 When binding with the TLR4/MD2/CD14 receptor complex, LPS stimulates macrophages to release a variety of pro-inflammatory cytokines, which mediates the inflammatory response and induces tissue injury. In addition, we made use of a piglet model—a good animal model for studying human nutrition and physiology.17,18 Our aim was to evaluate whether dietary supplementation of fish oil (rich in n-3 PUFAs, such as EPA and DHA) could alleviate LPS-induced liver injury via down-regulation of TLR4 and NOD signaling pathways.
Materials and methods
Animal care and diets
Ingredient composition of diets (as fed basis).
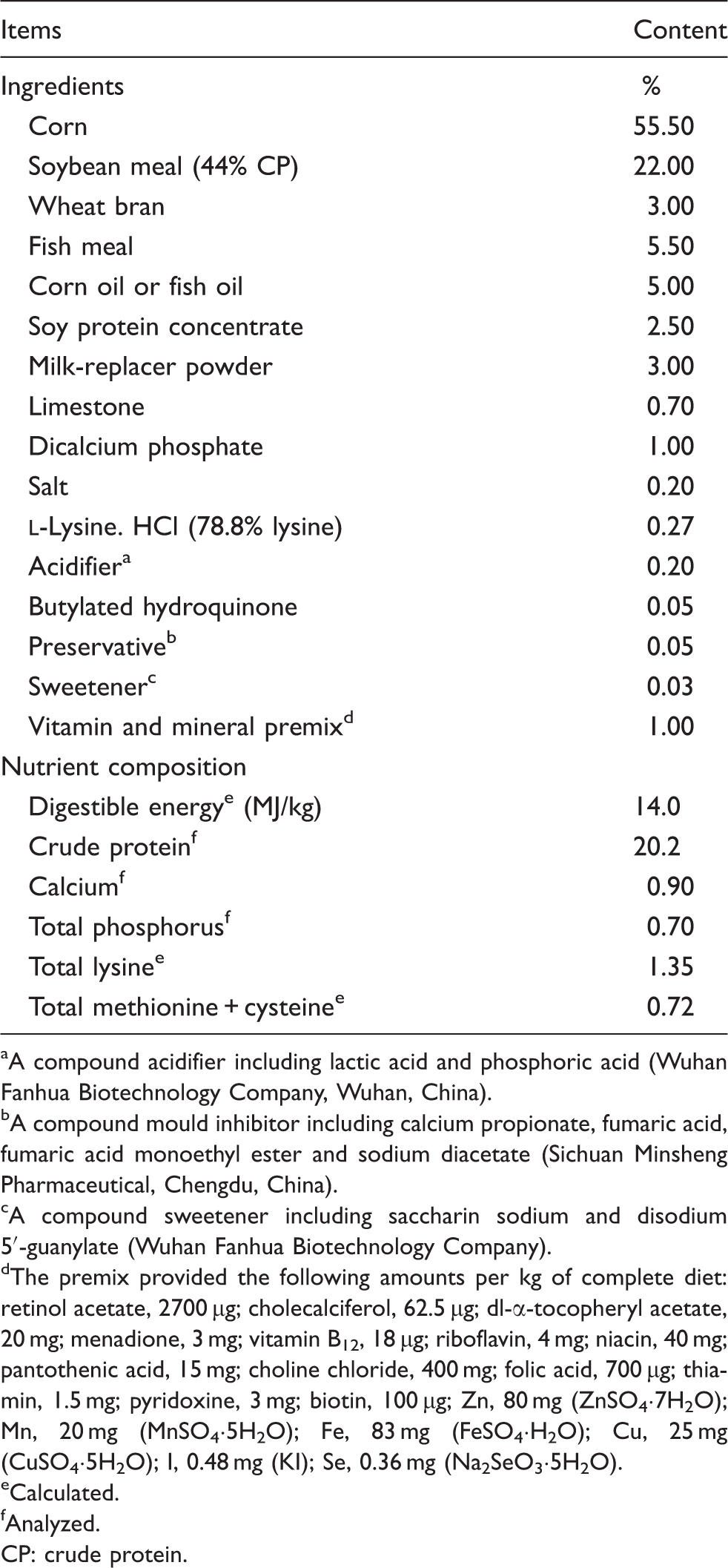
A compound acidifier including lactic acid and phosphoric acid (Wuhan Fanhua Biotechnology Company, Wuhan, China).
A compound mould inhibitor including calcium propionate, fumaric acid, fumaric acid monoethyl ester and sodium diacetate (Sichuan Minsheng Pharmaceutical, Chengdu, China).
A compound sweetener including saccharin sodium and disodium 5′-guanylate (Wuhan Fanhua Biotechnology Company).
The premix provided the following amounts per kg of complete diet: retinol acetate, 2700 µg; cholecalciferol, 62.5 µg; dl-α-tocopheryl acetate, 20 mg; menadione, 3 mg; vitamin B12, 18 µg; riboflavin, 4 mg; niacin, 40 mg; pantothenic acid, 15 mg; choline chloride, 400 mg; folic acid, 700 µg; thiamin, 1.5 mg; pyridoxine, 3 mg; biotin, 100 µg; Zn, 80 mg (ZnSO4·7H2O); Mn, 20 mg (MnSO4·5H2O); Fe, 83 mg (FeSO4·H2O); Cu, 25 mg (CuSO4·5H2O); I, 0.48 mg (KI); Se, 0.36 mg (Na2SeO3·5H2O).
Calculated.
Analyzed.
CP: crude protein.
Fatty acid composition of the 5% fish oil or 5% corn oil supplemented diets. a
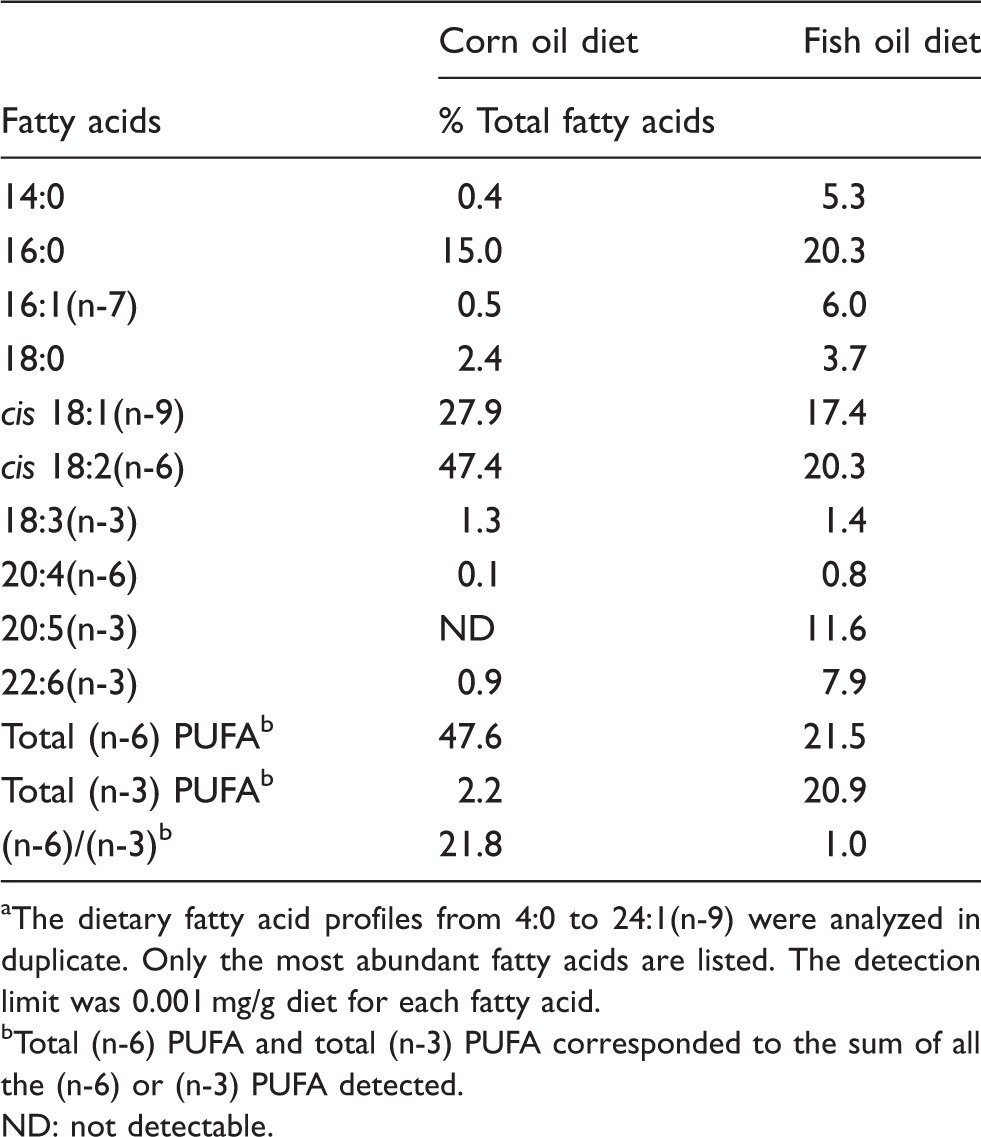
The dietary fatty acid profiles from 4:0 to 24:1(n-9) were analyzed in duplicate. Only the most abundant fatty acids are listed. The detection limit was 0.001 mg/g diet for each fatty acid.
Total (n-6) PUFA and total (n-3) PUFA corresponded to the sum of all the (n-6) or (n-3) PUFA detected.
ND: not detectable.
Experimental design
Pigs were fed fish oil or corn oil-supplemented diets for 21 d before LPS challenge. Before pigs were challenged with LPS, the experimental design was a randomized complete block. After pigs were challenged with LPS, the experiment became a 2 × 2 factorial design. The main factors consisted of diet (5% corn oil or 5% fish oil-supplemented diets) and immunological challenge (LPS or saline). On d 21, half of the pigs (n = 6) in each dietary treatment were injected i.p. with either E. coli LPS (E. coli serotype 055: B5; purity >99%; Sigma Chemical Inc., St. Louis, MO, USA) at 100 µg/kg body mass (BM) or the same amount of 0.9% (wt/vol) NaCl solution. The dose of LPS was chosen in accordance with our previous study. 1
Blood and liver sample collections
The procedures of blood and liver sample collections were the same as Li et al. 1 At 4 h following injection with LPS or saline, blood samples were collected into uncoated vacuum tubes and centrifuged (3500 g for 10 min) to collect serum. Plasma was stored at −80℃ until analysis of serum biochemical parameters. Following blood collection, all pigs were killed humanely by i.m. injection of sodium pentobarbital (80 mg/kg BM) and then the liver samples (0.5-cm 3 segments) were collected. One fragment of liver samples was stored in fresh 4% paraformaldehyde/PBS for at least 24 h and then embedded in paraffin; the remaining portions were frozen immediately in liquid nitrogen and then stored at −80℃ for further analysis. The time point of 4 h following LPS challenge was chosen according to our previous study, which showed that LPS caused acute liver injury and increased production of hepatic pro-inflammatory cytokines at 4 h after injection of LPS. 1
The frozen liver samples were weighed, homogenized in ice-cold PBS-EDTA (0.05 M Na3PO4, 2.0 M NaCl, 2 × 10−3 M EDTA, pH 7.4) with a 1:10 (wt/vol) ratio and then centrifuged (3500 g for 10 min at 4℃) to collect the supernatant. The supernatant was used for TNF-α and prostaglandin E2 (PGE2) analysis.
Hepatic fatty acid composition
Hepatic fatty acid profiles were determined by gas chromatography in accordance with the method of Nieto et al. 22
Serum biochemical parameters
The activities of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST) and alkaline phosphatase (AKP) were analyzed by the colorimetric method using commercial kits (Nanjing Jiancheng Biological Product, Nanjing, China). The activity of serum glutamyl transpeptidase (GGT) was analyzed by enzymatic rate method using a commercial kit (Shanghai Fenghui Medical Technology Company, Shanghai, China). The concentration of serum total bilirubin (TBIL) was measured using commercial kits (Nanjing Jiancheng Biological Product). All parameters were analyzed according to the protocols of the manufacturers.
Liver morphology
Tissue slides (5 µm) for liver morphology were deparaffinized and stained with hematoxylin and eosin. Histological analysis was performed in a blinded manner using a light microscope with a computer-assisted morphometric system (BioScan Optimetric, BioScan, Edmonds, WA, USA) by an experienced pathologist.
Hepatic TNF-α and PGE2 concentrations
The TNF-α concentration in liver supernatant was determined using a commercially available porcine ELISA kit (R&D Systems, Minneapolis, MN, USA). The PGE2 concentration in liver supernatant was analyzed using a commercially available 125I RIA kit (Beijing Sino-uk Institute of Biological Technology, Beijing, China). The results of TNF-α and PGE2 were expressed as pg/mg protein.
Protein expression analysis by Western blot
The methods for analysis of heat shock protein 70 (HSP70) and NF-κB p65 proteins by Western blot was the same as described in Hou et al.23,24 Briefly, the liver samples were homogenized in lysis buffer and centrifuged to collect the supernatants for Western blot. Hepatic proteins were separated on a polyacrylamide gel and transferred onto polyvinylidene difluoride membranes. Immunoblots were blocked for at least 60 min with 3% BSA in TBS/Tween-20 buffer. The membranes were incubated overnight (12–16 h) at 4℃ with primary Abs and then with the secondary Ab for 120 min at room temperature (21–25℃). Specific primary Abs included mouse anti-HSP70 (1:2000; #SPA-810, Stressgen, Farmingdale, NY, USA), mouse anti-NFκB p65 (1:1000; #6956, Cell Signaling, Danvers, MA, USA) or mouse anti-β-actin (1:10000; #A2228, Sigma Aldrich, St. Louis, MO, USA). The secondary Ab was goat anti-mouse IgG-HRP (1:5000; #ANT019, Antgene Biotech, Wuhan, China). Blots were developed using an enhanced chemiluminescence Western blotting kit (Amersham Biosciences, Uppsala, Sweden), visualized using a Gene Genome bioimaging system and analyzed using GeneTools software (Syngene, Frederick, MD, USA). The relative abundance of each target protein was expressed as target protein/β-actin protein ratio.
mRNA expression analysis by real-time PCR
Total RNA was isolated from liver samples using TRIzol reagent [TaKaRa Biotechnology (Dalian), Dalian, China] according to the the manufacturer’s instructions. Total RNA was quantified by determining the absorbance at 260 nm using a Nano-Drop 2000 Spectrophotometer (Thermo Scientific, Wilmington, DE, USA). RNA purity was assessed by determining the ratio of the absorbance at 260 and 280 nm, and the integrity was verified using agarose gel electrophoresis.
Primer sequences used for real-time PCR.

Statistical analysis
All data were analyzed by ANOVA using the GLM procedures of SAS (SAS, Cary, NC, USA). The statistical model included the main effects of immunological challenge (saline or LPS) and dietary treatment (5% corn oil or 5% fish oil), and their interactions. Data were expressed as means ± pooled SEM. P ≤ 0.05 was considered as statistically significant and 0.05 < P ≤ 0.10 as trends. If a significant interaction or a trend for interaction occurred, post hoc testing was performed using Bonferroni’s multiple comparison tests. The Bonferroni correction is used to reduce the possibility of making a type I error when multiple pair wise tests are performed on a single data set. To perform a Bonferroni correction, the critical P-value (α) is divided by the number of comparisons being made to get the new critical P-value. The statistical power of the study is then calculated based on this modified P-value. In our current study, the modified P-value was 0.0125 (i.e. 0.05/4 = 0.0125).
Results
Liver fatty acid composition
Effects of fish oil or corn oil supplementation on selected fatty acids in liver after 4 h E. coli LPS challenge in weaned pigs.
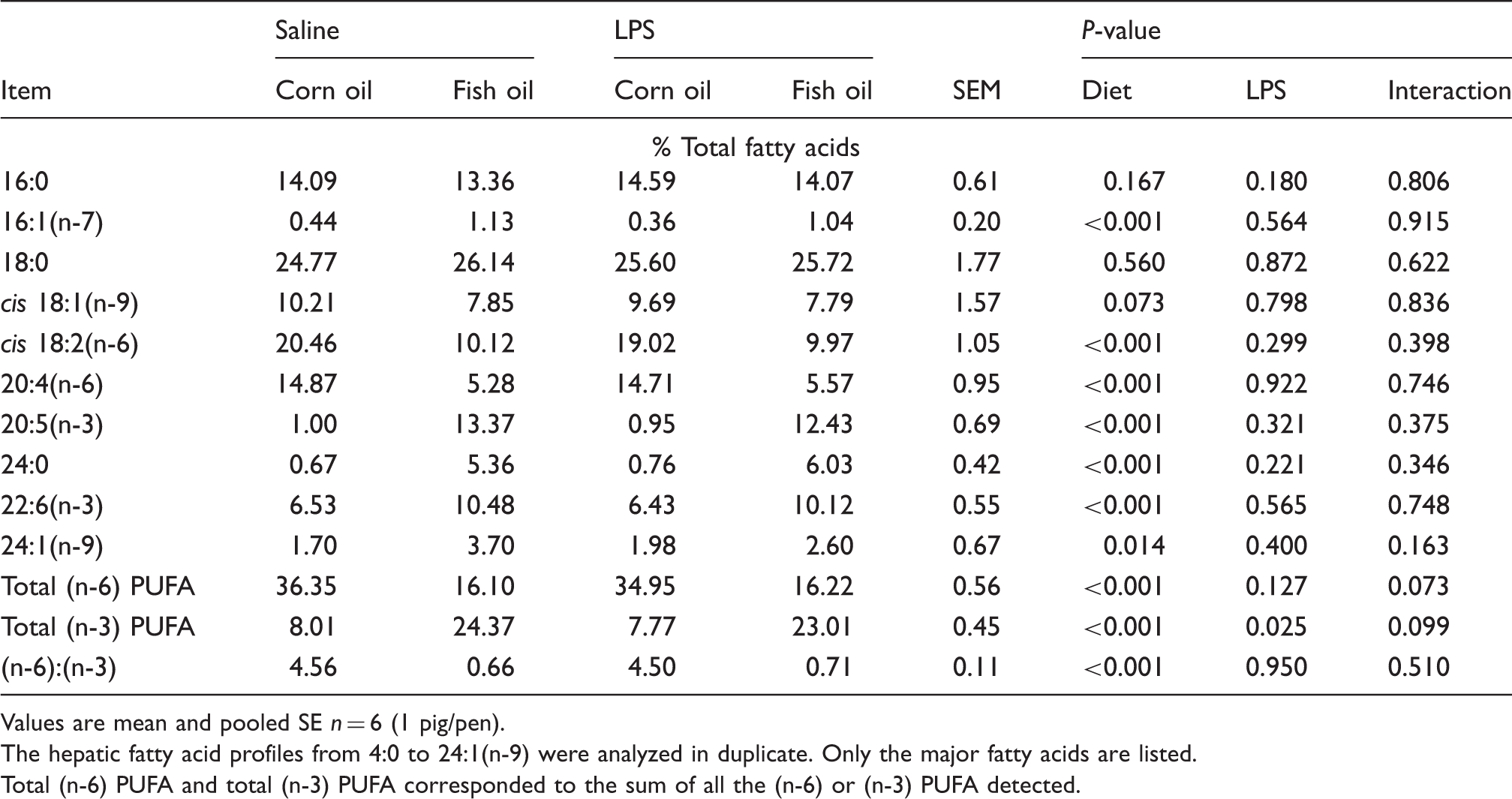
Values are mean and pooled SE n = 6 (1 pig/pen).
The hepatic fatty acid profiles from 4:0 to 24:1(n-9) were analyzed in duplicate. Only the major fatty acids are listed.
Total (n-6) PUFA and total (n-3) PUFA corresponded to the sum of all the (n-6) or (n-3) PUFA detected.
Liver morphology
Regarding hepatic morphology, no obvious morphologic changes were observed in the saline-injected pigs fed corn oil (Figure 1A) or fish oil (Figure 1B) diets. However, morphologic changes associated with hepatic injury, such as heptatocyte caryolysis, karyopycnosis, inflammatory cell infiltration, hyperaemia in hepatic sinusoids and hepatic cell cords arrangement in disorder, were observed in LPS-challenged pigs fed corn oil (Figure 1C). Compared with LPS-challenged pigs fed corn oil, liver injury was significantly attenuated in LPS challenged pigs fed fish oil (Figure 1D).
Effects of fish oil or corn oil supplementation on liver morphology after 4 h E. coli LPS challenge in weaned pigs. The representative photomicrographs of liver sections stained with hematoxylin and eosin are shown. (A) Piglets fed the corn oil diet and injected with saline. (B) Piglets fed the fish oil diet and injected with saline. No obvious morphologic changes were found. (C) Piglets fed the corn oil diet and challenged with LPS. Significant morphologic changes associated with liver injury, such as heptatocyte caryolysis (a), karyopycnosis (b), inflammatory cell infiltration (c), hyperaemia in hepatic sinusoids (d) and hepatic cell cords arrangement in disorder, were observed. (D) Piglets fed the fish oil diet and challenged with LPS. Liver injury was significantly attenuated. Original magnifications 400 × . Scale bars = 125 µm.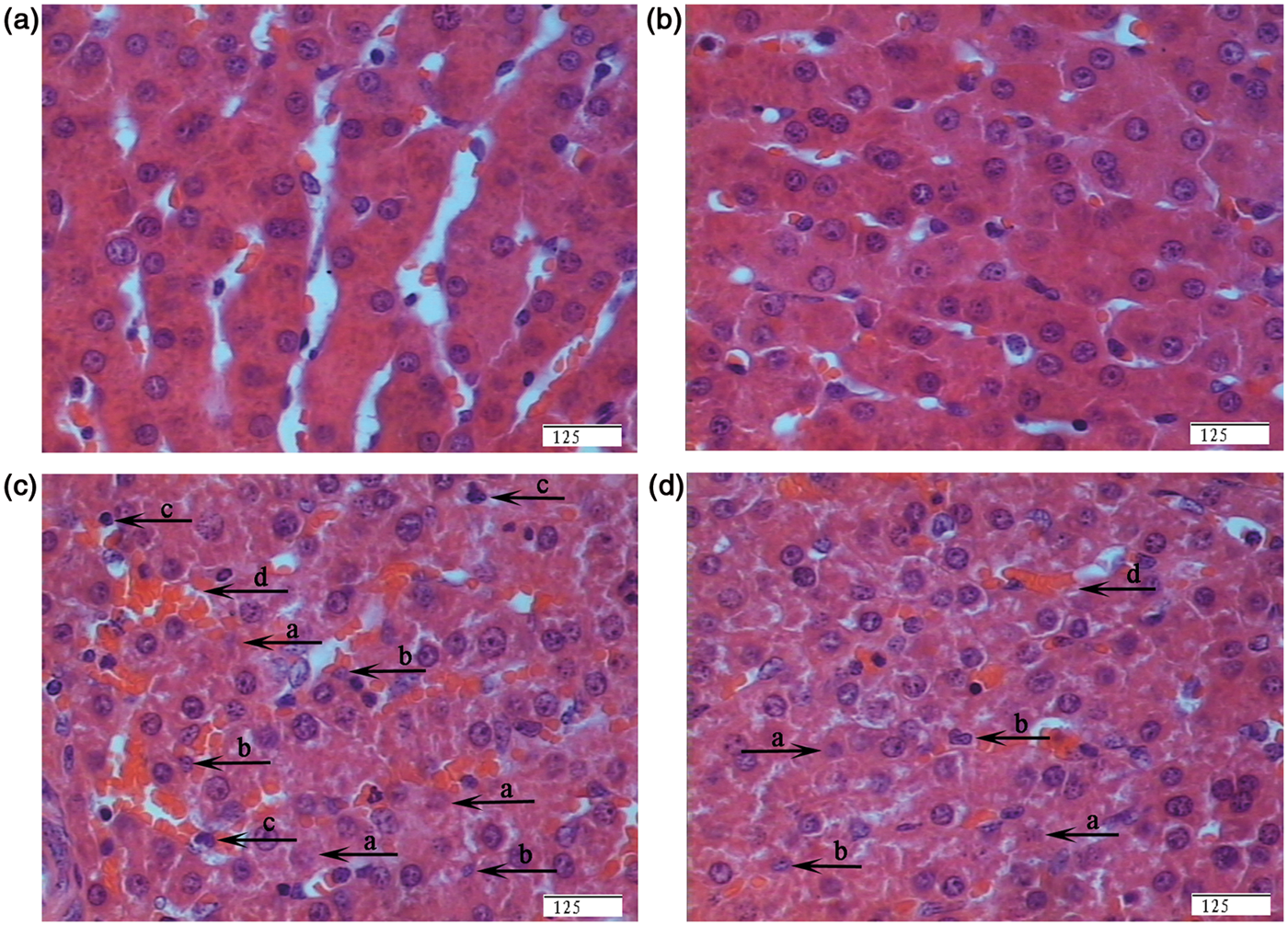
Serum biochemical parameters
Effects of fish oil or corn oil supplementation on serum biochemical parameters after 4 h E. coli LPS challenge in weaned pigs.
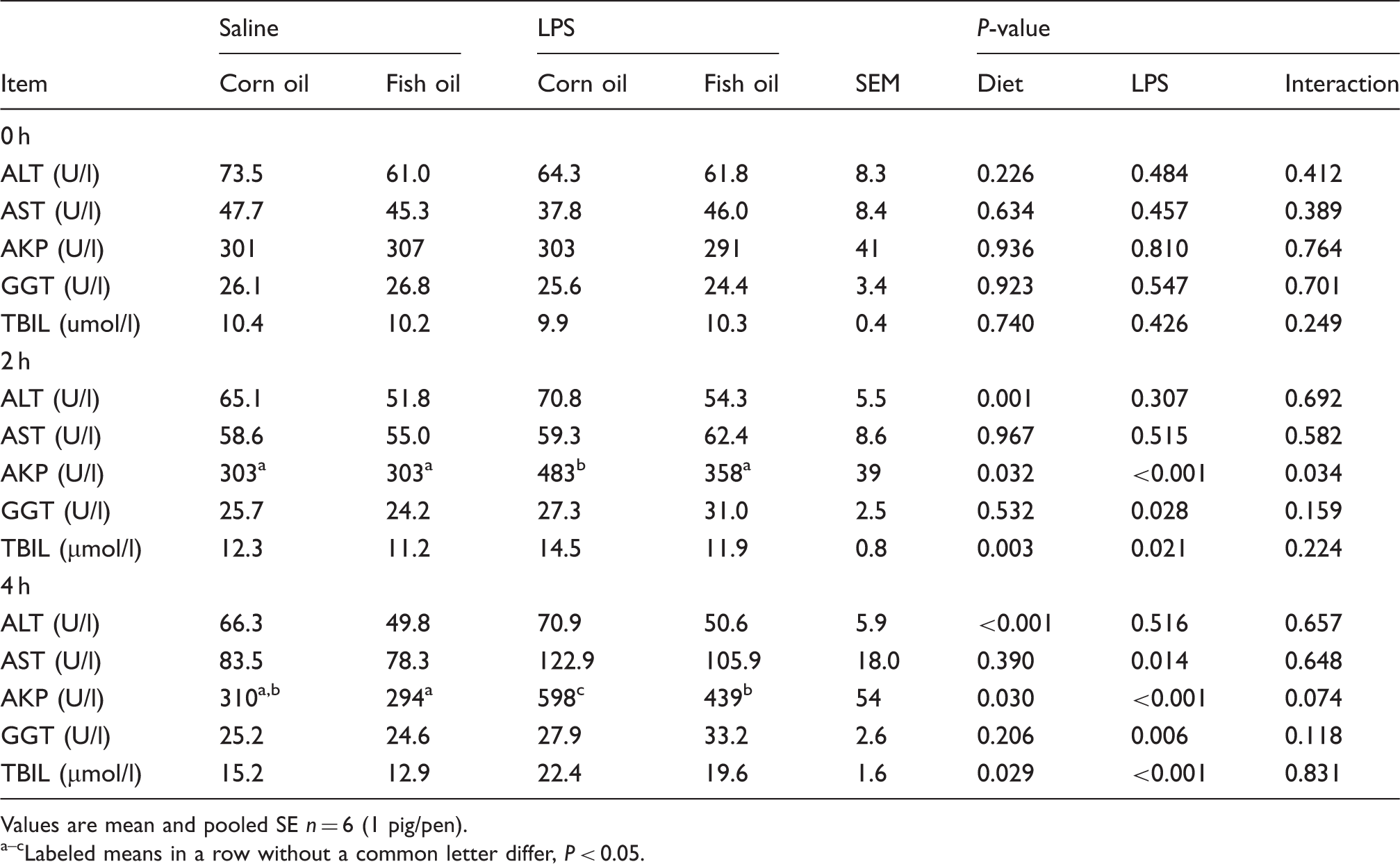
Values are mean and pooled SE n = 6 (1 pig/pen). a–cLabeled means in a row without a common letter differ, P < 0.05.
Liver tight junction proteins, proliferation cell nuclear antigen and caspase-3 expression
Effects of fish oil or corn oil supplementation on mRNA expression of tight junction proteins, PCNA and caspase-3 in liver after 4 h E. coli LPS challenge in weaned pigs.
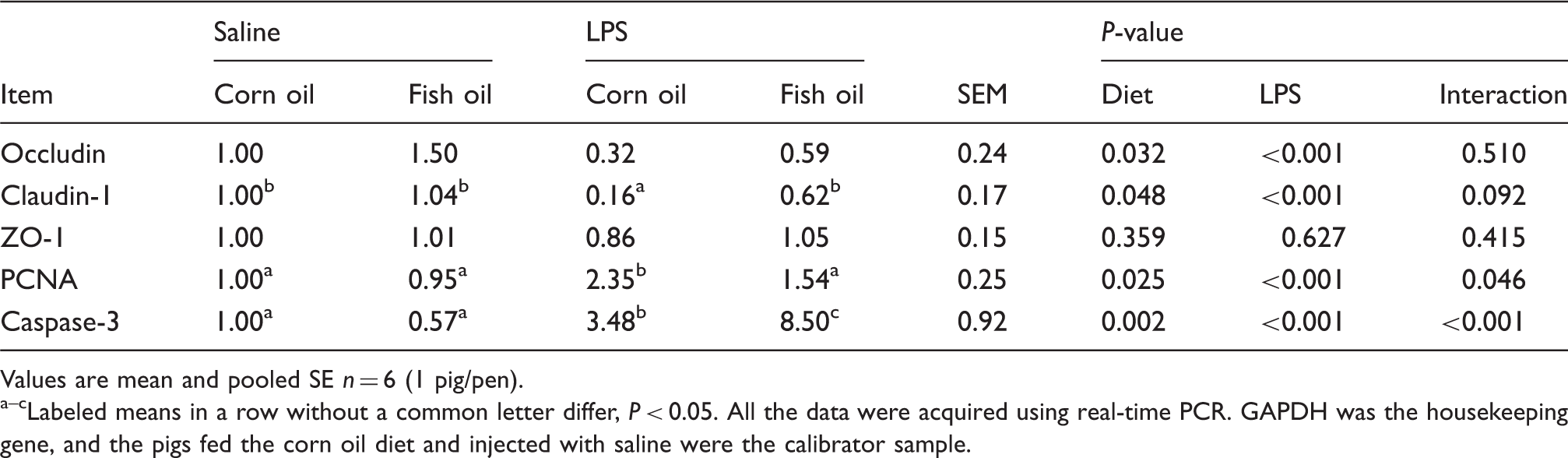
Values are mean and pooled SE n = 6 (1 pig/pen). a–cLabeled means in a row without a common letter differ, P < 0.05. All the data were acquired using real-time PCR. GAPDH was the housekeeping gene, and the pigs fed the corn oil diet and injected with saline were the calibrator sample.
Liver TNF-α, PGE2 and HSP70 levels, and TNF-α, COX2 and HSP70 mRNA expression
Effects of fish oil or corn oil supplementation on liver TNF-α and PGE2 concentrations, and HSP70 and NF-κB p65 protein expressions after 4 h E. coli LPS challenge in weaned pigs.
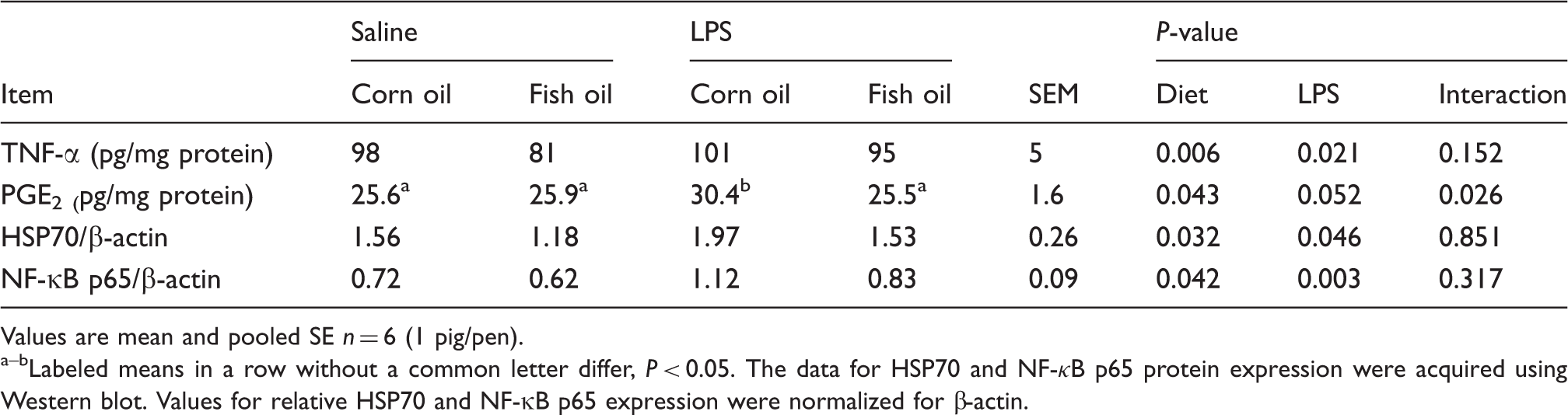
Values are mean and pooled SE n = 6 (1 pig/pen). a–bLabeled means in a row without a common letter differ, P < 0.05. The data for HSP70 and NF-κB p65 protein expression were acquired using Western blot. Values for relative HSP70 and NF-κB p65 expression were normalized for β-actin.
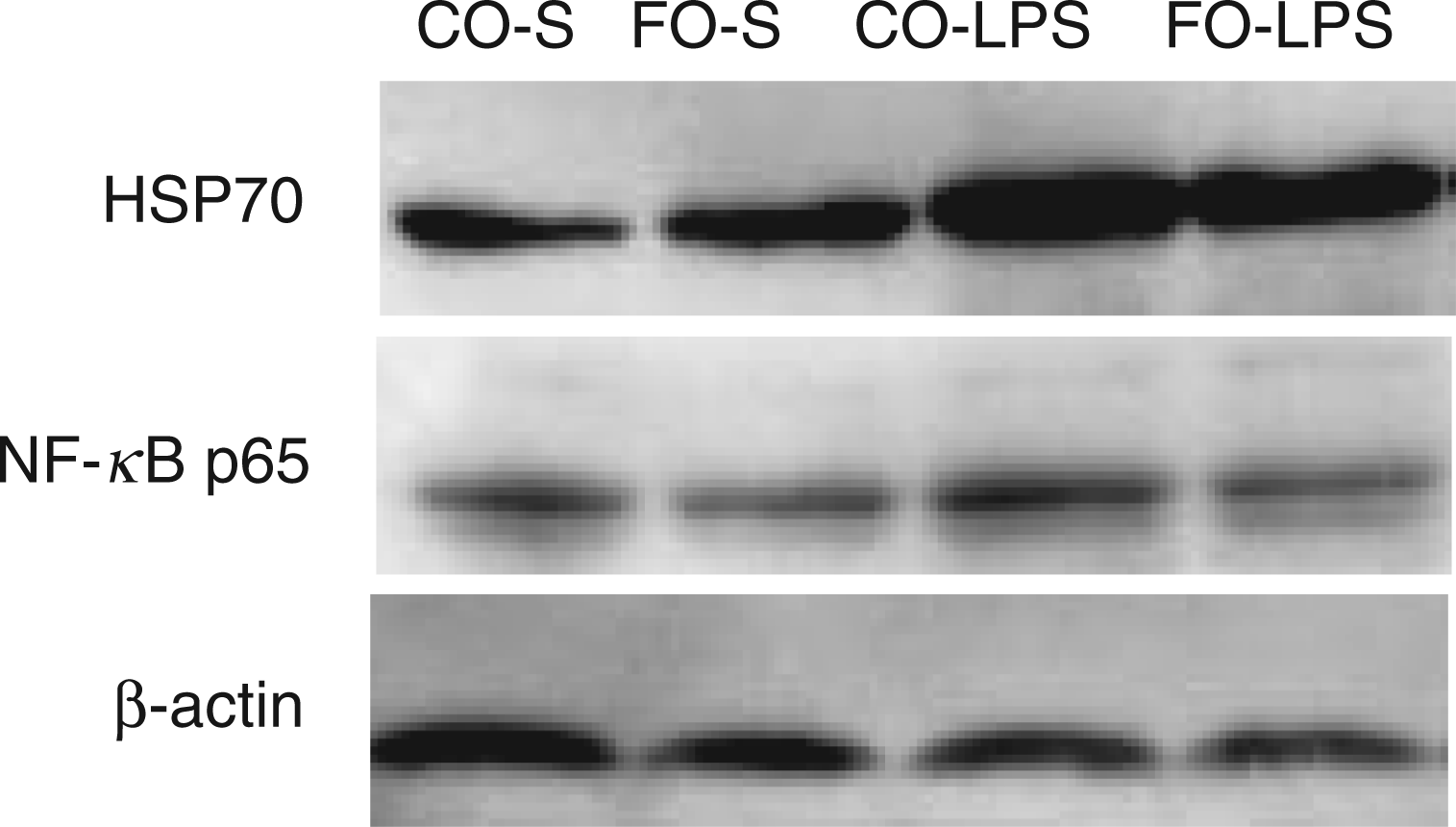
Effects of fish oil or corn oil supplementation on protein expression of HSP70 and NF-κB p65 in liver after 4 h E. coli LPS challenge in weaned pigs. The bands were the representative Western blot images of HSP70 (70 ku), NF-κB p65 (65 ku) and β-actin (42 ku) in liver. Column CO-S, piglets fed the corn oil diet and injected with saline; Column FO-S, piglets fed the fish oil diet and injected with saline; Column CO-LPS, piglets fed the corn oil diet and challenged with LPS; Column FO-LPS, piglets fed the fish oil diet and challenged with LPS.
Effects of fish oil or corn oil supplementation on mRNA expression of TLR4 and NODs and their downstream signals after 4 h E. coli LPS challenge in weaned pigs.
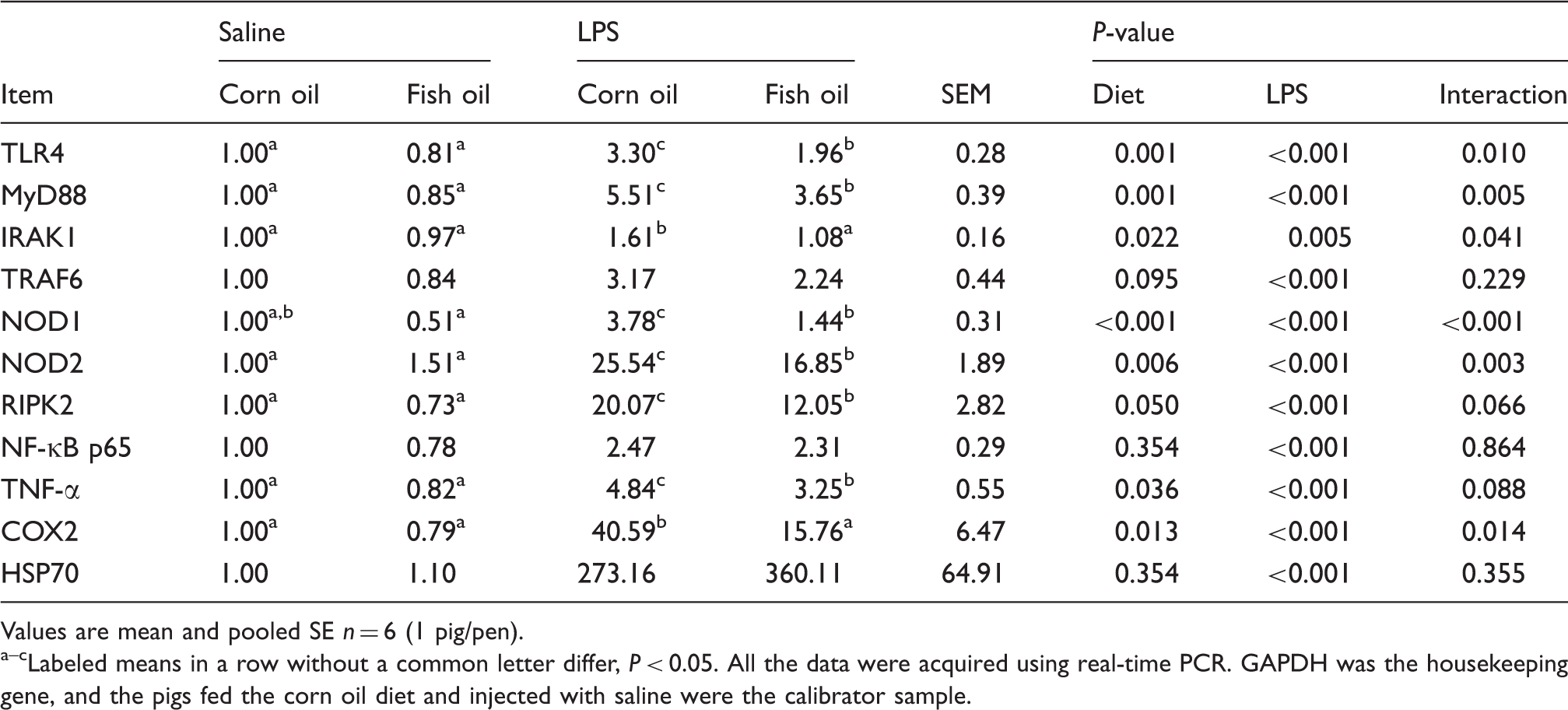
Values are mean and pooled SE n = 6 (1 pig/pen). a–cLabeled means in a row without a common letter differ, P < 0.05. All the data were acquired using real-time PCR. GAPDH was the housekeeping gene, and the pigs fed the corn oil diet and injected with saline were the calibrator sample.
mRNA expression of TLR4 and NODs, and their downstream signals
LPS challenge increased mRNA abundance (Table 8) of liver TLR4, MyD88, IL-1 receptor-associated kinase 1 (IRAK1), TNF-α receptor-associated factor 6 (TRAF6), NOD1, NOD2 and receptor-interacting serine/threonine-protein kinase 2 (RIPK2) (P < 0.01). There was a LPS challenge × diet interaction observed for liver TLR4, MyD88, IRAK1, NOD1 and NOD2 (P < 0.05), and a trend for interaction observed for liver RIPK2 (P = 0.066) such that the responses of these parameters to LPS challenge were lower in those pigs receiving the fish oil diet compared with the LPS challenged pigs fed the corn oil diet, whereas there was no difference for these parameters in saline-injected pigs. There was no LPS challenge × diet interaction observed for liver TRAF6. However, the pigs fed the fish oil diet tended to have lower TRAF6 mRNA abundance (P = 0.095) than pigs fed the corn oil diet.
There was no LPS challenge × diet interaction observed for liver NF-κB p65 mRNA (Table 8) and protein expression (Table 7 and Figure 2). LPS challenge increased mRNA and protein expression of NF-κB p65 in liver (P < 0.01). Fish oil had no effect on NF-κB p65 mRNA abundance, but decreased its protein expression (P < 0.05).
Discussion
In this study, to investigate whether dietary supplementation of fish oil could alleviate liver injury, we utilized a well-documented model for inducing liver damage by injecting E. coli LPS.1,2,16,26 This molecule (LPS), found in the outer membrane of all Gram-negative bacteria, is a potent endotoxin.2,16 It binds to Kupffer cells, the specialized macrophages located in liver, and activates them, resulting in the over-release of pro-inflammatory cytokines, such as TNF-α, IL-1β and IL-6. Of them, TNF-α is a critical mediator of liver damage in this model. 2 Studies have shown that, within 3–6 h of LPS injection, LPS caused acute liver histological injury and dysfunction, which was accompanied with enhanced hepatic pro-inflammatory cytokine production.2,16,26,27
As an excellent source of n-3 PUFAs such as EPA and DHA, fish oil exerts beneficial effects on inflammatory diseases in animal models and clinical trials. 3 Based on this, we investigated a protective effect of 5% fish oil supplementation against LPS-induced liver injury and the molecular mechanism for hepatoprotective effect of fish oil using a piglet model. In our study, as expected, we observed that EPA, DHA and total (n-3) PUFA were enriched in liver through dietary supplementation of 5% fish oil, which is consistent with the results of El-Badry et al. 28 and Schmöcker et al. 2
Elevation in serum transaminases (ALT and AST) is considered to be a sensitive indicator of liver injury.
1
In addition, serum AKP and GGT activity, and TBIL level, are also described as valuable parameters of liver injury.1,5 In this study, serum AST, AKP and GGT activities, and TBIL level, showed a marked increase in pigs subjected to LPS challenge. However, fish oil decreased ALT and AKP activities, and TBIL level, compared with corn oil. These results were also supported by histological observations; while liver obtained from the LPS-challenged pigs fed corn oil showed severe injury, such as heptatocyte caryolysis, karyopycnosis, inflammatory cell infiltration, hyperaemia in hepatic sinusoids and hepatic cell cords arrangement in disorder, only mild hepatic injury was observed in the LPS-challenged pigs fed fish oil. These results demonstrate that fish oil supplementation attenuated the damage of hepatic architecture and function after LPS challenge. Similarly, n-3 PUFAs have been reported to attenuate hepatic damage, as indicated by reduced serum AST, ALT, AKP and GGT levels, and less histological liver damage in a
The tight junction is a major cellular component for the maintenance of tissue integrity and has a barrier function. 29 The injury of liver architecture is due largely to changes in tight junctions. 29 Changes in tight junctions result in deteriorated liver barrier function and increased intercellular permeability. 29 Tight junctions are composed of at least 40 different proteins. Of them, occluding, claudins and ZO-1 are considered as the major integral membrane proteins which participate in tight junction structural integrity. 30 In our study, consistent with the improved hepatic architecture and function, fish oil increased the mRNA expression of liver occludin and claudin-1. In agreement with our study, Yan et al. 31 reported that n-3 PUFA supplementation enhanced the expression of occludin, claudin-3 and ZO-1, and protected hepatic tight junction architecture and hepatic function after 70% hepatectomy in rats. In the present study, fish oil may improve hepatic architecture and function partially via improving the expression of liver tight junction proteins.
Maintaining tissue integrity and homeostasis is determined mainly by a balance between cell proliferation and cell apoptosis. 32 However, in pathological conditions, the balance between proliferation and apoptosis is often altered, which results in changes in tissue integrity and homeostasis, thus resulting in the onset of liver diseases. 32 Insufficient apoptosis, with failure to remove the damaged cells or cells carrying mutated genes, and uncontrolled proliferation on inflammatory condition, can promote the development of liver cancer. 33 In the current study, LPS challenge induced hepatocyte proliferation indicated by higher PCNA mRNA abundance. This increase in proliferation was accompanied by an increase in apoptosis, which was indicated by higher caspase-3 mRNA abundance, suggesting a compensative trend toward tissue homeostasis. Fish oil inhibited LPS-induced hepatocyte proliferation and promoted hepatocyte apoptosis, which was indicated by lower PCNA mRNA abundance and higher caspase-3 mRNA abundance. Similarly, Chen et al. 34 reported that fish oil inhibited liver cell proliferation in spontaneously hypertensive rats. In addition, Kotzampassi et al. 6 reported that n-3 PUFAs promoted hepatocyte apoptosis, which resulted in faster detachment of damaged cell from the irradiated tissue, thus attenuating radiation-induced liver injury in the rat. In the present study, fish oil may improve the liver integrity partially via modulating hepatocyte proliferation and apoptosis.
We hypothesized that fish oil improved liver integrity by attenuating the liver inflammatory response. Increased levels of TNF-α, PGE2, COX2 (the key enzyme to produce PGE2) and HSP70 are thought of as important markers of inflammation.1,35,36 In our study, consistent with improved liver integrity, dietary supplementation of fish oil decreased TNF-α and PGE2 concentrations and HSP70 protein expression, and also decreased the mRNA expression of TNF-α and COX2 in liver. Several studies have implicated n-3 PUFA in the dampening of liver inflammation by reducing pro-inflammatory mediators, such as pro-inflammatory cytokines (especially TNF-α)2,7,37,38 and pro-inflammatory eicosanoids (e.g. PGE2).
39
For example, Schmöcker et al.
2
reported that n-3 PUFA alleviated
To explore the molecular mechanism(s) by which dietary supplementation of fish oil exerted an anti-inflammatory role in liver, we examined the roles of TLR4 and NOD signaling pathways. TLRs and NODs are two major forms of innate immune sensors, which play a central role in detection of invading pathogens and regulation of innate and adaptive immune responses by recognizing PAMPs.8,9 Of them, TLRs are transmembrane proteins which detect a variety of extracellular PAMPs, whereas NODs are cytoplasmic proteins which response to PAMPs in the cytosol. Among the TLR family, TLR4 is the most studied member; it is responsible for recognizing endotoxin (LPS) present in the outer membrane of Gram-negative bacteria and triggering an inflammatory response.10,11 When engaged by LPS, the TLR4/MD-2/CD14 receptor complex transduces a signal sensed by MyD88, which is further transmitted by a cascade of downstream signaling molecules, such as IRAKs, TRAF6 and NF-κB-inducing kinase, leading to activation of NF-κB. Activation of NF-κB leads to expression of the inflammatory genes, such as pro-inflammatory cytokines and COX2. 10 Among the NOD family, NOD1 and NOD2 are the best-characterized members. Different from TLR4, NODs possess the ability to connect with the bacterial LPS and peptidoglycan, and to transduce a TLR-independent signal, which also leads to NF-κB activation via the adaptor molecule RIPK2, and stimulates the synthesis of pro-inflammatory mediators. 9 In the present experiment, we found that liver mRNA abundances of TLR4 and its downstream signals (MyD88, IRAK1 and TRAF6), and NOD1, NOD2 and their adaptor molecule (RIPK2) were reduced, and the protein and mRNA levels of liver TNF-α were decreased simultaneously in the pigs fed a fish oil diet after LPS challenge. Surprisingly, mRNA expression of NF-κB p65 was not affected by fish oil. However, further protein expression analysis showed that NF-κB p65 protein expression was down-regulated by fish oil. Some research investigating whether mRNA and protein expression are correlated has shown that mRNA and protein are differentially expressed, which suggests frequent regulation of gene expression at the post-transcriptional level. 40 Currently, there are several reports about (n-3) PUFA regulating TLR signaling in cell models, such as monocyte/macrophage cells41–43 and intestinal microvascular endothelial cells. 44 However, data in vivo are lacking. Lee et al. 42 reported that saturated fatty acids (lauric acid) stimulated, but (n-3) PUFA (EPA or DHA) inhibited the activation of TLR4 and the expression of its target genes in macrophage cells (RAW 264.7) and 293T cells. Most recently, Wong et al. 43 reported that n-3 PUFAs inhibited LPS- or lauric acid-induced activation of TLR4 through inhibition of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. Relative to the research on n-3 PUFA modulating the TLR signaling pathway, research on n-3 PUFA regulating NOD signaling is even more limited. Only research from Zhao et al. 45 reported that saturated fatty acids (lauric acid) stimulated, but n-3 PUFAs (such as EPA or DHA), inhibited NOD signaling pathways, and thus differentially modulated NF-κB and IL-8 expression in human colonic epithelial HCT116 cells. In our current study, it is possible that the protective effects of fish oil on liver integrity were closely associated with decreasing the expression of hepatic pro-inflammatory cytokines through inhibition of the TLR4 and NOD signaling pathway.
In our study, owing to the dynamic changes that may occur in expression of hepatic pro-inflammatory mediators and signaling molecules, 26 measurements taken at one time point (4 h) are probably not adequate to confirm the roles of hepatic pro-inflammatory mediators and signaling molecules in LPS-induced liver injury. So, sample collections at more time points might be needed to analyze their dynamic changes in both mRNA and protein levels, which awaits further experimentation.
In summary, dietary treatment of fish oil attenuates LPS-induced liver injury in weaned pigs. It is possible that the hepatoprotective effects of fish oil may be closely associated with down-regulating the expression of hepatic pro-inflammatory cytokines via inhibition of TLR4 and NOD signaling pathways.
Footnotes
Funding
The authors express their gratitude to the National Natural Science Foundation of China (31172222 and 30972109), the Program for New Century Excellent Talents in University (NCET-10-0158), the National Basic Research Program of China (2012CB126305), the Project of Natural Science Foundation of Hubei Province (2010CDA050 and 2009CDB006), the Project of International Cooperation of Hubei Province (2011BFA008), the Hubei Provincial Department of Education (Q20111702), and Wuhan Science and Technology Program (201171034320) for the financial support.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
