Abstract
Emphysematous gastritis is a severe form of gastritis caused by gas-forming infectious organisms and is most frequently encountered in critically unwell patients. Diagnosis rests on the radiographic appearances of air within the gastric wall, which may extend into the portal venous system. Not previously described in the context of neutropenic sepsis, our case involves a 77-year-old patient with emphysematous gastritis who was admitted to the intensive care unit with a neutrophil count of 0.1 × 109/L and managed successfully with conservative treatment. Presenting complaints usually include abdominal pain, nausea, vomiting and occasionally haematemesis, in the context of systemic upset. Predisposing factors may include diabetes and immunosuppression, ingestion of corrosive substances, alcohol abuse, and abdominal surgery. The historical approach to management which previously involved urgent exploratory laparotomy with gastrectomy, has largely been replaced with conservative therapy, including broad-spectrum antimicrobials, gut rest and parenteral nutrition, with improved outcomes. Previously considered a commonly terminal diagnosis with mortality rates as high as 60%, this recent shift in approach to management has contributed to mortality rates being halved. The role of oesophago-gastro-duodenoscopy has not been established and is unlikely to be indicated in every case. Longterm complications may be of concern and include fibrosis and gastric contractures.
Keywords
Introduction
Emphysematous gastritis (EG) is a severe form of gastritis, characterised by gastric intramural air on computed tomography (CT), with associated systemic upset and toxicity.
It is a rare clinical condition, with high rates of mortality.
Case presentation
A 77-year-old male, with a background of Burkitt’s lymphoma, presented to the emergency department feeling generally unwell with fever, rigors and two episodes of vomiting without abdominal pain, on day 10 after his final cycle of R-CHOP chemotherapy. He was found to have a neutrophil count of 0.1 × 109/L and computed tomography (CT) pulmonary angiogram showed consolidation of right upper and lower lobes.
He had septic shock, presumed secondary to pneumonia, and was admitted to the intensive care unit (ICU) for respiratory support with 100% high flow nasal oxygen (HFNO) and cardiovascular support with double strength noradrenaline at 0.27 µg/kg/min. After only 4 h in ICU, he developed worsening type 1 respiratory failure (T1RF) and required tracheal intubation and mechanical ventilation. A 14 G nasogastric tube (NG) was placed for enteral feeding without apparent complication.
Despite increasing levels of organ support, recovery of the neutrophil count with administration of granulocyte colony stimulating factor and 4 days of six hourly (QID) intravenous (IV) piperacillin/tazobactam and daily gentamicin at 360 mg (5 mg/kg), the patient’s clinical condition failed to improve, with increasing vasopressor requirements, climbing inflammatory markers and a blood culture result positive for a pan-sensitive Pseudomonas aeruginosa.
Abdominopelvic (A/P) CT was performed to explore the possibility of abdominal sepsis or ischaemia due to concerns surrounding a rising lactate level (from 1.5 to 3.2 mmol/L) and evidence of enteral malabsorption with high NG aspirates, despite the abdominal examination revealing a soft and non-tender abdomen. The CT A/P showed significant gastric intramural gas, with extension into the portal venous system and right lobe of the liver, in keeping with emphysematous gastritis (Figure 1).
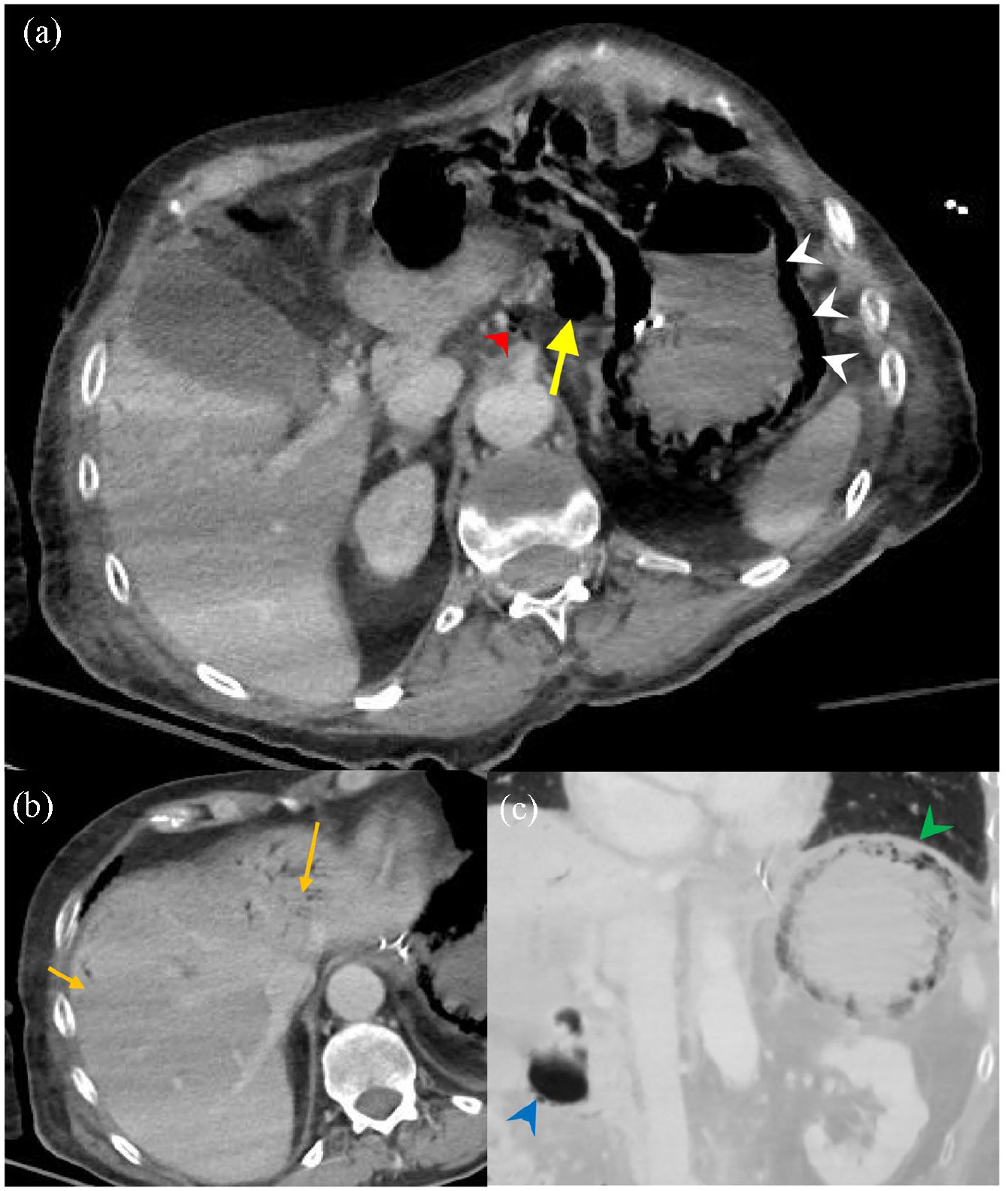
(a) The first post intravenous contrast CT demonstrates diffuse intramural gas (white arrowheads) with translocation/perforation into the gastrohepatic ligament (yellow arrow) and into portal vein tributaries along the left gastric supply (red arrowhead). (b) Bottom left picture demonstrates portal vein gas lying peripherally in the liver in both lobes (orange arrows). (c) Bottom right picture is an oblique coronal minimum intensity projection which demonstrates the detail of intramural locules of gas on the stomach (green arrowhead) and beneath the mucosa of the duodenal cap (blue arrowhead).
A diagnosis of emphysematous gastritis had not been encountered on our unit before, and gastroenterology and upper gastrointestinal surgery consults were sought, specifically regarding the preferred approach to management. Opinion from both specialities yielded a bleak picture. Both opinions suggested minimal prospect of survival and that any further clinical deterioration may herald a terminal event. The advice was to aim for complete gut rest, with provision of parenteral nutrition and continuation of broad spectrum antibiotic therapy and proton pump inhibition therapy. Surgical intervention was deemed futile. The advice was followed and metronidazole was added to enhance anaerobic cover, after discussion with microbiology. Due to multiple negative Beta-d-Glucan tests, anti-fungal therapy was not added.
There was slow but steady clinical improvement and despite prior expectations, on day 10 the patient was extubated and weaned to room air. A repeat CT scan performed on day 13 showed full resolution of the gastric intramural and portal venous air (Figure 2), and the patient was deemed medically fit for ICU stepdown to the ward on day 14. The patient was reviewed on the ward at day 40, and was found to be much improved. Nasogastric feeding had been implemented with evidence of good enteral absorption. The patient was planned for step down to a local rehabilitation facility for ongoing physical rehabilitation.
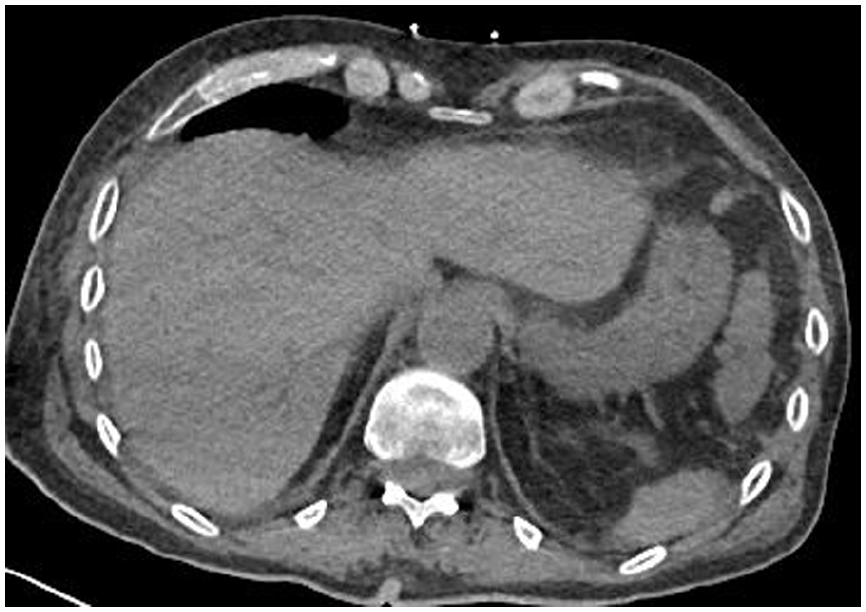
Follow-up non-contrast CT which demonstrates a collapsed stomach without intramural gas and resolution of the hepatic portal venous gas.
Discussion
Emphysematous gastritis was originally encountered by Dr E. Fraenkel in 1889 who, during autopsy of a young man who had unexpectedly died following severe abdominal complaints after debridement of a septic finger, described ‘intensively erythematous and oedematous gastric mucosa’ with evidence of submucosal air. 1 It was hypothesised that a gas-forming organism was responsible for these histopathological findings, after rod shaped bacteria were found between the mucosa and submucosa.1,2
Due to the rarity of this condition, (in 2021 there were fewer than 100 cases of emphysematous gastritis reported in English language literature 3 ) there is a paucity of evidence available to guide decision making around optimal management strategies.
Presenting complaints may be non-specific, but usually include abdominal complaints, such as abdominal pain, nausea, vomiting and occasionally haematemesis.4,5 Paradoxically, our patient presented predominantly with chest sepsis and was noted to have a soft and non-tender abdomen throughout his critical care stay. The only evidence of abdominal upset after intubation and invasive ventilation, were the non-specific findings of rising lactate levels and progressively increasing NG aspirates, which prompted abdominal imaging. Most commonly, patients with emphysematous gastritis are found to have a leukocytosis, which has been reported as high as 67 × 109/L 3 . To our knowledge, our case is the first reported example of emphysematous gastritis in the context of neutropenic sepsis.
The accepted pathogenesis involves a disruption in the gastric mucosal barrier. Despite the gastric mucosa, under normal circumstances, being relatively resistant to invasion by microorganisms due to low gastric pH and a rich blood supply, certain conditions may lead to a breach in these inherent defence mechanisms. These include ingestion of corrosive substances, alcohol abuse, abdominal surgery, diabetes and immunosuppression. 6 Proton pump inhibitors (PPI), frequently prescribed for critically ill patients, could reasonably be assumed to further promote microorganism invasion but there is no literature to confirm or refute this assumption.
In a literature review conducted on 59 reported cases of emphysematous gastritis prior to 2014, the most common organisms implicated were Clostridium Sp., Candida Sp., Streptococcus Sp., Klebsiella Sp. and polymicrobial infections. 2 A wide variety of isolated organisms has been described in literature, including Pseudomonas aeruginosa, 6 as was the case in our patient. However, in almost half of cases an organism is not identified.2,4
The diagnosis of emphysematous gastritis is a radiological one. Computed tomography of the abdomen shows air within the wall of the stomach. There may be portal venous air, free air within the peritoneal cavity, and gastric and bowel wall thickening. 7 It is notable that present-day imaging is more sensitive to intramural gas in the stomach and bowel. Modern multidetector CT is much more accurate than historic plain films at detecting signs of ischaemia and infection. As such, there exists a detection bias for less severe disease which previously would have been radiologically occult. These patients are more likely to survive with improved mortality with respect to historic cases – when the changes had to be severe enough to be seen on plain radiographs.
The differential diagnosis of gastric intramural air includes gastric emphysema.
Distinguishing between emphysematous gastritis and gastric emphysema is important, as management and outcomes differ significantly. Gastric emphysema is caused by simple introduction of air into the stomach wall through a mechanical cause and is usually a benign condition. Causes include bowel obstruction, increased gastric intraluminal pressure secondary to vomiting, penetrating gastric ulcers, instrumentation (endoscopy with air insufflation) or following extensive cardiopulmonary resuscitation. 7 The management of gastric emphysema is conservative, and the condition resolves spontaneously in those patients who survive.7,8 Radiologically gastric emphysema is associated with a rounded appearance of intramural gas and there is no portal or intrahepatic gas, whereas in emphysematous gastritis intramural gas may appear more streaky and linear, with associated thickening of the gastric wall and the presence of gas in the portal veins and liver. 7 If radiological findings do not aid in distinguishing the two, the clinical condition of the patient will likely point towards the correct diagnosis, as patients with emphysematous gastritis are generally severely ill with signs of sepsis associated with bacterial translocation.3,8
The role of oesophago-gastro-duodenoscopy (OGD) in diagnosis has not been established, as the diagnosis of emphysematous gastritis rests on clinical and radiological findings. 3 Whilst OGD may be considered to obtain tissue for microbiological analysis, particularly in treatment-resistant cases, the lack of correlation between OGD appearances and the need for operative management, together with the risks of the procedure itself mean that routine OGD is not indicated. 4
The diagnosis of emphysematous gastritis carries with it a poor prognosis with mortality rates reported to be as high as 60%.2,5,6 It is likely that this is, in part, related to the patients underlying conditions that that predispose them to the condition. Higher mortality rates also appear to be associated with more historic reports and management with an aggressive surgical approach.2,4 More recent data from a study assessing outcomes of emphysematous gastritis after the year 2000, found lower mortality rates, of around 33%. 2 This, in part, may be attributable to higher rates of conservative management, more sensitive imaging modalities (as discussed above) and improvements in the holistic care of critically ill patients, particularly those with sepsis. 9 However, in the absence of a trial comparing treatments these conclusions must be considered speculative.
Management options include surgical (with exploratory laparotomy and consideration of total gastrectomy), or conservative therapy with IV antimicrobials, IV fluids, gut rest and parenteral nutrition.
As mentioned, there appears to be an increasing trend since 2000 to follow a non-invasive approach. A retrospective review of 59 cases of emphysematous gastritis reported fewer cases taken for exploratory laparotomy after the year 2000 (22.2%. vs 62.5%; p = 0.002) and that conservative management was associated with a lower mortality rate (33.3% vs 59.4%, p = 0.046). 2 There is potential for bias in these results -as noted above. Even in cases where oesophago-gastro-duodenoscopy (OGD) reveals necrotic or ischaemic-looking gastric mucosa, findings on exploratory laparotomy do not always correlate, with normal appearances of bowel and intra-abdominal organs sometimes seen.4,10 Surgical resection is likely to have a high risk of anastomotic leak amongst other post-operative complications, leading to mortality. 11 Surgery may therefore be reserved for patients who develop complications, such as perforation, strictures, bowel ischaemia, or those failing medical management. 8
For those patients undergoing conservative management, early antimicrobial therapy has been associated with reduced mortality. 12 The duration of treatment has not been well established, however a 7-day course has been described in successful cases.4,8 Whether or not anti-fungal treatment should be added is another area of uncertainty, as there have been cases managed successfully which included3,8,12 and omitted4,12 it. As Candida species and polymicrobial infections account for 8.5% and 20% of organisms respectively, 2 a pragmatic approach would be to add empiric antifungal cover pending Beta-d-Glucan testing when there is concern of fungal sepsis.
Long-term sequalae have not been well described. After the initial insult, healing may occur with hyperplasia of the connective tissue with residual fibrosis. 12 In one report, 25% of patients who survived the initial insult, went on to develop gastric contractures. 6 The implications of this have yet to be described, though malabsorptive states and chronic discomfort or pain may be of concern.
Conclusion
Emphysematous gastritis is a rare condition and our report describes the first reported case in the context of neutropenic sepsis. There is usually an identifiable ‘inciting event’ or predisposing factor, such as immunosuppression. Patients commonly exhibit systemic toxicity and may present with septic shock. The diagnosis rests on the presence of gastric intramural air on imaging and may include hepatic portal gas. Conservative management is likely the preferred approach with early antibiotic therapy, anti-fungals when there is concern of fungal sepsis, gut rest, and parenteral nutrition. Whether PPI therapy should be continued is uncertain and likely to require a case by case decision. Prognosis is likely not as poor as has been described historically. Surgical intervention may be considered for patients with complications, such as perforation or for those failing to respond to medical management, but the evidence base for this is weak. The role for OGD is not established, and may not be indicated for the majority of patients. Long term sequelae may include fibrosis and gastric contractures, but more data from surviving patients is needed to clarify this further.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
