Abstract
Hyaluronan (HA) oligosaccharides stimulate pro-inflammatory responses in different cell types by modulating both cluster determinant 44 (CD44) and TLR4. The activation of these receptors is also mediated by collagen-induced arthritis (CIA) that, via two different pathways, culminates in the liberation of NF-κB. This then stimulates the production of pro-inflammatory cytokines, including IL-18 and IL-33, that are greatly involved in rheumatoid arthritis. The aim of this study was to investigate the effects of 6-mer HA oligosaccharides on mouse synovial fibroblasts obtained from normal DBA/J1 mice or mice subjected to CIA. Compared with normal synovial fibroblasts (NSF), rheumatoid arthritis synovial fibroblasts (RASF) showed no up-regulation of CD44 and TLR4 mRNA expression and the related proteins, as well as no activation of NF-κB. Very low levels of both mRNA and related proteins were also detected for IL-18 and IL-33. Treatment of NSF and RASF with 6-mer HA oligosaccharides significantly increased all the parameters in both fibroblast groups, although to a greater extent in RASF. The addition of hyaluronan binding protein to both NSF and RASF inhibited HA activity and was able to reduce the effects of 6-mer HA oligosaccharides and the consequent inflammatory response.
Introduction
Hyaluronan (HA), a common component of connective tissue, can be depolymerized into small oligosaccharides via oxygen radicals and enzymatic degradation by hyaluronidases, β-glucuronidases, chondroitinases and hexosaminidases. 1 Similarly, inflammatory cytokines, such as TNF-α, IFN-γ and IL-1β, can stimulate fibroblasts to produce increased numbers of HA fragments.2,3 In its highly polymerized form, HA is believed to play a homeostatic role. However, in the context of tissue destruction, highly polymerized HA is broken down into its lower molecular mass components, which are able to induce the inflammatory response.4–6
Cluster determinant 44 (CD44) is the best-known receptor of HA. 7 CD44 is a trans-membrane glycoprotein that is widely distributed on leukocytes and several other cell types. The stimulation of CD44 with HA plays a role in various physiologic functions, as well as in pathologic processes, such as inflammation and metastasis. 8 Initial studies indicated that inflammatory signaling from HA degradation products involved CD44 alone. 8 However, subsequent works reported other signaling pathways to be involved, particularly through TLRs.9,10
TLRs are a class of proteins that play a key role in the innate immune system. Although each TLR recognizes specific ligands, they may trigger other molecules, such as small HA oligosaccharides which are able to cause the release of pro-inflammatory cytokines.9,10
Recently, several reports have shown that small fragments of HA or HA at low molecular mass can interact with both TLR4 and CD44, thereby stimulating inflammation or increasing the inflammatory mechanism previously induced by other agents in different cell types.11,12
The hallmark of rheumatoid arthritis (RA) is synovial inflammation; this directly drives extracellular matrix (ECM) degradation with consequent joint cartilage destruction and bone alterations. 13 There are no long-term effective treatments to counteract RA and current research is focused on understanding how the imbalance between specific ECM molecules may influence disease progression. Evidence is emerging to show that endogenous ECM molecules supply signals to damaged cartilage and chondrocytes in order to promote further cartilage degradation.6,14
The collagen-induced arthritis (CIA) model is a widely used experimental animal model of inflammation polyarthritis with clinical and pathological features that are similar to those of human RA and are dependent on both humoral and cellular immunity to the immunizing antigen. 15 Human rheumatoid arthritis synovial fibroblast (RASF) cultures originated from RA patients are able to activate a greater inflammatory response after stimulation with different cytokines than in normal synovial fibroblasts (NSF),16,17 as well as in RASF obtained from CIA mice. 18 In this context, CIA has the ability to promote inflammation by inducing the release of reactive oxygen species, cytokines, such as TNF-α and IL-1β, and destructive enzymes like matrix metalloproteases (MMPs). IL-18 and IL-33 are recognized as members of the IL-1 family. Interestingly, they exhibit closest sequence homology to each other. 19 IL-18 seems to play a role in a variety of early inflammatory responses and has been identified in different cell types, including dendritic cells and synovial fibroblasts.20,21 IL-33 has been identified as a potent inflammatory cytokine and it is also widely expressed in many tissues, including synovial fibroblasts. 21 Both of these cytokines seem to be responsible for initiating inflammation by activating other inflammatory cytokines, such as IL-1β, IL-6, TNF-α and IFN-γ.21,22
The objectives of the current study were to investigate the inflammatory effects of small fragments of HA (6 mer HA) in NSF obtained from normal DBA/J1 mice and RASF obtained from DBA/J1 mice subjected to CIA, with particular regard to IL-18 and IL-33 production.
Materials and methods
Animals
Male mice DBA/J1 6–7 weeks old with a mean weight of 25–30 g were used in our study. The mice, purchased from Harlan (Correzzana, Italy), were kept under climate-controlled conditions with a 12-h light/dark cycle. The animals were fed standard rodent chow and provided water ad libitum. The health status of the animal colony was monitored in accordance with Italian Veterinary Board guidelines. Mice were divided into two groups: (1) Control (n = 7) and (2) CIA (n = 10).
Materials
HA 6-mer oligosaccharides as sodium salt (Cat. CSR-11007) were obtained from Cosmo Bio Co. Ltd (Tokyo, Japan). Medium molecular mass HA (MHA), Select-HA™ hyaluronan 1000 K, molecular mass 800–1200 ku (cat S9825), was provided by Sigma-Aldrich Srl, (Milan, Italy). The products were endotoxin-free (endotoxin content <0.1 ng/mg). Mouse TLR4 (cat. ABIN424269) and CD44 (cat. ABIN457122) commercial ELISA kits were provided by Antibodies-online.com GmbH, (Aachen, Germany). HA binding protein (HAPB) (cat H7980-30) was supplied by US Biological (Swampscott, MA, USA). Mouse IL-18 (cat. 7625) and IL-33 (cat. M3300) ELISA kits were purchased from R&D Systems (Minneapolis, MN, USA). All other reagents used were purchased from Fluka (division of Sigma-Aldrich Srl).
Induction of CIA and arthritis evaluation
Mice were injected intradermally at the tail base with 100 μl of 2.0 mg/ml type II collagen emulsion in Freund’s complete adjuvant containing 2.5 mg/ml heat-killed Mycobacterium tuberculosis H37Ra. Mice were immunized a second time 7 days later.
The severity of the arthritis in each limb was graded daily on a scale of 0–4 as follows: 0, no macroscopic signs of arthritis; 1, swelling of one group of joints (e.g. knee or ankle joints); 2, two groups of swollen joints; 3, three groups of swollen joints; 4; swelling of the entire limb. The maximum score for each mouse was 16. Clinical severity was also assessed by quantifying changes in limb volume. Measurements were performed using a dial gauge caliper. At the end of the experimental period (day 35), the mice with maximum score (16) were collected, anesthetized with ethyl ether and then sacrificed to remove their hind limbs.
Isolation and culture of synovial fibroblasts
Synovial fibroblasts were isolated and cultured as previously described.
23
In brief, on day 35, the synovium of the knee joint from control mice and CIA mice was dissected and then digested with 1.0 mg/ml collagenase in DMEM with 1% v:v FCS medium at 37°C for 2 h. The tissues were pooled, digested and then suspended and passed through nylon mesh, in order to remove tissue debris. Dissociated cells were then washed with DMEM medium containing 50.0 μ
Synovial fibroblasts were plated at a density of 105 cells/well in a 96-well plate in DMEM medium with 1% v:v FCS and cultured either with or without the addition of 6 mer HA. Synovial fibroblasts cultures were used between the forth and the ninth passage, during which time they constituted a homogeneous population of cells.
6-mer HA treatment
Twelve hours after plating (time 0), 6-mer HA was added to both NSF and RASF at two different doses of 10 and 20 µg/ml. The cells and medium underwent biochemical evaluation 24 h later. As it was for experimental use, 6-mer HA was diluted to a final concentration of 10–20 µg/ml, with a maximum estimated endotoxin content of 0.002 ng/ml for a 20 µg/ml concentration. The study therefore included the following groups of cells: a first set of six groups of NSF (NSF; NSF + MHA 20 µg/ml; NSF + HABP; NSF + HA 10 µg/ml; NSF + HA 20 µg/ml; NSF + HA 20 µg/ml + HABP) and a second set of six groups of RASF treated in the same way (RASF; RASF + MHA 20 µg/ml; RASF + HABP; RASF + HA 10 mg/ml; RASF + HA 20 µg/ml; RASF + HA 20 µg/ml + HABP). A 250 µg/ml dose of HABP was added to NSF or RASF 5 min before the 6-mer HA treatment.
RNA isolation, cDNA synthesis and real-time quantitative PCR amplification
Total RNA was isolated from NSF and RASF for reverse-PCR real time analysis of TLR4, CD44, IL-18 and IL-33 (RealTime PCR system, Mod. 7500, Applied Biosystems, Foster City, CA, USA) using an Omnizol Reagent Kit (Euroclone, West York, UK). The first strand of cDNA was synthesized from 1.0 µg total RNA using a high capacity cDNA Archive kit (Applied Biosystems). β-Actin mRNA was used as an endogenous control to allow the relative quantification of TLR4, CD44, IL-18 and IL-33. Real-time PCR was performed by means of ready-to-use assays (Assays on demand, Applied Biosystems) on both targets and endogenous controls. The amplified PCR products were quantified by measuring the calculated cycle thresholds (CT) of TLR4, CD44, IL-18, IL-33, and β-actin mRNA. The CT values were plotted against the log input RNA concentration in serially-diluted total RNA of the synovial fibroblast samples and used to generate standard curves for all mRNAs analyzed. The amounts of specific mRNA in samples were calculated from the standard curve and normalized with β-actin mRNA. After normalization, the mean value of NSF levels became the calibrator (one per sample) and the results are expressed as the n-fold difference relative to normal controls (relative expression levels).
TLR4 and CD44 ELISA assay
Samples of cell culture in the presence of protease inhibitor cocktail were first lysed using a specific lysing buffer and then centrifuged at 10,000 g at 4°C for 10 min. The analysis of CD44 and TLR4 was carried out using a specific commercial kit. In brief, 100 μl of standards, blank or samples were added to each well of each specific coated microplate. The microplates were then covered with the plate sealer and incubated for 2 h at 37°C. After incubation, the liquid from the wells was discarded without washing and 100 μl of Biotin-Ab was added. After covering with plate sealer, the microplates were further incubated for 60 min at 37°C. At the end of the incubation, the liquid was aspirated from the microplates and the wells were washed three times with wash buffer. Then, after adding 100 μl of HRP-avidin to each well, the microplates were again covered with plate sealer and incubated for 60 min at 37°C. After washing again three times and adding 90 μl of TMB substrate solution to each well, the microplates were allowed to incubate for a further 30 min at 37°C, with protection from light. After incubation and the addition of 50 μl of stop solution, the absorbance of each well was read spectrophotometrically at λ 450 nm. TLR4 values are expressed as ng/ml, while CD44 values are expressed as pg/ml.
NF-κB p50/65 transcription factor assay
NF-κB p50/65 DNA binding activity in nuclear extracts of NSF and RASF was evaluated in order to measure the degree of NF-κB activation. The analysis was carried out following the manufacturer’s protocol for a commercial kit (NF-κB p50/65 EZ-TFA Transcription Factor Assay Colorimetric, cat. no. 70-510; Millipore, Billerica, MA, USA). In brief, cytosolic and nuclear extraction was performed by lysing the cell membrane with an apposite hypotonic lysis buffer containing protease inhibitor cocktail and tributylphosphine (TBP) as reducing agent. After centrifugation at 8000 g, the supernatant containing the cytosolic fraction was stored at −80°C, while the pellet containing the nuclear portion was then re-suspended in the apposite extraction buffer and the nuclei were disrupted by a series of drawing and ejecting actions. The nuclei suspension was then centrifuged at 16,000 g. The supernatant fraction was the nuclear extract. After determining protein concentration and adjusting to a final concentration of approximately 4.0 mg/ml, this extract was stored in aliquots at −80°C for the subsequent NF-κB assay. After incubation with primary and secondary Abs, color development was observed following the addition of the substrate TMB/E. Lastly, the absorbance of the samples was measured using a spectrophotometric microplate reader set at λ 450 nm. Values are expressed as relative optical density (OD) per mg protein.
IL-18 and IL-33 ELISA assay
Samples of cell-secreted protein extracted from the culture media in the presence of 1 nM PMSF and protease inhibitor cocktail were centrifuged at 13,000 g, at 4°C for 10 min. The analysis of IL-18 and IL-33 was carried out using two specific commercial kits. Briefly, 100 μl or 50 μl of each sample, standards and control were added to each well of the coated microplate for the analysis of IL-18 or IL-33 respectively. After 60 min or 120 min incubation at room temperature (25°C), (for IL-18 or IL-33 respectively) the supernatant of each well was aspirated and the plate was washed four times. Wells were filled with 100 μl of each specific peroxidase-conjugate antibody. After 60 min or 120 min incubation at room temperature (for IL-18 and IL-33 respectively) the liquid from the wells was discarded and the plate washed four times. One-hundred microliters of substrate solution was then added to each well. After a further 30 min incubation of all plates, 100 μl of a stop solution was added to each well. Within 30 min, the absorbance was then read spectrophotometrically at λ 450 nm. IL-18 and IL-33 values are expressed as pg/ml.
Protein determination
The amount of protein was determined using the Bio-Rad protein assay system (Bio-Rad Lab., Richmond, CA, USA) with BSA as a standard in accordance with the published method. 24
Statistical analysis
Data are expressed as the mean ± SD values of at least seven experiments for each test. All assays were repeated three times to ensure reproducibility. Statistical analysis was performed by ANOVA followed by the Student-Newman-Keuls test. The statistical significance of differences was set at P < 0.05.
Statement of animal care
The studies reported in this manuscript have been performed in accordance with the Helsinki declaration and the NIH guidelines for the Care and Use of Laboratory Animals.
Results and discussion
TLR4 and CD44 mRNA evaluation (Figures 1 and 2, panel A) and ELISA assay (Figures 1 and 2, panel B) were performed in order to estimate the degree of TLR4 and CD44 activation in the presence/absence of 6-mer HA in NSF and RASF. The results showed a marked dose-dependent increase in mRNA expression and the protein synthesis of both the TLR4 and CD44 receptors in NSF treated with 6-mer HA (Figures 1 and 2). In NSF, the addition of HABP together with 6-mer HA abolished the increment in TLR4 and CD44 expression and the related protein production exerted by 6-mer HA. Compared with NSF, RASF showed no difference in CD44 and TLR4 mRNA expression and related protein production. The addition of 6-mer HA to RASF increased TLR4 and CD44 expression and also protein production to a greater extent than for NSF treated with 6-mer HA. Once again, the addition of HABP to RASF treated with HA also significantly reduced the expression of the two receptors. These results show that 6-mer HA, by interacting with TLR4 and CD44, was able to increase TLR4 and CD44 expression and protein synthesis in NSF. In RASF, however, the basal expression of TLR4 and CD44 was unchanged with respect to NSF, the addition of 6-mer HA produced an over-expression of these two receptors, as if they had previously been up-regulated by CIA. The addition of the HABP was also able to reduce 6-mer HA effects in NSF and also in RASF, but to a lesser extent. The reduction of TLR4 and CD44 expression, stimulated by 6-mer HA, in both NSF and RASF through the addition of HABP confirms the involvement of 6-mer HA fragments in the mediation of inflammatory activation of TLR4 and CD44. The evidence that TLR4 and CD44 expression was not fully abolished in RASF (as it was in NSF) may be explained by the fact that other molecules may take part in the activation of these receptors. No effect was observed by treating both NSF and RASF with MHA.
Effect of 6-mer HA treatment on NSF and RASF TLR4 mRNA expression (A) and related protein production (B) in normal mice or mice subjected to CIA. Values are the mean ± SD of no less than seven experiments and are expressed as the n-fold increase with respect to the Control (A) and as ng/ml (B) for the TLR4 protein levels. MHA = HA at medium molecular mass (1000 ku). *P < 0.001 vs Control; °P < 0.001 vs NSF + HA; #P < 0.001 vs RASF; ^P < 0.001 vs RASF + HA. Effect of 6-mer HA treatment on NSF and RASF CD44 mRNA expression (A) and related protein production (B, C) in normal mice or mice subjected to CIA. Values are the mean ± SD of no less than seven experiments and are expressed as the n-fold increase with respect to the Control (A) and as pg/ml (B) for the CD44 protein levels. MHA = HA at medium molecular mass (1000 ku). *P < 0.001 vs Control; °P < 0.001 vs NSF + HA; #P < 0.001 vs RASF; ^P < 0.001 vs RASF + HA.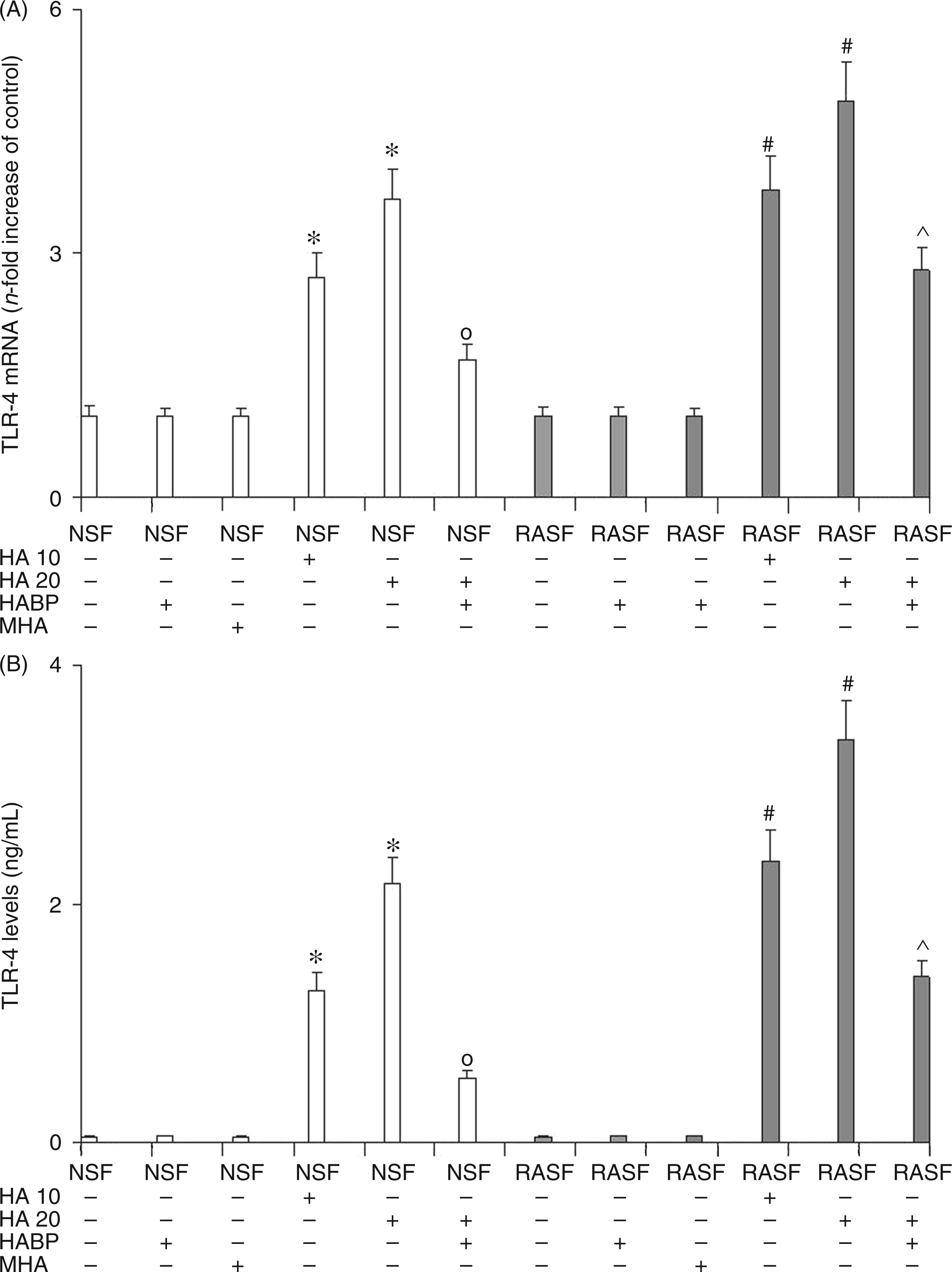
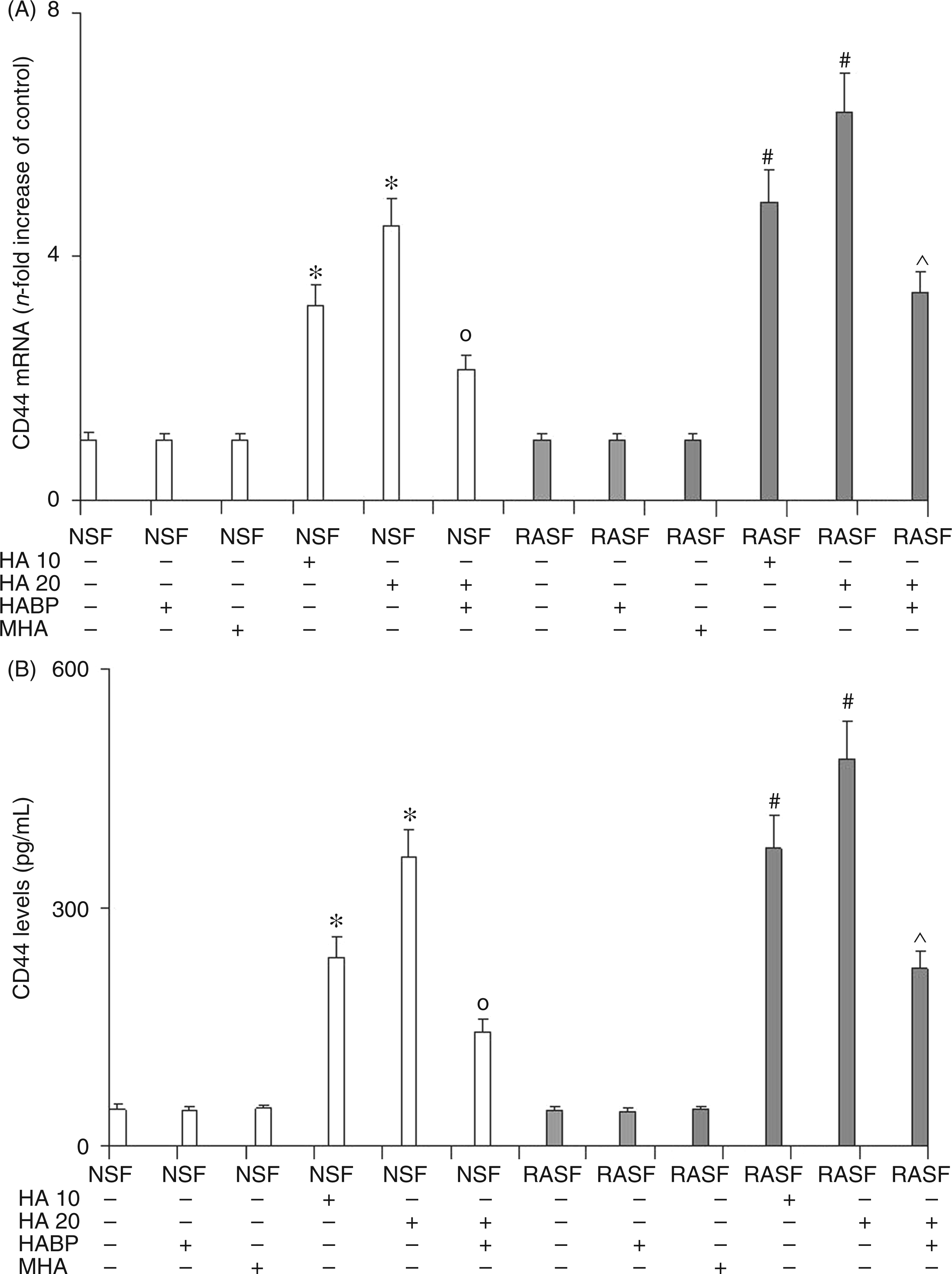
Figure 3 shows the changes in the NF-κB p50/p65 heterodimer translocation in NSF and RASF over the entire course of the experiment. This assay was also carried out in order to estimate the starting point of the inflammatory process, as NF-κB factor can be activated by both the TLR4 and CD44 pathways and these, in turn, may converge to stimulate the expression of several genes that prime/amplify inflammation. The results obtained from evaluating NF-κB factor confirmed that 6-mer HA was able to activate NF-κB expression in a significant dose-dependent manner via both TLR4 and CD44 receptors. Once again, the addition of HABP abolished the effect of 6-mer HA on NSF while, in RASF, HA effects were only reduced. The reduction of 6-mer HA effects caused by adding HABP confirms the involvement of NF-κB in the mediation of the TLR4 and CD44-activated inflammatory response.
Effect of 6-mer HA treatment on NSF and RASF NF-κB p50/65 transcription factor DNA binding activity in normal mice or mice subjected to CIA. White bars represent the p/50 subunit, grey bars represent the p/65 subunit. Values are the mean ± SD of no less than seven experiments and are expressed as OD at λ 450 nm/mg protein of nuclear extract. MHA = HA at medium molecular mass (1000 ku). *P < 0.001 vs Control; °P < 0.001 vs NSF + HA; #P < 0.001 vs RASF; ^P < 0.001 vs RASF + HA.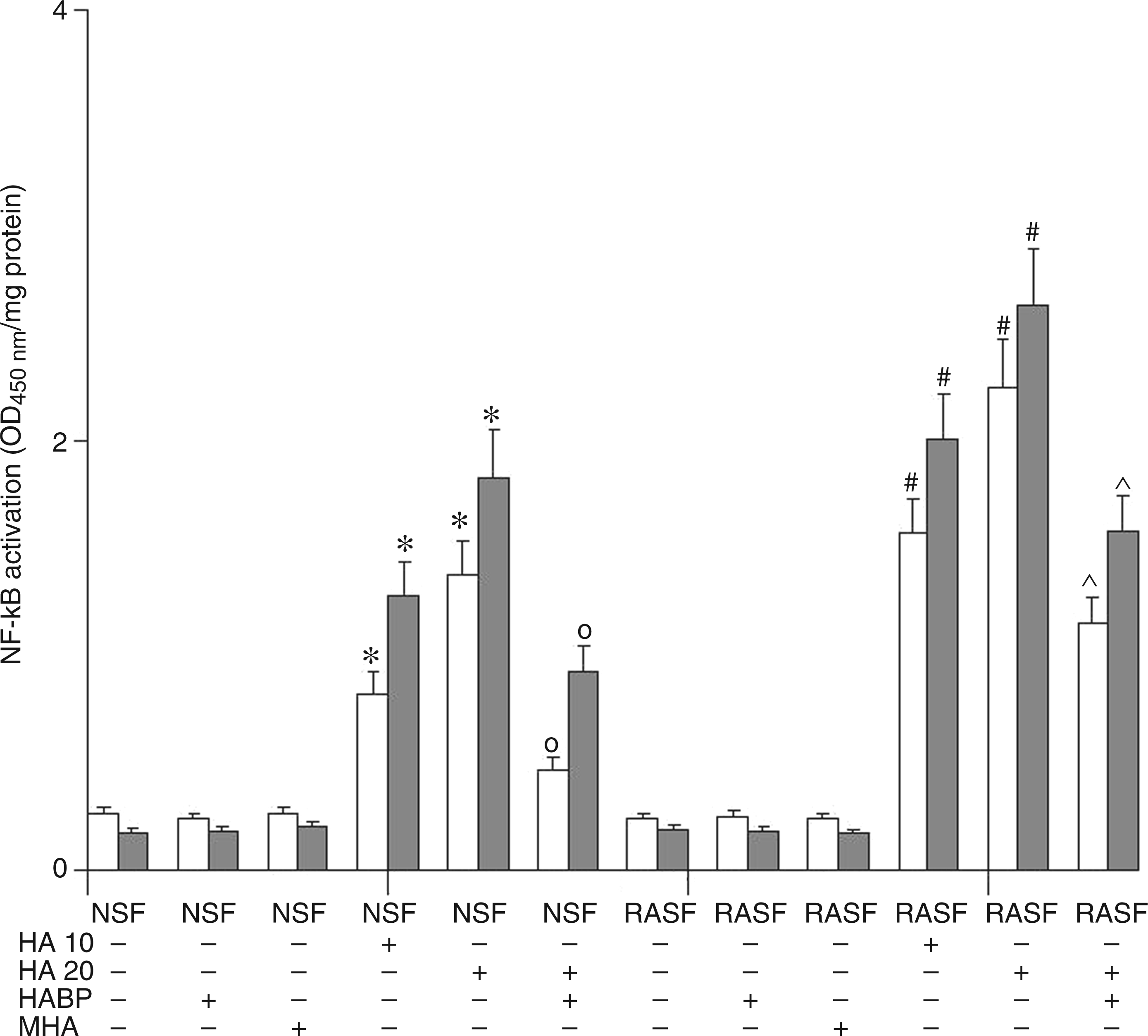
IL-18 (Figure 4) and IL-33 (Figure 5) mRNA evaluation (panel A), and ELISA assay (panel B) confirmed the previous data obtained by evaluating NF-κB factor. In fact, these results showed a marked dose-dependent increase in the expression and protein synthesis of both these inflammatory cytokines in NSF treated with 6-mer HA. This increase is a direct consequence of NF-κB activation by 6-mer HA-induced stimulation of TLR4 and CD44 receptors. The treatment of NSF with HABP was able to reduce the levels of these inflammatory cytokines as a consequence of the reduction in 6-mer HA-induced stimulation of TLR4 and CD44 receptors and therefore of NF-κB activation. Analogous results were obtained in RASF where Il-18 and IL-33 levels rose significantly after the addition of 6 mer HA to a greater extent than the increase seen in NSF. The addition of HABP was, again, able to limit, but not to abolish, increased IL-18 and IL-33 expression in RASF. The reduction of IL-18 and IL-33 expression and protein levels exerted by the addition of HABP in NSF and RASF treated with HA, confirms the key role played by 6-mer HA fragments on the mediation of inflammation during RA. Also, for IL-18 and IL-33, no effect was observed by treating both NSF and RASF with MHA.
Effect of 6-mer HA treatment on NSF and RASF IL-18 mRNA expression (A) and related protein production (B) in normal mice or mice subjected to CIA. Values are the mean ± SD of no less than seven experiments and are expressed as the n-fold increase with respect to the Control (A) and as pg/ml (B) for the IL-18 protein levels. MHA = HA at medium molecular mass (1000 ku). *P < 0.001 vs Control; °P < 0.001 vs NSF + HA; #P < 0.001 vs RASF; ^P < 0.001 vs RASF + HA. Effect of 6-mer HA treatment on NSF and RASF IL-33 mRNA expression (A) and related protein production (B) in normal mice or mice subjected to CIA. Values are the mean ± SD of no less than seven experiments and are expressed as the n-fold increase with respect to the Control (A) and as pg/ml (B) for the IL-33 protein levels. MHA = HA at medium molecular mass (1000 ku). *P < 0.001 vs Control; °P < 0.001 vs NSF + HA; #P < 0.001 vs RASF; ^P < 0.001 vs RASF + HA.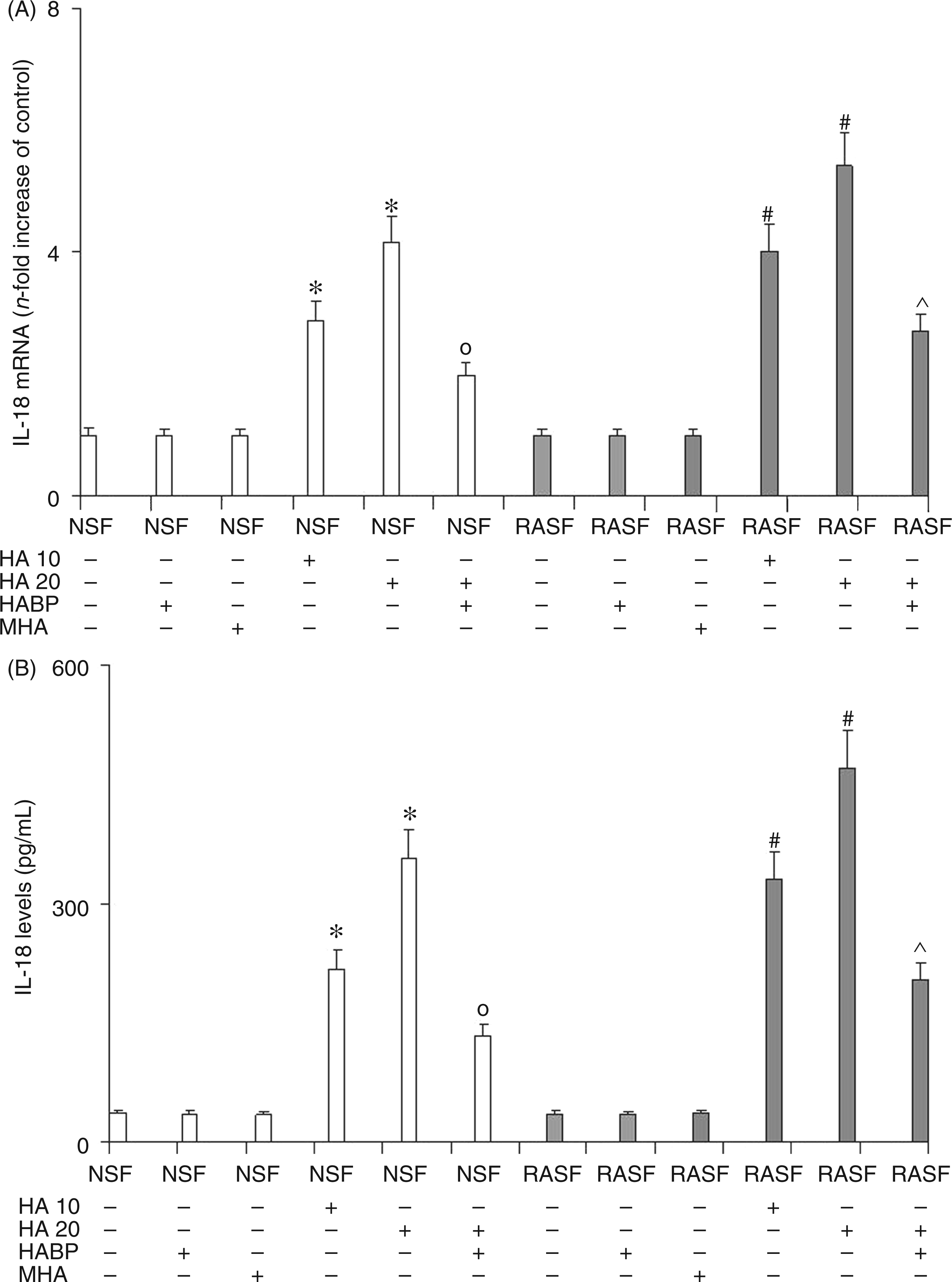
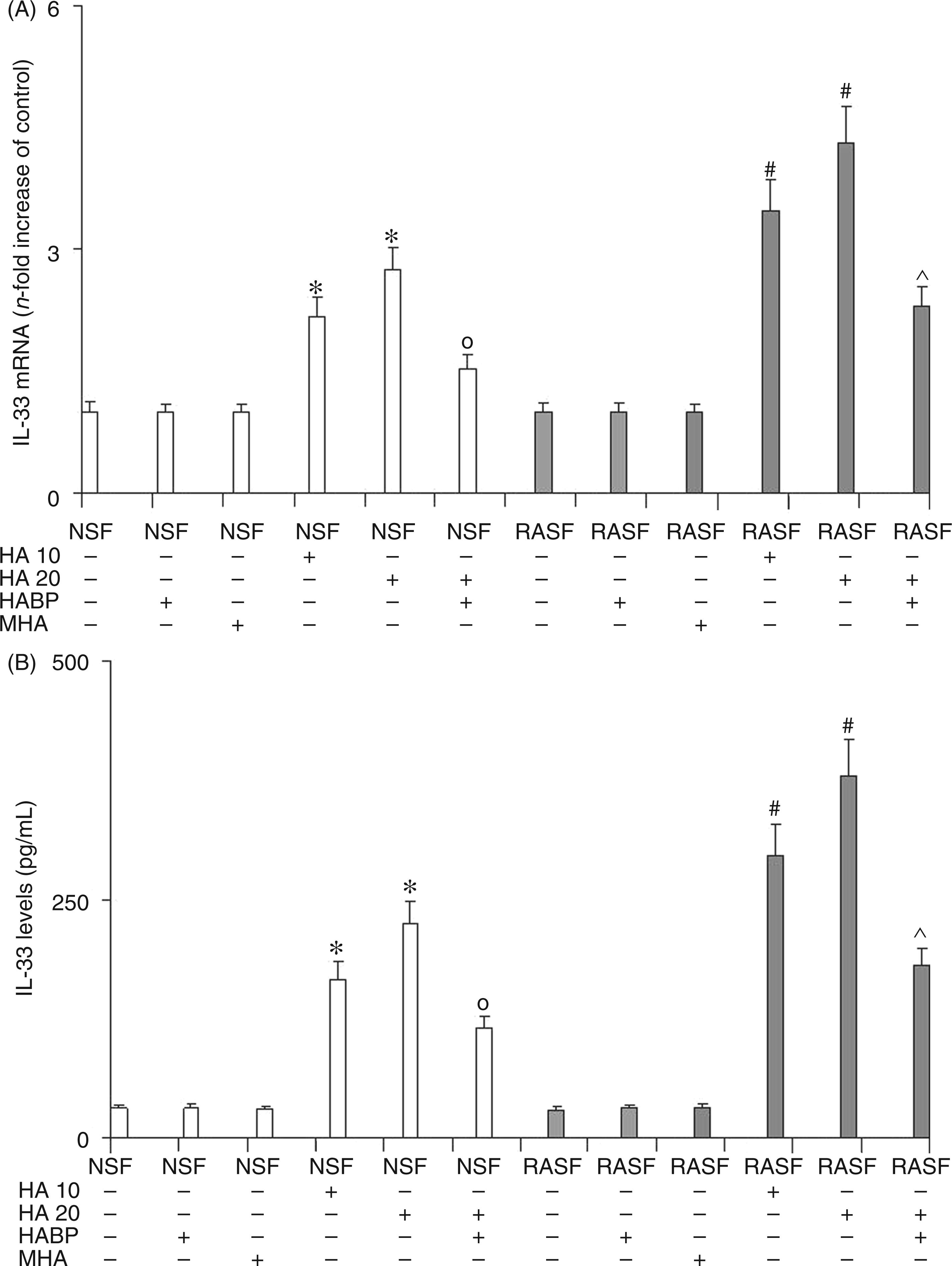
Taken together, these results may mean that 6-mer HA oligosaccharides prime inflammation, in part, by interacting with both TLR4 and CD44 receptors as the block of these HA fragments may alter the production of the inflammation mediators IL-18 and IL-33.
The destruction of articular cartilage is a prominent feature of inflammatory joint diseases such as RA. 13 The mechanism underlying tissue damage remains largely unknown. There is compelling evidence that products secreted by inflammatory cells and hyper-proliferation of synovial cells are responsible for the loss of proteoglycans, denaturation of collagen and bone destruction. Hyper-proliferative synovial fibroblasts play a critical role in the pathogenesis of RA by directly invading bone and cartilage, producing pro-inflammatory cytokines such as TNF-α and IL-1β, 25 destroying cartilage through the production of metalloproteinase and activating NF-κB, which augments osteoclast activity for bone destruction. 26
We recently reported that low molecular mass HA is able to stimulate inflammation in normal articular mouse chondrocytes.27,28 In these studies, cytokines and other pro-inflammatory mediators were shown to be produced as the consequence of the interaction either between HA and TLR4 or between HA and CD44 receptors. Although HA stimulation of TLR4 and CD44 receptors activated two distinct pathways, in the end they both converged in NF-κB activation.27,28 The aim of the present study was to evaluate TLR4 and CD44 involvement in both NSF and RASF following stimulation with 6-mer HA oligosaccharides which had been shown in previous studies to have a great capacity to prime inflammation through these receptors.
The results of the present study prompt the following considerations: on one hand, the inflammatory effect exerted by 6-mer HA is the sum of each inflammatory effect individually stimulated by both receptors; on the other hand, both TLR4 and CD44 receptors contribute to IL-18 and IL-33 production. Furthermore, as the inhibition of HA effects in RASF did not totally suppress TLR4 and CD44 over-expression and the increment in pro-inflammatory IL-18 and IL-33, then other inflammatory pathways are presumed to activate TLR4 and CD44. For instance, other matrikines could stimulate TLR4 and CD44 activation. 14 In conclusion, we believe that the ECM degradation pathways, in particular HA depolymerization, should be carefully considered for future anti-inflammatory strategies, although further studies are needed to fully confirm these hypotheses.
Footnotes
Funding
This study was supported by a PRA grant (Research Athenaeum Project 2008-2009) from the University of Messina, Italy.
Conflict of Interest Statement
The authors declare that there is no conflict of interest.
