Abstract
We reported a case of portal vein pneumatosis caused by a nasojejunal nutrition tube that was successfully treated conservatively. This patient was discharged after 1 week of treatment with imipenem/cilastatin sodium for anti-infection, gastrointestinal decompression, fluid replacement, and pain relief, and the patient’s abdominal pain was significantly relieved. The main pathogenesis of hepatic portal venous gas is as follows: (a) increased pressure in the digestive tract leading to gas entering the portal vein circulation through the intestinal wall and finally into the liver; (b) destruction of the intestinal mucosa causing the gas formed by intestinal bacteria to enter the portal vein system; and (c) diffusion of gas produced by bacteria in intraperitoneal abscesses or portal vein and mesentery, causing pyelitis. The successful treatment of this patient highlights that addressing physical factors, providing gastrointestinal decompression, and administering anti-infection therapy can aid in the management of such cases. Bacterial culture of gastric juice and drug sensitivity testing can guide effective anti-infection treatment.
Introduction
Hepatic portal venous gas (HPVG) refers to the presence of gas in the portal vein, its intrahepatic branches, and gastrointestinal branches. 1 This rare and concerning finding is occasionally identified on computed tomography (CT) and requires prompt medical attention. 2 This article reports a case of portal vein pneumatosis caused by a nasojejunal nutrition tube that was successfully treated conservatively.
Case report
The patient was a female in her early 60s, with a height of 147 cm, weight of 33 kg, body mass index of 15.27. She was admitted to Ganzhou People’s Hospital in August 2023 due to recurrent upper abdominal distension and pain accompanied with vomiting for 1 year, which worsened over the last month. The patient presented with upper abdominal distension and pain 1 year ago, which was particularly noticeable after meals. The pain was localized and did not radiate to other areas, and it was accompanied with symptoms such as nausea, vomiting, acid regurgitation, heartburn, and nonprojectile vomiting of gastric contents. The abdominal pain subsided after vomiting. Following a visit to a local hospital, the patient underwent gastroscopy, revealing antral ulcer, gastric retention, duodenal bulb ulcer with bulbar stenosis, non-atrophic gastritis with gastric antral erosion, and esophageal ulcer. A positive 14-carbon breath test was performed, leading to the administration of acid suppression therapy for stomach protection and eradication of Helicobacter pylori. The patient’s condition improved; however, recurrence was observed. Recurrence and worsening of symptoms were noted 1 month ago, accompanied with a weight loss of 10 kg, necessitating admission to our hospital. An upper gastrointestinal tract iodine contrast revealed pyloric obstruction, gastroptosis, and gastric retention, with no contrast agent shadow in the duodenum (Figure 1(a)). Additionally, the patient underwent various examinations, including abdominal CT revealing pyloric obstruction, gastric retention, and gastroptosis (Figure 1(b)). Blood routine tests indicated a decrease in the white blood cell count (3.35 × 109/L), neutrophil count (1.76 × 109/L), red blood cell count (3.04 × 1012/L), hemoglobin level (88 g/L), and hematocrit level (26.2%), along with a normal platelet count (312 × 109/L). Furthermore, the ferritin level was low at 4.77 ng/mL. Liver and kidney function, electrolytes, C-reactive protein, alpha-fetoprotein, carcinoembryonic antigen, and cancer antigen (CA19-9) levels were all within normal ranges. As the patient’s gastroscopy revealed duodenal bulb stenosis, gastric retention, gastroptosis, and reflux esophagitis (LA-C), balloon dilatation of duodenal stenosis and obstruction were performed under gastroscopy (Figure 2(a)). However, the patient still exhibited repeated nausea and vomiting after duodenal balloon dilation. The patient underwent nasojejunal nutrition tube placement under gastroscopy combined with digital subtraction angiography (DSA), and the nasojejunal nutrition tube was inserted at a depth of 108 cm (Figure 2(b)). DSA revealed that the nasojejunal nutrition tube only ran along the greater curve of the stomach, and the head was located at the level of the duodenum (Figure 3). The patient was treated with 1000 mL of enteral nutrition solution. However, approximately 12 h after the operation, the patient suddenly experienced the following symptoms: (a) severe pain in the upper abdomen, accompanied with nausea and vomiting (the vomit comprised foul-smelling stomach contents); (b) whole body cold; and (c) heavy sweating. The following signs were also observed: (a) body temperature of 36.5°C; (b) pulse of 97 beats/min; (c) respiratory rate of 19 beats/min; (d) blood pressure level of 97/67 mmHg; (e) abdominal muscle tightness; (f) liver dullness boundary; (g) upper abdominal tenderness; (h) no rebound pain; and (i) bowel sound frequency of 4 times/min. Enhanced CT of the whole abdomen revealed multiple gas in the portal vein, superior mesenteric vein, and proximal segment of the splenic vein; gastric dilatation; and gastric emphysema (GE) (Figure 4). Klebsiella pneumoniae, extended-spectrum beta-lactamases: Pos µg/mL, cultures were positive in vomit; this pathogen is sensitive to imipenem/cilinstatin sodium and other antibiotics. The patient underwent laboratory tests again, which indicated a white blood cell count of 6.82 × 109/L↓, neutrophil count of 3.78 × 109/L↓, red blood cell count of 3.46 × 1012/L↓, hemoglobin level of 85 g/L↓, hematocrit level of 25.6%↓, platelet count of 320 × 109/L, and calcitonin level of 0.06 ng/mL↑. Postoperative CT at 24 h revealed a small amount of gas in the portal vein compared with the previous scan, gastric dilatation, and GE (Figure 5(a)). Moreover, postoperative CT on day 5 revealed almost complete disappearance of the portal venous gas, gastric dilatation, and GE. Gastric dilation and GE improved significantly compared with the findings of the previous scan (Figure 5(b)). The patient was discharged after 1 week of treatment with imipenem/cilastatin sodium for anti-infection, gastrointestinal decompression, and fluid replacement; the patient showed pain relief, and her abdominal pain was significantly relieved. The reporting of this study conforms to the Case Report (CARE) guidelines. 3 This study was approved by the Ethics Committee of Ganzhou People’s Hospital, Jiangxi Province, China (approval No. PJB2025-099-01). We have de-identified all patient details. Written informed consent was obtained from the patient for publication of this case report.

(a) Digestive tract iodine–water angiography: pyloric obstruction, gastric ptosis, gastric retention, and no contrast material shadow in the duodenum and (b) abdominal computed tomography enhancement on admission: pyloric obstruction combined with gastric retention and gastric ptosis.

(a) Gastroscopy: balloon dilation of duodenal stricture with obstruction, gastric retention, and gastric ptosis and (b) gastroscopy: duodenal stricture with obstructive reflux esophagitis (LA-B), gastroptosis, failure of jejunal nutrient tube implantation under gastroscopy, indentation of guide wire.

Nasojejunal nutrition tube placement under digital subtraction angiography along the gastroscope indwelling guide wire.

Enhanced computed tomography of the whole abdomen showing multiple pneumatosis in the portal vein, superior mesenteric vein, and proximal splenic vein; gastric dilatation; and gastric emphysema.
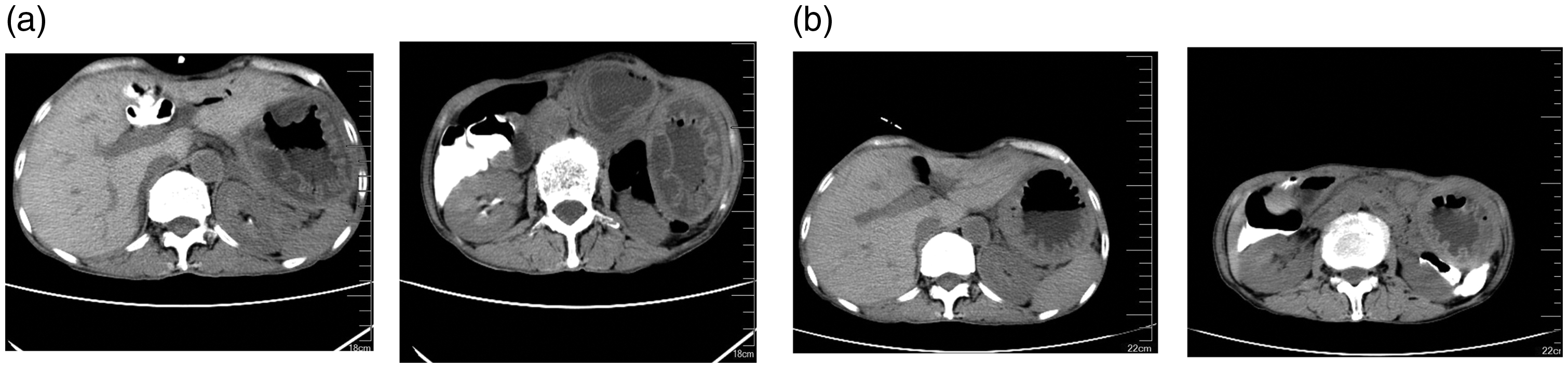
(a) Computed tomography at postoperative 24 h showing a small amount of gas in the portal vein compared with the previous scan, gastric dilatation, and gastric emphysema (GE) and (b) computed tomography at postoperative day 5 showing almost complete disappearance of the portal venous gas, gastric dilatation, and GE. Furthermore, gastric dilation and GE improved significantly compared with the findings of the previous scan.
Discussion
HPVG is a rare radiological finding that can indicate inflammatory bowel disease, obstructive gastrointestinal disease, mesenteric ischemia, and other diseases. 4 Currently, the main pathogenesis of HPVG is as follows: (a) increased pressure in the digestive tract leading to gas entering the portal vein circulation through the intestinal wall and finally into the liver; (b) destruction of the intestinal mucosa causing the gas formed by intestinal bacteria to enter the portal vein system; and (c) diffusion of gas produced by bacteria in intraperitoneal abscesses or portal vein and mesentery, causing pyelitis. 5 In previous reports, the fatality rate of HPVG was as high as 75%, while in recent years, the fatality rate decreased to 29%–56%. 6 Historically, mesenteric vascular occlusion and intestinal necrosis were considered the primary causes of HPVG. However, with advancements in CT technology, HPVG is more commonly attributed to gastrointestinal dilatation, inflammation, sepsis, endoscopic surgery, peptic ulcer disease, blunt trauma, and abdominal and pelvic malignancy. Among this subset of patients, early active etiological intervention has significantly increased the success rate of conservative treatment, resulting in a decrease in mortality rates. Previous studies have suggested a scientific approach to diagnosing and treating HPVG using radiology, laboratory examinations, and clinical symptoms. Emergency surgery should be considered when HPVG is suspected based on ordinary X-ray or CT findings indicating intestinal ischemia, increased lactate levels, and peritonitis. 7 In cases where intestinal ischemia is not present, conservative treatment and close monitoring may be appropriate. The presence of ascites (CT), peritoneal irritation (physical examination), and shock (vital sign examination) during conservative treatment is considered life-threatening, warranting emergency surgery. Advances in improved HPVG evaluation and treatment systems may also be a major reason for the decline in the associated mortality rates.
The patient developed HPVG 12 h following gastroscopy and DSA-guided jejunal nutrition tube placement, characterized by gastric dilatation and GE. Notably, preprocedural upper gastrointestinal iodine–water angiography revealed no significant gastric dilatation. This case highlights the importance of prompt abdominal CT for patients experiencing sudden abdominal pain, nausea, and vomiting following endoscopic procedures, to detect HPVG and potential digestive tract perforations. Based on the literature review, five cases of HPVG following gastrostomy have been reported, with three cases successfully treated using conservative methods (two cases with anti-infection therapy and one case without anti-infection therapy). The patient in this case exhibited a similar pathophysiological process to HPVG following gastrostomy, with GE detected during CT.5,8–11 The pathogenesis of HPVG in these cases may involve physical or infectious factors causing GE, leading to the entry of gas into the gastric veins through capillaries and subsequent return to the portal vein. 5 The GE in this patient resolved within 12 h of the onset of HPVG, with noticeable swelling. The patient was diagnosed with gastroptosis. The jejunal nutrition tube, measuring 145 cm in length, was inserted along the greater curvature of the stomach and advanced into the duodenum. Post-insertion X-ray imaging revealed that the tension exerted by the nutrition tube within the stomach, which could potentially lead to gastric distension. Studies have demonstrated that GE is observed in patients with organic lesions in the upper gastrointestinal tract, with the most common cause being trauma induced by nasogastric tube insertion or endoscopic intubation. Additionally, invasive infections (such as those caused by Escherichia coli, Streptococcus, Enterobacter, Clostridium, and Pseudomonas aeruginosa), anorexia nervosa, neonatal emphysematous gastritis, and acute gastric volvulus are recognized as causes of GE. 12 Therefore, we propose that the presence of K. pneumoniae infection and acute gastric dilatation indicates that the pathogenesis of HPVG involves a combination of physical and infectious factors. The successful treatment of this patient highlights the importance of promptly removing the nasal jejunal nutrition tube, addressing physical factors, providing gastrointestinal decompression, and administering anti-infection therapy. Bacterial culture of gastric juice and drug sensitivity testing can guide effective anti-infection treatment, underscoring the need for gastric juice collection for early etiological detection and adjustment of the anti-infection treatment plan based on drug sensitivity results.
Conclusions
The presence of nasojejunal nutrition tubes may lead to acute gastric dilation, which can result in HPVG. Critical treatment measures in patients with HPVG include removal of the nasojejunal nutrition tube, alleviation of physical factors contributing to the dilation, implementation of gastrointestinal decompression, and administration of etiology-guided anti-infection therapy.
Main points
We report the successful management of a case of portal vein pneumatosis caused by a nasojejunal nutrition tube with gastrointestinal decompression and anti-infection therapy. We summarized the main pathogenesis of hepatic portal venous gas and briefly described its main treatment options.
Footnotes
Acknowledgements
Not applicable.
Author contributions
Study conceptualization: Youshun Liu; Study design: Youshun Liu; Supervision: Yong Li; Resources: Ji Huang; Materials: Ji Huang; Data collection and/or processing: Ji Huang; Analysis and/or interpretation: Yong Li; Literature search: Haifeng Liu; Manuscript writing: Haifeng Liu; Critical review: Chunping Zhu.
Data availability statement
This is a clinical case, and publicly available data have not been uploaded.
Declaration of conflicting interests
We declare that we have no financial or personal relationships with other people or organizations that can inappropriately influence our work. Furthermore, there is no professional or other personal interest of any kind in any product, service, and/or company that could be construed as influencing the results of this case report.
Funding
This research was supported by the project of Ganzhou Municipal Health Commission of Jiangxi Province (2020-2-43), project of Science and Technology of Ganzhou City, Jiangxi Province (2023LNS17419), Jiangxi Provincial Bureau of Traditional Chinese Medicine (2020B0117, 2023A0198, and 2019A485).
