Abstract
The effects of
Introduction
Weaning age has reduced dramatically over the past 20 years. Because of decreased weaning age, pigs are smaller, and have less mature immune and digestive systems at weaning. 1 Additionally, the weaning transition involves complex social, environmental, and nutritional changes for piglets, and it is a stressful event. 2 Weaning removes young pigs from passive immune protection from the milk of the sow and increases their susceptibility to enterotoxigenic Escherichia coli infection.3,4 Moreover, early weaning stress impairs development of mucosal barrier function in the porcine intestine. 5 Therefore, the intestinal mucosa immune barrier function is especially important at the time of weaning. This has augmented the calls for nutritional strategies that provide preservation of intestinal mucosa immune barrier function.
Recent studies show that specific nutrients, such as amino acids, can prevent the damage of intestinal mucosal immune barrier function.6–9
In the current study, we used a well-established model to induce intestinal injury in weaned pigs by administering E. coli LPS. 17 Our objective was to test the hypothesis that dietary Arg supplementation would facilitate intestinal mucosal immune barrier function restitution.
Materials and methods
Pigs and diets
Ingredient composition of the basal diet (as-fed basis).
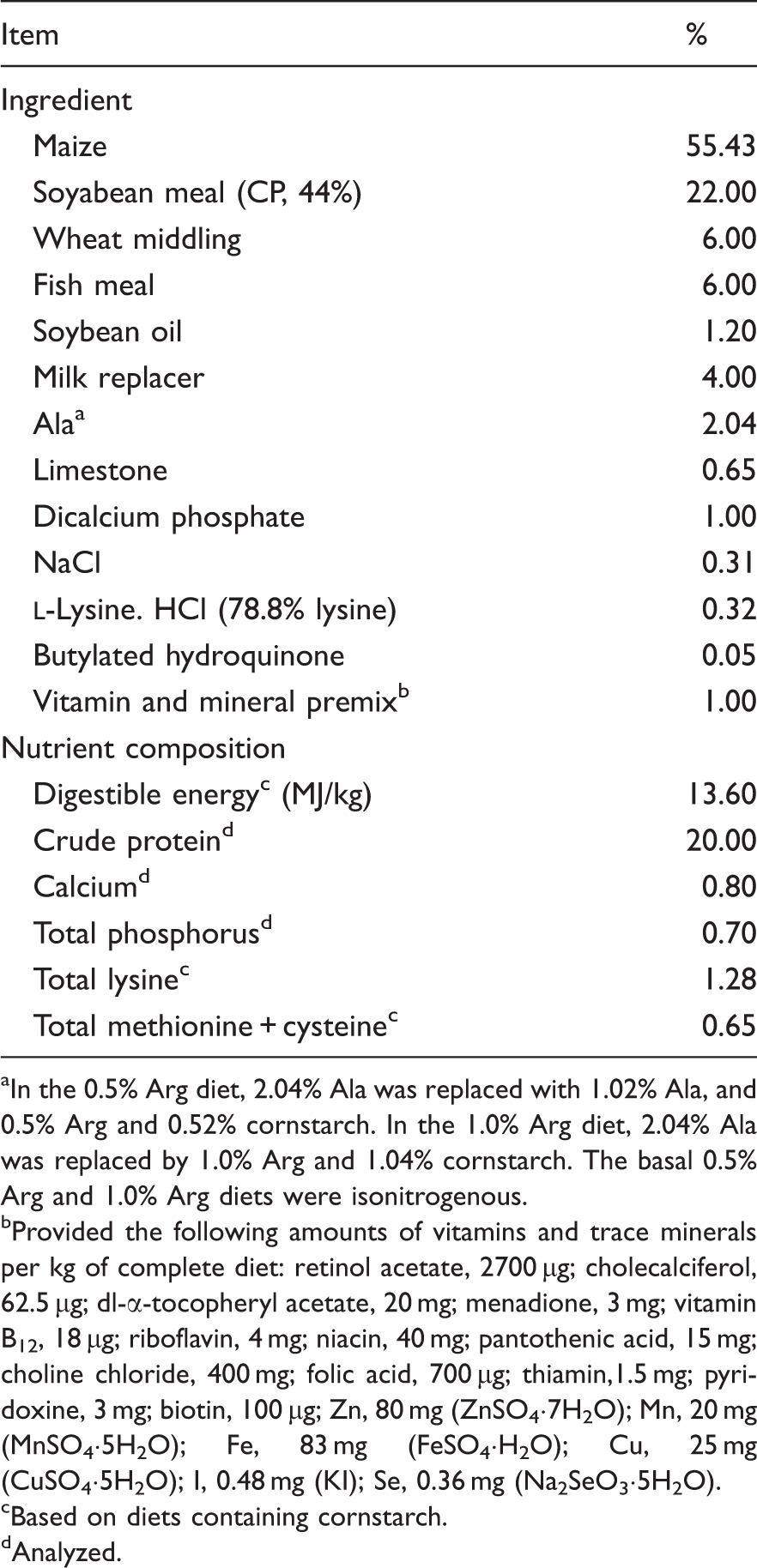
In the 0.5% Arg diet, 2.04% Ala was replaced with 1.02% Ala, and 0.5% Arg and 0.52% cornstarch. In the 1.0% Arg diet, 2.04% Ala was replaced by 1.0% Arg and 1.04% cornstarch. The basal 0.5% Arg and 1.0% Arg diets were isonitrogenous.
Provided the following amounts of vitamins and trace minerals per kg of complete diet: retinol acetate, 2700 µg; cholecalciferol, 62.5 µg; dl-α-tocopheryl acetate, 20 mg; menadione, 3 mg; vitamin B12, 18 µg; riboflavin, 4 mg; niacin, 40 mg; pantothenic acid, 15 mg; choline chloride, 400 mg; folic acid, 700 µg; thiamin,1.5 mg; pyridoxine, 3 mg; biotin, 100 µg; Zn, 80 mg (ZnSO4·7H2O); Mn, 20 mg (MnSO4·5H2O); Fe, 83 mg (FeSO4·H2O); Cu, 25 mg (CuSO4·5H2O); I, 0.48 mg (KI); Se, 0.36 mg (Na2SeO3·5H2O).
Based on diets containing cornstarch.
Analyzed.
Experimental design
Twenty-four pigs [initial body mass (BM) = 5.53 ± 0.24 kg] were blocked by initial mass and sex, and assigned to one of four treatments. The treatments included: (i) non-challenged control (CONTR; pigs fed a control diet and injected with sterile saline); (ii) LPS-challenged control (LPS; pigs fed the same control diet and challenged by injection with E. coli LPS); (iii) LPS + 0.5% Arg treatment (pigs fed a 0.5% Arg diet and challenged by injection with E. coli LPS; and (iv) LPS + 1.0% Arg treatment (pigs fed a 1.0% Arg diet and challenged by injection with E. coli LPS). The doses of Arg (
On d 16 of the trial, the challenged groups were administrated E. coli LPS intraperitoneally at 100 µg/kg BM and the unchallenged group was injected with the same amount of 0.9% (wt/vol) saline. The LPS (E. coli serotype O55:B5; Sigma Chemical Inc., St Louis, MO, USA) was dissolved in sterile 0.9% saline. To avoid the possible effects of LPS-induced feed intake reduction on gastrointestinal characteristics, the control, LPS, LPS + 0.5% Arg and LPS + 1.0% Arg were fed the same amount of feed/kg BM during d 16–18 of the trial (post-challenge). According our preliminary experiment and previous study, 17 all pigs among four treatments received 20 g feed/kg BM during d 16–18 of the trial (post-challenge). BM and feed intake were measured at 08.00 h of d 0, 16 and 18.
Sample collection
On d 18 of the trial, all pigs were sacrificed with sodium pentobarbital (80 mg/kg BM). The small intestine, from the ligament of Treitz to the ileocecal junction, was removed and flushed with ice-cold saline. A 1–2-cm-long cross section of intestinal tissue was obtained from the mid-jejunum and mid-ileum, and fixed in 4% paraformaldehyde in PBS for subsequent histologic analysis.
Intestinal morphology and cell count
Paraformaldehyde-fixed intestinal samples were dehydrated and embedded in paraffin. Four-µm sections were cut and mounted on polylysine-coated slides. The sections were then stained with routine hematoxylin and eosin (H&E) stain.
Intestinal morphological measurements included villus height, crypt depth and villus area. These criteria were determined using a microscope with an ocular micrometer. Villus area was quantitated from the perimeter and height of the villi. 19 The villus height:crypt depth ratio (VCR) was calculated. The 10 longest and straightest villi and their associated crypts from each segment were measured. The same villus and crypt were used to determine the number of intraepithelial lymphocytes (IELs). 19 The variables were expressed per 100 enterocytes. Goblet cells were counted in 10 villi and 10 crypts on two intestinal sections (mid-jejunal and mid-ileal sections) combination stained with the periodic acid Schiff (PAS)/alcian blue pH 2.5 procedure. 20 The goblet cell numbers were determined per 100 enterocytes. The mean value of each sample was calculated.
To visualize mast cells, intestinal tissue sections (mid-jejunal and mid-ileal sections; 4 µm) were cut, de-waxed, rehydrated, and stained with an improved toluidine blue (pH 4.0) and analyzed by light microscopy. The mast cells were counted in 10 random field of view using a 400 × objective on each section. The mean of mast cells per field in each sample was given.
Lymphocyte immunohistochemistry
Intestinal CD8+, CD4+ T lymphocytes (T cells) and IgA-secreting cells (ASCs), which participate in effectors mechanisms of intestinal mucosal immunity, were monitored in this study by immunohistochemistry. Intestinal tissues (mid-jejunum and ileum) were cut into 4 -µm-thick sections onto glass slides to use for ASCs, CD4+ and CD8+ T cell enumeration by an immunoperoxidase technique.20,21 Slides were de-paraffinized in xylene and then rehydrated through a series of washes in alcohol, water and PBS. Endogenous peroxidase activity was extinguished by incubation with 0.3% H2O2-PBS (pH 7.3, 0.01 M) for 10 min. The sections were digested with complex enzymes for 5 min at room temperature (21–25℃). Then, slides were washed three times in PBS and blocked by goat serum (1:50 dilution) at 37℃ for 20 min in a humidified chamber. When the serum had been shaken off, the sections were first incubated with 1:100 dilution of polyclonal rabbit anti-swine-IgA Ab (kindly donated by Professor Qian Yang, College of Veterinary Medicine, Nanjing Agricultural University, Nanjing, China) or 1:20 dilution of anti-human CD4+, CD8+ Ab (Wuhan Boster Biological Technology Ltd, Wuhan, China) at 4℃ in a humidified chamber overnight (12–16 h), then with a biotinylated goat anti-rabbit-IgG Ab (SABC kit provided by Wuhan Boster Biological Technology Ltd, Wuhan, China) at 37℃ for 20 min. Subsequently, peroxidase-conjugated streptavidin labels were added. Diaminobensidine was used as substrate. Omission of the primary Abs with the corresponding Ag served as negative control. ASC counts were conducted with light microscopy. As ASCs were distributed mainly in the jejunal and ileal lamina propria between glandular cavities, ASCs and interstitial cells (mainly lymphocytes) were counted in 10 fields of the ASCs well-distributed lamina propria in each section, examined under 400 × magnification. The mean percentage of ASCs to interstitial cells in each sample was calculated. Jejunal and ileal CD4+ or CD8+ T cells were counted in stained sections at 400 × magnifications in 10 fields. The interstitial cells were counted at the same time. The mean percentage of CD4+ or CD8+ T cells to interstitial cells was calculated.
Lymphocyte proliferation in ileal Peyer’s patches was measured as described previously. 22 Two hours before slaughter, 5-BrdU (Roche Diagnostics, Indianapolis, IN, USA) was injected intraperitoneally with 25 mg/kg BM of 5-BrdU. BrdU-labeled cell were detected by immunohistochemistry in paraformaldehyde-fixed, paraffin-embedded sections and expressed as a percentage of total nuclei per lymphoid follicles observed in approximately 10–12 lymphoid follicles from 3 tissue sections from each animal.
Measurements of apoptosis were made according to the instructions supplied by the manufacturer [dUTP-digoxgenin nick end labeling (TUNEL) kit, Wuhan Boster Biological Technology Ltd, Wuhan, China). Apoptotic cell counts were conducted with a light microscope. Apoptotic cells were expressed as a percentage of the total lymphocytes per lymphoid follicles of the same sections; approximately 10–12 lymphoid follicles were counted from 3 tissue sections per animal.
Statistical analyses
All data were analyzed by ANOVA using the general linear model procedure of SAS. 23 A post hoc least significant difference test was used to determine differences between treatments following ANOVA. Differences were considered significant when P < 0.05.
Results
Performance and intestinal morphology
Overall, there was no difference in average daily gain (ADG) (137, 125, 120 and 141 g respectively) and average daily feed intake (ADFI) (256, 246, 234 and 276 g respectively) among four treatments during d 0–16 (pre-challenge). To avoid the possible effects of LPS-induced feed intake reduction on intestinal characteristics, we fed the same amount of feed/kg BM during 16–18 d (post-challenge), which caused no significant difference of ADFI (155, 148, 147 and 159 g) among four treatments over 16–18 d. However, LPS pigs had a significant decrease in ADG compared with CONTR pigs (LPS, −176 g; CONTR, −18 g, P < 0.001). Dietary supplementation of 0.5% Arg or 1.0% Arg had no effect on ADG compared with LPS pigs (0.5% Arg, −110 g; 1.0% Arg, −70 g; LPS, −176 g) over 16–18 d.
Effect of
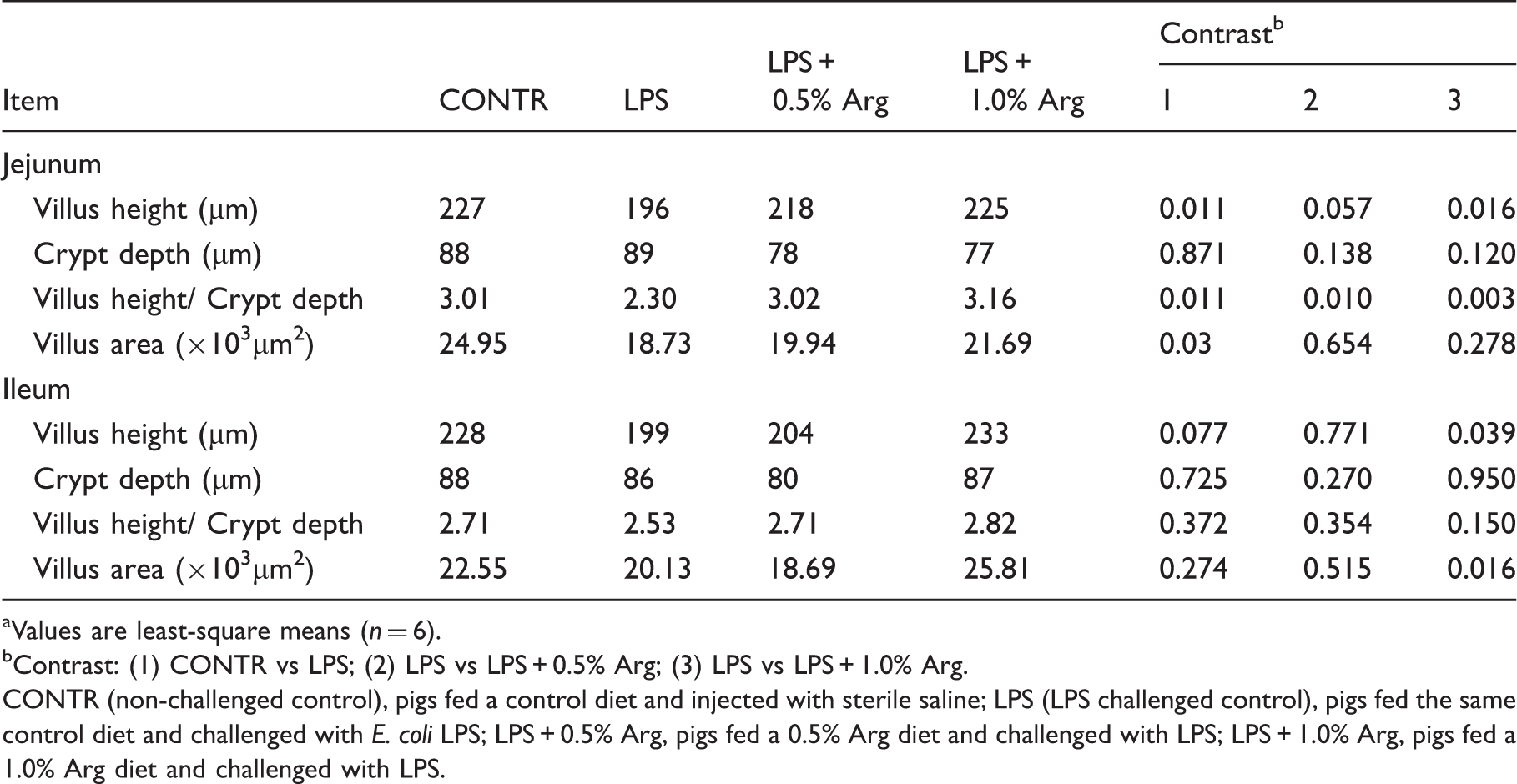
Values are least-square means (n = 6).
Contrast: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg; (3) LPS vs LPS + 1.0% Arg.
CONTR (non-challenged control), pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control), pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg, pigs fed a 0.5% Arg diet and challenged with LPS; LPS + 1.0% Arg, pigs fed a 1.0% Arg diet and challenged with LPS.
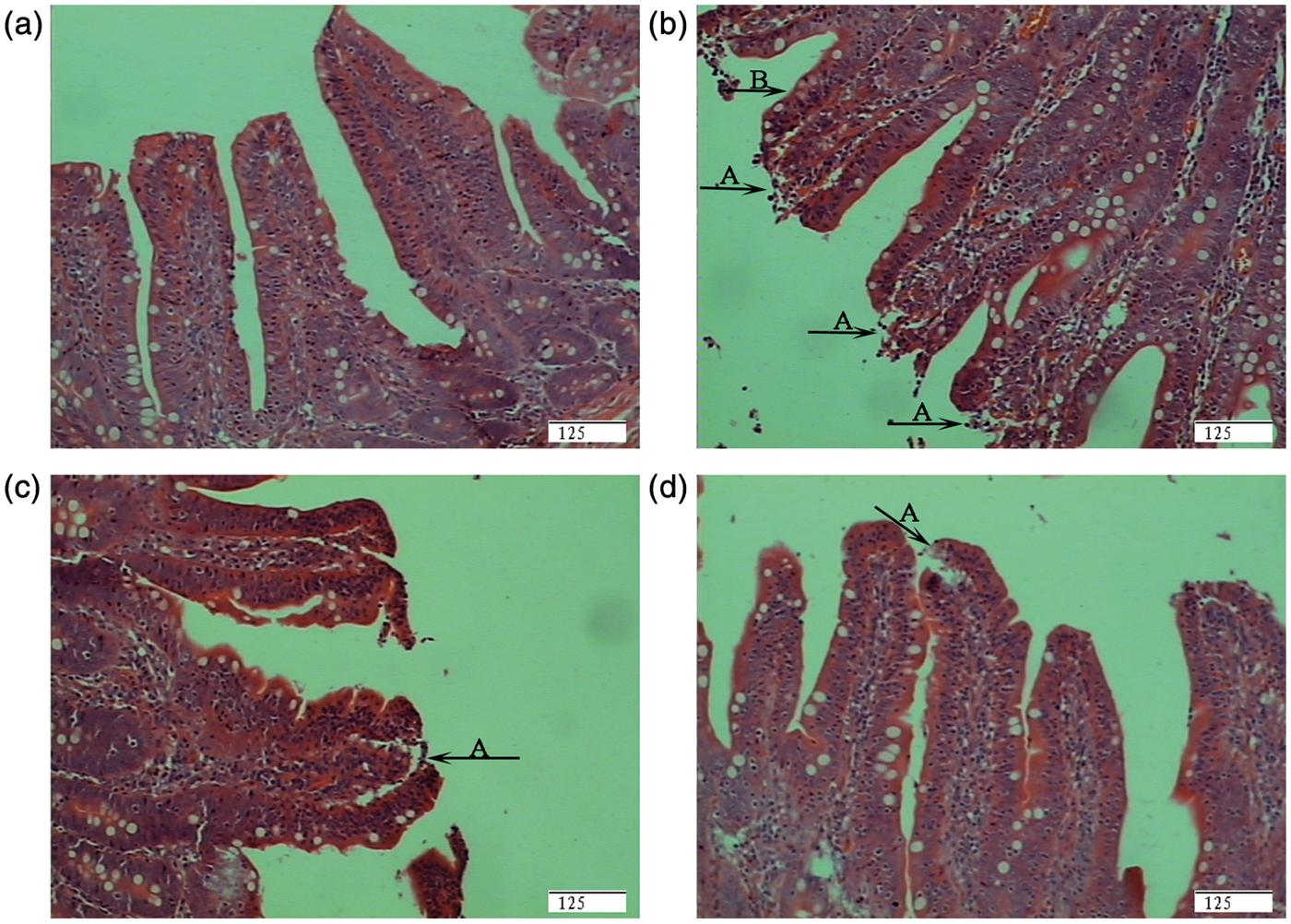
Morphological characterization of jejunum. (A) Pigs fed a control diet and injected with sterile saline (CONTR). No obvious changes were found. (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). Morphologic changes associated with intestinal mucosal injury, such as lifting of epithelium at the tip of the villus (a), and villous atrophy (b). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). Intestinal mucosal injury was attenuated. (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). Intestinal mucosal injury was significantly attenuated. Sections were stained with hematoxylin and eosin. Original magnification 200 ×. Scale bar = 125 µm.
Intestinal intraepithelial lymphocytes (IELs), goblet cells and mast cells
Effect of Arg on intraepithelial lymphocytes (IELs), goblet cells, mast cells and IgA-secreting cells counts in the small intestine of weaned pigs after E. coli LPS challenge. a
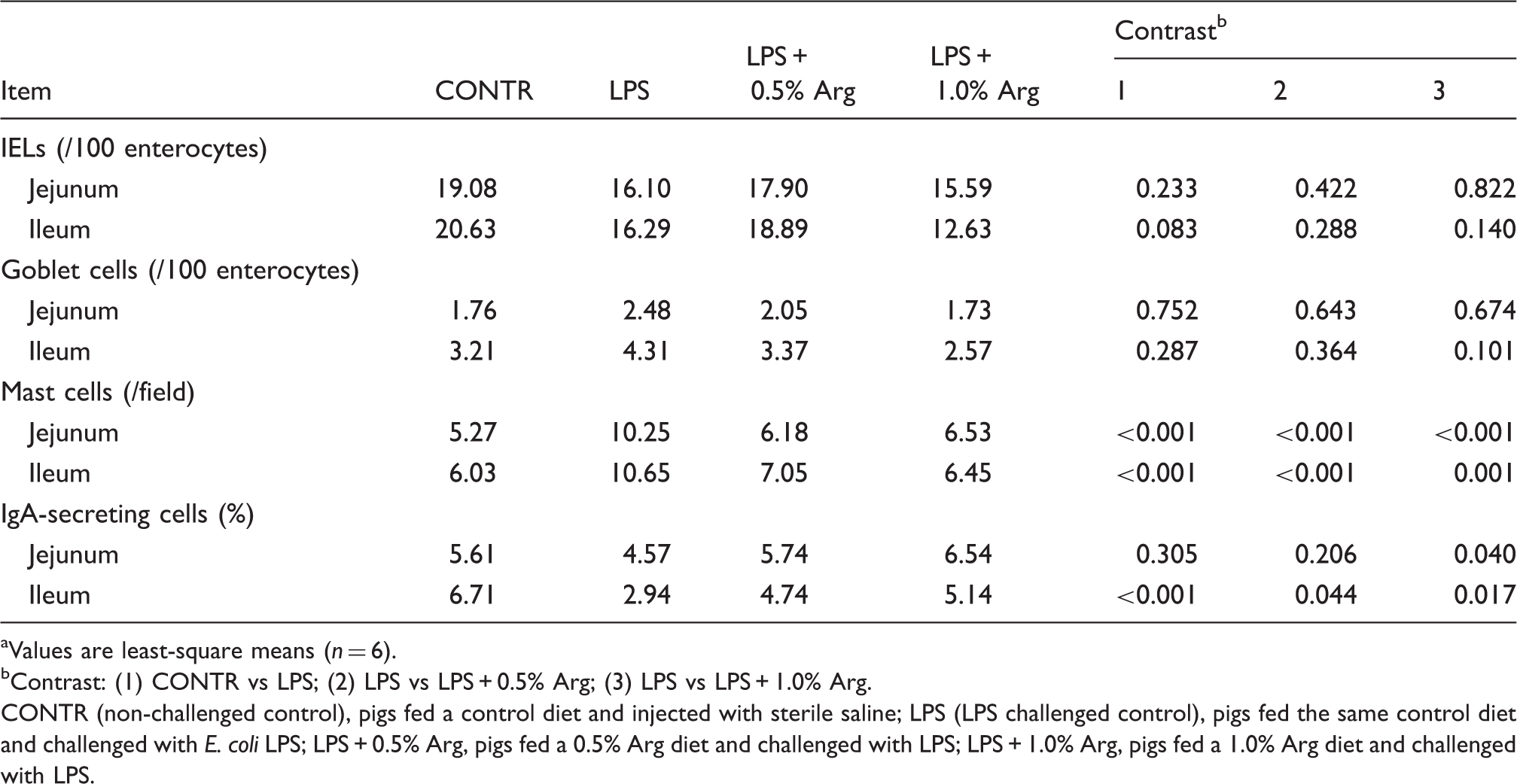
Values are least-square means (n = 6).
Contrast: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg; (3) LPS vs LPS + 1.0% Arg.
CONTR (non-challenged control), pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control), pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg, pigs fed a 0.5% Arg diet and challenged with LPS; LPS + 1.0% Arg, pigs fed a 1.0% Arg diet and challenged with LPS.
Intestinal mast cell numbers are presented in Table 3 and Figure 2. LPS challenge increased the numbers of mast cells in the jejunum (94% higher, P < 0.001) and ileum significantly (77% higher, P < 0.001) compared with CONTR pigs (Figure 2). Pigs fed a 0.5% or 1.0% Arg diet had decreased mast cell numbers in the jejunum (0.5% Arg, 40% lower; 1.0% Arg 36% lower; P < 0.001) and ileum (0.5% Arg, 34% lower; 1.0% Arg, 39% lower; P < 0.001) compared with LPS pigs (Figure 2).
Photomicrographs of pig ileal mucosa showing mast cells stained with toluidine blue. (A) Pigs fed a control diet and injected with sterile saline (CONTR). (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). LPS challenge increased the number of mast cells significantly compared with CONTR pigs. Arg (0.5% or 1.0%) supplementation decreased the number of mast cells compared with LPS pigs. Original magnification 400 ×. Scale bar = 125 µm.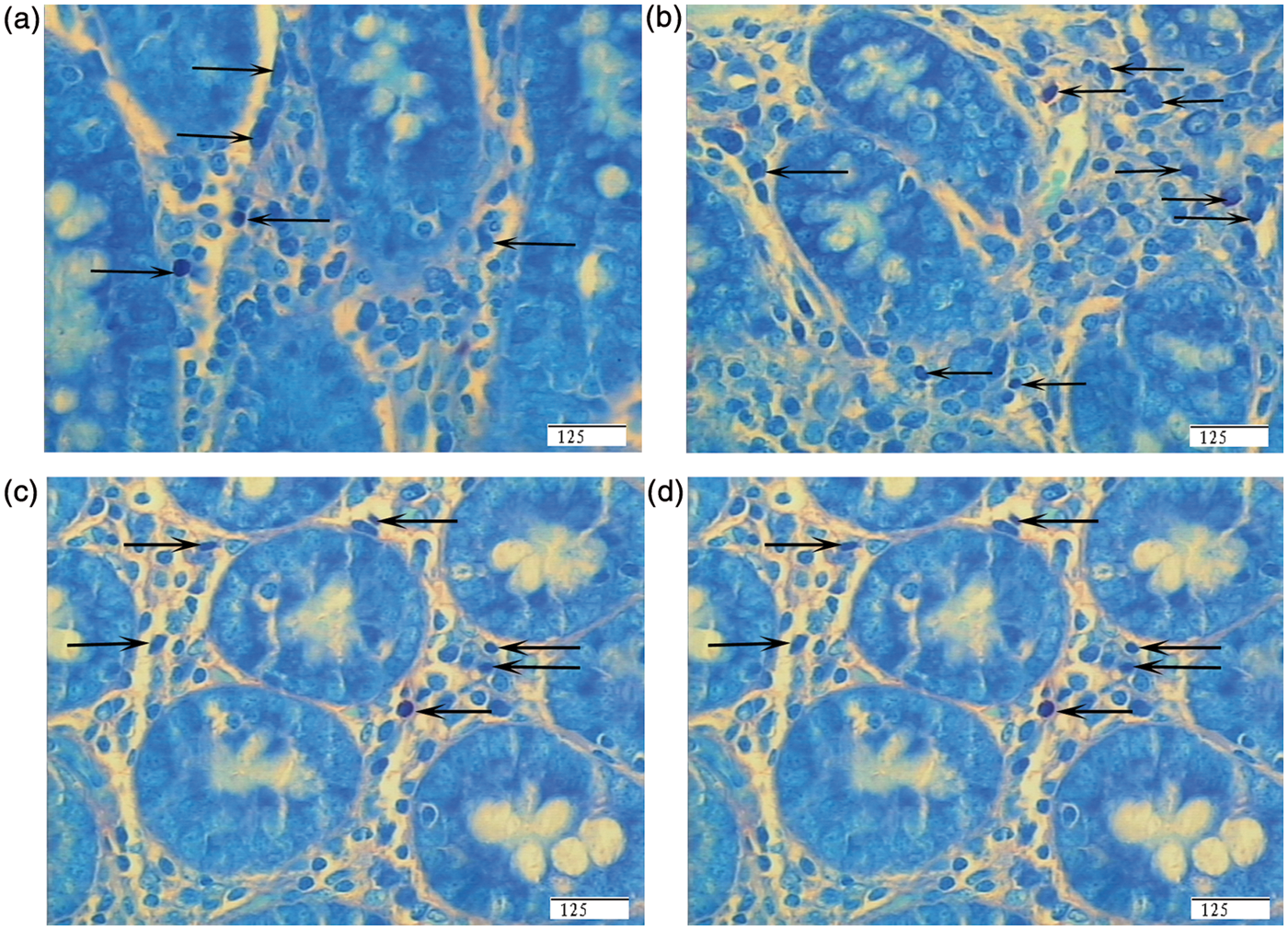
Intestinal IgA secreting cells (ASCs)
The numbers of intestinal ASCs are shown in Table 3. LPS challenge did not affect jejunal ASCs. However, a significant reduction was observed in ileal ASCs compared with CONTR pigs (56% lower, P < 0.001) (Figure 3). Dietary supplementation of 1.0% Arg increased ASCs in the jejunum (P = 0.040) and ileum significantly (P = 0.017) compared with LPS pigs (Figure 3). In addition, 0.5% Arg supplementation increased ileal ASCs (P = 0.044) relative to LPS pigs (Figure 3).
Representative photomicrographs of pig ileal mucosa showing immunostained IgA-secreting cells (ASCs). (A) Pigs fed a control diet and injected with sterile saline (CONTR). (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). The number of ASCs was decreased in LPS pigs compared with CONTR pigs. Arg (0.5% or 1.0%) supplementation increased ASCs in the ileum significantly compared with LPS pigs. Original magnification 400 ×. Scale bar = 125 µm.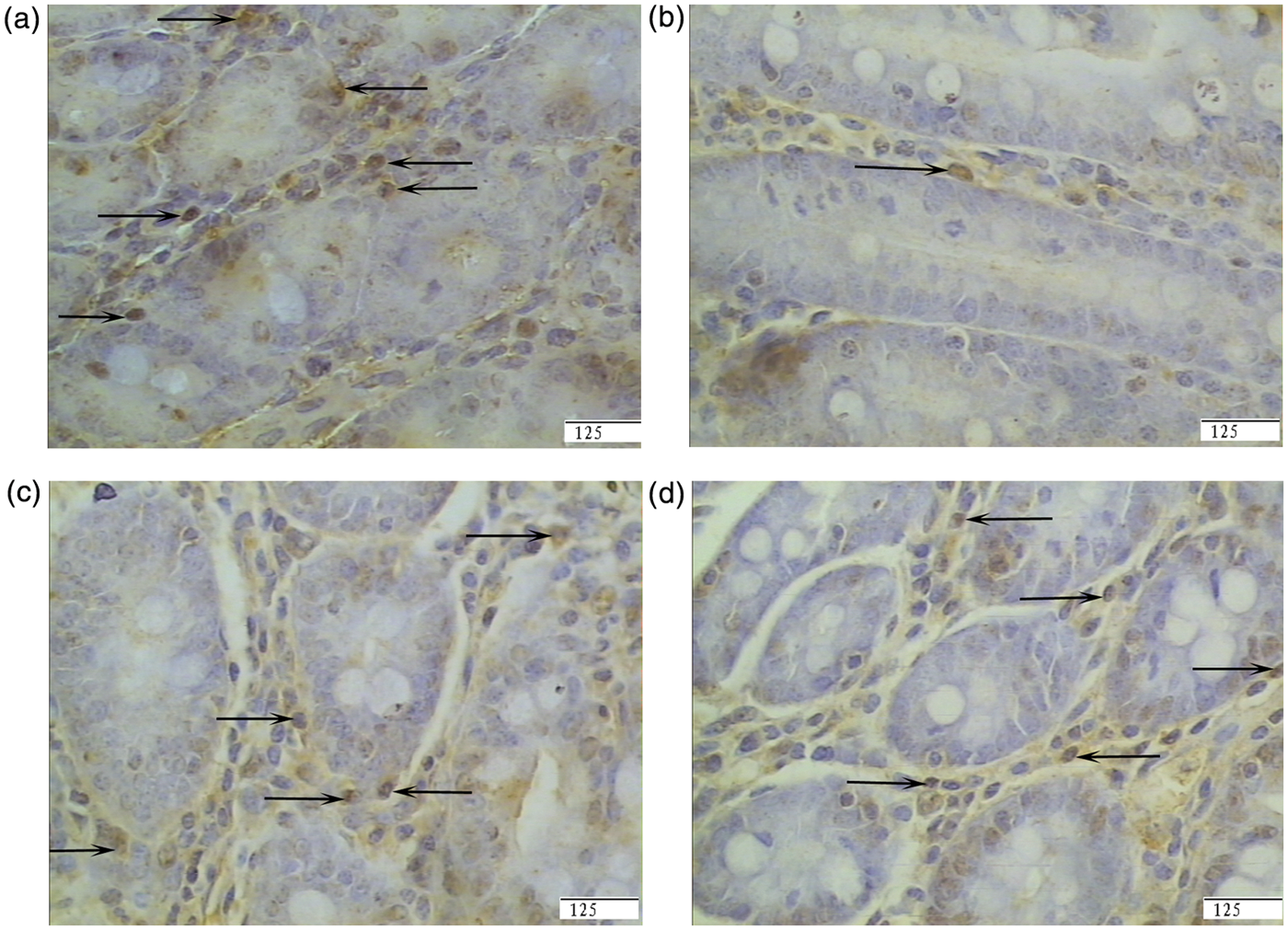
Intestinal CD4+ and CD8+ T cells
Effect of
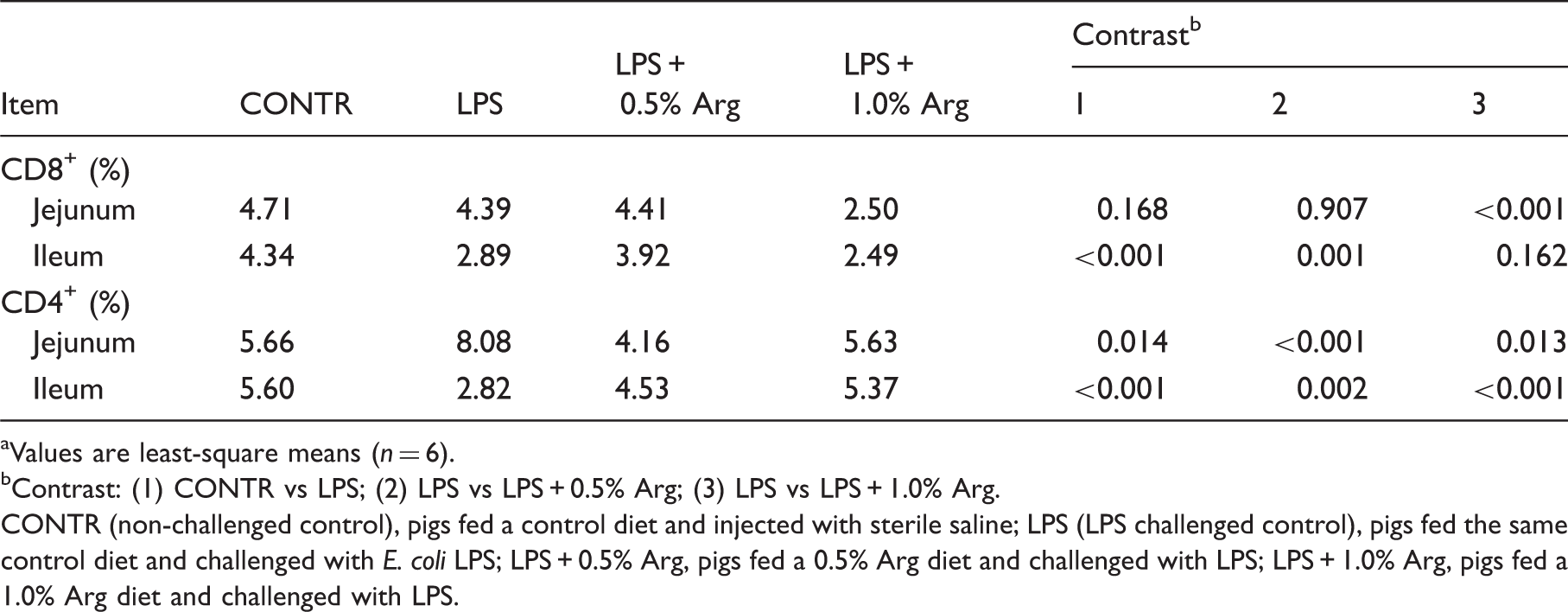
Values are least-square means (n = 6).
b Contrast: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg; (3) LPS vs LPS + 1.0% Arg.
CONTR (non-challenged control), pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control), pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg, pigs fed a 0.5% Arg diet and challenged with LPS; LPS + 1.0% Arg, pigs fed a 1.0% Arg diet and challenged with LPS.
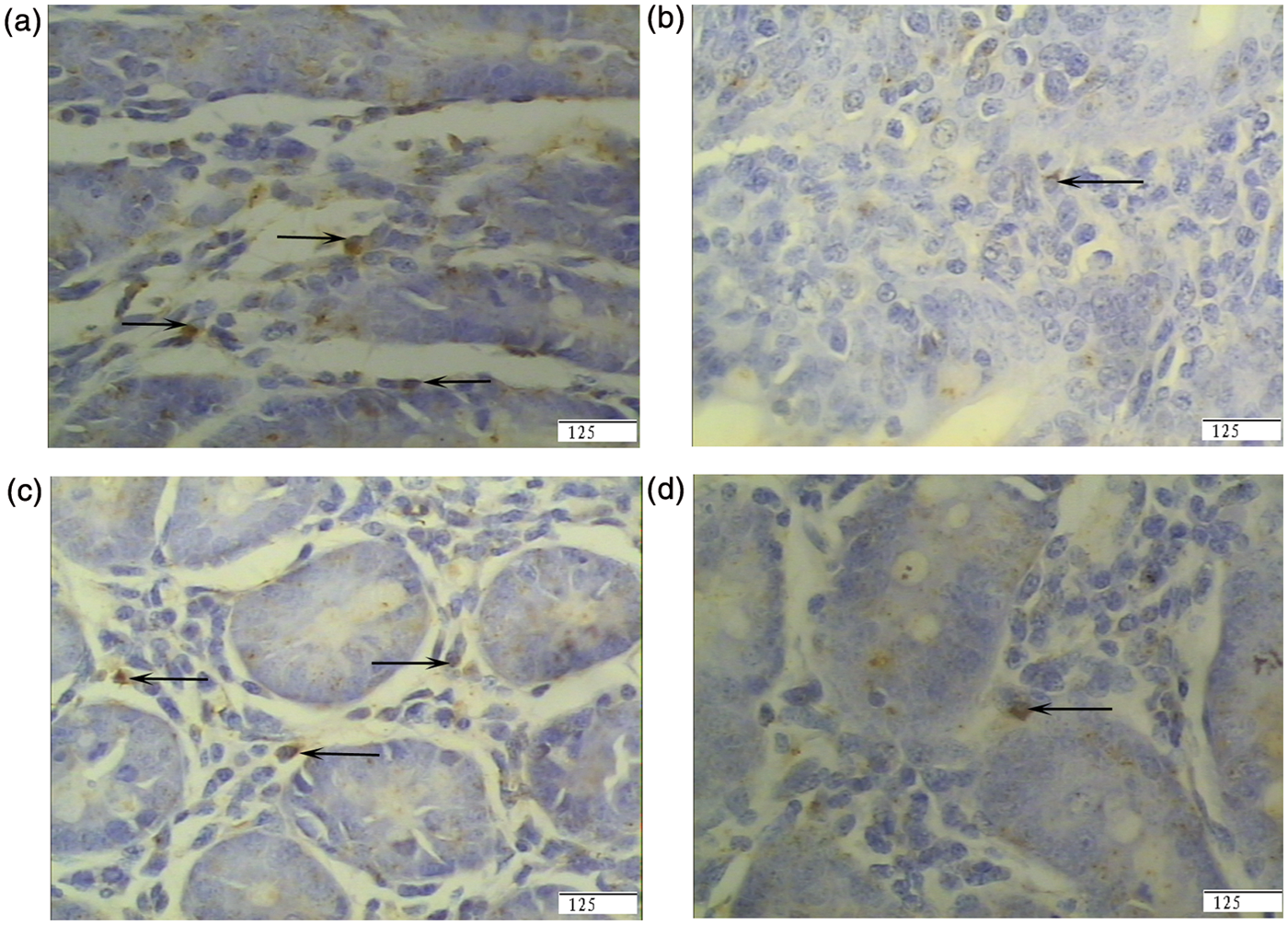
Representative photomicrographs of pig ileal mucosa showing immunostained CD8+ T cells. (A) Pigs fed a control diet and injected with sterile saline (CONTR). (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). The number of CD8+ T cells was decreased in LPS pigs compared with CONTR pigs. Arg (0.5%) supplementation increased CD8+ T cells in the ileum compared with LPS pigs. Original magnification 400 ×. Scale bar = 125 µm.
The number of jejunal CD4+ T cells in LPS pigs was higher than that of CONTR pigs (P = 0.014) (Table 4). However, Arg supplementation inhibited jejunal CD4+T cell expansion relative to LPS pigs (0.5% Arg, P = 0.001; 1.0% Arg, P = 0.013). Compared with CONTR pigs, LPS challenge decreased the number of CD4+ T cells in the ileum significantly (50% lower, P < 0.001) (Figure 5). Arg supplementation increased ileal CD4+ T cell number (0.5% Arg, P = 0.002; 1.0% Arg, P = 0.001) compared with LPS pigs (Figure 5).
Representative photomicrographs of pig ileal mucosa showing immunostained CD4+ T cells. (A) Pigs fed a control diet and injected with sterile saline (CONTR). (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). The number of CD4+ T cells was decreased in LPS pigs compared with CONTR pigs. Arg (0.5% or 1.0%) supplementation increased CD4+ T cells in ileum compared with LPS pigs. Original magnification 400 ×. Scale bars = 125 µm.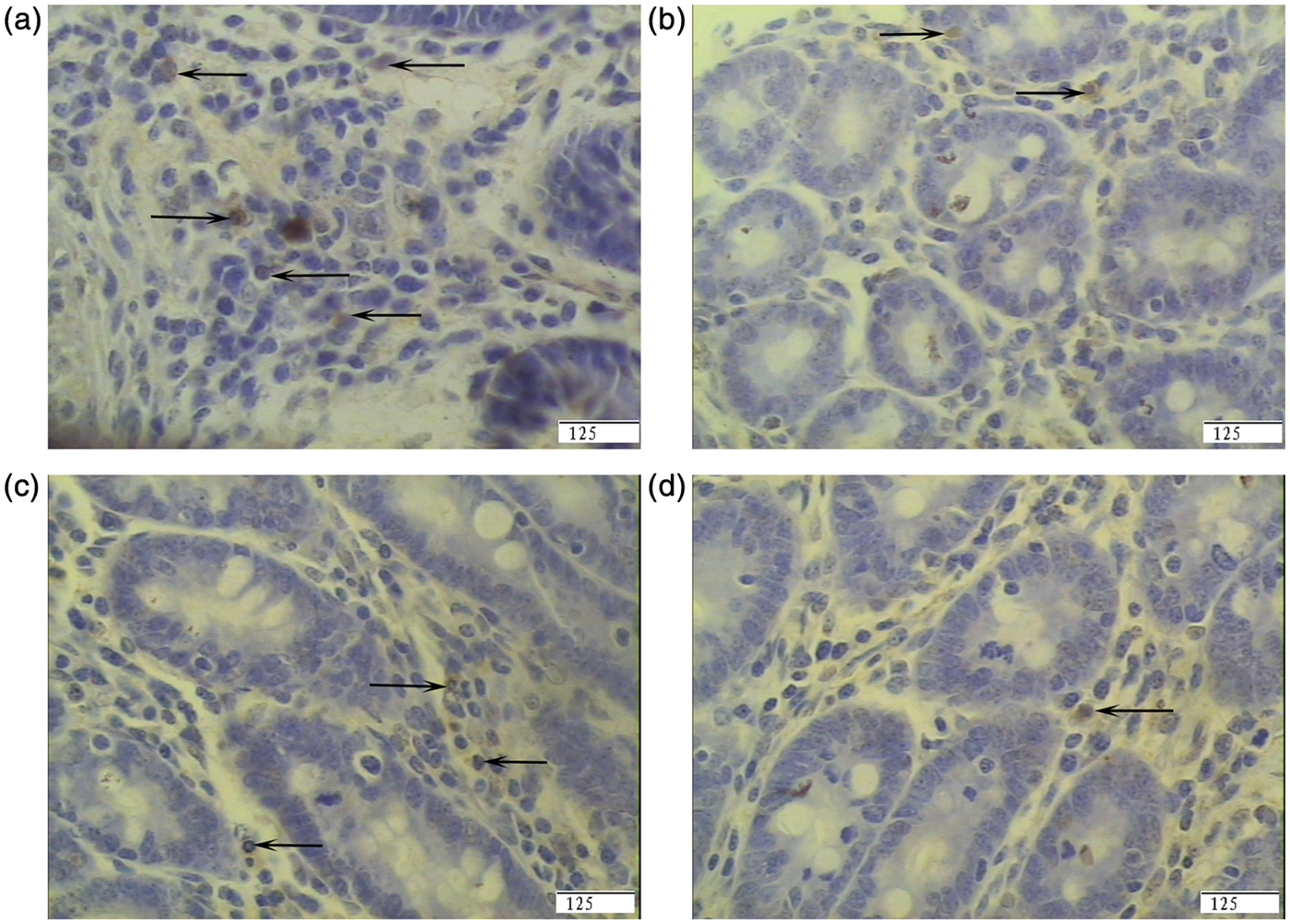
Lymphocyte proliferation and apoptosis in intestinal Peyer’s patches
Effect of
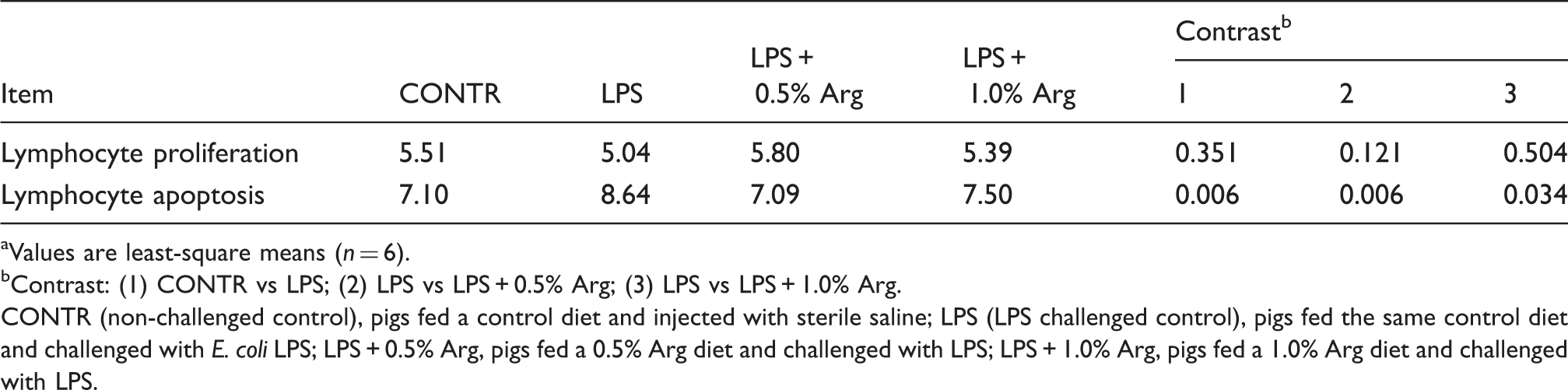
Values are least-square means (n = 6).
Contrast: (1) CONTR vs LPS; (2) LPS vs LPS + 0.5% Arg; (3) LPS vs LPS + 1.0% Arg.
CONTR (non-challenged control), pigs fed a control diet and injected with sterile saline; LPS (LPS challenged control), pigs fed the same control diet and challenged with E. coli LPS; LPS + 0.5% Arg, pigs fed a 0.5% Arg diet and challenged with LPS; LPS + 1.0% Arg, pigs fed a 1.0% Arg diet and challenged with LPS.
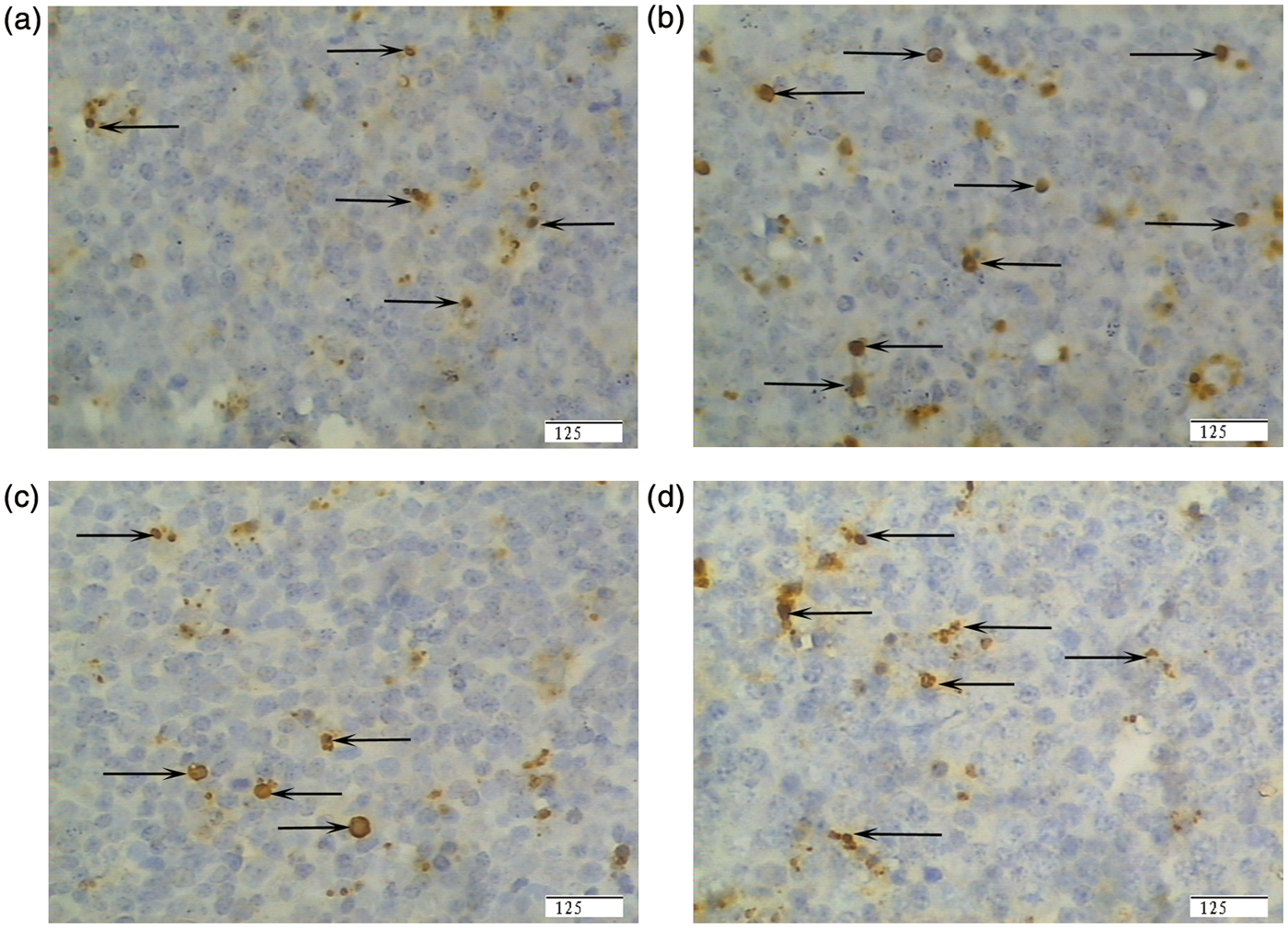
Representative photomicrographs of pig intestinal Peyer’s patches showing immunostained lymphocyte apoptosis. (A) Pigs fed a control diet and injected with sterile saline (CONTR). (B) Pigs fed the same control diet and challenged with E. coli LPS (LPS). (C) Pigs fed a 0.5% Arg diet and challenged with LPS (LPS + 0.5% Arg). (D) Pigs fed a 1.0% Arg diet and challenged with LPS (LPS + 1.0% Arg). LPS challenge increased lymphocyte apoptosis compared with CONTR pigs. Arg (0.5% or 1.0%) supplementation decreased lymphocyte apoptosis compared with LPS pigs. Original magnification 400 ×. Scale bar = 125 µm.
Discussion
The present experiment was conducted to evaluate intestinal mucosal immune response to E. coli LPS challenge and to determine if Arg supplementation could improve intestinal mucosal immune barrier function in weaned pigs. LPS, a potent endotoxin, can enter the systemic circulation via the gastrointestinal tract to cause inflammation and sickness. 24 In addition, in vivo and in vitro studies in animals and humans indicate that LPS can cause damage to intestinal barrier function,25,26 thereby causing diarrhea and low food intake. Consequently, a LPS model has been used for studying the effects of dietary regimes and stressors on the immunity of weaned pigs.27,28
In the present study, we observed that LPS caused epithelium lifting and villous atrophy. This is in accordance with most of the published studies on the effect of LPS on intestinal morphology.1,17,26 In our conventional outlook it is assumed that epithelium lifting and atrophy of villi are accompanied by impaired nutrient absorption and increased permeability in intestinal mucosa.29,30 Our study showed that Arg alleviated villous epithelium lifting caused by LPS, and increased villous height and villous area. Similarly, Sukhotnik et al. 15 reported that oral Arg reduced gut mucosal injury caused by LPS in rat. After acute mucosal injury, rapid restoration of epithelial continuity depends on migration of uninjured epithelial cells to cover denuded sections of basement membrane. Arg stimulates intestinal cell migration and enhances intestinal restitution. 31 Therefore, Arg protected the integrity of intestinal mucosa, which was a key element in maintaining gut barrier function.
The gastrointestinal tract is not only an organ for nutrient digestion and absorption, but also an organ for systemic immunity and barrier function. The immune system plays a major role in protecting the host from the external environment. IELs are the first immune cells to be exposed to foreign Ags and have the potential to be critical in the host defense. 30 In the present study, 0.5% Arg supplementation resulted in a numerical increase in IEL number. Currently, there are few studies on the regulation of IEL through dietary Arg supplementation. Sukhotnik et al. 15 reported that Arg decreased enterocyte apoptosis induced by LPS in rat, which suggests that Arg exerted a beneficial influence on intestinal inflammatory response. Moreover, Arg increases specific lymphocyte subsets in the gut-associated lymphoid tissue and enhances the proliferative response of IEL. 32 In addition, previous studies in mice showed that a decrease in the number of IELs was associated with decreasing tight junction integrity in intestine. 33 This finding supported the current study that Arg maintained epithelial barrier integrity by inducing the proliferation of IELs. We also found that 1.0% Arg reduced ileal IEL numbers. The reason for the discrepancy might be that Arg efficacy was abolished at supraphysiological levels. 31
In order to understand the effect of Arg on the distribution of CD4+ and CD8+ T cells in intestinal mucosa, subpopulations of lymphocytes were evaluated. Our study showed that 0.5% Arg attenuated a LPS-induced CD8+ T cell number decrease. Abdukalykova et al. 34 also showed that Arg supplementation enhanced CD8+ T cells of peripheral blood proliferation in chickens after bursal disease virus vaccination. CD8+ T cells display cytotoxic activity, which plays an important defensive role in preventing microbial invasion. It was reported that the increase in CD8+ T cells was correlated with decreased infection. 35 In the present study, Arg may have improved immune barrier function of the intestinal mucosa by increasing the number of CD8+ T cells—which may be beneficial host resistance.
In this study, LPS administration decreased CD4+ T cell and ASC numbers in the ileum. Similarly, Kono et al. 36 and Ding et al. 37 found that LPS reduced secretory IgA levels in plasma and small intestinal mucosa in rat. Arg supplementation increased the numbers of CD4+ T cells and ASCs. In agreement with our findings, Fan et al. 38 reported Arg supplementation resulted in a numerical increase in CD4+ T cells of Peyer’s patches in severely burned mice. Moreover, previous studies showed that Arg supplementation enhanced the numbers of ASCs and the secretion of intestinal IgA in burned rats.10,39 CD4+ T cell, the T helper cell, plays an important role in stimulating ASC activation. Activated ASCs produce intestinal mucosal secretory IgA. Intestinal mucosal secretory IgA is the primary specific immunologbulin that protects the intestinal mucosal surfaces. 38 In the present study, Arg attenuated the decrease of CD4+ T cell and the number of ASCs induced by LPS. It indicated that Arg supplementation enhanced proliferation of CD4+ T cells and ASCs, which might, consequently, protect the immune barrier function of intestinal mucosa. In this study, LPS challenge resulted in higher jejunal CD4+ T cell numbers compared with CONTR pigs. However, LPS did not affect ASC numbers in the jejunum. It is a complex process for ASCs in the intestine to proliferate and produce secretory Abs. It needs more factors to participate. Thus, the number of CD4+ T cells is not always correlated directly with the number of ASCs. Interestingly, LPS increased the number of CD4+ T cells in the ileum. The variable response in the number of CD4+ T cells and ASCs between the jejunum and ileum was unknown—further study is needed to elucidate it.
It is well known that IELs, mast cells and goblet cells are the major intestinal immunocompetent cells. Mast cells are positioned strategically at the epithelial barrier, signifying their important role in mucosal surveillance. 5 Our histological study showed that LPS increased the number of mast cells in intestinal mucosa. The number of mast cells increased along with mast cell activation in the jejunum of diarrhea-predominant irritable bowel syndrome patients. 40 Mast cell activation leads to degranulation and release of several mediators, such as histamine, eicosanoids, cytokines and chemokines, which injure the intestinal structure and increase intestinal permeability. 41 Interestingly, our data showed Arg alleviated the increase of the number of mast cells induced by LPS. Similarly, Kwasniewski et al. 42 indicated that Arg reduced the intestinal hermorrhagic lesions associated with allergy, which was triggered by mast cells and platelet-activating factor. In the present study, Arg might have stabilized the mast cells and alleviated the effect of LPS-induced mast cell increase.
In this study, Arg supplementation attenuated the increase of Peyer’s patch lymphocyte apoptosis caused by the LPS challenge. Previous studies also demonstrate that Arg supplementation promotes proliferation of lymphocytes in Peyer’s patches in severely burned mice. 35 Peyer’s patches act as the primary inductive sites for mucosal immunity, where naive lymphocytes are sensitized, migrate via the thoracic duct to the lamina propria of various sites and perform the mucosal immune function. 43 Increased lymphocyte apoptosis in Peyer’s patches means a decrease in the number of Peyer’s patch lymphocytes, which may impair the intestinal immune barrier. Therefore, our findings indicate that Arg could alleviate the intestinal immune barrier injury caused by the LPS via decreasing Peyer’s patch lymphocyte apoptosis.
In conclusion, our study demonstrated that Arg supplementation modulated intestinal inflammatory response to LPS by increasing the number of IELs, CD4+ T cells, CD8+ T cells and ASCs, and decreasing Peyer’s patch cell apoptosis and mast cell number in the intestinal mucosa of weaned pigs. These findings suggest that Arg supplementation protects and improves mucosal immune barrier function and maintains normal intestinal integrity.
Footnotes
Funding
The authors express their gratitude to the National Natural Science Foundation of China (31172222 and 30972109), the Program for New Century Excellent Talents in University (NCET-10-0158), the Key Project of Chinese Ministry of Education (209082), the Project of Natural Science Foundation of Hubei Province (2010CDA050 and 2009CDB006), the Project of International Cooperation of Hubei Province (2011BFA008), the Hubei Provincial Department of Education (Q20111702), and Wuhan Science and Technology Program (201171034320) for the financial supports.
Conflict of interest
The authors declare there are no conflicts of interest.
