Abstract
Background:
Despite advances in sepsis management, the relationship between appropriate empiric antibiotic therapy and acute kidney injury (AKI) in sepsis remains unclear. This study aimed to examine the association of appropriate empiric antimicrobial therapy with AKI in early onset sepsis caused by gram-negative bloodstream infections.
Methods:
We conducted a retrospective study of gram-negative bloodstream infection episodes in adult patients with early onset sepsis, using the Premier Healthcare Database from 2016 to 2020. The exposure was appropriate empiric antibiotic therapy determined by antibiotic regiments and antimicrobial susceptibilities of pathogens. The primary outcome was development of AKI or death by day 7 after the onset of sepsis. AKI was defined using the Kidney Disease Improving Global Outcome criteria based on serum creatinine levels, as urine output data were not available. The multivariable regression analysis was used to examine the association between appropriate empiric antibiotic therapy and the outcomes.
Results:
We identified 8565 patients with gram negative sepsis. In the total sample, the proportion of appropriate empiric antibiotic therapy was 93.2%, and the prevalence of AKI was 85.3%. Appropriate empiric antibiotic therapy was associated with decreased risk of AKI or death (adjusted odds ratio 0.70, 95% CI 0.52–0.94). For secondary outcomes, appropriate empiric antibiotic therapy was associated with lower AKI, shorter hospital LOS, lower C. difficile infections. However, it was not associated with in-hospital mortality.
Conclusion:
Appropriate empiric antibiotic therapy was associated with lower AKI in gram-negative sepsis. Early administration of appropriate antibiotics may prevent development of AKI.
Keywords
Introduction
Sepsis is a life-threatening condition characterized by a dysregulated host response to infection, leading to organ dysfunction. 1 Gram-negative bloodstream infections (GNBSIs) are a common cause of sepsis, with pathogens such as Escherichia coli, Klebsiella species, and Pseudomonas aeruginosa being contributors.2 –5 Acute kidney injury (AKI) is a serious complication of sepsis, including gram-negative sepsis, and is associated with significant morbidity and mortality. 6 The incidence of sepsis-associated acute kidney injury (SA-AKI) varies widely, ranging from 14% to 87%, with mortality rates between 11% and 77%. 7 A meta-analysis and systematic review revealed that gram-negative bacterial infections are more likely to result in AKI than gram-positive bacterial infections. 6 AKI in sepsis leads to worse outcomes, including prolonged hospital stays, increased healthcare costs, and higher mortality rates.8 –10
Despite advances in sepsis management, the relationship between appropriate antibiotic therapy and AKI in sepsis, particularly gram-negative sepsis, remains an area of interest and uncertainty. Appropriate empiric antibiotic therapy is vital in sepsis to promptly and effectively target the causative pathogens and reduce mortality rates.4,11 –13 However, the specific impact of appropriate antibiotic therapy on the development of AKI in sepsis is not well-defined. Previous studies have not conclusively demonstrated whether the use of appropriate antibiotics modifies the risk of developing AKI in sepsis. AKI in sepsis can result from various factors, including direct renal injury due to systemic inflammation, hemodynamic instability, nephrotoxic medications, and sepsis-induced microvascular dysfunction. 14 Identifying the role of appropriate antibiotic therapy in preventing or mitigating septic AKI could have significant implications for patient care and outcomes. Therefore, this study aims to examine the association of appropriate empiric antimicrobial therapy with the development of AKI in early onset sepsis caused by GNBSIs.
Methods
Study population
We conducted a retrospective cohort study using the Premier database (Premier Inc, Charlotte, NC) between January 2016 and March 2020. Adult patients (⩾18 years old) were included if they were diagnosed with sepsis within 2 days of admission. Sepsis was defined using the Centers for Disease Control and Prevention (CDC) adult sepsis event surveillance criteria.3,15 The onset of sepsis was defined as the timing of blood culture collection. Patients with end-stage renal disease, length of hospital stay < 2 days, and no serum creatinine measurements within 2 days of admission were excluded. The cohort was restricted to patients with monomicrobial gram-negative bloodstream infections from the first blood culture collection within 2 days of admission. We focused on common GNB (Escherichia coli, Klebsiella species, Proteus species, Enterobacter species, Pseudomonas aeruginosa, Serratia species, Acinetobacter species, and Citrobacter species) because the susceptibility for other GNB was not generally reported. This study was approved by the Duke University Institutional Review Board (IRB number: Pro00110302) with a waiver of informed consent.
Exposure
We defined an appropriate antibiotic treatment as initiation of at least one active antibiotic agent against the pathogens based on in vitro susceptibility.12,16 A microbial result that was either resistant or intermediate was considered as nonsusceptibility. When susceptibilities to antibiotics administered to patients were not reported, imputed interpretation tables for each pathogen were used. 12
Outcome
The primary outcome was AKI (stage 1, 2, or 3) or death occurring within 7 days from the onset of sepsis, in order to account for the competing risk of death. Using the KDIGO AKI criteria with daily serum creatinine from the onset of sepsis, AKI was categorized into 3 stages: stage 1 (creatinine level that was 1.5–1.9 times the baseline level or increased by ⩾ 0.3 mg/dL), stage 2 (creatinine level that was 2.0–2.9 times the baseline level), and stage 3 (creatinine level that was ⩾ 3.0 times the baseline level, AKI with a creatinine level ⩾ 4.0 mg/dL, or receipt of RRT). 17 The AKI urine output criteria were not used because urine output was unavailable in the dataset. We used the lowest serum creatinine value during the hospital admission as baseline, because serum creatinine records before admission were unavailable.18 –21
Secondary outcomes included any AKI occurring within 7 days from the onset of sepsis, in-hospital mortality, and C. difficile infections (CDIs). CDIs were identified using the International Classification of Diseases,10th Revision (ICD-10) diagnosis codes.
Statistical analysis
Data were presented as means ± standard deviation, medians and interquartile ranges (IQRs), or percentages, as appropriate. To estimate the association of appropriate empiric antibiotics, we fit multivariable logistic regression for binary outcomes. We fit generalized estimating equations with the negative binomial distribution for continuous outcomes. Random intercepts for individual hospitals were incorporated into the models to account for clustering within hospitals. Covariates included in the model were age, sex, transfer from anther hospital, surgery, intensive care unit admission within 2 days of admission, the van Walraven score (a weighted summary score based on Elixhauser comorbidities using ICD-10 codes), class of empiric antibiotics, infection sites (Supplemental Table 1)4,12, pathogens, septic shock, vasopressor use, mechanical ventilation, total bilirubin, and platelet count, hospital characteristics (bed size and teaching status). Septic shock was defined as use of at least one vasopressor and lactate >2 mmol/L on the day of sepsis onset. 22 A complete case analysis for the regression models was used because laboratory data had small percentages of missing information (0.1% for platelet count, and 2.1% for total bilirubin). Subgroup analysis was performed based on stratifying the cohort according to severity markers and pathogen type. An alpha level of less than 0.05 was considered statistically significant. Analyses were performed using SAS Version 9.4 (SAS Institute, Cary, NC).
Results
Patient inclusion and characteristics
In the cohort of 8565 patients with sepsis caused by GNBSIs, 7998 (93.4%) received appropriate empiric antibiotic therapy (Figure 1). Patient characteristics, including demographic information, comorbidities, and organ dysfunction, are detailed in Table 1.
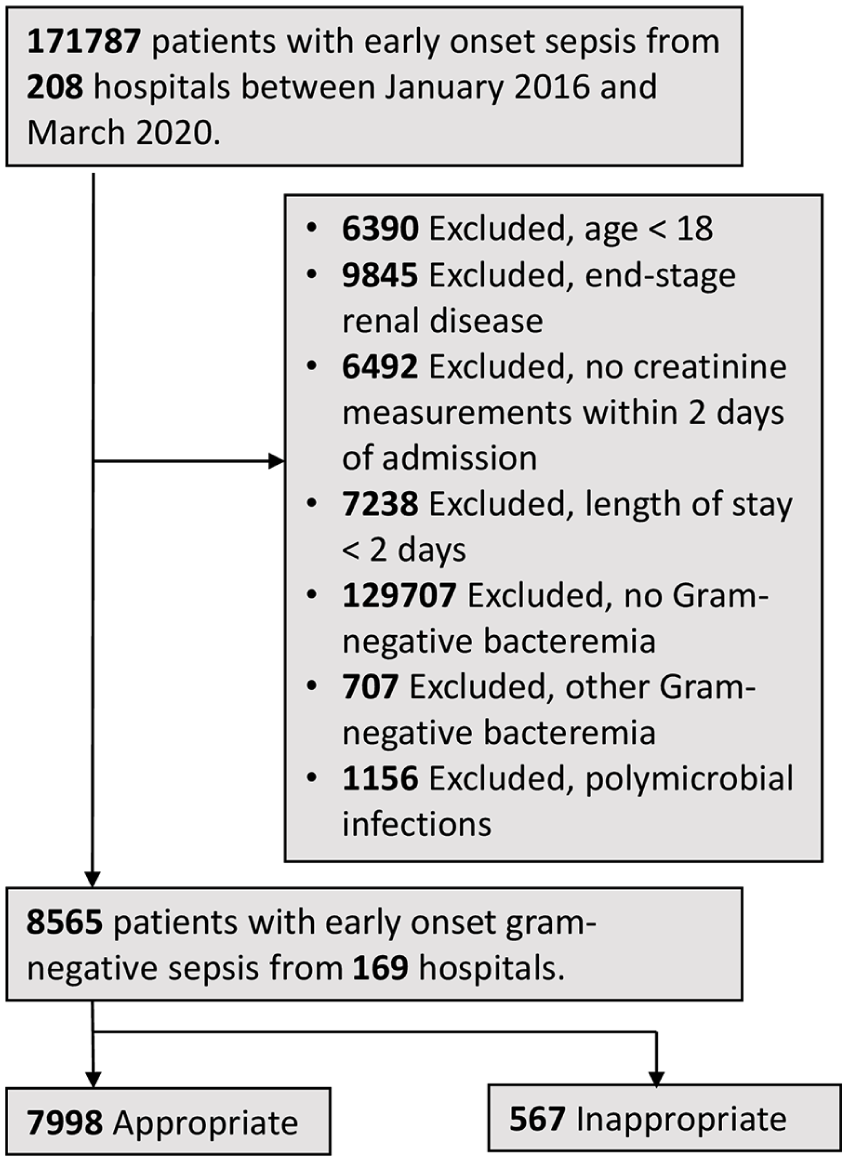
Patient flow.
Characteristics of patients with early onset gram-negative sepsis.
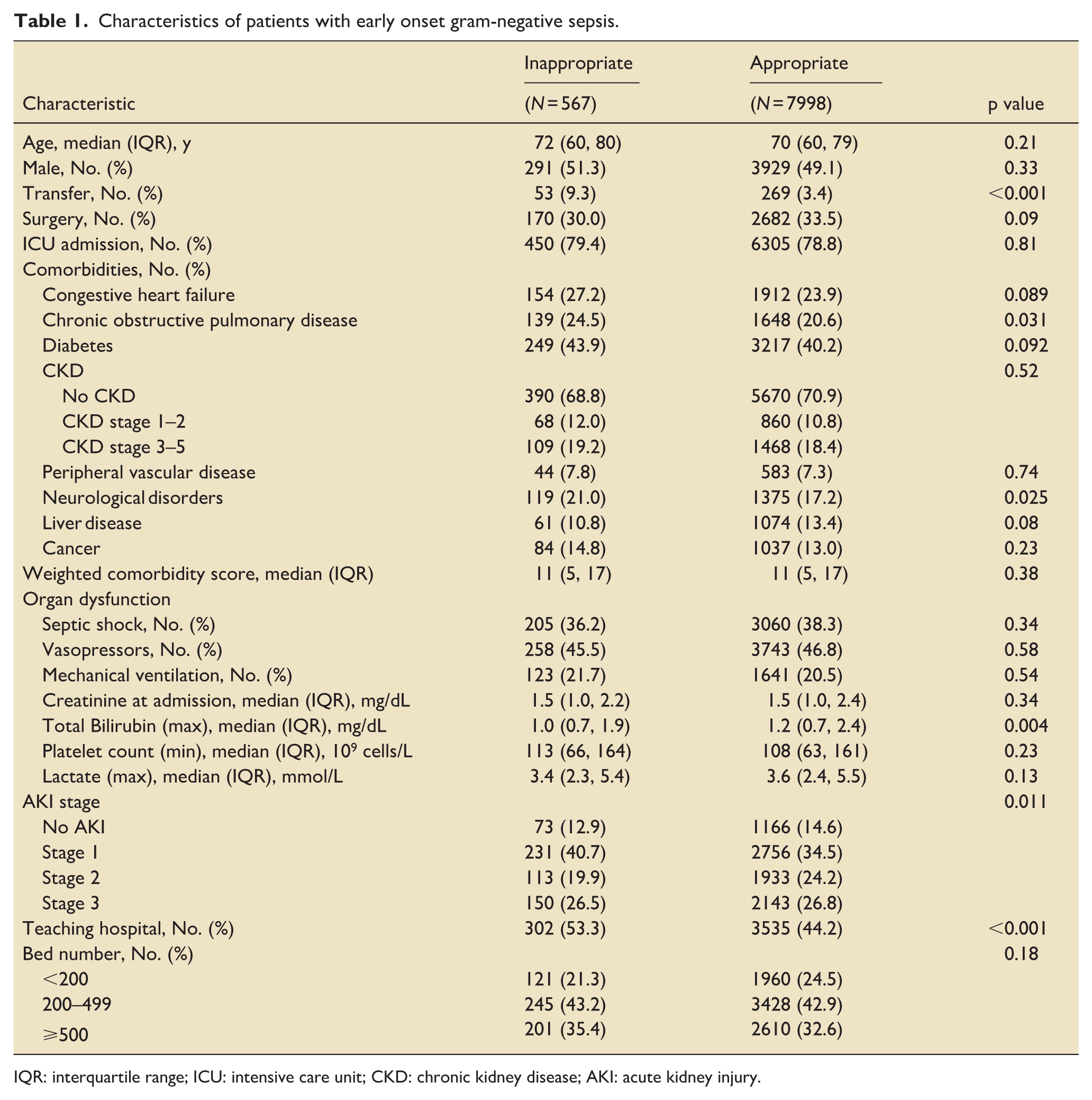
IQR: interquartile range; ICU: intensive care unit; CKD: chronic kidney disease; AKI: acute kidney injury.
The median age of patients in the inappropriate therapy group was 72 years (IQR 60–80), compared to 70 years (IQR 60–79) in the appropriate therapy group. The proportion of male patients was 51.3% in the inappropriate group and 49.1% in the appropriate group. COPD (24.5% vs 20.6%) and neurological disorders (21.0% vs 17.2%) were more prevalent in the inappropriate therapy group.
Infection profiles and antibiotic use
The infection profiles of patients with early-onset gram-negative sepsis are shown in Table 2. The most common sites of infection in both groups were genitourinary and pulmonary. Specifically, 48.7% of patients in the inappropriate therapy group and 47.1% in the appropriate therapy group had genitourinary infections, while 22.4% and 21.4%, respectively, had pulmonary infections.
Infection profiles of patients with early onset gram-negative sepsis.
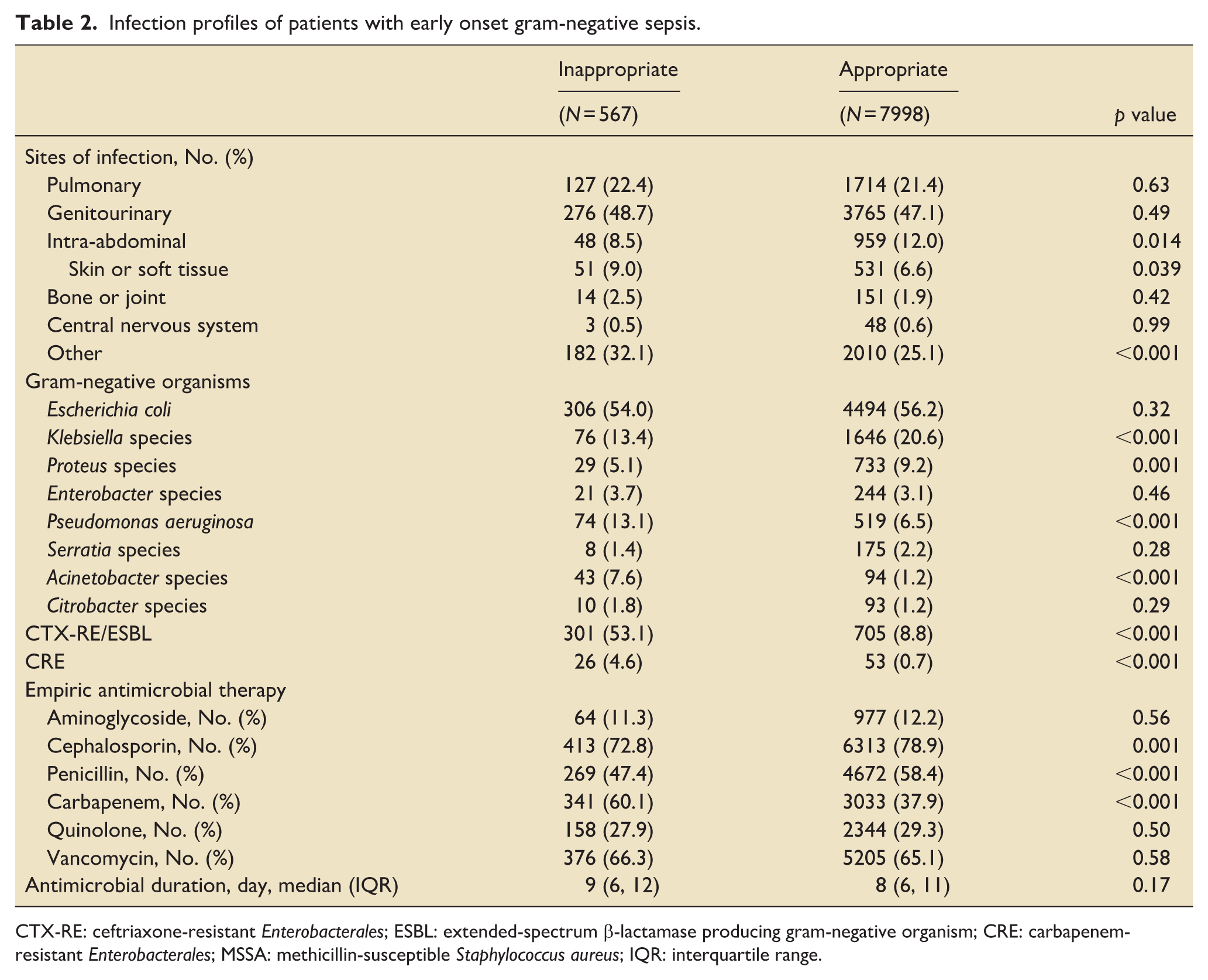
CTX-RE: ceftriaxone-resistant Enterobacterales; ESBL: extended-spectrum β-lactamase producing gram-negative organism; CRE: carbapenem-resistant Enterobacterales; MSSA: methicillin-susceptible Staphylococcus aureus; IQR: interquartile range.
Regarding the specific gram-negative organisms identified, Escherichia coli was the most prevalent in both groups (54.0% in inappropriate vs 56.2% in appropriate). However, significant differences were noted with Klebsiella species (13.4% vs 20.6%), Proteus species (5.1% vs 9.2%), Pseudomonas aeruginosa (13.1% vs 6.5%), and Acinetobacter species (7.6% vs 1.2%). The presence of CTX-RE/ESBL-producing organisms was notably higher in the inappropriate therapy group (53.1% vs 8.8%).
Cephalosporin use was lower in the inappropriate group (72.8% vs 78.9%), while carbapenem use was higher (60.1% vs 37.9%). The median duration of antimicrobial therapy was longer in the inappropriate therapy group (9 days vs 8 days).
Outcomes associated with appropriate empiric antibiotic therapy
The analysis of outcomes associated with appropriate empiric antibiotic therapy is presented in Table 3 and Figure 2. Among the patients, the combined outcome of AKI or death by day 7 occurred in 88.4% in the appropriate therapy group compared to 86.1% in the appropriate therapy group. The unadjusted odds ratio (OR) for this outcome was 0.81 (95% CI: 0.62–1.06), and the adjusted OR was 0.70 (95% CI: 0.52–0.94). For the outcome of AKI by day 7, appropriate empiric antibiotic therapy was associated with lower AKI (adjusted OR 0.75, 95% CI: 0.56–0.99). In the subgroup analysis for the primary outcome, there was no association between the appropriate empiric antibiotic therapy and the primary outcome by severity markers or pathogen type (Figure 2).
Outcomes associated with appropriate empiric antibiotic therapy.
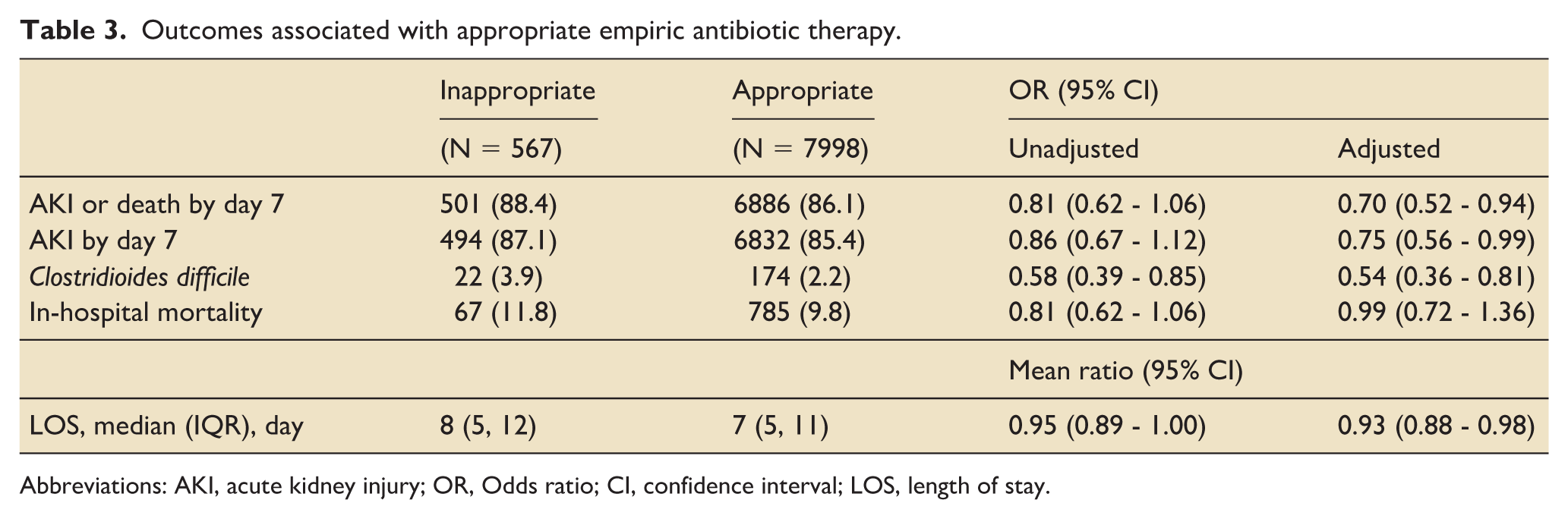
Abbreviations: AKI, acute kidney injury; OR, Odds ratio; CI, confidence interval; LOS, length of stay.
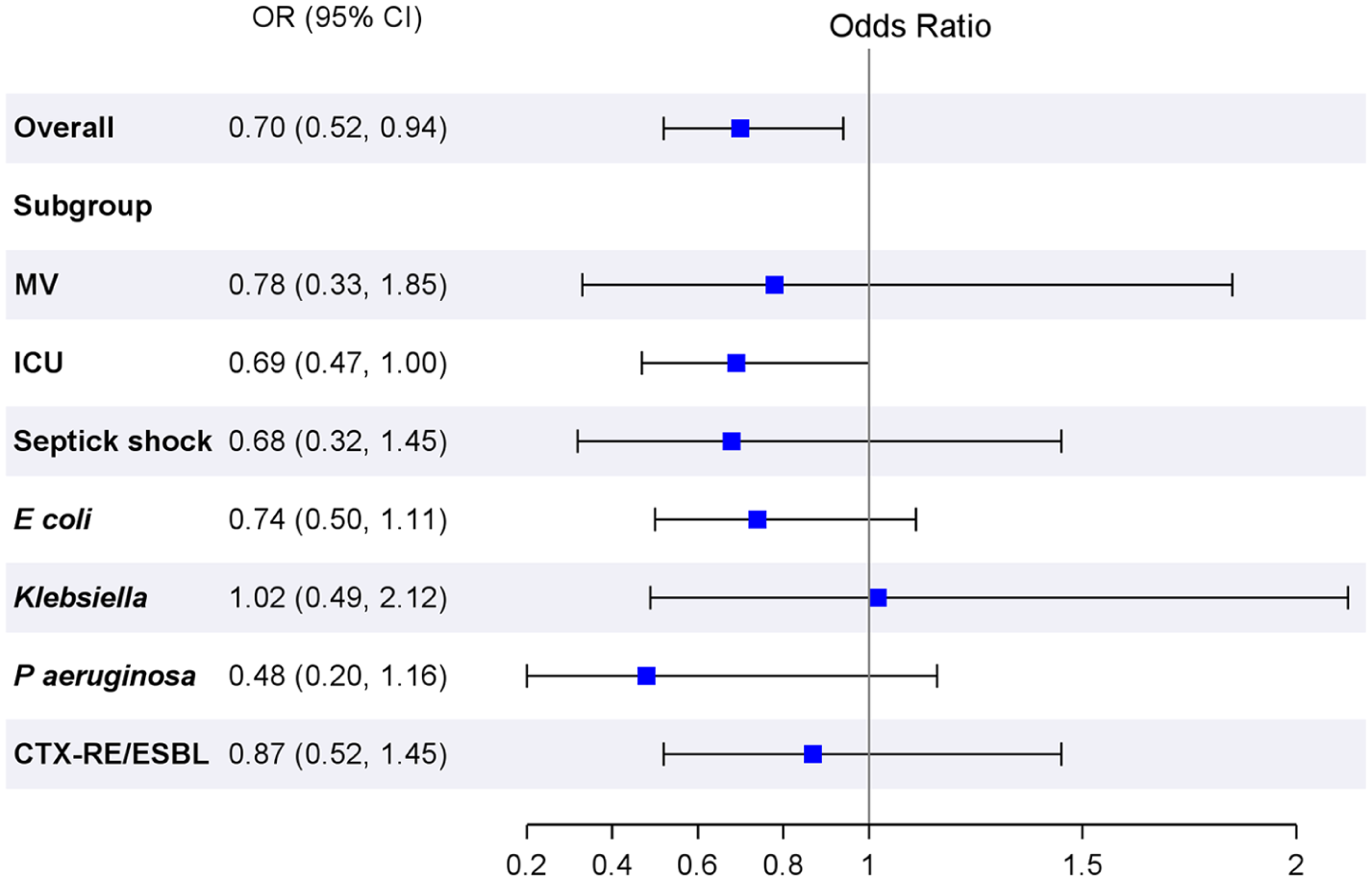
Forest plot showing odds ratios of acute kidney injury or death within 7 days associated with appropriate antibiotic therapy across different subgroups.
Appropriate empiric antibiotic therapy was associated with decreased odds of Clostridioides difficile infection (adjusted OR 0.54, 95% CI: 0.36–0.81) as well as shorter LOS (adjusted mean ratio 0.93, 95% CI: 0.88 – 0.98). There was no association between appropriate empiric antibiotic therapy and in-hospital mortality after adjustment. Similarly, there was no association for in-hospital mortality across the subgroups (Supplemental figure 1).
Discussion
This study examined the impact of appropriate empiric antibiotic therapy on the development of AKI and other outcomes in patients with early-onset gram-negative sepsis. Our findings highlight that appropriate empiric antibiotic therapy is associated with lower AKI or death by day 7, shorter LOS, and lower CDI, while there was no association for in-hospital mortality.
Approximately 9 of 10 patients in our study received appropriate empiric antibiotic therapy. This proportion is slightly higher than the recent studies including patients with sepsis.4,13,23 A multicenter retrospective study including 17,430 patients with culture-positive sepsis using the Cerner database from 2009 to 2015 demonstrated that 82% of the patients received adequate empiric antimicrobial therapy. 4 This finding may be explained by the fact that more critically ill patients with sepsis might be more likely to receive broad-spectrum antibiotics, but the prevalence of resistance pathogens are generally not high in the United States.4,24,25
Early appropriate antimicrobial administration and source control remain a cornerstone of sepsis treatment, which may lead to prevention of sepsis associated with AKI. 14 In our study, appropriate empiric antibiotic therapy was associated with deceased odds of AKI. Although there is little evidence directly looking at the relationship between appropriate antibiotic therapy, a large retrospective study reported that there was no association between appropriate antibiotic therapy and hospital-onset AKI in patients with community-onset sepsis. 4 The discrepancy might be explained by differences in the definition of AKI, as they defined AKI as an increase in creatinine during the hospitalization. Sepsis-associated AKI can occur due to either direct mechanisms (infection or the host response to infection) or indirect mechanisms (sepsis therapies including antimicrobials). 7 Therefore, in addition to early administration of appropriate antimicrobial agents to prevent AKI, the future studies will focus on the choice of empiric antimicrobial therapy and the strategy of antimicrobial de-escalation in patients with sepsis.
Although we observed a trend toward lower in-hospital mortality in patients treated with appropriate empiric antibiotic therapy, the result was not statistically significant. In contrast, several studies showed that appropriate empiric antibiotic therapy among severe infections or sepsis was associated with lower mortality.11,26 There are possible explanations for this inconsistency. One possibility is that our findings might be confounded by indication, because clinicians might be more likely to administer broad-spectrum antibiotics to patients with more severe status, which would make the treatment appear worse. 27 Moreover, the definition of appropriate empiric antibiotic therapy in this study was based only on the relationship between antibiotic regimens administered to patients and in-vitro susceptibility tests. However, antibiotic concordance is not necessarily contributing to therapeutic appropriateness, because the overall effectiveness of sepsis treatment includes multiple factors such as the pathogen, site of infection, severity of illness, the timing of antibiotic use, and baseline patient characteristics. 28
Limitation
This study has several limitations that need to be acknowledged. First, the generalizability of our findings may be limited due to the specific patient population and healthcare settings included in the study. Second, the definition of sepsis used in our analysis did not adhere to the Sepsis-3 criteria, as the qSOFA score was unavailable due to a lack of Glasgow Coma Scale (GCS) scores in the Premier database. This discrepancy could affect the consistency and comparability of our findings with other studies using the Sepsis-3 criteria. Third, baseline creatinine levels were not available for all patients. In addition, some patients had fewer creatinine measurements available for calculating AKI. These factors may have affected the accuracy of the assessment of AKI development. Fourth, the definition of appropriate antibiotic therapy relied only on in vitro susceptibility data. Dosage and frequency of antibiotics as well as minimum inhibitory concentration were not considered in the analysis, so that antibiotic agents that were susceptible to a pathogen might not achieve effective concentrations at the infection site. Also, antibiotic timing and duration were not considered in the definition of appropriateness, which might lead to the potential for misclassification bias. Fifth, AKI was defined according to the KDIGO criteria based solely on serum creatinine data, as urine output data were unavailable. These limitations underscore the need for cautious interpretation of our results and highlight the importance of further research with more comprehensive data to validate our findings.
Conclusion
In this retrospective cohort study including 8,565 cases of gram-negative bloodstream infections in adults with sepsis, we found that administration of appropriate antibiotics was associated with a reduced risk of AKI or death within seven days of sepsis onset. Notably, while there was no association between appropriate antibiotic therapy and in-hospital mortality, its impact on mitigating AKI underscores its importance in sepsis management. Further prospective studies are warranted to validate our findings across diverse patient populations and healthcare settings, ensuring broader applicability and guiding optimal treatment strategies in clinical practice.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251377990 – Supplemental material for Association of appropriate empiric antimicrobial therapy with acute kidney injury in gram-negative sepsis
Supplemental material, sj-docx-1-inc-10.1177_17511437251377990 for Association of appropriate empiric antimicrobial therapy with acute kidney injury in gram-negative sepsis by Tetsu Ohnuma, Shreya Khandelwal, Shingo Chihara, Miriam Treggiari, Jamie R. Privratsky, Pattrapun Wongsripuemtet, Julia A. Messina, Karthik Raghunathan and Vijay Krishnamoorthy in Journal of the Intensive Care Society
Footnotes
Ethical considerations
This study was approved by the Duke University Institutional Review Board (IRB number: Pro00110302) with a waiver of informed consent.
Author contributions
Study design was performed by TO, VK, and MMT. Data collection and analysis were performed by TO. Literature search and manuscript preparation were performed by SK and TO. The manuscript was reviewed by all authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
