Abstract
Emphysematous gastritis is a rare, life-threatening condition characterized by inflammation of the stomach lining and gas within the gastric wall, with a mortality rate ranging from 55% to 61%. Emphysematous gastritis is caused by gas-producing bacteria and fungi that can infect both gastric and bowel walls. Risk factors include diabetes, corticosteroid use, prolonged nonsteroidal anti-inflammatory drug use, neoplasia, recent abdominal surgery, and other conditions that compromise immune function. Emphysematous gastritis can present with nonspecific symptoms such as abdominal pain, nausea, vomiting, diarrhea, and fever; however, severe manifestations such as hematemesis and sepsis may also occur. Symptoms alone are insufficient for establishing a definitive diagnosis; computed tomography is the most effective imaging modality to identify characteristic findings. Management typically involves conservative treatment with bowel rest, hydration, and intravenous antibiotics. Surgical intervention may be required in cases of clinical deterioration; however, its role remains controversial. Recent studies have questioned the necessity of surgery, emphasizing early medical management to improve outcomes. This narrative review aims to provide an overview of emphysematous gastritis, focusing on its pathophysiology, clinical features, diagnostic methods, and treatment strategies. It highlights the significance of timely intervention to improve survival and reduce the high mortality associated with this rare condition.
Keywords
Introduction
Emphysematous gastritis (EG) is a rare and potentially life-threatening condition characterized by inflammation of the gastric lining and gas within the gastric wall. It represents a severe form of gastric pneumatosis, which includes a range of conditions, from benign conditions such as gastric emphysema (GE) to EG. 1 A concise comparison of EG and GE is presented in Table 1,1,2 which highlights their distinguishing features. EG has a reported mortality rate of 55%–61% and was first described by Fraenkel in 1889.3,4 Although gas is normally present within the lumen of bowel loops, its presence in the parenchyma of solid organs or the walls of hollow viscera is considered pathological. 5
EG is most commonly caused by gas-producing gram-positive and gram-negative bacteria and occasionally by fungi. Patients often present with nonspecific symptoms such as abdominal pain, nausea, vomiting, diarrhea, and fever, with some progressing to more severe manifestations such as hematemesis and sepsis. Such nonspecific presentation makes diagnosis challenging, particularly in advanced stages, emphasizing the need for early detection and treatment. 6 Symptoms alone are insufficient for diagnosis; computed tomography (CT) is the most effective diagnostic tool for EG. 7 Treatment options may include conservative management or surgical resection of the stomach, depending on the patient’s clinical status. 8 This narrative review summarizes the pathophysiology, clinical features, diagnosis, and treatment of EG.
Methods
This narrative review summarizes the current evidence on the pathophysiology, clinical features, diagnostic approaches, and management of EG following the Scale for the Assessment of Narrative Review Articles (SANRA) guidelines. 9 A comprehensive literature search was conducted across PubMed, Scopus, DynaMed, and Google Scholar for English-language studies published up to August 2025 using the search terms “emphysematous gastritis,” “gastric pneumatosis,” “gas-forming infections of the stomach,” and “management of emphysematous gastritis.” References of retrieved articles were also screened. Eligible studies included case reports, case series, retrospective analyses, review articles, and systematic reviews that discuss the etiology, pathogenesis, clinical presentation, diagnosis, and treatment of EG. Studies focused solely on GE, pediatric populations, or unrelated gastric disorders were excluded unless they provided comparative data. Two authors independently extracted data on patient demographics, risk factors, causative organisms, imaging findings, endoscopic features, therapeutic interventions, and outcomes. Discrepancies were resolved through discussion, and data were summarized narratively and presented in tables, where appropriate. The primary outcomes of interest were mortality rates, clinical recovery, and complications associated with EG. Secondary outcomes included the diagnostic accuracy of imaging modalities and effectiveness of conservative versus surgical management approaches.
Clinical presentation
EG causes severe systemic impairment by releasing endotoxins and inflammatory mediators. Common symptoms include vomiting, diarrhea, abdominal pain, ileus, leukocytosis, fever, and hemodynamic instability. According to a systematic review of 121 patients by Elnaggar et al., the most common symptoms were abdominal pain (90%) and vomiting, nausea, and diarrhea (80%). 10 In immunocompromised individuals, such as those with diabetes and renal or hepatic failure, the condition may manifest subacutely with only fever or milder symptoms. 11 In rare cases, it can present with sudden epigastric pain, hematemesis, or melena. 3 Complications include gastric perforation, septic shock, gastrointestinal bleeding, abscess formation, and disseminated infection. 12 Prognosis depends on early recognition and timely intervention, often involving antibiotic use, supportive care, and, in some cases, surgical debridement.
Pathophysiology
The pathophysiology of GE, which can progress to EG, involves a complex interplay of predisposing factors, including mucosal injury, microbial invasion, ischemic changes, and immune system compromise. 13 The stomach’s mucosal barrier, comprising a thick layer of mucus, tight epithelial junctions, and a robust blood supply, serves as a critical defense mechanism against microbial invasion. 14 However, any disruption to this barrier, due to ulceration, trauma, instrumentation, or use of medications such as nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, predisposes the stomach wall to microbial infiltration. In addition, alcohol consumption and smoking can impair the gastric blood supply, weaken mucosal defenses, and increase susceptibility to bacterial invasion. 6 Once the mucosal barrier is compromised, gas-forming microorganisms, particularly Escherichia coli, Klebsiella pneumoniae, Staphylococcus aureus, Clostridium perfringens, Enterococcus species, and Streptococcus species can infiltrate the stomach wall. In immunocompromised patients, fungal pathogens such as Candida and Aspergillus species may also be involved. 15
These microorganisms produce enzymes such as urease and protease, which generate gases such as nitrogen, hydrogen, and carbon dioxide that accumulate in the gastric mucosa. 15 Increased intraluminal pressure, as observed in conditions such as pyloric obstruction and severe vomiting, can facilitate deeper penetration of these organisms into the tissue. Loss of epithelial integrity and poor perfusion from septic shock, cardiac arrest, or severe dehydration further facilitate this invasion. Ischemia exacerbates the process by impairing immune responses and creating a hypoxic condition that favors the proliferation of anaerobic bacteria, such as Clostridium species.16,17
Chronic illnesses such as diabetes and kidney disease as well as immunosuppressive therapy weaken host defenses through impaired neutrophil and phagocyte function, increasing the risk of EG. 18 Typical features include gas bubbles in the submucosa and muscularis propria, accompanied with necrosis caused by bacterial toxins and ischemia. 7 This disrupts the gastric architecture and function. Neutrophil and macrophage infiltration causes fluid buildup, thereby weakening the wall and increasing the risk of perforation. 19 This has been demonstrated visually in Figure 1.

Pathogenesis of emphysematous gastritis showing mucosal injury, microbial invasion, and gas production within the gastric wall, leading to intramural gas accumulation in the submucosa and muscularis propria.
Discussion
Trends in EG management
The mechanisms underlying EG highlight the complex interplay between microbial invasion, tissue ischemia, and systemic inflammation. Building on this foundation, the following section explores the infectious agents and predisposing conditions responsible for the development of EG as well as the evolving approaches to diagnosis and treatment.
Infectious etiologies and predisposing conditions
A retrospective analysis by Watson et al. reviewed 59 adult cases of EG documented before 2013 and identified Clostridium species, Streptococcus species, Escherichia coli, Klebsiella, and Pseudomonas as the most common causative organisms. 20 Similar trends were noted by Ono et al. in their analysis of 40 cases published between 2019 and 2022, with a rare case caused by Bacteroides uniformis. 18 Fungal pathogens such as Candida and Mucorales have also been reported as causative agents, with their effects attributed to their angio-invasive properties. 13 ,21 Sarcina ventriculi, also known as Clostridium ventriculi, is a rare causative organism of EG found on gastric biopsy in two cases. 22 Al Rasheed and Senseng reported Sarcina in patients with delayed gastric emptying, emphasizing its association with severe complications. 22 Elnaggar et al., in their systematic review of 121 cases, observed that Sarcina ventriculi was the most frequently reported gas-producing microorganism associated with gastric wall infections. 10
These organisms have been identified through gastric aspirates or tissue biopsy culture. In some cases, no organism was isolated. Watson et al. found that no causative organism was isolated in 25 of the 59 cases (42.4%) reviewed. 20 Although the precise pathogenesis of EG remains unclear, it is believed to involve mucosal injury and systemic illness. Contributing factors include alcohol abuse, ingestion of corrosive substances, abdominal surgery, and NSAID use. 21
In a recent case report, Fatima et al. described a young male patient without any comorbid conditions who developed EG following a binge-drinking episode. 23 A comprehensive literature review conducted by Matsushima et al. included 39 EG cases reported up to 2012; they found that diabetes mellitus was the most common predisposing factor, with a positive medical history in 10 patients. A history of malignancy was noted in five patients, while renal failure and organ transplant were implicated in seven and three cases, respectively. 15 Although EG is commonly observed in individuals with underlying predisposing conditions, a few rare cases have been reported in otherwise healthy adults. Yu et al. reported a case of EG in a young, healthy man, possibly caused by infection secondary to the consumption of contaminated deer meat while hunting. 24 In another case involving a young, healthy man with no prior comorbidities, EG developed after the consumption of pickles a day before symptom onset. It was hypothesized that pickle juice, an acid-containing food item, contributes to histopathological changes in the gastric mucosa, creating a conducive environment for the gas-forming bacteria to invade and cause infection. 25
In immunocompromised patients, such as those with diabetes, renal failure, or cirrhosis, the presentation of EG may be more severe and rapid. These patients are at higher risk of systemic sepsis and multiorgan failure due to their impaired immune status. For instance, diabetic patients may present with altered mental status and severe leukocytosis, as seen in the case of an 82-year-old diabetic man. Similarly, patients with renal failure, such as those on hemodialysis, may exhibit more pronounced systemic toxicity and a higher risk of mortality.26,27
Patients with cirrhosis may present with additional complications, such as hepatic encephalopathy and worsening hepatic function, as seen in the case of a patient who developed EG status post partial hepatectomy. The presence of portal venous gas in these patients can further complicate the clinical picture. 28
Collectively, these cases highlight several key trends in the epidemiology and clinical spectrum of EG. Bacterial pathogens, particularly Clostridium, Streptococcus, Sarcina ventriculi, and E. coli, remain the most commonly identified causative organisms; however, fungal infections are increasingly being recognized in immunocompromised patients.10,13,20,21 Most cases occur in individuals with underlying conditions such as diabetes, renal failure, malignancy, and recent surgery; however, a subset of reports have documented EG in previously healthy adults, often following mucosal insult from alcohol or acidic food intake.26,27 The variability in causative organisms, absence of identifiable pathogens in some cases, and the broad range of predisposing factors underscore the heterogeneous nature of EG and the importance of maintaining a high index of suspicion in both high-risk and atypical patient populations.
Diagnostic approach and management
EG presents with nonspecific symptoms such as fever, abdominal pain, nausea, vomiting, and diarrhea, making radiological imaging essential for differentiating it from its more benign predecessor, GE. Both conditions are characterized by the presence of gas in the gastric wall; however, in GE, this typically appears as thin, linear intramural air collections, while in EG, it presents as cystic gas pockets and is associated with a considerably worse prognosis. 12
EG is very rare; therefore, there are no established guidelines for the diagnosis and management of EG patients. However, most case reports published to date emphasize prompt diagnosis and aggressive treatment. 29 Although plain radiographs and ultrasound can detect air in the gastric mucosa, they have low sensitivity and specificity. CT is the definitive imaging modality for diagnosing EG because it can confirm the intramural location of the gas and the extent of mucosal involvement. 5 CT characteristically demonstrates the presence of cystic air pockets trapped within the gastric wall, hypertrophic mucosal folds, and free air within the peritoneal cavity and portal vein. 30 The presence of portal venous gas is a poor prognostic indicator, associated with 75% mortality, and approximately all patients require surgical management. 31 According to a comprehensive review, CT was performed for 31 of the 39 cases reported between 1982 and 2009, of which 41% showed radiographic findings of intramural gas in the stomach. 15
Esophagogastroduodenoscopy (EGD) plays an essential role in assessing the extent of mucosal damage and obtaining tissue biopsies for microbiological analysis in treatment-resistant cases. 7 Matsushima et al. found that EGD was performed in 48.7% of suspected EG cases, and >50% of these cases exhibited erosion of the gastric mucosa and necrosis of the stomach wall. 15 In a recent systematic review, endoscopy revealed gastric necrosis in 40% of the cases and inflammation in 30%, characterized by mucosal erythema and edema. Ulcerations were observed in 25% of the cases, while perforation occurred in 10%, often linked to ischemic damage. Normal or nonspecific findings were reported in 10% of the cases, usually due to incomplete evaluations. 10 EGD is primarily indicated when signs of severe sepsis are present or when microbiologic confirmation is required. 7
Management depends on the severity of the presentation. Patients with normal-appearing mucosa benefit from conservative measures such as bowel rest, intravenous (IV) fluid administration, and close hemodynamic monitoring. Patients should be started on broad-spectrum antibiotics covering gram-negative and anaerobic bacteria; antifungal agents may be added to the treatment regimen if fungal etiology is suspected. 32 Surgery is indicated in patients with evidence of perforation, stricture, or clinical deterioration despite medical therapy.15,33
Although surgery may be lifesaving, it is associated with postoperative complications, including bleeding, worsening infection, anastomotic leak, bowel obstruction, ileus, ischemia, fistula and stricture formation, adhesions, stenosis, and wound dehiscence, all contributing to increased morbidity and mortality.34,35 There are no set protocols for the duration of antibiotic therapy in these patients. Yusef et al., based on their study on a patient with EG, reported the effectiveness of a 14-day course of antimicrobial treatment. 36
After assessing cases of EG published from 2019 to 2022, Ono et al. found that surgical treatment was performed for 13 of the 40 patients, while the remaining majority was managed conservatively. 18 Matsushima et al. and Watson et al. similarly noted that the use of surgical intervention has declined over time, with laparotomies performed in 62.5% of the cases before 2000 versus only 22.2% after 2000.15,20 According to a systematic review by Elnaggar et al., involving 121 cases of EG, management primarily involved medical therapy, which was used in 75% of cases, with antibiotics such as vancomycin, piperacillin-tazobactam, and metronidazole, along with supportive care. Nasogastric decompression was used in 11% of the patients. Surgical intervention was required in 25% of the cases; the most common surgical intervention was gastrectomy (32%), followed by laparotomy (21%) and splenectomy (4%), reconfirming the trend toward conservative management. Overall, the recovery rate was 70%, with a mortality rate of 15%. 10
Long-term sequelae of EG remain poorly understood. Jenkins et al. hypothesized that EG sequelae involve connective tissue hyperplasia accompanied with residual fibrosis. Reportedly, 25% of surviving patients developed gastric contractures. The clinical implications of this outcome remain unclear; potential concerns include malabsorptive conditions and chronic pain. 12
A clear contrast emerges between conservative and surgical management. Conservative therapy, consisting of bowel rest, broad-spectrum antibiotics, proton pump inhibitors, and supportive care, has shown favorable outcomes in hemodynamically stable patients without evidence of perforation or necrosis, as reported by Nasser et al. and Sánchez-Hernández et al.4,11 In contrast, surgical intervention is typically reserved for cases with extensive gastric necrosis, perforation, or uncontrolled sepsis. These patients often have a poorer prognosis, with high mortality rates reported in studies by Huang et al. and Costa et al.30,34 The shift from early aggressive surgical management to an initial conservative approach reflects improvements in diagnostic imaging, early recognition, and antibiotic therapy.
In summary, EG is an extremely rare and potentially life-threatening condition, with only a few cases documented in the literature. The cases analyzed in this review are summarized in Table 2. The scarcity of data and published studies presented several challenges in the preparation of this review. Nevertheless, the aim was to consolidate the available information into a single, comprehensive resource for researchers, clinicians, and students. We hope this review not only serves as a consolidated reference but also contributes toward shaping future understanding and management strategies for EG.
Comparison of key features, pathogens identified, management, and outcomes from published cases.
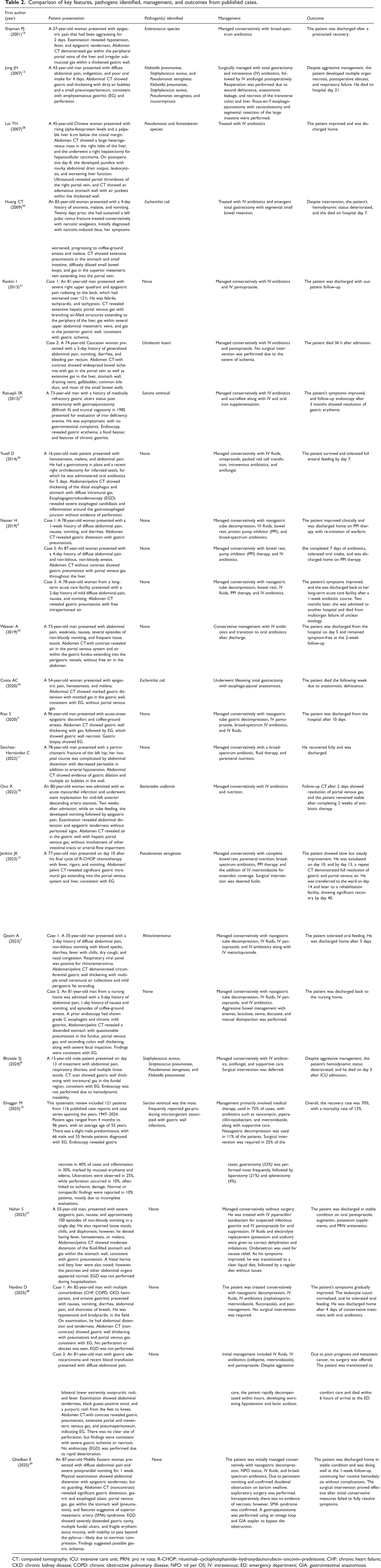
CT: computed tomography; ICU: intensive care unit; PRN: pro re nata; R-CHOP: rituximab–cyclophosphamide–hydroxydaunorubicin–oncovin–prednisone; CHF: chronic heart failure; CKD: chronic kidney disease; COPD: chronic obstructive pulmonary disease; NPO: nil per OS; IV: intravenous; ED: emergency department; GIA: gastrointestinal anastomosis.
Conclusion
EG is a rare, life-threatening condition caused by gas-forming organisms that infect the gastric wall, often in the presence of risk factors such as diabetes, immunosuppression, and mucosal injury. Although surgery is historically the primary treatment option, recent trends favor conservative management, including bowel rest, IV fluid administration, and broad-spectrum antibiotics, especially in the absence of ischemia. However, there is no standardized diagnostic pathway or clear criteria for selecting surgical versus nonsurgical approaches. The rarity and variability of EG continue to hinder the development of evidence-based protocols. To address these gaps, multicenter research and consensus guidelines are needed to facilitate early diagnosis, guide treatment decisions, and reduce the morbidity and mortality associated with EG.
Footnotes
Acknowledgements
We would like to extend our gratitude to Namita Ruhela, MD, for her expert editing and invaluable feedback, which greatly enhanced the readability and clarity of this manuscript.
Author contributions
CSA conceived the idea and made significant contributions to drafting the manuscript's Discussion section. SD contributed substantially to the pathophysiology content and was involved in collecting the initial data for review. SK assisted in writing the Introduction section, editing the tables and figures, and critically revising the manuscript. KA drafted the abstract and participated in manuscript editing. LE contributed to the conceptualization of the work and coordinated team administration. TJ conducted critical revisions and helped improve the visualization of the final publication. RJ led the core conceptualization and provided the oversight and leadership necessary to plan and execute this research project.
Consent to participate
Not applicable because this is a review article, and there was no new participation.
Consent for publication
Not applicable as data from an individual person were used.
Data availability
Not applicable to this article because no datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
As this is a review article, no ethical approval was needed.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.

