Abstract
The efficacy and tolerability of eribulin mesylate, a synthetic halichondrin B analog, in patients with metastatic breast cancer (MBC) previously treated with anthracyclines and taxanes have been established. Acute-on-chronic liver failure (ACLF) is a clinical syndrome manifesting as acute and severe hepatic derangement resulting from varied insults in patients with established chronic liver disease or cirrhosis who did not previously receive eribulin. A middle-aged woman diagnosed with MBC and diffuse liver metastases who was pretreated with multi-line chemotherapy received eribulin as eighth-line chemotherapy and presented with hepatic encephalopathy, rapid bilirubin elevation, and significant coagulation dysfunction on day 4 in cycle 1. The patient was diagnosed with ACLF induced by eribulin. Therefore, ACLF may be a lethal and rare adverse event when patients with chronic liver metastases receive eribulin treatment, and clinicians’ awareness should be increased for optimal prevention and prompt diagnosis and treatment.
Keywords
Introduction
Metastatic breast cancer (MBC) remains a lethal threat to women globally, with a poor prognosis. 1 Treatments with survival benefits are urgently needed for women with MBC, particularly those with heavily pretreated MBC.
Eribulin mesylate is a synthetic analog of halichondrin B, which is a natural product isolated from the marine sponge Halichondria okadai. Eribulin is a non-taxane, microtubule dynamics inhibitor that is markedly distinct from other tubulin-targeting agents because it binds to high-affinity sites on the growing ends of microtubules, suppressing migration and invasion and reversing epithelial–mesenchymal transition.2–7 Practically, substantial evidence has proven that eribulin has a unique advantage in the treatment of heavily pretreated MBC. Initially, EMBRACE, 8 a phase III open-label, randomized, parallel-group, global multicenter clinical trial, established that eribulin significantly prolonged overall survival (OS) compared with the treatment of physician choice (TPC), and the drug was well tolerated in patients with heavily pretreated or anthracycline- and taxane-resistant MBC. Then, Study 301 demonstrated that for patients with MBC who were pretreated with anthracyclines and taxanes, eribulin prolonged median OS compared with capecitabine, with the effects being especially pronounced in the triple-negative breast cancer (TNBC), human epidermal growth factor receptor 2 (HER-2)–negative, and non-visceral subgroups, and overall treatment-related adverse event (TRAE) rates were similar between the study arms.9–11 In addition, a post hoc subgroup analysis by metastatic lesion site in the EMBRACE study illustrated that eribulin significantly increased progression-free survival (PFS; hazard ratio [HR] = 0.65; 95% confidence interval [CI] = 0.53–0.81; P < 0.0001), OS (HR = 0.74; 95% CI =0.58–0.96; P = 0.02), and objective response rates (31.5% vs. 14.6%) compared with TPC among patients with liver metastases. 12 Because the liver has a rich blood supply and it plays a vital role in the metabolism of medications, it is not surprising that chemotherapy causes hepatic impairment. However, hepatic failure is relatively rare and noteworthy.
Liver failure can present as acute liver failure (ALF) in the absence of any pre-existing liver disease, acute-on-chronic liver failure (ACLF, an acute deterioration of known and unknown chronic liver disease), or acute decompensation (AD) of end-stage liver disease. ACLF is a clinical syndrome manifesting as acute and severe hepatic derangement resulting from varied insults. Specifically, it is a condition in which two insults affect the liver simultaneously, including one chronic and one acute insult. In other words, it is a distinct entity in which acute hepatic decompensation occurs in a patient with established chronic liver disease or cirrhosis on exposure to an acute insult in a defined period.
In this study, we report the case of a patient diagnosed with triple-negative MBC who ultimately developed ACLF after treatment with eribulin, which has not been reported previously, in an effort to increase clinicians’ awareness of this potentially fatal adverse for optimal prevention and prompt diagnosis and treatment.
Case presentation
Case narration
The reporting of this study conforms to CARE guidelines. 13 A Chinese woman in her early 40s with sinus tachycardia as her past medical history was diagnosed with estrogen receptor-positive, progesterone receptor-positive, HER-2–negative locally advanced breast cancer (pT2pN3aM0), and she underwent surgery in August 2005. After surgery, the patient received six cycles of adjuvant cyclophosphamide, doxorubicin, and fluorouracil followed by tamoxifen for 5 years, and adjuvant loco-regional radiotherapy was not performed.
In June 2012, she presented with left cervical and supraclavicular lymph node metastases. First-line chemotherapy was initiated with six cycles of docetaxel and capecitabine, with the best response of partial remission (PR), followed by maintenance therapy with capecitabine until December 2012. Then, the patient underwent loco-regional radiotherapy and began endocrine therapy with exemestane and ovarian function suppression by bilateral ovariectomy.
In January 2016, a new progression with solitary liver metastasis was observed, and treatment with fulvestrant was initiated. Three new liver metastases were observed after two cycles of fulvestrant. Second-line chemotherapy with paclitaxel liposome and capecitabine commenced in May 2016, which resulted in PR until September 2016 (six cycles), and capecitabine maintenance therapy was performed again until progression with new right pleural and peritoneal metastases in February 2018. Cyclophosphamide and methotrexate were administered orally as third-line chemotherapy, with the best response being stable disease (SD) until disease progression again in August 2018. Hence, systemic therapy was replaced with cyclophosphamide, etoposide, megestrol.
In February 2019, progressive disease with new bone metastasis and liver and peritoneum evolution was evaluated. Bisphosphonate and endocrine therapy with fulvestrant and palbociclib were provided, achieving SD until December 2019, when liver and peritoneal metastases progressed. Peritoneal exfoliating cytology demonstrated high-grade ductal carcinoma metastasis from the breast with a subtype of TNBC. Liver lesions developed despite two cycles of per oral vinorelbine and apatinib as fifth-line chemotherapy, and cisplatin peritoneal perfusion was performed.
In February 2020, the patient started sixth-line chemotherapy with six cycles of nab-paclitaxel and apatinib and maintained SD until peritoneal metastases progressed and new intracranial metastases developed in July 2020. Therefore, treatment was switched to gemcitabine and bevacizumab. Two cycles later, the peritoneal lesion progressed and produced massive ascites. Eighth-line chemotherapy was implemented with eribulin mesylate 0.7 mg/m2 on days 1 and 8 of a 21-day cycle adjusted according to the patient’s Child–Pugh class B hepatic impairment. The patient’s treatment history is summarized in Figure 1.

Schematic illustration of the patient’s treatment history.
On September 4, 2020, the patient was conscious, and she agreed to accept 1 mg eribulin on day 1. On September 7, 2020, sharp clinical and laboratory deterioration was observed, and the patient presented with a liver failure syndrome. Clinically, she had anorexia, and she rapidly progressed from somnolence to coma. The positive signs were mainly as follows: Eastern Cooperative Oncology Group performance status 4, coma, stable vital signs, moderate yellow skin and sclera, scattered bleeding points on the trunk and limbs, chemosis and mydriasis 6 mm in diameter, slight dull papillary light reflex, a grossly distended abdomen with hepatomegaly and shifting dullness positive, and moderate pitting edema of the lower extremities. Moreover, remarkable elevations of bilirubin and ammonia levels as well as coagulation parameter abnormalities in blood tests were noted, indicating severe liver anabolic and metabolic function impairment. Her alanine aminotransferase (ALT) level was normal, whereas her aspartate aminotransferase (AST) level had increased from 63.1 U/L to 136.7 U/L (upper limit of normal [ULN], 32.0 U/L). In addition, her total bilirubin level was dramatically increased from 24.2 µmol/L to 106.2 µmol/L (ULN, 21.0 µmol/L), her direct bilirubin level had increased from 15.6 µmol/L to 75.6 µmol/L (ULN, 5.0 µmol/L), and her albumin (ALB) level was 29.8 g/L. The patient’s prothrombin time (PT) was 24 s, and the prothrombin time activity (PTA) was 36%. The activated partial thromboplastin time was 48.4 s, and her D-dimer and fibrin degradation product levels were 9.45 and 33.79 µg/mL, respectively. Her blood ammonia level was sharply evaluated at 187.7 µmol/L (ULN, 51.0 µmol/L). Changes in major laboratory tests before and after eribulin treatment are presented in Table 1.
Changes in major laboratory test data after eribulin treatment.

ALT, alanine aminotransferase; AST, aspirate aminotransferase; ALP, alkaline phosphatase; GGT, gamma-glutamyl transpeptidase; TBIL, total bilirubin; DBIL, direct bilirubin; IBIL, indirect bilirubin; ALB, albumin; WBC, white blood cell; NEUT, neutrophil; Hb, hemoglobin; PLT, platelet; PT, prothrombin time; INR, International Normalized Ratio; PTA, prothrombin time activity; APTT, activated partial thromboplastin time; FDP, fibrin degradation products; BA, blood ammonia.
Differential diagnosis
Two aspects needed to be carefully analyzed in this case, and the differential diagnosis revolved around the following main issues.
First, it was of great importance to distinguish ACLF from AD on the basis of cirrhosis. Patients with ACLF are distinct from those with cirrhosis, who are already decompensated and who experience a sudden worsening of their condition. 14 According to consensus recommendations from the Asian Pacific Association for the Study of the Liver (APASL), 15 AD often occurs in patients with cirrhosis, and the time from insult to presentation is up to 3 months. The disease can have hepatic presentations, such as jaundice, ascites, or hepatic encephalopathy (HE), or extra-hepatic presentations, such as acute variceal bleeding (AVB), acute kidney injury (AKI), or sepsis, and ascites, HE, and AVB may precede jaundice. However, ACLF is defined as an acute hepatic insult manifesting as jaundice (serum bilirubin ≥5 mg/dL [85 µmol/L] and coagulopathy [international normalized ratio ≥1.5 or PTA <40%]) complicated within 4 weeks by clinical ascites and/or encephalopathy in a patient with diagnosed or undiagnosed chronic liver disease/cirrhosis. Non-hepatic organ failure, such as AVB, AKI, or sepsis, often develops after ACLF depending on the severity of liver failure. Moreover, reversibility is a feature of ACLF as defined by APASL. With mitigation of the acute insult and the progression of time, the hepatic reserve improves, fibrosis regresses, and the portal pressure decreases. Differential key points between ACLF and AD including the basic disease, time of onset, presentation, and reversibility are presented in Table 2. As this case implied, the patient had chronic liver disease caused by long-term liver metastasis originating from breast carcinoma. Her AST levels and PT were slightly elevated at baseline (63.1 U/L and 16.1 s, respectively). Beyond that, other laboratory parameters concerning routine blood, cardiac, and kidney tests were normal. On the fourth day after eribulin administration, she exhibited cerebral failure with grade IV HE, hyperammonemia, jaundice, and coagulopathy. Apart from these hepatic disturbances, cardiac, kidney and lung function were intact. Finally, she recovered from liver failure. Combined with the underlying condition (chronic liver dysfunction), time of onset (4 days, within 4 weeks), presentation (hepatic insults such as HE, jaundice, and coagulopathy and no extra-hepatic organ failure), and reversibility, ACLF was diagnosed rather than cirrhotic AD.
Differential key points between ACLF and AD*.
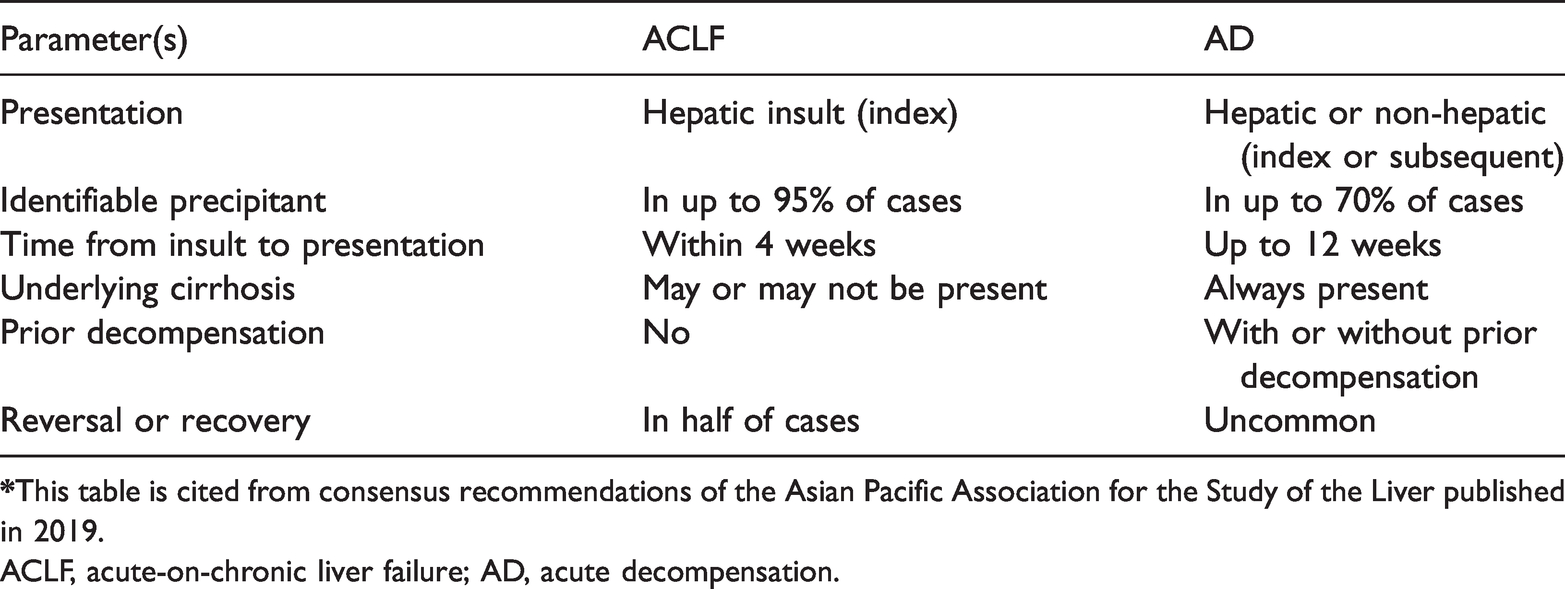
ACLF, acute-on-chronic liver failure; AD, acute decompensation.
The second issue was the cause of the acute insult. According to the APASL consensus, ACLF can be caused by hepatotropic and non-hepatotropic infections, drugs, alcohol, autoimmune liver disease, AVB, vascular liver disease, and liver tumor. In this case, it was critical to determine whether eribulin or liver metastasis progression precipitated ACLF. To answer this question, computed tomography was performed to evaluate the liver lesions, which illustrated that the liver metastases had partly shrunk rather than grown compared with their pre-eribulin size (Figure 2). Workups were also performed to eliminate other chronic liver diseases in the differential diagnosis (Table 3). According to her medical history, changes in symptoms, and signs and tests before and after eribulin, eribulin-related ACLF was diagnosed. Regrettably, liver biopsy could not be performed owing to the patient’s poor condition.
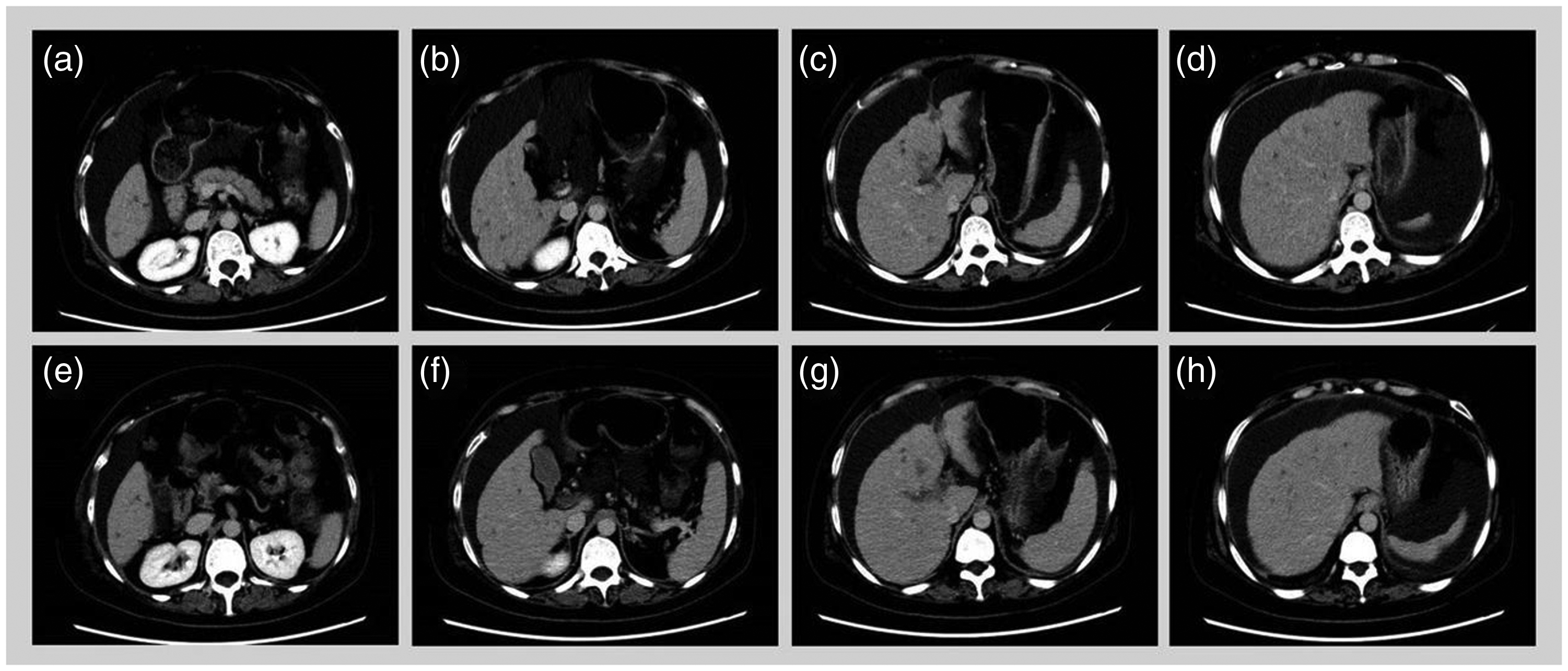
Response of hepatic metastatic lesions to eribulin. (a–d) Cervical–thoracic–abdominal–pelvic computed tomography scan (25 August 2020) revealing various hepatic metastatic lesions and (e–h) Cervical–thoracic–abdominal–pelvic computed tomography scan (15 September 2020) revealing disease stabilization (smaller) after one cycle of eribulin.
Workups of chronic liver diseases.

Treatment and outcome
Based on the consensus recommendations of APASL, the principal treatment modalities for liver failure are generalized in three aspects: etiological treatment, supportive treatment, and comprehensive therapy for complications. The first and foremost step was to cease eribulin immediately. Supportive care, such as close monitoring of vital signs and consciousness, total enteral nutrition support, ALB supplementation, homeostasis maintenance, reducing enzyme levels, and eliminating jaundice, was performed to facilitate the patient’s recovery. Finally, combination therapies were of equal importance, and they were mainly administered as follows: i) mannitol was used to relieve cerebral and bulbar conjunctival edema; ii)
On 9 September 2020, the patient gradually regained consciousness and started to speak to her family. Meanwhile, laboratory examination revealed gradual improvement in AST, ammonia, and bilirubin levels and coagulation function. On 12 September 2020, the patient had regained clear consciousness, and her Glasgow Coma Scale had improved from 3 to 15. Although her white blood cell and platelet counts had decreased as expected, they returned to normal with recombinant human granulocyte colony-stimulating factor and thrombopoietin administration. Finally, she experienced esophagogastric variceal bleeding and hypovolemic shock. Thereafter, the patient and her family, on discussion with physicians, selected palliative care and declined fluid resuscitation, blood product transfusion, and vasopressor and hemostatic use. Vital signs and laboratory monitoring were discontinued, whereas pain medication, oxygen, and hospice palliative care were continued. She died on 4 November 2020.
Discussion
A novel agent to improve the clinical outcomes of MBC, the most common and threatening malignancy of women globally, is urgently needed. As mentioned previously, emerging evidence illustrates that eribulin exhibits high potency and further improves OS compared with cytotoxic agents that have been approved for taxane-resistant MBC.
The main dose-limited toxicities of eribulin in Phase I Study were neutropenic events.16,17 The pharmacokinetic profile revealed that most of eribulin is eliminated by bile excretion, with only a small fraction of the drug (5%–7%) excreted unchanged in urine. 18 Therefore, the dose of eribulin should be adjusted in relation to basic liver function to ensure efficiency and safety.
ACLF, a liver failure syndrome characterized by jaundice and coagulation disorders in patients with previously diagnosed or undiagnosed chronic liver disease provoked by various precipitating factors, can lead to dose delays, dose reductions, discontinuation, and even death, and it is associated with a high 28-day mortality rate of up to 33%. 15 The patient was certainly diagnosed with MBC accompanied by diffuse liver metastases that resulted in chronic liver disease with Child–Pugh Class B liver function, and she accepted eribulin as eighth-line chemotherapy. Shortly afterward, she experienced ACLF induced by eribulin. To date, regarding eribulin TRAEs, different clinical trials have obtained distinct outcomes, and several clinical trials reported eribulin-related liver impairment. Nevertheless, there are no data demonstrating eribulin-associated liver failure, especially acute liver failure based on chronic hepatic disorders. In the EMBRACE study, adverse events with an incidence higher than 10% associated with eribulin treatment were exclusive to hepatic dysfunction, and liver failure was not cited. 8 Subsequently, the rates of all-grade and grade ≥3 liver impairment manifested as increased ALT levels were 8.5% and 2%, respectively, in Study 301. 11 Moreover, in an open-label, randomized, parallel, two-arm, multicenter clinical trial in China, AST and ALT elevation occurred in 41.7% and 35.2% of patients in the eribulin treatment group, respectively. 19 Nevertheless, no bilirubin abnormality, coagulopathy, or liver failure was described. Although eribulin, as a chemotherapeutic drug, might be potentially hepatotoxic, given the wide use of eribulin and prevalence of liver metastases in MBC and the threat of ACLF lethality, it is crucial to increase the awareness of this fatal and rare disease to ensure timely diagnosis and treatment. Furthermore, the identification of patients with a predisposition to hepatic failure when exposed to eribulin is especially important and beneficial for early prevention. A phase II study of eribulin in Japanese patients with heavily pretreated MBC reported a rate of AST elevation of 37%. 20 Available regional and global randomized clinical trials concluded that eribulin was hepatotoxic, but the incidence of hepatotoxicity appeared to vary by region. The incidence of liver aminotransferase aberrancy in clinical trials conducted in Asia, especially China and Japan, was much higher than that globally. Liver failure, which is considered the ‘high edition’ of liver dysfunction, is logically similar to liver dysfunction. Hence, whether eribulin-related liver failure is correlated with ethnicity is unknown. In addition, a post hoc analysis of Study 301 and Study 305 concluded that mild elevations of bilirubin levels were associated with increased eribulin toxicity. 21 For patients with breast cancer, metastatic disease commonly involves the liver, which made mild hepatic impairment at the start of therapy become prevalent. In this case, the patient had mild hepatic impairment before eribulin therapy, and she developed liver failure after eribulin treatment. What role did the baseline liver function play? Whether patients with basal liver dysfunction more easily develop liver failure when receiving eribulin administration is unclear, and further exploration is needed.
Conclusion
Currently, eribulin is widely used in patients with MBC, and its safety was verified in several pivotal studies. The case report presented a new and fatal adverse event called ACLF precipitated by eribulin in a patient with chronic liver disease caused by long-term liver metastases. Therefore, caution is needed when administering eribulin to patients with chronic and diffuse liver metastases, especially those with liver dysfunction at baseline.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221090097 - Supplemental material for Acute-on-chronic liver failure following eribulin treatment for metastatic breast cancer: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221090097 for Acute-on-chronic liver failure following eribulin treatment for metastatic breast cancer: a case report by Xiao-Feng Xie, Jia-Yi Huang, Li-Ping Chen, Xiao-Feng Lan, Qiu-Yi Zhang, Lin Song, Xue- Bai and Cai-Wen Du in Journal of International Medical Research
Footnotes
Acknowledgements
We are grateful to the patient and all the researchers, including the physicians, pathologists, and technicians, who participated in this case.
Author contributions statement
Each of the authors significantly contributed to this manuscript. All authors are responsible for the contents, and they have read and approved the manuscript for submission.
Ethics statement
All procedures performed involving human participants were in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent was obtained from patient and her family for the treatment regimen and publication of this manuscript. The patients’ detailed information was de-identified. Moreover, this manuscript was published with approval of the Ethics Committee of National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital (approval number: 2020-03-10; approval date: January 10, 2021).
Declaration of conflicting interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from the Shenzhen Science and Technology Program (2018, JCYJ20180306171227129) and the National Natural Science Foundation of China (No. 81671750, 2016).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
