Abstract
Acute on chronic liver failure (ACLF) is a clinical syndrome characterised by acute hepatic decompensation, multi-organ failure and high mortality, in patients with cirrhosis.
Organ dysfunction in ACLF is often reversible and when necessary these patients should be considered appropriate candidates for admission to an intensive care unit (ICU). The yearly increase in numbers of patients with ACLF admitted to ICU has been matched with an improvement in survival.
ACLF has only been recently defined. In the absence of evidence-based guidelines we outline a systems-based approach to care which encompasses accepted ICU practice and evidence from trials in this cohort. We advocate for timely referral to specialist liver centres and consider the complexities of proceeding with liver transplantation. Equally, in a proportion of patients who continue to deteriorate, appropriate ceilings of care should be established.
Future clinical trials may change treatment paradigms but care of patients with ACLF is undoubtedly becoming an integral part of an intensivist’s practice. We hope that this review is a welcome starting point when managing this complex clinical syndrome.
Introduction
Acute-on-chronic liver failure (ACLF) is a clinical syndrome, which occurs in patients with cirrhosis. Acute liver decompensation results in both hepatic and extra-hepatic organ failure, has an associated high mortality and when progressing to ACLF prompts referral to intensive care services, with over 4500 ICU admissions per year in the UK.1,2
ACLF is characteristically a systemic inflammatory disease, driven by a dysregulated immune system. 3 The clinical sequelae of multi-organ failure and high short-term mortality delineate this syndrome from an isolated acute decompensation (AD). 4 A diagnosis of ACLF is prevalent in approximately a third of patients hospitalised with chronic liver disease (CLD) and as such presents a significant burden on ICUs. 4 Definitive evidence-based guidance on how best to manage patients with ACLF is currently lacking raising concerns over which patients benefit most from organ support. This review describes the main clinical presentation, priorities in care and current treatment strategies to guide the intensivist managing a patient with ACLF.
Clinical presentation and severity
AD can manifest with ascites, hepatic encephalopathy (HE), gastro-intestinal haemorrhage and bacterial infection 5 and is commonly managed in medical ward or specialist liver services. A diagnosis of ACLF may represent the first episode of decompensation in over 20% of patients and the differentiation between AD and ACLF only recently formalised. The landmark CANIONIC study elucidated diagnostic criteria and a scoring system to grade ACLF severity. 4 This has now been validated and is accepted within European practice although not universally (Figure 1).1,4,6,7
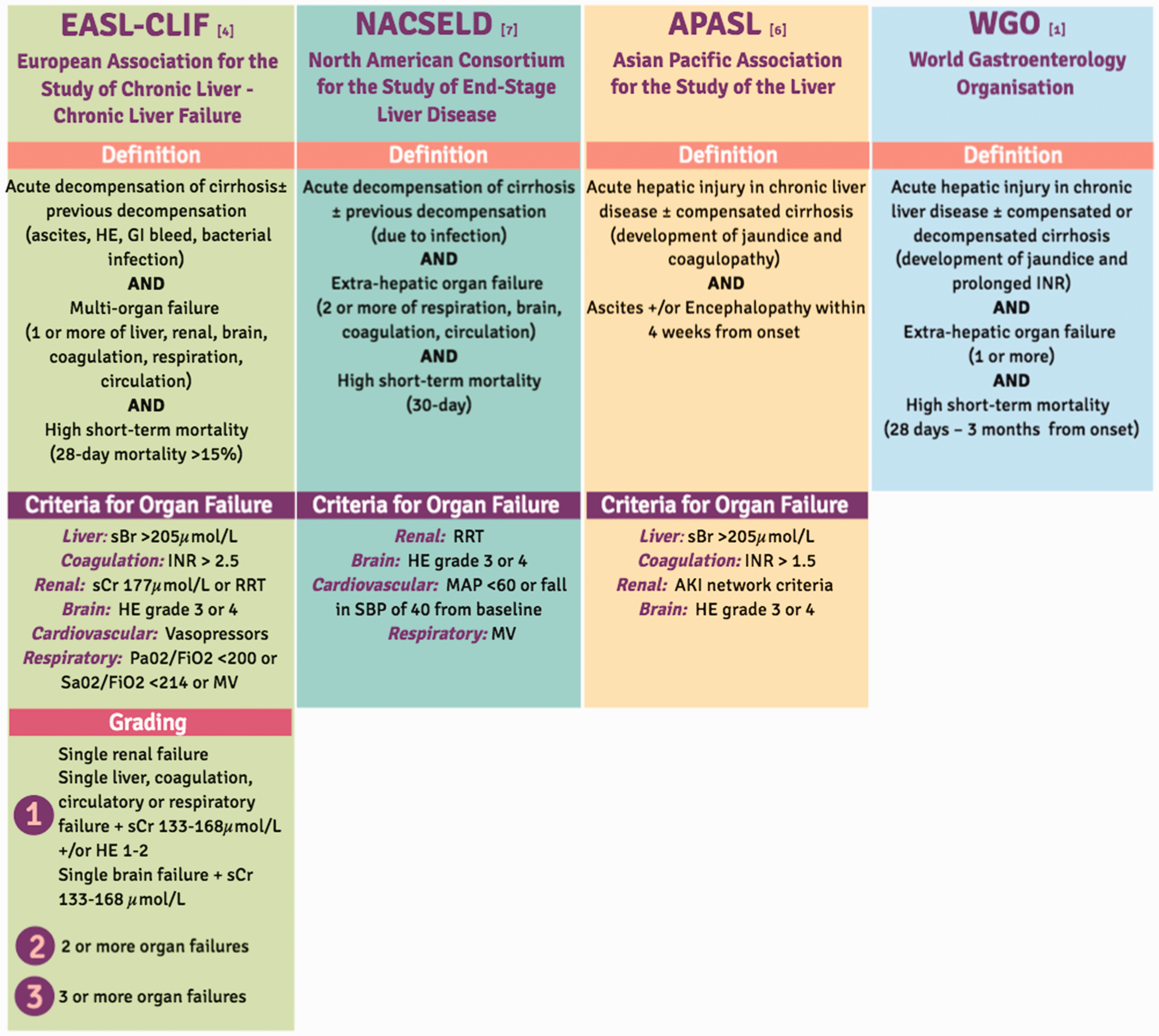
Current definitions and diagnostic criteria for ACLF.
The most common precipitating events are bacterial infection, active alcohol abuse and reactivation of, or, superimposed viral hepatitis. 5 In around 40% of patients a precipitating event is not identified, but the precipitant has neither been linked to disease severity, nor mortality. 4 Accompanying organ failures are defined using a modified version of the Sequential Organ Failure Assessment (SOFA) score, known as the Chronic Liver Failure (CLIF)-SOFA score (or Chronic Liver Failure Consortium, CLIF-C score).4,8 The CLIF-C score can better predict mortality in ACLF when compared with the model for end-stage liver disease (MELD), MELD-Sodium and Child-Pugh scoring systems. 8 Patients are assigned to one of three grades of ACLF severity based on the number of organ failures present. In the CANIONIC study 28- and 90-day mortality across all grades was 31% and 52%, respectively. 4
Admission to ICU
The number of patients with CLD admitted to ICU has increased annually and represents a small (2–3%), but significant, proportion of all admissions. 9 Survival has also improved.9–11 A recent analysis of Intensive Care National Audit and Research Centre (ICNARC) data has demonstrated that 68% and 52% of cirrhotic patients admitted to ICU will now survive to discharge from ICU and hospital, respectively. 10 Increased survival may be explained by improvements in quality of care and earlier admission, with identification of organ failure before it is irreversible. Whilst admissions have increased there has been a corresponding decrease in the severity of extra-hepatic organ failure on admission. 9 Patients with ACLF have a greater degree of organ dysfunction on admission when compared to the general ICU population and this may explain their increased mortality. 11 Nonetheless, in ACLF organ dysfunction is often reversible and these patients should be considered candidates for admission to ICU.9–11
Organ failure and supportive care
Best clinical practice in ICU involves a comprehensive multi-disciplinary and systems-based approach. In ACLF this is based on a combination of accepted ICU practice and evidence from trials in this cohort (Figure 2). Nevertheless, universally accepted pharmacological therapy for these patients is inadequate and a major focus of ongoing research. In the absence of this, attention to supporting the major organ systems remains paramount while reversible causes of ACLF are sought and addressed.
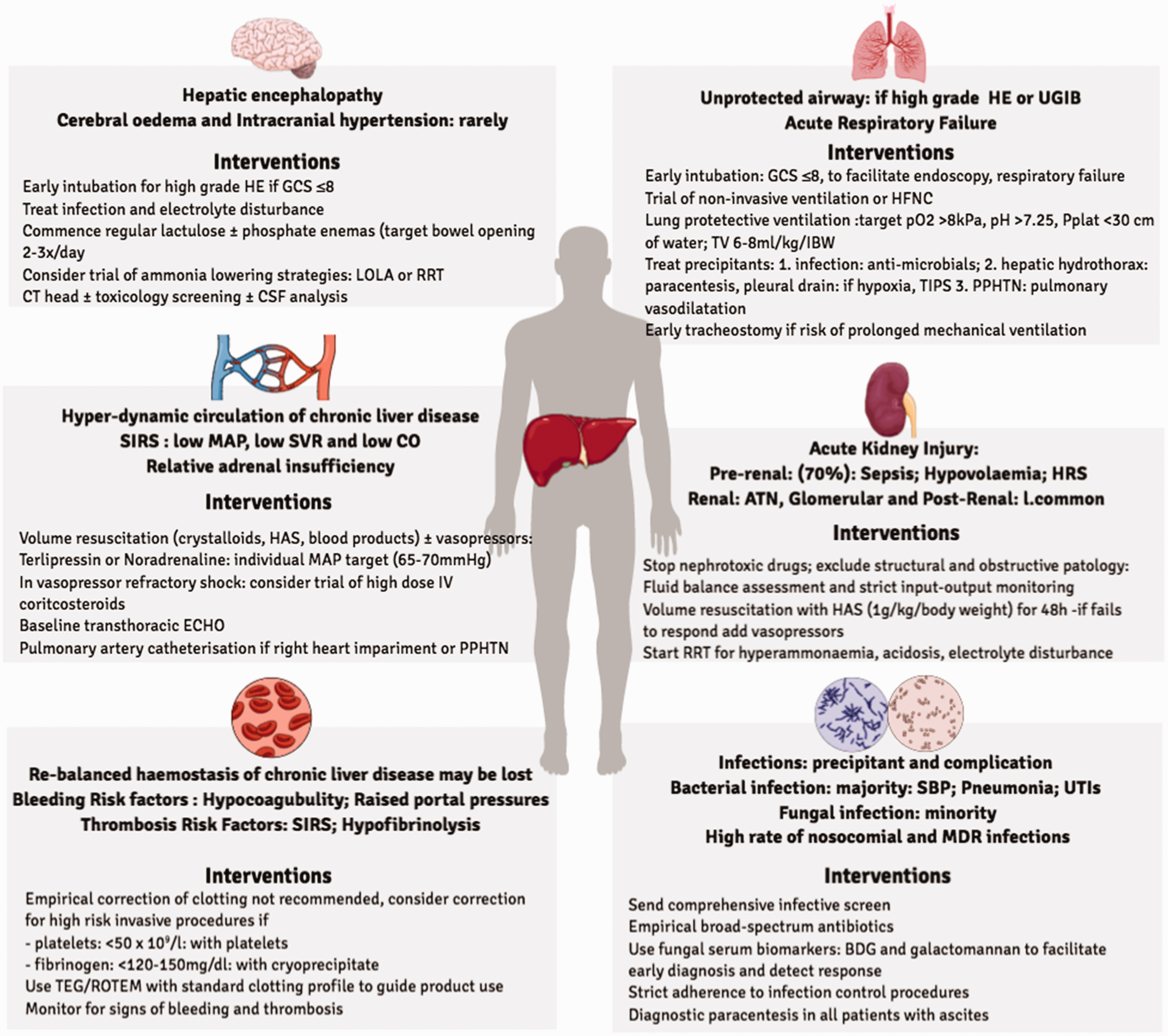
The organ specific problems and approach to care for the patient with ACLF on ICU.
Airway and ventilation
Intubation and mechanical ventilation is indicated in patients with severe HE (Glasgow Coma Score <8, West Haven grade >2) and to facilitate endoscopy following a variceal bleed. Administration of sedation to tolerate a definitive airway should be minimized in HE given the prolonged hepatic clearance of some agents. 12
Acute respiratory failure secondary to pulmonary pathology, predominantly infection and acute lung injury, often requires ventilatory support. 12 Pulmonary pathology may be pre-existing and can precipitate or exacerbate respiratory failure. Porto-pulmonary hypertension (PPHTN) and hepato-pulmonary syndrome (HPS) are specific to cirrhosis but are rare causes of hypoxaemia. HPS is characterised by intra-pulmonary arterio-venous dilatations, which lead to shunting and hypoxaemia. It is an important differential to consider in patients in whom hypoxaemia is either out of proportion to, or persists beyond the acute pathology. 13
Ascites and hepatic hydrothorax can equally impede ventilation and drainage of either is indicated where respiratory embarrassment is obvious.
Non-invasive ventilation or high-flow nasal cannula are initial strategies to support oxygenation, but if respiratory failure is worsening invasive ventilation should not be delayed. 13 Where invasive ventilation is indicated, in the absence of other evidence, a lung-protective strategy should be employed using lower tidal volumes (6-8ml/kg based on ideal body weight), plateau pressure limitations (<30cmH20) and positive end-expiratory pressure titration. 14 This may require adjustment in view of existing lung pathology and intraabdominal hypertension. Targeting normoxia (Pa02 >8) and normocapnia (PaC02 4.5-6.0) is justified but not validated. Early tracheostomy should be considered if prolonged mechanical ventilation is likely, especially where recovery from high grade HE is not expected to be prompt. 12 Concomitant anti-microbials for infection, diuresis and paracentesis for ascites and hepatic hydrothorax and pulmonary vasodilatation for PPHTN should be employed.12,13
The circulation
A hyper-dynamic (high cardiac-output(CO)) circulation caused by decreased systemic vascular resistance (SVR) and subsequent low mean arterial pressure (MAP), characterises circulatory dysfunction in CLD. 15 Total blood volume increases but effective circulating volume is decreased, secondary to marked vasodilatation. The systemic inflammatory reaction driving ACLF is likely to explain the further fall in SVR and MAP, alongside a corresponding rise in CO. 15 In such a finely balanced system further changes in vascular tone or volume status, which occur in ACLF, may precipitate circulatory failure. 16 Cirrhotic cardiomyopathy impedes ventricular contractility and diastolic function, although its impact on the clinical sequelae of ACLF is unknown.5,15 Relative adrenal insufficiency occurs in >50% of patients. The improvement in haemodynamic status following corticosteroid replacement suggests it contributes to circulatory dysfunction. 17
Volume resuscitation is the first priority in management. Current clinical practice includes the use of crystalloids, human albumin solution (HAS) and if necessary, blood.13,16 In cirrhosis serum albumin concentration decreases and albumin may be qualitatively impaired with its proposed anti-oxidant and anti-inflammatory properties lost. 18 This may be restored following supplementation and in turn improve haemodynamic disturbance. HAS is therefore often preferred12,13,16 although the immunological benefits are not well defined. In patients with refractory hypotension vasopressor support, with noradrenaline or vasopressin-receptor agonists, is required. The MAP target should be individualised to the patient and account for their pre-morbid physiology. A target of 65–70 mmHg is generally accepted and should be used to titrate vasopressors.12,13,16 The ideal fluid and vasopressor in ACLF management is still unknown. Similarly, whether corticosteroid administration improves survival is unclear and their use is physician dependent.16–18 A prospective trial has demonstrated a survival benefit, which was not replicated in a randomised controlled trial (RCT).17,19
Haemodynamic monitoring with an arterial line should be instituted in patients with circulatory disturbance.12,13 A trans-thoracic echo is helpful in all patients with circulatory dysfunction and pulmonary artery catheterisation may be required with right-heart impairment or PPHTN. 16 Utilisation of stroke volume variation, CO monitoring, central venous monitoring and mixed-venous oxygen saturations may be informative.12,13
Renal failure
Renal failure is the most common extra-hepatic organ failure in ACLF and occurs in over 55% 4 of cases. The International Club of Ascites recently changed the definition of acute kidney injury (AKI) in cirrhosis to include both absolute serum creatinine and its change from baseline. 20 This change has not lead to revision of CLIF-SOFA and ACLF grading. The accuracy of creatinine as a surrogate of renal function is limited in cirrhosis. 21 Measurement of creatinine alongside urine output, urinary fractional extraction of sodium and urea and novel biomarkers may resolve the current under-diagnosis of renal impairment.21,22 In ACLF, AKI is predominantly a pre-renal problem, accounting for 60-70% of cases. Whilst hepato-renal syndrome (HRS) is a pre-renal cause, it only accounts for 15–20% of all cases of AKI. 21 HRS is diagnosed following exclusion of shock, structural kidney disease and recent exposure to nephrotoxics, in patients with cirrhosis and ascites, presenting with an AKI. 20
The approach to management is the same independent of cause. It includes: removing nephrotoxic medications, excluding obstructive pathology, prompt identification and treatment of infections and timely assessment of fluid status, with institution of appropriate volume resuscitation. 20 The choice of fluids includes crystalloid, HAS and blood, but volume expansion with HAS (1 g/kg/body weight), for 48 hours, is recommended.16,20 If a patient fails to respond to volume resuscitation and meets the other criteria of HRS, a diagnosis is likely. 20 Earlier use of vasopressors may be necessary in multi-organ dysfunction.12,13,16,21 Recent Cochrane reviews are inconclusive to choice of vasopressor of choice.23,24 A recent RCT has shown a greater reversal of HRS and decreased requirement for renal replacement therapy (RRT) when terlipressin, administered as an infusion, was compared with a noradrenaline infusion 25 but this finding has not been validated. Nonetheless the poor response to treatment that is consistently observed in patients with HRS may be because HRS progresses to a non-HRS AKI. This is an area of importance in the management of ACLF. 21
RRT is utilised in approximately half of patients with ACLF and is a poor prognostic marker. Standard indications for RRT, including electrolyte disturbance, hyperammonemia and acidosis, remain applicable. RRT is not recommended in HRS unless a reversible aetiology is identified or, it is used as a bridge to liver transplantation.12,13,21
The brain
In ACLF, HE is the most common cerebral disturbance and Grade III/IV HE defines cerebral organ failure. 4 Previous HE is more common in those who develop it in ACLF. 4 The most common precipitating events are bacterial infection, active alcoholism and hyponatraemia. 26 Hyperammonaemia is invariable, but it does not correlate with HE severity. 27 Nevertheless, the latest guidance suggests that a normal plasma ammonia makes the diagnosis of HE unlikely. The development of cerebral oedema and intra-cranial hypertension are rarely clinical sequelae. 28
No trials have specifically looked at therapies for HE in ACLF. 29 As described, protection of the airway is the first priority. Precipitants, notably infection and electrolyte disturbance, should be promptly identified and treated. Treatment with lactulose ± phosphate enemas is routine practice and administration should be titrated to ensure regular bowel opening (2–3 times/day).5,16 Other ammonia-lowering therapies include L-ornitine L-aspartate (LOLA) and ornithine-phenylacetate await validation in clinical trials although the efficacy of LOLA in ammonia reduction is well established.29,30 In view of the relative infrequency of cerebral oedema and intra-cranial hypertension neuroprotection is not routinely instituted. 29 Other causes of a reduced GCS should be excluded and CT imaging is recommended in all patients. Where relevant further investigations e.g. toxicology screening and CSF analysis, should be performed. 16 At present Rifaximin is not indicated in the treatment of acute HE in the context of ACLF. 5
Infection
Infection occurs in over two thirds of patients with ACLF. It is both a precipitant and complication of this syndrome. 31 Half of the patients who present without an infection will develop later secondary infection. The most common presentations are spontaneous bacterial peritonitis, pneumonia and urinary tract infections. Bacterial infections dominate, whilst fungal infections represent the minority (5-15%).31,32 Fungal infection is more common in those who develop an infection after ACLF diagnosis and risk factors include invasive ventilation, RRT and previous antibiotic use. 32 There is a high prevalence of both nosocomial and multi-drug resistant (MDR) infections in all patients.31,33 Furthermore, severe sepsis occurs in over half of the patients who develop an infection. 31 This overwhelming burden of infection reflects the immune dysregulation which is a key component of ACLF. 34
At the point of diagnosis all patients should be considered as having underlying infection driving progression to ACLF. 34 A comprehensive infective screen should be sent and empirical broad-spectrum antibiotic therapy commenced. To enhance treatment efficacy and minimise the development of MDR, which is increasing, antibiotics should be reviewed after 48–72 hours and modified according to culture results.33,34 Empirical anti-fungal use is not recommended. 31 Where fungal infection is suspected serum biomarkers (beta-D glucan and galactomannan) can facilitate earlier diagnosis and subsequently detect treatment response. 32 Adherence to infection control procedures is essential. 31 Diagnostic paracentesis even in the absence of classical sepsis clinical features is mandated for all patients with cirrhosis and ascites who require emergency admission to hospital. 35
Coagulation
Despite concerns over increased bleeding tendency haemostasis is re-balanced in patients with stable cirrhosis. In primary haemostasis, thrombocytopenia and abnormal platelet function is offset by an increase in von Willibrand factor and a concomitant reduction in both pro-coagulant and anti-coagulant factors. In fibrinolysis, reduced plasminogen is counteracted by elevated tissue plasminogen activator and reduced factor VIII, alpha-2 anti-plasmin and thrombin-activatable fibrinolysis inhibitor.36,37 In ACLF, patients frequently demonstrate hypocoagubility and hypofibrinolysis. In a prospective trial, hypocoagubility persisted or worsened over the course of illness, but was not linked to increased risk of bleeding. 37 Worsening portal HTN may be the more important precipitant.36–38 Hypo-fibrinolysis occurred later and may have contributed to worsening organ failure due to the inflammatory response. 38 A systemic inflammatory response may alternatively manifest with thrombosis.36–38
Clotting parameters, including prothrombin time (PT), international normalized ratio (INR), fibrinogen and platelet count, are invariably abnormal in ACLF.5,12 INR correlates with disease severity but the significance of abnormal values, with regards to bleeding or thrombosis, is unclear.4,5,12 Empirical correction of clotting abnormalities is not recommended. In patients undergoing invasive procedures, if platelets <50×109/l consider platelet transfusion and if fibrinogen <120–150 mg/dl consider replacement with cryoprecipitate. 37 Thrombo-elastography (TEG) is preferred to stratify bleeding risk. 37 In small RCTs the use of TEG reduced blood product transfusions, in cirrhotic patients undergoing invasive procedures, without increased bleeding complications.39,40 TEG may more accurately reflect the haemostatic balance in ACLF, although definitive trials are awaited. Its use should be considered, alongside a standard clotting profile, to guide transfusion for high risk procedures.12,13
Referral to specialist liver centres
On admission, patients who are on the transplant waiting list should be discussed with their transplant centre. 12 They must be informed of any changes in the patient’s clinical condition as offers are accepted at any time and the status on the list may improve or worsen with the clinical situation in critical care. There should be a low-threshold for discussing patients, not listed for transplantation, to determine if they are appropriate candidates. An assessment in the ICU and, or, transfer to a specialist centre may be recommended. If the clinical trajectory is improving assessment is likely to be delayed until discharge from ICU or hospital. Referrals are centred around transplantation discussions but, if necessary, specialist centres can provide expert advice on management of patients (Figure 3). 41 An alternative reason for referral to specialist centres is refractory variceal bleeding in particular where the ability to provide injection therapy for gastric varices or TIPSS placement is not possible locally.
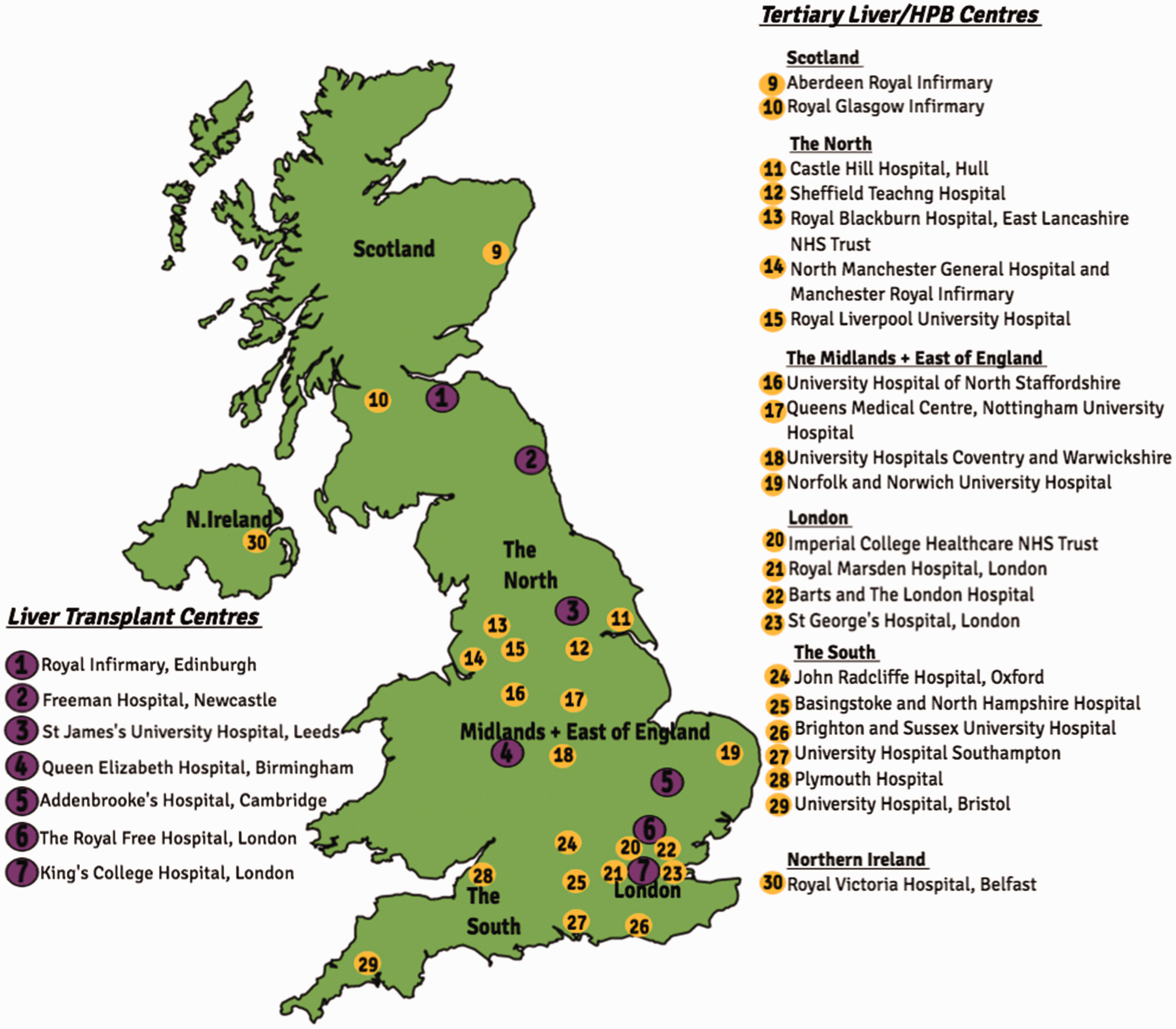
Liver transplant and tertiary liver centres in the UK. 41
Liver transplantation
There is increasing evidence to suggest that the approach to liver transplantation in ACLF should be reconsidered and that following initial stabilisation a prompt transplant assessment should be performed. 42 This is particularly true of patients with Grade-3 ACLF in whom one-year transplant-free survival is consistently less than 10%. 43 In patients with Grade-3 ACLF, listed and successfully transplanted, there is an approximately 80% one-year survival. This mirrors survival in ALF, although is significantly lower than one-year survival in patients electively transplanted.5,43–45 These patients have a greater post-transplant morbidity which translates to longer ICU and hospital stay and higher rates of post-operative complications, when compared with patients without ACLF or with lower grades of ACLF.43,45 Furthermore, these single centre experiences have not been widely replicated
The ideal time frame with which to perform semi-emergent transplantation for ACLF is unknown. In studies that have observed acceptable one-year survival, transplantation occurred within a month from listing.43–45 Even earlier transplantation may be favoured as the percentage that die or are removed from the waiting list significantly increase from 7–21 days 42 following ICU admission. It has been suggested that transplant assessment should be performed within one week and listed patients transplanted within a critical window of 7–14 days. 42 This is a non-trivial undertaking as assessing patients and reaching a decision, in the acute setting, is more complicated where thorough psychological or functional assessment is not possible.
The validity of MELD scores in this process is questioned. 45 Factors which have been linked to increased survival post-transplantation include downgrading to lower ACLF grades and the absence of both mechanical ventilation and circulatory failure.44–46 Survival is associated with younger age. Alcoholic liver disease is a more prevalent aetiology amongst survivors. 45 Marginal organs are associated with decreased survival. Earlier transplantation with a marginal organ may outweigh waiting for a better organ in view of diminished survival over time. 45 Overall the incidence of graft complications post-transplant is exceeded by complications pertaining to organ failure. 43
No futility scoring systems have accurately identified contra-indications to transplantation. 47 Established contra-indications for super-urgent transplant are active bleeding, control of sepsis <24hours, rising vasopressor requirements, severe acute respiratory distress syndrome. These remain valid exclusions applied in this cohort and consensus growing on those too sick to transplant.43,48
Extra-corporeal liver support
In ACLF the synthetic and metabolic capacities of the liver are severely impaired, leading to the accumulation of endogenous toxins and multi-organ failure. 49 The theoretical benefit of albumin dialysis with the molecular adsorbent reticulating system (MARS) is that this can be resolved with removal via an extra-corporeal circuit. In a RCT comparing medical management alone with MARS, improvements in hyperbilirubinaemia, renal function and HE were not significant enough to confer a survival benefit. 49
In ALF, high volume plasma exchange (PLEX) has demonstrated improved transplant-free survival, when compared with standard medical therapies in a RCT, 50 with a dampening of the innate immune response in recipients of PLEX. Whether this translates to ACLF awaits investigation in ongoing major interventional trials. 5 At present the use of extra corporeal liver support devices is not warranted outside the trial setting.
Futility ± establishing ceilings of care
In patients with Grade-3 ACLF, who are not candidates for transplantation, continued supportive care in the absence of improvement, has been suggested to be futile in view of the high mortality. 51 The time period with which to allow for a degree of reversibility and recovery to lower ACLF grades is not well defined. CLIF-C and CLIF-SOFA scores have demonstrated accuracy in outcome prediction, but are not validated futility scoring systems. Those with persistent MOF after 3–7 days of ICU care are however very unlikely to survive. Establishing ceilings of care is appropriate in patients in this scenario9,51 but the CLIF SOFA or indeed any scoring system is not suitable for assessing futility pre-ICU admission.
The future and conclusions
ACLF was defined and characterised only recently and further improvement on the definition is required to provide a description framework for research and clinical decision making. 4 Many unanswered questions regarding the optimum next stage of defining ACLF and in the management of these patients exist.21,52 Prospective or randomised multi-centre trials in novel therapies and liver transplantation are needed to improve the increasing survival seen with standard of care management. Immunological based therapies targeting immune dysregulation are anticipated, as understanding of ACLF pathophysiology is elucidated (Figure 4).
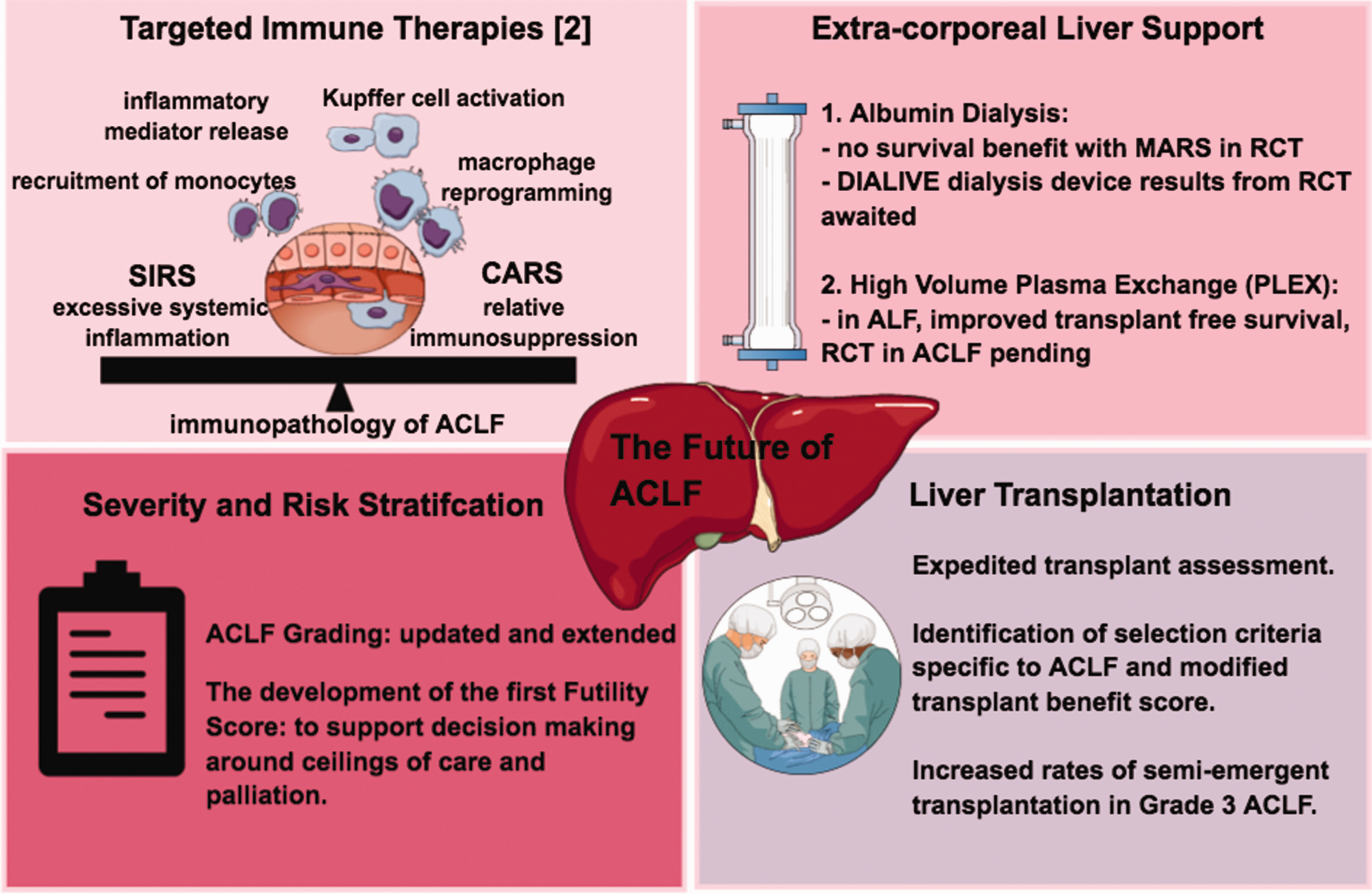
The future of ACLF management.
Admission to ICU and survival of patients with ACLF is increasing. The intensivist can therefore expect to see both more patients and more clinical trials within this cohort and should be aware of initial management paradigms and when to refer to specialist centres.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
