Abstract
Objective
Hepatitis B virus–related acute-on-chronic liver failure is a rapidly progressive syndrome associated with high mortality. The aim of this study was to develop a dynamic prognostic score for precise outcome prediction in patients with hepatitis B virus–related acute-on-chronic liver failure.
Methods
Data from a single-center retrospective cohort were used to develop a dynamic prognostic score. The score was subsequently validated in an external multicenter prospective cohort.
Results
A total of 124 patients with hepatitis B virus–related acute-on-chronic liver failure were included in the derivation cohort. The change in the delta Chinese Severe Hepatitis B Research Group-acute-on-chronic liver failure II score between day 7 and baseline (δCOSSH-ACLF II 7-0) showed better predictive performance than the baseline Chinese Severe Hepatitis B Research Group-acute-on-chronic liver failure II score. Multivariate Cox regression analysis identified baseline total bilirubin, baseline prothrombin time international normalized ratio, and δCOSSH-ACLF II 7-0 as independent predictors of 90-day survival. We proposed a dynamic prognostic score calculated as follows: Dynamic prognostic score = 0.005 × total bilirubin + 0.609 × prothrombin time international normalized ratio + 1.234 × δCOSSH ACLF II 7-0. The area under the curves of the new score were 0.923 and 0.925 for predicting 28- and 90-day mortality, respectively. These findings were replicated in the validation cohort.
Conclusion
The new prognostic score, based on dynamic changes in the clinical course, can accurately predict short-term mortality in patients with hepatitis B virus–elated acute-on-chronic liver failure.
Keywords
Introduction
Acute-on-chronic liver failure (ACLF) is a complex clinical syndrome characterized by acute decompensation of chronic liver disease, multiple organ failure, and a high mortality rate. 1 A variety of factors, such as alcoholic liver disease and hepatitis virus infection, can cause ACLF. In many Asian countries, more than 70% of ACLF cases are related to hepatitis B virus (HBV) infection, and in China, this proportion may reach 80%.2–4 Currently, liver transplantation is the most effective treatment for selected patients with ACLF. 5 However, the shortage of donor organs, high costs, and the need for lifelong immunosuppressive therapy pose significant challenges associated with liver transplantation. Therefore, early diagnosis of ACLF and rapid prognostic assessment are crucial for identifying appropriate transplant candidates and reducing the high mortality rate.
Several models have been developed to assess the severity of ACLF and predict prognosis. The model of end-stage liver disease (MELD) has been widely used to evaluate the severity of liver disease and to allocate and manage organs for liver transplantation in patients with liver failure. 6 A large prospective multicenter cohort of patients with ACLF conducted by the European Association for the Study of the Liver-Chronic Liver Failure (EASL-CLIF) has proposed the use of the chronic liver failure-sequential organ failure assessment (CLIF-SOFA) score, which can more accurately predict outcomes. This score was subsequently simplified to the Chronic Liver Failure Consortium (CLIF-C)-ACLF score.7,8 Other large prospective multicenter cohort studies on Chinese Severe Hepatitis B Research Group (COSSH) have proposed COSSH-ACLF and COSSH-ACLF II scores, especially for HBV-ACLF. 9 The CLIF-C-ACLF and COSSH-ACLF II scores have been reported to have a predictive accuracy of approximately 80% for 28- and 90-day mortality. 10
These scoring models are generally based on clinical data collected at the time of hospital admission of patients with ACLF to assess prognosis. However, the clinical course of ACLF is highly dynamic. In addition to disease severity at admission, the response to treatment and the reversibility of the disease are strongly associated with outcomes. 11 Prognostic models based on a single initial time point may not be appropriate for patients with rapidly progressive ACLF or for those whose condition significantly improves after effective therapy. To provide a more accurate prediction of 90-day mortality, we developed a new prognostic model incorporating baseline indices and the change in COSSH-ACLF II scores between admission and day 7. The new model was subsequently validated in a multicenter prospective cohort.
Materials and methods
Study design
First, clinical characteristics and prognostic indicators at admission and on days 3 and 7 were collected to validate the MELD, CLIF-C ACLF, and COSSH-ACLF II scores and to develop a new prognostic score. The newly developed score was subsequently validated in a multicenter prospective cohort. Detailed clinical data and outcomes for all enrolled patients were collected and recorded in case report forms (see Supplementary Materials) at admission, on days 3 and 7, and at 90 days of follow-up. The study was conducted in accordance with the latest version of the Declaration of Helsinki (2024) and was approved by the Ethics Committee of The First Affiliated Hospital of Anhui Medical University, China (PJ2022-07-18). The requirement for written informed consent by individual patients was waived by the Ethics Committee of The First Affiliated Hospital of Anhui Medical University because of the retrospective nature of the study. The reporting of this study conforms to the Standards for Reporting of Diagnostic Accuracy (STARD) guidelines. 12
Patients
We established a retrospective derivation cohort and a prospective validation cohort. Patients with ACLF were initially screened at the Department of Infectious Disease, The First Affiliated Hospital of Anhui Medical University, from July 2021 to June 2022. The diagnosis of ACLF was made based on the COSSH-ACLF diagnostic criteria. The exclusion criteria are summarized in Figure 1. During hospitalization, all patients received treatment consistent with that used in the COSSH-ACLF study, including standard medical treatment (SMT; the detailed SMT protocols are provided in the Supplementary Materials). The artificial liver support system was administered according to the Chinese guidelines for liver failure published in 2020. Patients in the validation cohort were enrolled using the same inclusion and exclusion criteria (Figure 1). They received the same treatment protocols as those in the derivation cohort and were recruited from five liver units across different hospitals.

Flowchart of the study. Patients were screened, enrolled, and excluded according to the COSSH-ACLF criteria. ACLF: acute-on-chronic liver failure; COSSH: Chinese Group on the Study of Severe Hepatitis B; HBV-ACLF: hepatitis B virus–related ACLF; LT, liver transplantation.
Development and validation of the HBV-ACLF prognostic score
We aimed to develop a new prognostic score to predict 90-day outcomes in patients with HBV-ACLF. Four main steps were undertaken. First, we calculated the difference between the COSSH-ACLF II score at baseline and at day 7, and the delta value of the two scores was used as an index for subsequent analyses. Second, univariate and multivariate Cox proportional hazards regression analyses were performed, including δCOSSH-ACLF II and other laboratory indices as candidate risk factors, to identify variables most strongly associated with 90-day mortality. The independent risk factors identified by multivariate Cox regression were used to construct the new prognostic score. Third, the performance of the new prognostic score was compared with that of the three previously described prognostic scoring systems using receiver operating characteristic (ROC) curve analysis. The new score was further evaluated by calibration curve and decision curve analyses and was ultimately presented in the form of a nomogram. Fourth, the new prognostic score was validated in an external validation cohort that included data from five centers.
Risk stratification
The optimal cutoff value of the new prognostic score was determined using X-tile software (version 3.6.1; Yale University, New Haven, CT, USA) by selecting the largest χ2 value to stratify patients into low- and high-risk groups for mortality.
Statistical analysis
Statistical analyses were performed using SPSS Statistics version 25.0 (SPSS, Chicago, IL, USA), GraphPad Prism version 10.0 (GraphPad Software, San Diego, CA, USA), and R software version 4.4.0 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were presented as the mean ± standard deviation (SD) for normally distributed data or as the median with interquartile range (IQR) for non-normally distributed data. Categorical variables were expressed as absolute frequencies and percentages. Intergroup differences for continuous variables were assessed using Student’s t test or the Mann–Whitney U test, as appropriate. Categorical variables were compared using the χ2 test. A two-tailed p value <0.05 was considered statistically significant. Survival curves across different ACLF grades or risk strata were generated using GraphPad Prism. The area under the ROC, decision curve analysis (DCA), and calibration curves were constructed using R software.
Results
Patients and clinical characteristics
Among 242 patients hospitalized with HBV-ACLF, 70 were excluded from the study, including 46 patients with hepatocellular carcinoma or other tumors, 6 with severe extrahepatic diseases, 6 receiving immunosuppressive drugs for other conditions, 4 with uncontrolled mental illness, 2 aged >80 years, 2 pregnant patients, and 4 for other reasons. In addition, 48 patients who underwent liver transplantation or were lost to follow-up during the 90-day period were excluded from the analysis of 90-day mortality (Figure 1). Among the 124 included patients, 27 (21.8%) died within 7 days, and a cumulative total of 71 (57.3%) died within 90 days. The K-M curves showed the survival probability of patients with HBV-ACLF stratified by ACLF grade at 7, 28, and 90 days (Figure 2). The clinical characteristics of all enrolled patients are summarized in Table 1. Most patients with HBV-ACLF in both the deceased and survival groups were male. The mean age of patients in the deceased group was 50 ± 12 years, which was not significantly different from that in the survival group (48 ± 12 years). Laboratory analyses demonstrated that compared with the survival group, the deceased group had significantly higher levels of total bilirubin (TBIL), serum creatinine, serum urea, white blood cell count, neutrophils, international normalized ratio (INR), and prothrombin time and significantly lower levels of albumin (ALB), glutamyl transpeptidase (γ-GT), triglycerides (TG), total cholesterol (Tch), low-density lipoprotein cholesterol (LDL-C), and serum sodium. The COSSH-ACLF II, CLIF-C-ACLF, and MELD scores for patients with HBV-ACLF in the deceased group were 7.5 (7.1–8.3), 43.7 (38.5–47.0), and 25.4 (22.3–30.3), respectively, which were significantly higher than those in the survival group (CLIF-C-ACLF: 39.3 (34.2–43.5), COSSH-ACLF II: 6.8 (6.4–7.3), and MELD: 21.1 (18.5–23.8)).

Kaplan–Meier curves for overall ACLF and ACLF grades 1, 2, and 3 over 90 days. ACLF grade 1 includes four subgroups: (1) patients with kidney failure alone; (2) patients with isolated liver failure with INR ≥1.5 and/or kidney dysfunction and/or HE grade 1 or 2; (3) patients with a single organ failure involving the coagulation, circulatory, or respiratory system and/or kidney dysfunction and/or HE grade 1 or 2; and (4) patients with cerebral failure alone plus kidney dysfunction. ACLF grade 2 includes patients with failure of two organ systems. ACLF grade 3 includes patients with failure of three or more organ systems. Organ failure definitions were as follows: kidney dysfunction, serum creatinine 1.5–1.9 mg/dL; kidney failure, serum creatinine ≥2 mg/dL; liver failure, total bilirubin ≥12 mg/dL; cerebral failure, HE grade 3 or 4; coagulation failure, INR >2.5; circulatory failure, use of vasoactive agents; respiratory failure, PaO2/FiO2 ≤200 or SpO2/FiO2 ≤200. ACLF: acute-on-chronic liver failure; HE: hepatic encephalopathy; INR: international normalized ratio.
Clinical characteristics of patients in the deceased and survival groups in the derivation cohort.

The data are expressed as medians (IQR), mean ± (SD) or number of patients (%).
ALB: albumin; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BI: bacterial infection; CLIF-C ACLFs: Chronic Liver Failure Consortium ACLF score; COSSH-ACLF П: Chinese Group on the Study of Severe Hepatitis B-ACLF score II; DD: D dimer; FIB: fibrinogen; GIH: gastrointestinal hemorrhage; GLO: globulin; Glu: fasting blood glucose; HDL-C: high-density lipoprotein cholesterol; HE: hepatic encephalopathy; HGB hemoglobin; Hs-CRP: high-sensitivity C-reactive protein; K+: serum potassium; LDL-C: low-density lipoprotein cholesterol; MAP: mean arterial pressure; MELDs: model for end-stage liver disease score; Na+: serum sodium; PCV: packed cell volume; PLT: platelet count; PT: prothrombin time; PT-INR: prothrombin time international normalized ratio; TBIL: total bilirubin; TBA: total bile acid; Tch: total cholesterol; TG: triglycerides; TP: total protein; WBC: white blood cell count; γ-GT: glutamyl transpeptidase; HBV-DNA: hepatitis B virus DNA.
ROC curves were used to validate the predictive performance of the model and compare it to the abovementioned prognostic scores. The area under the curve (AUC) values for the MELD score were 0.801, 0.621, and 0.673 for predicting 7-, 28-, and 90-day mortality, respectively. The corresponding AUCs for the CLIF-C-ACLF score were 0.705, 0.696, and 0.640, respectively. The AUCs for the COSSH-ACLF II score were 0.790, 0.774, and 0.733, respectively. Overall, the COSSH-ACLF II score showed the best predictive performance for 7-, 28-, and 90-day outcomes. Notably, the 90-day predictive performance of all three models was lower than that of the 7-day model (Figure 3(a)), indicating that baseline disease severity alone was insufficient to predict 3-month outcomes.
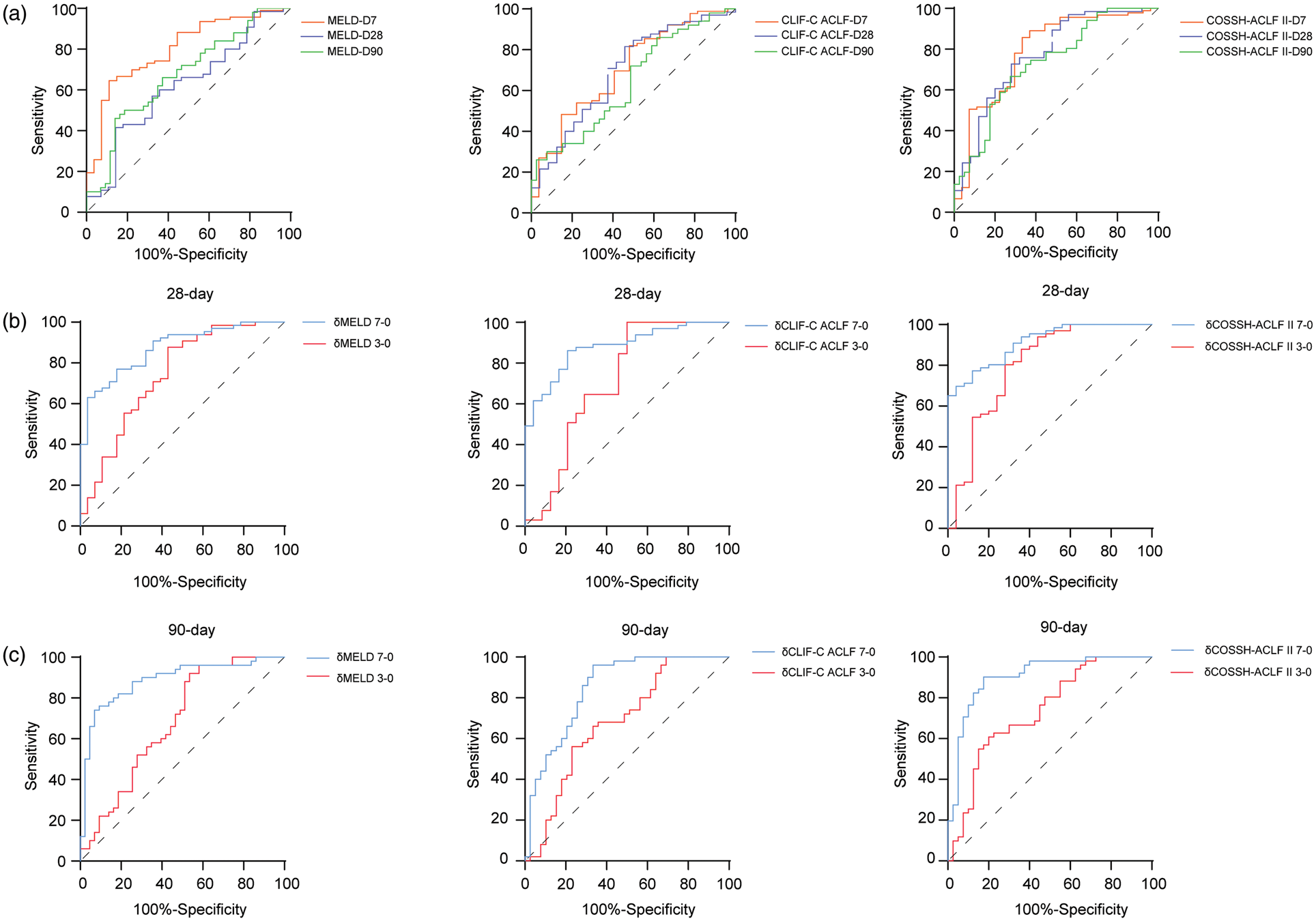
ROC of prognostic scores. (a) ROC curves of MELD, CLIF-C-ACLF, and COSSH-ACLF II scores for predicting 7-, 28-, and 90-day mortality. (b) ROC curves of δMELD, δCLIF-C-ACLF, and δCOSSH-ACLF II (day 7–baseline and day 3–baseline) for predicting 28-day mortality. (c) ROC curves of δMELD, δCLIF-C-ACLF, and δCOSSH-ACLF II (day 7–baseline and day 3–baseline) for predicting 90-day mortality. MELD: model of end-stage liver disease; CLIF-C-ACLF: chronic liver failure consortium acute-on-chronic liver failure; COSSH-ACLF: Chinese Group on the Study of Severe Hepatitis B–acute-on-chronic liver Failure; ROC: receiver operating characteristic.
We subsequently calculated the difference between the three scores on day 3 and day 7 and compared it with the baseline scores. ROC analysis showed that δMELD 7-0 was better than δMELD 3-0 (AUC: 0.872 vs. 0.741; p = 0.0518) and that δCLIF-C ACLF 7-0 and δCOSSH-ACLF II 7-0 were significantly better than δCLIF-C ACLF 3-0 and δCOSSH-ACLF II 3-0 (AUC: 0.879 vs. 0.712, p = 0.0062; and 0.913 vs. 0.800, p = 0.0277, respectively) for predicting 28-day mortality (Figure 3(b)). For predicting 90-day mortality, δMELD 7-0, δCLIF-C ACLF 7-0, and δCOSSH-ACLF II 7-0 (AUC: 0.887, 0.848, and 0.905, respectively) also performed significantly better than δMELD3-0, δCLIF-C ACLF3-0, and δCOSSH-ACLF II 3-0 (AUC: 0.686, 0.681, and 0.737; p = 0.0001, 0.0012, and 0.0001, respectively) (Figure 3(c)). Furthermore, based on the obtained AUC values, δCOSSH-ACLF II 7-0 demonstrated superior predictive performance compared with δCLIF-C ACLF 7-0 and δMELD 7-0 in predicting both 28-day (AUC: 0.913, 0.872, and 0.879, respectively) and 90-day mortality (AUCs: 0.905, 0.848, and 0.887, respectively; Figure S1). Therefore, δCOSSH-ACLF II7-0 was selected for inclusion in the subsequent analyses.
Development of a prognostic score
Univariate Cox regression analysis revealed that TBIL, ALB, Tch, serum sodium, serum creatinine, serum urea, prothrombin time (PT)-INR, FIB, and δCOSSH-ACLF II 7-0 were associated with 90-day mortality. Multivariate Cox regression analysis further demonstrated that TBIL, INR, and COSSH-ACLF II 7-0 were independent risk factors for 90-day mortality (Table S1). We subsequently established a prognostic score using the following formula: 7-day dynamic score model (DSM) = 0.005 ×TBIL + 0.609 × INR + 1.234 × COSSH-ACLF II 7-0. Compared with the MELD, CLIF-C ACLF, and COSSH-ACLF II scores, the new score demonstrated significantly improved predictive performance, with the highest AUC values for predicting 28- and 90-day mortality in patients with HBV-ACLF (0.923 and 0.925, respectively; p < 0.05) (Figure 4). ROC curve analysis further showed that the 7-day DSM outperformed δCOSSH-ACLF II 7-0 alone (28- and 90-day AUCs: 0.913 and 0.905, respectively) (Figure S2).
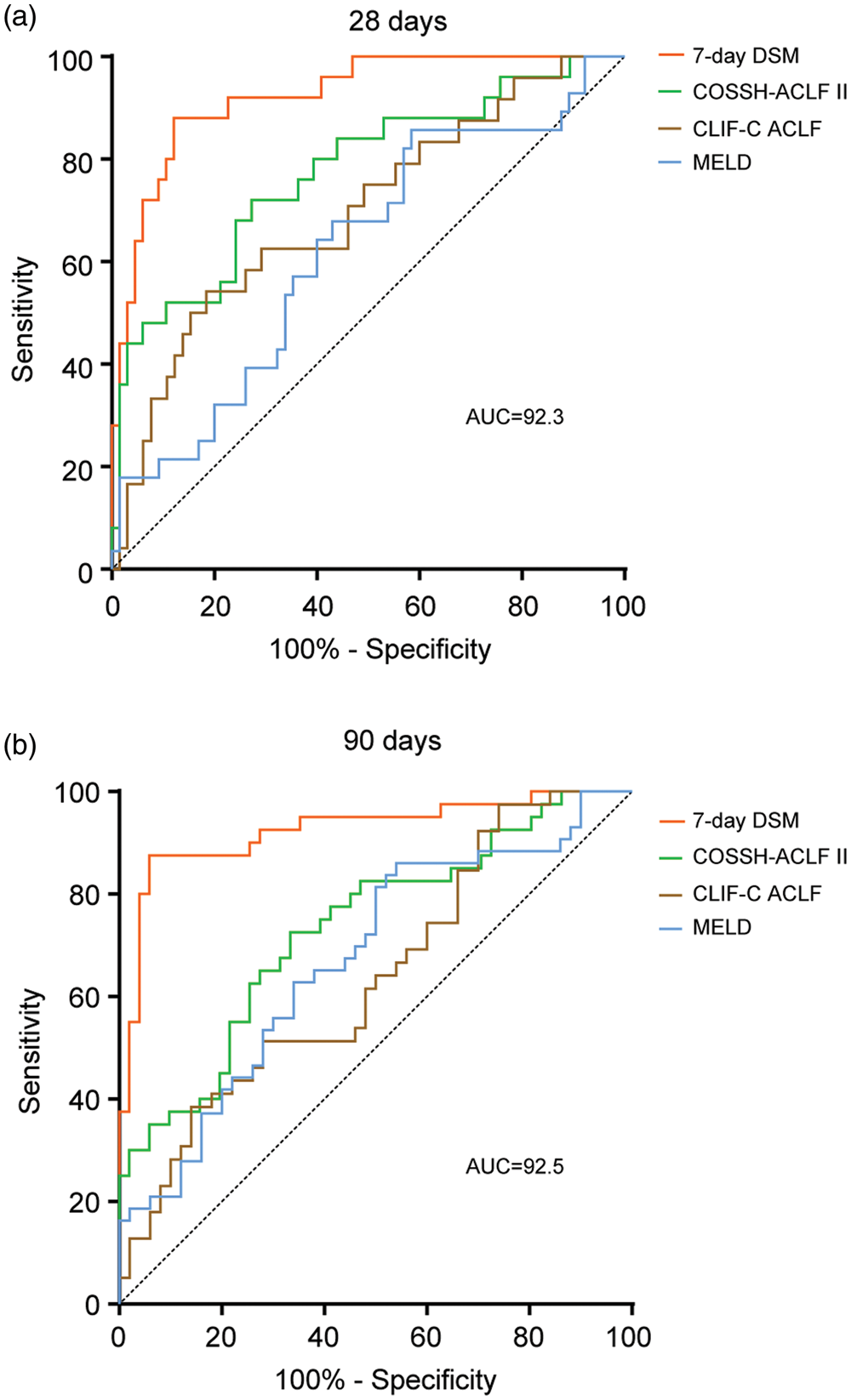
ROC curves of the 7-day dynamic scoring model. (a) ROC curves of the 7-day dynamic scoring model, MELD, CLIF-C-ACLF, and COSSH-ACLF II scores for predicting 28-day mortality. (b) ROC curves of the 7-day dynamic scoring model, MELD, CLIF-C-ACLF, and COSSH-ACLF II scores for predicting 90-day mortality. MELD: model of end-stage liver disease; ROC: receiver operating characteristic; CLIF-C-ACLF: chronic liver failure consortium acute-on-chronic liver failure; COSSH-ACLF: Chinese Group on the Study of Severe Hepatitis B–acute-on-chronic liver failure.
Estimation, validation, and visual and risk stratification of the new score
To validate the performance of the 7-day DSM, an external validation cohort comprising 72 patients from 5 centers was enrolled; detailed exclusion criteria are shown in Figure 1. The clinical characteristics are summarized in Table 2. The main indices, including severity scores and mortality rates, did not significantly differ from those in the derivation cohort. Although certain indicators, such as HDL-C, LDL-C, fasting blood glucose (Glu), ferritin, and D dimer (DD), differed between the derivation and validation cohorts, these differences did not affect the validation outcomes. Calibration analysis demonstrated that the 7-day DSM showed good overall performance and promising predictive accuracy for death at 90 days in both the derivation and validation groups (Figure 5(a) and (b)). DCA also revealed a good net benefit of the 7-day DSM in both groups, indicating its high utility for clinical application (Figure 5(c) and (d)). ROC curve analysis revealed that the 7-day DSM score was more accurate than COSSH-ACLF II (Figure 5(e) and (f)) and δCOSSH-ACLF II 7-0 (Figure S3) in predicting 28-day and 90-day mortality in the validation group (28-day AUCs: 0.8424 vs. 0.7998, 0.8424 vs. 0.8163; 90-day AUCs: 0.8798 vs. 0.8186, 0.8798 vs. 0.8404). Finally, to visually assess the 7-day DSM, we constructed a nomogram to predict the probability of 90-day survival (Figure 6).
Clinical characteristics of patients in the derivation and validation cohorts.

The data are expressed as medians (IQR), mean ± (SD) or number of patients (%).
ALB: albumin; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BI: bacterial infection; COSSH-ACLF П: Chinese Group on the Study of Severe Hepatitis B-ACLF score; DD: D dimer; FIB: fibrinogen; GIH: gastrointestinal hemorrhage; GLO: globulin; Glu: fasting blood glucose; HDL-C: high-density lipoprotein cholesterol; HGB hemoglobin; INR: international normalized ratio; K+: serum potassium; LDL-C: low-density lipoprotein cholesterol; MAP: mean arterial pressure; Na+: serum sodium; PLT: platelet count; PT: prothrombin time; TBIL: total bilirubin; TBA: total bile acid; Tch: total cholesterol; TG: triglycerides; TP: total protein; WBC: white blood cell count; γ-GT: glutamyl transpeptidase.

Estimation and validation of the 7-day dynamic scoring model. (a, b) Calibration curves of the 7-day DSM in the derivation and validation cohorts. (c, d) DCA of the 7-day DSM in the derivation and validation cohorts. (e, f) ROC curves of the 7-day DSM and COSSH-ACLF II for predicting 28- and 90-day mortality in the validation cohort. DCA: decision curve analysis; DSM: dynamic score model; ROC: receiver operating characteristic; COSSH-ACLF: Chinese Group on the Study of Severe Hepatitis B–acute-on-chronic liver failure.

Nomogram of the 7-day dynamic scoring model for predicting 90-day outcomes.
We also performed risk stratification based on the 7-day DSM. Risk stratification of the 7-day DSM with an X-tile plot revealed that patients with HBV-ACLF could be divided into two 90-days mortality risk strata based on the optimal cutoff value of 3.52: low risk (<3.52) and high risk (≥3.52). The 90-day mortality rates of each group significantly differed (low risk, 14.0%; high risk, 94.1%). Compared with the low-risk group, the hazard ratio for 90-day mortality in the high-risk group reached 18.328 (p < 0.001) (Figure 7(a)). These findings indicate that the new prognostic score provides a simple and accurate method for risk stratification and prediction of disease severity. In the external validation cohort, the 90-day mortality rates also significantly differed between the two groups (low risk, 28.2%; high risk, 91.6%). Compared with the low-risk group, the hazard ratio for 90-day mortality in the high-risk group was 6.564 (p < 0.001) (Figure 7(b)).
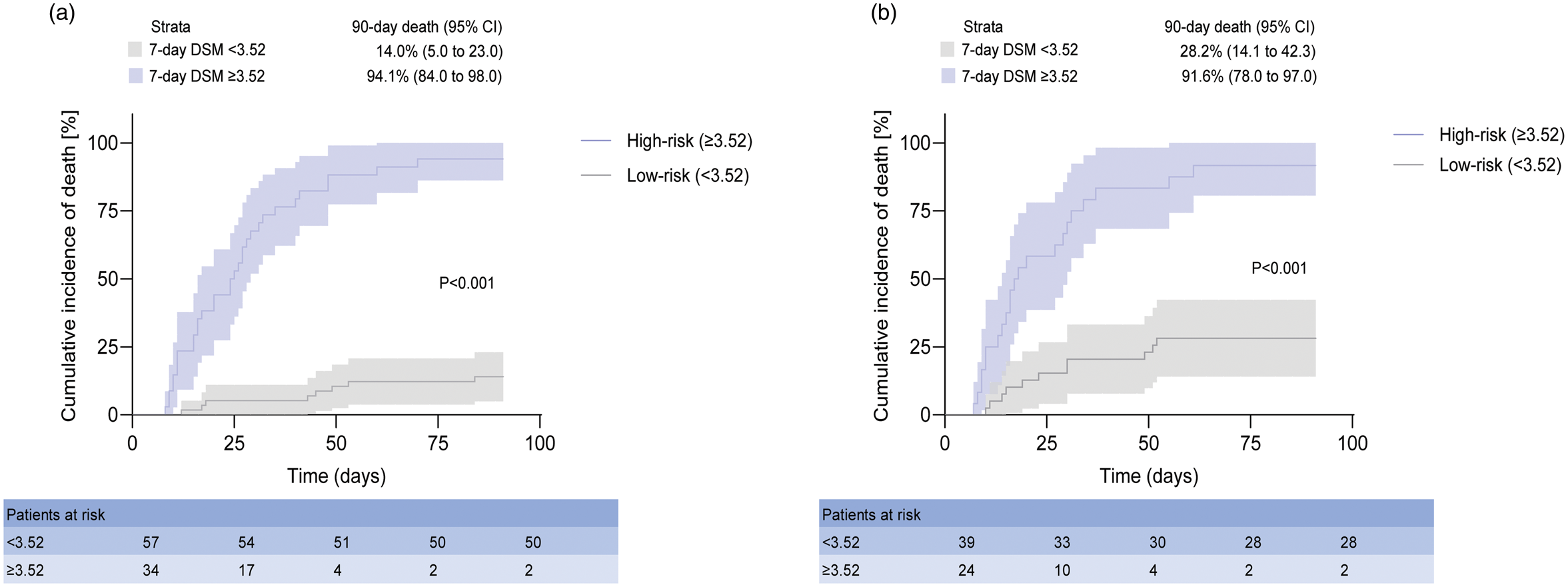
Risk stratification based on the 7-day DSM. (a) Derivation group. (b) Validation group. Cumulative incidence of 90-day mortality stratified according to the 7-day DSM classification rule (low risk: <3.52; high risk: ≥3.52). p < 0.001 (log-rank test) for comparisons between the two risk strata.
Discussion
Because of its complexity and high mortality, early prognostication remains a pivotal topic in ACLF research. The classic MELD score, originally proposed for liver transplantation assessment, is often used to predict outcomes in patients with ACLF. 13 An ACLF-specific prognostic model, the CLIF-C ACLF score, has been shown to accurately predict outcomes in patients with all-cause ACLF. 14 Compared with other models, the more recently proposed COSSH-ACLF II score has been demonstrated to be superior in patients with HBV-ACLF. 10 In this study, we found that the current prognostic models strongly predicted outcomes in patients with ACLF, particularly among those who died shortly after disease onset. However, in patients who survived for a relatively longer duration, the predictive performance of these models based on baseline indices decreased. This finding suggests that disease severity at admission is not the sole determinant of prognosis. Based on these observations, we hypothesize that, in addition to disease severity, the degree of reversibility and the response to treatment also influence the final outcomes.
Approximately 60% of patients with ACLF experience precipitating events. 15 Both hepatic and nonhepatic precipitating events can trigger systemic inflammatory response syndrome (SIRS) through damage-associated molecular patterns or pathogen-associated molecular patterns, ultimately leading to multiple organ failure. 16 The SIRS phase typically lasts approximately one week and is referred to as the “golden window.” 17 A study of patients with ACLF reported that the cumulative incidence of new-onset SIRS was 29% by day 4 but sharply increased to 92.8% by day 7; moreover, the absence of SIRS during the first week was associated with a lower incidence of organ failure. 18 Another study demonstrated that the lactate clearance rate measured over the first week after admission was significantly greater in the survival group than in the deceased group. 19 In our study, we compared scores on day 3 and day 7 and found that the dynamic difference in scores on day 7 compared with baseline was more accurate than that on day 3 relative to baseline for predicting 90-day outcomes. We believe that effective therapy during the first week after symptom onset is critical throughout the clinical course and is strongly associated with patient outcomes. Reversibility is a characteristic feature of ACLF that distinguishes it from end-stage liver disease. The CANONIC study reported that ACLF resolved or improved in 49.2% of patients. 11 A study of HBV-ACLF reported that patients with a prior history of decompensation diagnosed according to the APASL-ACLF Research Consortium criteria exhibited favorable reversibility and maintained a stable condition after receiving nucleoside analogs. 20 Given the reversibility of ACLF, increasing attention has been directed toward dynamic assessments of disease severity in recent years. A previously mentioned study evaluated the clinical course by comparing CLIF-C ACLF scores on day 3 and day 7 and demonstrated that dynamic changes in CLIF-C ACLF scores were strongly associated with prognosis. 11 A recent multicenter study of patients with HBV-ACLF established a novel multistate model by assessing ACLF grade at different time points, which performed better than traditional prognostic scores. 21 Another study identified age, World Gastroenterology Organization type, underlying etiology, TBIL level, creatinine level, prothrombin activity, and hepatic encephalopathy stage as independent prognostic factors in patients with ACLF and proposed a DP-ACLF score based on the dynamic trends of these indicators. 22 However, these dynamic scoring systems were derived from retrospective data, and most are too complex for routine clinical application. In the present study, we validated mainstream prognostic scores in a prospective cohort and developed a new dynamic score based on baseline TBIL level, PT-INR, and the dynamic δCOSSH-ACLF II scores between day 7 and baseline. We subsequently validated this score in a multicenter prospective cohort and further evaluated its performance using discrimination and calibration analyses.
This study has several limitations. The sample sizes of both the derivation and validation cohorts were small, which may limit the generalizability and statistical power of the findings. Therefore, larger and more diverse cohorts are needed to further validate the new score in future studies.
Conclusion
We developed a new dynamic prognostic score based on changes in the clinical course, which accurately predicts short-term mortality in patients with HBV-ACLF and outperforms existing scoring systems.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261429829 - Supplemental material for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure
Supplemental material, sj-pdf-1-imr-10.1177_03000605261429829 for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure by Jiemenglu Li, Jian Yang, Chunyan Jiang, Qingting Zhao, Yufeng Gao and Jiang Li in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605261429829 - Supplemental material for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure
Supplemental material, sj-pdf-2-imr-10.1177_03000605261429829 for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure by Jiemenglu Li, Jian Yang, Chunyan Jiang, Qingting Zhao, Yufeng Gao and Jiang Li in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605261429829 - Supplemental material for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure
Supplemental material, sj-pdf-3-imr-10.1177_03000605261429829 for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure by Jiemenglu Li, Jian Yang, Chunyan Jiang, Qingting Zhao, Yufeng Gao and Jiang Li in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605261429829 - Supplemental material for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure
Supplemental material, sj-pdf-4-imr-10.1177_03000605261429829 for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure by Jiemenglu Li, Jian Yang, Chunyan Jiang, Qingting Zhao, Yufeng Gao and Jiang Li in Journal of International Medical Research
Supplemental Material
sj-pdf-5-imr-10.1177_03000605261429829 - Supplemental material for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure
Supplemental material, sj-pdf-5-imr-10.1177_03000605261429829 for Development and validation of a dynamic prognostic score for hepatitis B virus–related acute-on-chronic liver failure by Jiemenglu Li, Jian Yang, Chunyan Jiang, Qingting Zhao, Yufeng Gao and Jiang Li in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank Dr Nan Xu (Department of Infectious Diseases, The First Affiliated Hospital of Anhui Medical University) for providing helpful comments during manuscript preparation.
Author contribution statement
The study was designed by Jiang Li and supervised by Jiemenglu Li, Chunyan Jiang, and Jian Yang. The manuscript was written by Jiang Li and Jiemenglu Li. Data collection, analysis, and interpretation were performed by Jiemenglu Li, Chunyan Jiang, and Yufeng Gao. All authors participated in the critical revision of the manuscript and approved the final version.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors report no conflicts of interest in this work.
Funding
This study was supported by the National Natural Science Foundation of China Youth Program (Funding number: 82102293), the National Natural Science Foundation of China (Funding number: 82370608), and the Anhui Province Clinical Medical Research Transformation Special Fund (Funding number: 202304295107020040).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
