Abstract
Background
Acute hepatic dysfunction in the critically ill population with pre-existing liver cirrhosis is associated with a high mortality. Several prediction models have been developed to risk stratify patients with liver disease.
Methods
This UK dual-centre non-specialist hospital retrospective study (2015–2019) externally validated the Liver injury and Failure evaluation score (incorporating lactate, bilirubin and International Normalised Ratio), alongside two other general intensive care unit prediction models (Intensive Care National Audit and Research Centre and Acute Physiology and Chronic Health Evaluation II). Inclusion criteria matched a recent UK-wide study including at least one of biopsy proven cirrhosis, imaging suggestive of cirrhosis, hepatic encephalopathy or portal hypertension.
Results
One hundred and ninety-nine admissions met inclusion criteria over the study period (n = 169), mean age 57( ±13). In-hospital mortality was 40% in this cohort compared to 18% of all intensive care unit individuals during the same period. Variceal bleeding was associated with a lower short-term (18% versus 47%, P < 0.001, odds ratio 0.3 (95% confidence interval 0.1–0.5)) and longer-term mortality (log rank P = 0.015). In-patient mortality was higher in cases requiring renal replacement therapy (82% versus 29%, odds ratio 11.1 (95% confidence interval 4.6–26.9), P < 0.001) or ventilation (47% versus 32%, odds ratio 1.9 (1.1–3.4), P = 0.03). For in-patient mortality, area under the receiver operating characteristic curves were Liver injury and Failure evaluation 0.69 (95% confidence interval 0.62–0.77), Intensive Care National Audit and Research Centre 0.80 (0.74–0.86) and Acute Physiology and Chronic Health Evaluation II 0.73 (0.65–0.81). Forty-one per cent of cases were alive at one-year follow-up. Area under the receiver operating characteristic curves for one-year survival were Liver injury and Failure evaluation 0.69 (0.61–0.77), Intensive Care National Audit and Research Centre 0.75 (0.67–0.82) and Acute Physiology and Chronic Health Evaluation II 0.69 (0.61–0.77).
Conclusion
This first Liver injury and Failure evaluation score validation in a UK non-specialist hospital setting suggests this parsimonious, easy to calculate model may have utility in prediction of short-term and one-year mortality. As with previous studies variceal haemorrhage was associated with lower mortality.
Keywords
Introduction
Acute hepatic dysfunction in the critically ill population with pre-existing liver cirrhosis is associated with high mortality. 1 A spectrum of acute decompensation of cirrhosis exists which, at its most severe, includes organ failure and is referred to as acute on chronic liver failure (ACLF). 2 This has a higher associated short-term mortality.2–5 Acute decompensation is characterised by the rapid development of one or more major complications of liver disease: ascites, hepatic encephalopathy (HE), gastrointestinal haemorrhage or bacterial infection, 6 and is the main cause of hospitalisation in patients with cirrhosis. 1 Intensive care unit (ICU) admissions related to cirrhosis are on the increase in the UK, rising from 1.6 to 3.1% of all admissions between 1998 and 2012. 7
The LiFe scoring system.
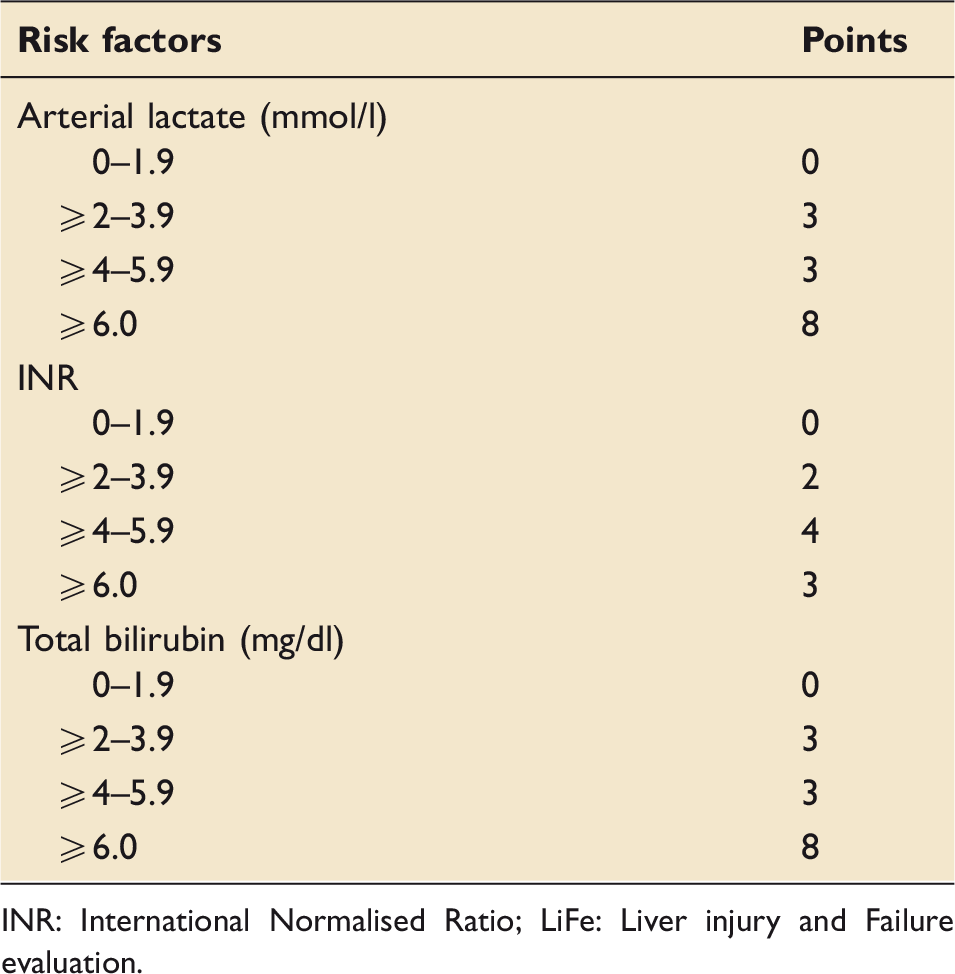
INR: International Normalised Ratio; LiFe: Liver injury and Failure evaluation.
The risk score, calculated by adding the points for each of the denoted risk factors, has a minimum of 0 and maximum of 20 points. The prognostic index was categorised into four groups: low risk (0 points), intermediate risk (1–3 points), high risk (4–8 points) and very high risk (>8 points).
First, this study aimed to validate the LiFe score in a non-specialist UK setting which reflects the majority of UK intensive care capacity. 18 Second, the study provides contemporaneous short- and longer-term outcomes for this cohort.
Methods
This UK dual-centre non-specialist hospital retrospective study was conducted between 2015 and 2019. The two hospitals comprised 22 beds with a mix of level 2 and level 3 care. In-patient and one-year mortality were recorded, alongside the presenting feature of decompensation (such as variceal bleeding or HE) and organ support (mechanical ventilation and renal replacement therapy (RRT)). Prospectively collected data were retrieved for analysis from the Reporting database of the ICU Clinical Information System (Philips IntelliSpace Critical Care and Anaesthesia). This database contains information on all ICU admissions and is used for contemporaneous medical record keeping during ICU stays. Information was retrieved by a hospital analyst not part of the research team in an anonymised fashion. The data on mortality and hospital length of stay were collected retrospectively. Following discussion with our local research department the study was considered a service evaluation of routinely collected fully anonymised data.
Inclusion criteria matched a recent UK-wide study 1 including at least one of biopsy proven cirrhosis, imaging suggestive of cirrhosis, HE or portal hypertension. Exclusion criteria included those under 18 years. Demographic, clinical and laboratory data were collected (including age, sex and ICU length of stay) alongside the primary and secondary reason for admission. Scores from data within 24 h of ICU admission were calculated for the LiFe, APACHE II and ICNARC models.
Statistical analysis was performed using SPSS V25. Continuous variables were reported as mean with standard deviation (SD) if normally distributed or median with interquartile range (IQR), as appropriate. Performance of the prognostic scores on admission were reported according to Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis recommendations including discrimination (area under the receiver operating characteristic curves (AUC)) and calibration (Hosmer–Lemeshow (H–L) test). 19 There were no missing data fields for the LiFe score. T-tests or Mann–Whitney U tests were employed for continuous dependent variables whereas categorical variables were analysed using the chi-squared test. Comparisons were reported as odds ratios (ORs) with 95% confidence intervals (CI) and/ or two-sided p values.
Results
Demographics of patient population, admission characteristics, organ support, admission blood parameters and mortality scores.
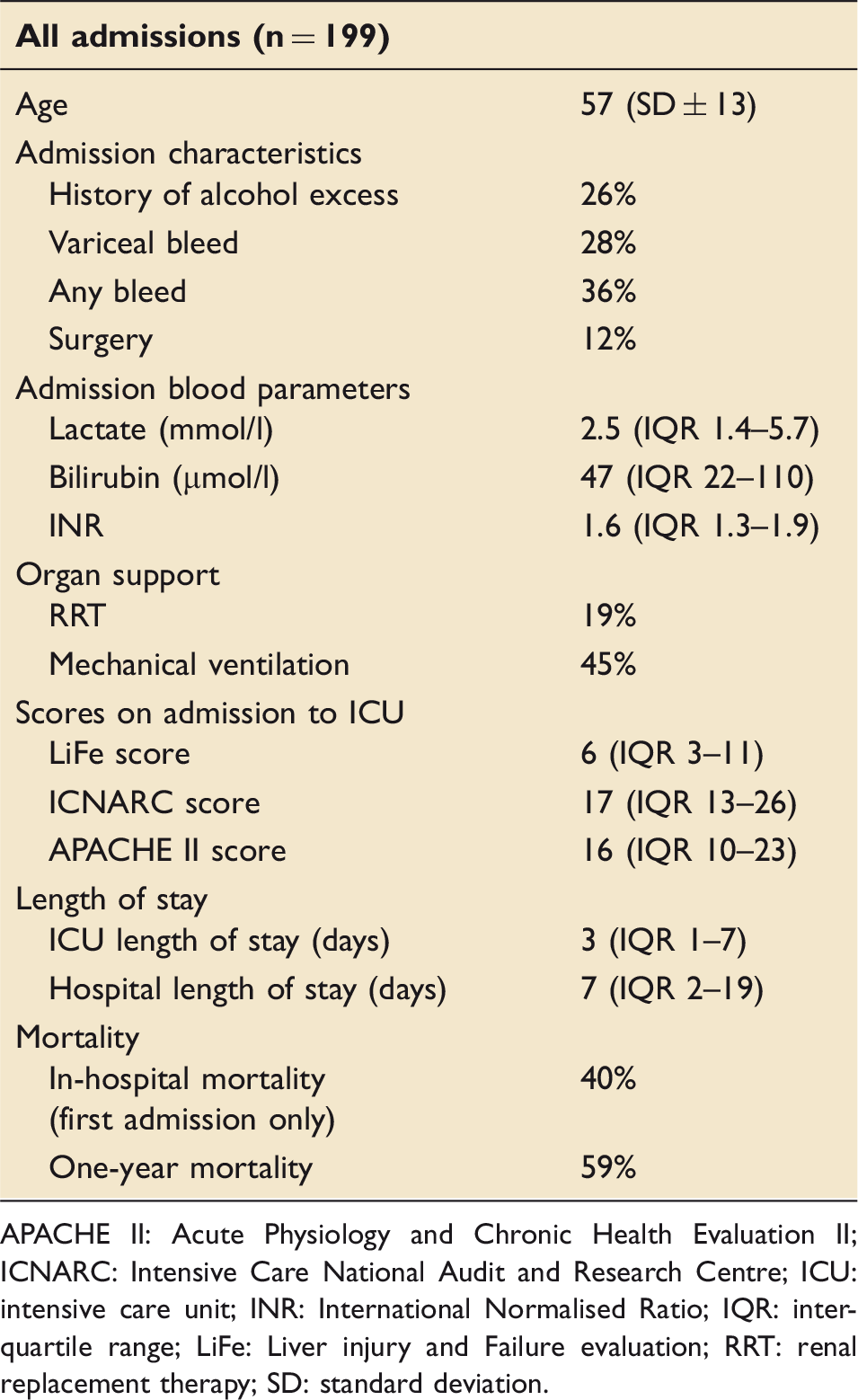
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICNARC: Intensive Care National Audit and Research Centre; ICU: intensive care unit; INR: International Normalised Ratio; IQR: interquartile range; LiFe: Liver injury and Failure evaluation; RRT: renal replacement therapy; SD: standard deviation.
Increasing LiFe score was associated with increasing in-hospital mortality. The low risk group (0 points, n = 7/40) had an in-hospital mortality of 17.5% compared to 30.6% for the intermediate risk group (1–3 points, n = 11/36) (OR 2.1 (95% CI 0.7–6.1), P = 0.280); 33.3% for high risk (4–8 points, n = 19/57) (OR 2.4 (95% CI 0.9–6.3), P = 0.105); and 60.6% for very high risk ( > 8 points, n = 40/66) (OR 7.3 (95% CI 2.8–18.8), P < 0.001).
When comparing the scoring systems for in-patient mortality, AUCs were LiFe 0.694 (95% CI 0.62–0.77), ICNARC 0.80 (0.74–0.86), APACHE II 0.73 (0.65–0.81) and lactate 0.68 (0.60–0.76). The H–L test P-value for calibration were LiFe P = 0.764, ICNARC P = 0.377, APACHE II P < 0.001 and lactate P = 0.607. AUCs for one-year survival were LiFe 0.69 (0.61–0.77), ICNARC 0.75 (0.67–0.82), APACHE II 0.69 (0.61–0.77) and lactate 0.64 (0.55–0.72). Comparative AUCs for each score are shown in Figure 1. A comparison of blood parameters and scores for LiFe, ICNARC and APACHE II between those who subsequently survived or did not survive to one year is shown in Table 3.
AUCs of LiFe, ICNARC, APACHE II and lactate for (a) in-hospital mortality and (b) one-year mortality. APACHE II: Acute Physiology and Chronic Health Evaluation II; ICNARC: Intensive Care National Audit and Research Centre; ROC: Receiver Operating Characteristic. Comparisons between one-year survivors and non-survivors (from time of first admission). APACHE II: Acute Physiology and Chronic Health Evaluation II; ICNARC: Intensive Care National Audit and Research Centre; INR: International Normalised Ratio; LiFe: Liver injury and Failure evaluation.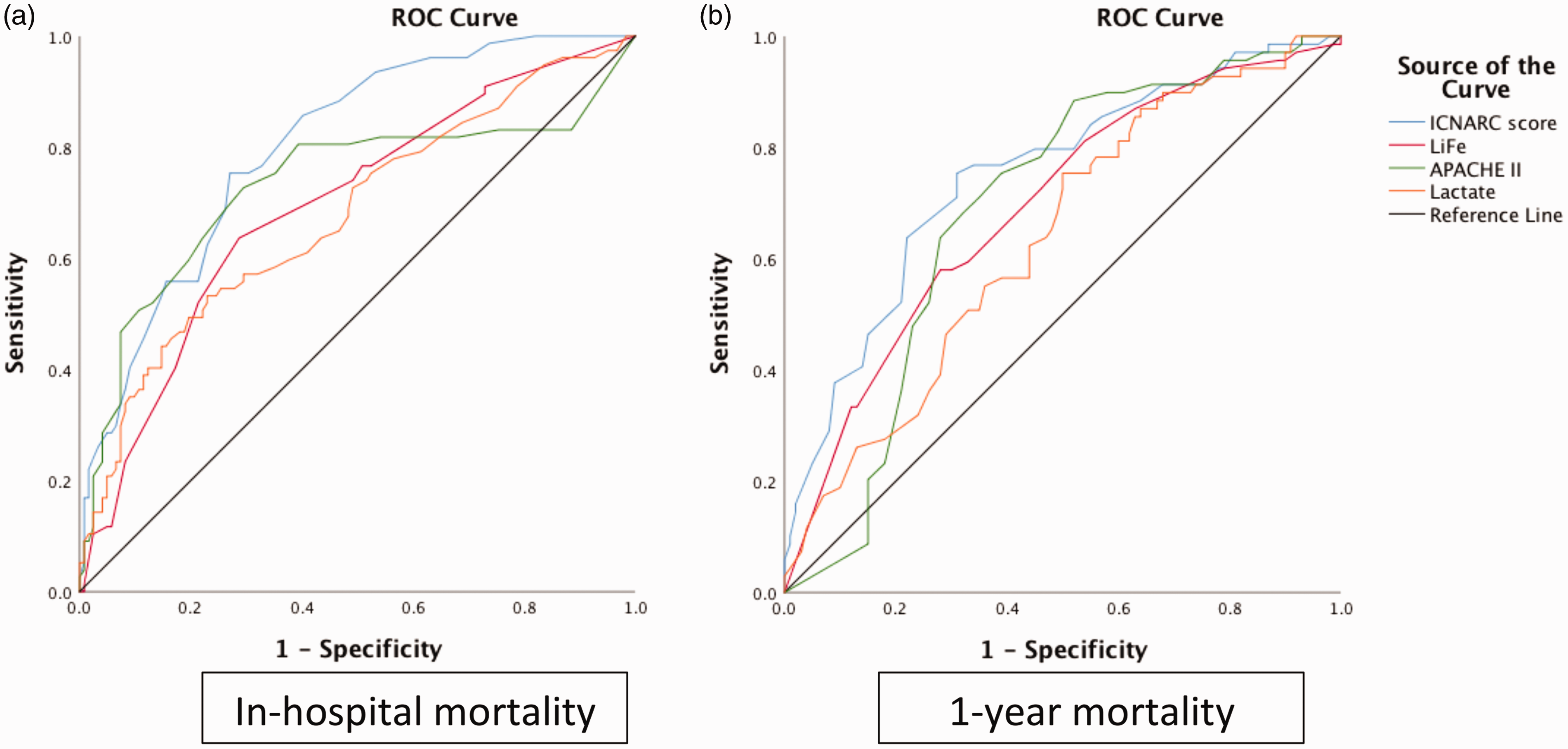

Of the 38 instances where RRT was initiated, in-patient mortality was 82% versus 29% (P < 0.001, OR 11.1 (4.6–26.9)), with only two patients in this group alive at one year following admission. The LiFe score at ICU admission was significantly higher in those who went on to receive RRT 11 (6–14) versus 6 (3–10), P < 0.001).
Requirement for ventilation was associated with increased in-hospital mortality (47% versus 32%, P = 0.03, OR 1.9 (1.1–3.4)), but not at one year (44% versus 38% alive, P = 0.53, OR 0.8 (0.4–1.5)).
Variceal bleeding accounted for 28% of admissions (n = 55) with a lower mortality (18% versus 47%, P < 0.001, OR 0.3 (95% CI 0.1–0.5)). At one-year follow-up there was no statistically significant difference in survival between those with variceal bleed (54% alive) or another cause of admission (37% alive) (OR 2.0 (0.99–4.0)); however, Kaplan–Meier survival curves did demonstrate a statistically significant increase in survival on follow-up to five years (log rank P = 0.015) for the variceal bleed group (Figure 2).
Kaplan–Meier survival curves for patients admitted with variceal bleeding or another cause of admission.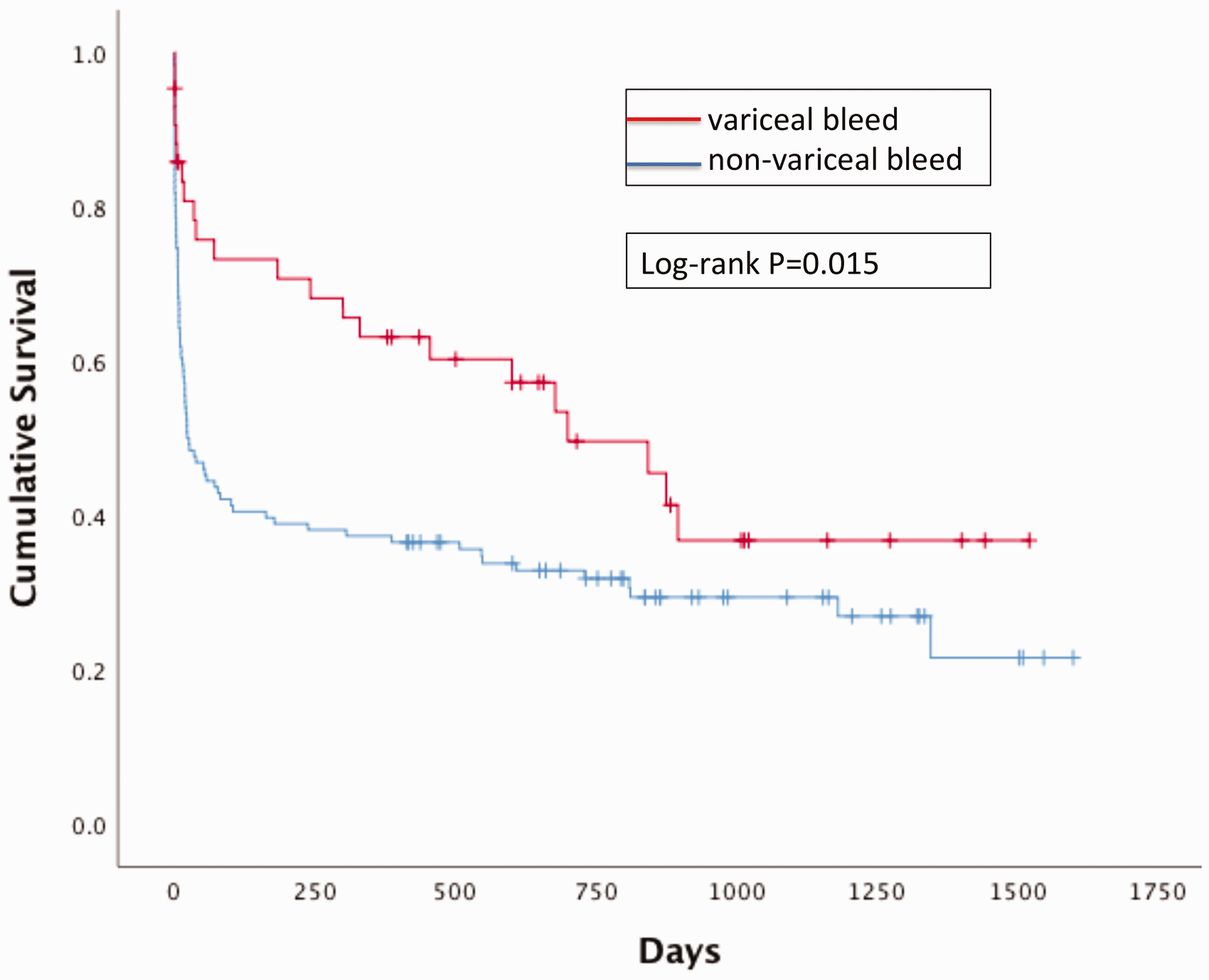
Discussion
Critically ill patients with cirrhosis have high mortality rates despite aggressive treatment and organ support.3,7,13,20 This is the first UK study to externally validate the LiFe score in a non-specialist centre and supports previous studies suggesting utility to predict mortality in critically ill patients with chronic liver disease.3,13,17 For both in-hospital and one-year mortality, the study demonstrated the LiFe score compared well with the established APACHE II score whilst the more complex ICNARC score demonstrated superior discrimination. Although the AUC for the LiFe score was only marginally higher than lactate alone, the AUC is well known to have poor sensitivity to reflect the additional value of variables 21 and all three components of the LiFe score were independent predictors of mortality.
In the UK, the majority of patients with cirrhosis are cared for on general ICUs and therefore validation of predictive tools in this setting is warranted. Prominent scoring systems such as CLIF-C ACLF were derived from teaching hospitals with transplant facilities and not exclusively from the ICU population.2,5 Unlike some of the other more complex scoring systems, the laboratory-based LiFe score is simple and convenient to calculate at the bedside on ICU admission and could help to provide objective evidence to estimate prognosis.
Risk of in-hospital death in the development cohort, validation cohorts, our cohort and the risk of in-ICU death in the Yao et al. 3 cohort, according to risk category.
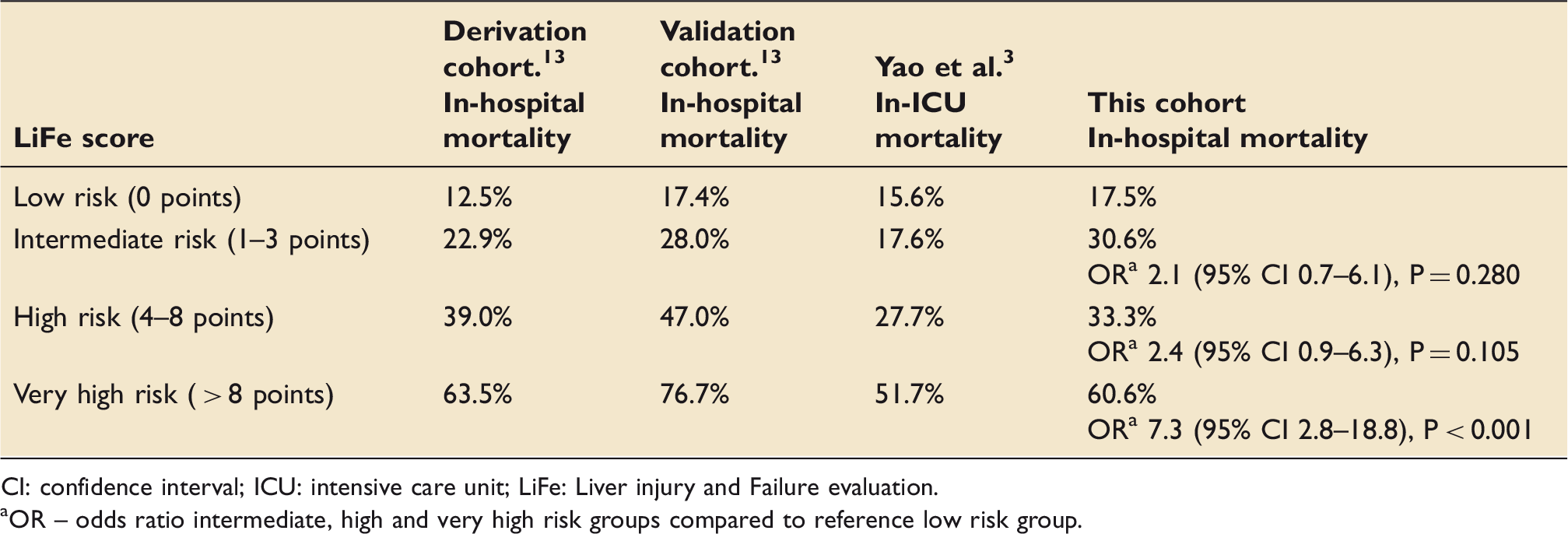
CI: confidence interval; ICU: intensive care unit; LiFe: Liver injury and Failure evaluation.
OR – odds ratio intermediate, high and very high risk groups compared to reference low risk group.
A small single centre study from Scotland (n = 83) proposed a similar score to LiFe which also incorporated albumin and found a good discrimination to predict one-year survival. 24 This study also showed that patients with variceal bleeding had a lower mortality, as reported in previous studies. 25 In addition, a recent large review of variceal bleeding admissions in Australia and New Zealand ICUs suggested improving outcomes over time. 26 This improvement may be because patients meet inclusion criteria by having a variceal bleed but may not necessarily have ACLF. 27
Our study supports previous work that suggests patients requiring RRT have significantly higher in-patient mortality. One such study found RRT to be associated with a 7-fold increase in mortality, with 50% of patients dying within one month and only 1 of 38 patients alive at one-year follow-up. 28 Requirement for ventilation was associated with increased in-hospital mortality, but not with a significant decrease in one-year survival.
The study has several weaknesses shared with others of its type, namely being partly retrospective, comprising a relatively small cohort size and being limited geographically to the south coast of England. Further such studies would be welcomed given there is some evidence to suggest regional variation in outcomes across the UK. 29 In spite of this, the population was comparable to a recent UK-wide study with similar mean ICNARC scores (this study 20.3 ± 10.6 versus 21.9 ± 10.2) 7 and in-patient mortality (40% versus 46%). We have not stratified for ACLF grades as per CLIF-SOFA2,5 and compared mortality and scores for each group. In addition, we have only included the LiFe, APACHE II and ICNARC scores and not compared them to others such as CLIF-C ACLF, 5 MELD or Child–Pugh.
Several areas of research exist that would complement this work and the existing literature on patients with cirrhosis in ICU. First, morbidity and quality of life after discharge is central to patient-centred decision-making. Whilst strong evidence for a low likelihood of survival lends additional weight to clinical experience when deciding whether to escalate care, it is not necessarily the most appropriate outcome measure. All the models for cirrhotic patients on the ICU lack the ability to quantify the likelihood of significant dependence for activities of daily living and this may mean more than their risk of dying to many patients and their families. Although the LiFe score and more complex models can help risk stratify on a population level, individual decision-making will continue to remain difficult and additional information such as pre-existing nutritional and physical status incorporating frailty assessments may further aid clinical judgement.30–32 A national validation and comparison of prediction models within the UK encompassing tertiary and non-specialist hospitals could establish the optimal model for different settings. Finally, resource allocation at the level of transplant services could also be impacted by refining the prediction models, as discussed by Galbois et al. 8 prior to the publication of the CANONIC study. 2 Gathering evidence that organ failure scores are superior at predicting outcome in cirrhotic patients could assist optimising prioritisation of recipients.3,13,33–35
In conclusion, this first UK validation of the LiFe score in a dual-centre non-specialist hospital setting suggests this parsimonious and easy to calculate model has utility in prediction of short-term and one-year mortality in patients with liver disease admitted to ICU.
Supplemental Material
sj-pdf-1-inc-10.1177_1751143720924352 - Supplemental material for Predicting outcome in liver patients admitted to intensive care: A dual-centre non-specialist hospital external validation of the Liver injury and Failure evaluation score
Supplemental material, sj-pdf-1-inc-10.1177_1751143720924352 for Predicting outcome in liver patients admitted to intensive care: A dual-centre non-specialist hospital external validation of the Liver injury and Failure evaluation score by Eleanor de Sausmarez, Paul Crowest, Steve Fry and Luke Hodgson in Journal of the Intensive Care Society
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
