Abstract
Objective:
Patients with acute-on-chronic liver failure (AoCLF) are prone to various infections, including invasive pulmonary aspergillosis (IPA). This retrospective study investigated the relationship between AoCLF and IPA in a large patient cohort.
Methods:
Twenty-nine out of 470 patients with AoCLF, who were diagnosed with definite or probable IPA by clinical and laboratory parameters and were treated with voriconazole, were analysed for predisposing factors for IPA and clinical outcome.
Results:
Imaging findings, such as the halo sign and increased white blood cell count, contributed to the early diagnosis of IPA. Patients with AoCLF and IPA experienced fever, cough and chest pain and, despite treatment with antifungal therapy, most (25/29 patients) died within 7 days. Possible risk factors for IPA included prolonged antibiotic therapy and dexamethasone exposure.
Conclusions:
AoCLF with prolonged antibiotic therapy and dexamethasone exposure carries a high risk for IPA. AoCLF patients with IPA involved in this study exhibited fever, cough, chest pain and increased white blood cell count, and their imaging findings were useful for the early diagnosis of IPA.
Keywords
Introduction
Aspergillus spp. cause a wide spectrum of lung diseases, and are commonly isolated from soil, plant debris and indoor environments, including hospitals. Invasive pulmonary aspergillosis (IPA) is a potentially severe and frequently fatal early complication in certain immuno -compromised and immunosuppressed patients, such as transplant recipients and chemotherapy patients, with a mortality rate exceeding 90% in these patients.1–4 Chronic necrotizing pulmonary aspergillosis and Aspergillus spp. pneumonia, often with dissemination, are two well-known forms of this invasive disease.5,6 IPA mainly occurs in patients with malignancy who experience deep and prolonged neutropenia, but there are other established risk factors (such as high-dose, long-term corticosteroid therapy or advanced forms of acquired immunodeficiency syndrome). 7 Patients with liver disease, especially liver failure, are often immunocompromised and are also prone to various infections including bacterial, viral and fungal infections such as IPA.8–12 Prodanovic et al. 13 reported four cases of IPA in patients with Child-Pugh class C liver cirrhosis in a critical care setting, which is considered a possible risk factor for IPA. The diagnosis of IPA can be elusive because the fungus is difficult to isolate; in addition, its symptoms and signs are not specific, and the effects of antifungal agents and duration of treatment for IPA remain inadequate. Together, these are major complicating factors for immuno -compromised patients and might be associated with increased mortality rates. Although IPA is the major factor associated with poor prognosis, there has been no large-scale analysis of IPA in patients with liver disease, especially acute-on-chronic liver failure (AoCLF), which denotes an acute deterioration of known or unknown chronic liver disease or a gradual decompensation of end-stage liver disease.
The current study determined the clinical presentation, predisposing factors and outcome of IPA in patients with AoCLF. Possible risk factors for IPA and treatment regimens were analysed in these patients, in an effort to determine the basis for the high mortality rate observed.
Patients and methods
Study Population
This retrospective study included patients with AoCLF who were admitted to the State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, Department of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, China, between July 2007 and December 2010. Patients with acute deterioration in liver function over 2 – 4 weeks with a defined inflammation-related precipitant (infection or alcoholic hepatitis), leading to: severe progressive clinical deterioration despite > 48 h supportive care; increasing jaundice (serum bilirubin > 100 μmol/l); evidence of abnormal coagulation (defined as an international normalized ratio [INR] 1.5); and either encephalopathy (grade 2) or hepatorenal syndrome (HRS) in those with clinical, radiological, biochemical and histological evidence of cirrhosis, were diagnosed with AoCLF.14,15 Medical and microbiological records, together with chest radiographic imaging of all patients with AoCLF and accompanying lung infection, were reviewed. Patient demographics, duration of admission, predisposing factors, clinical features, investigations, treatment and outcome were noted.
The study was approved by the Ethical Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University, China. Written informed consent was obtained from all participants prior to enrolment.
Diagnostic Criteria for IPA
The definitions of invasive aspergillosis used in this study were similar to the criteria of the European Organization for Research and Treatment of Cancer/Mycoses Study Group. 7 Definite or probable IPA was diagnosed according to previous studies and based on clinical, radiological and mycological data.7,16–19 In brief, the appearance of pulmonary consolidation or infiltrate and rapid progression on thoracic computed tomography (CT) scan, with antibiotic-resistant fever in the appropriate host setting, was diagnosed as probable IPA. Single-bed hospital rooms were provided for patients with a high index of suspicion of IPA. The following contributed to the diagnosis of IPA: positive culture of Aspergillus spp. from sputum or bronchoalveolar lavage (BAL) specimens; bilateral nodular infiltrates with central cavitation, or bilateral alveolar infiltrates or homogeneous infiltrates of right upper lobe, or infiltrates of left upper lobe and left lower lobe with empyema on chest X-ray; halo sign or the air-crescent sign (a macronodule surrounded by a perimeter of ground-glass opacity) on CT scan; 20 at least two minor clinical findings (signs of lower respiratory tract infection, pleural rub and presence of any new infiltrate in a patient who did not fulfil the major criteria, but for whom no alternative diagnosis was available). To confirm the diagnosis, nucleic acid detection in serum or BAL were assessed, as described previously. 21 Serological diagnoses, such as galactomannan or (1→3)-β-D-glucan, which are associated with a high false-positive rate, were not performed in the clinical laboratory of The First Affiliated Hospital, College of Medicine, Zhejiang University.
Laboratory parameters were also used to diagnose IPA. These included haemoglobin, white blood cell count, differential leucocyte count, platelet count, albumin, globulin, cholinesterase, total bilirubin, blood glucose, prothrombin time, INR, creatinine, blood urea nitrogen (BUN) and hepatitis B virus (HBV) viral load, which were all measured using standard techniques. On the basis of these criteria, patients were diagnosed with definite or probable IPA.
Statistical Analyses
Statistical analyses were carried out using the SPSS® statistical package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Continuous data were expressed either as mean ± SD or median and range, based on the distribution. Quantitative data were assessed by one-way analysis of variance and, if the data showed a normal distribution, Bonferroni multiple analyses were used. If the data did not show a normal distribution, Dunnett's multiple comparison test was used. A P-value < 0.05 was considered statistically significant for all analyses.
Results
A total of 470 patients were diagnosed with AoCLF between July 2007 and December 2010, and were admitted to the State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, Department of Infectious Diseases, The First Affiliated Hospital, College of Medicine, Zhejiang University, China. Of these, 29 patients were also diagnosed with probable or definite IPA, according to the diagnostic criteria for IPA (see Patients and Methods). The demographic and clinical characteristics of the 29 patients with IPA and AoCLF are presented in Table 1.
Demographic and clinical characteristics of patients with acute-on-chronic liver failure, who were diagnosed with invasive pulmonary aspergillosis (n = 29)

Data presented as n patients unless stated otherwise.
All patients were treated in conventional wards, not in intensive care units, and had underlying liver disease with hepatitis B, indicating that infection with HBV was one of the most important risk factors for AoCLF. 22 In most of the patients with IPA and AoCLF, disease was associated with one or several complications such as hepatic encephalopathy, HRS, ascites, hydrothorax or haemorrhage. With so many severe complications, most patients received antibiotic therapy (regimens included 4.5 g piperacillin–tazobactam every 8 h, 0.5 g meropenem every 8 h, 0.2 g ciprofloxacin twice daily, 0.5 g vancomycin every 8 h, 4 g phosphomycin twice daily or 2.0 g cefoperazone twice daily, either alone or in combination). Most patients also had dexamethasone exposure, to prevent allergic reactions or to decrease persistent hyperbilirubinaemia before IPA diagnosis. In addition, 22 patients with IPA were treated with plasma exchange to reduce various complications and ameliorate the symptoms. Despite treatment with antifungal therapy (30 mg/kg per day voriconazole), which started immediately following the confirmed diagnosis of IPA, 25/29 patients with IPA died within 7 days; three patients improved and one underwent liver transplantation.
Clinical and imaging findings, and laboratory parameters, for patients with acute-on-chronic liver failure who were diagnosed with invasive pulmonary aspergillosis (IPA; n = 29)
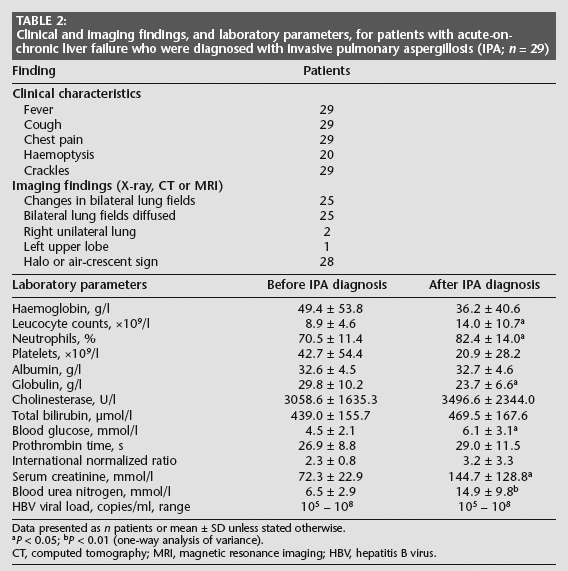
Data presented as n patients or mean ± SD unless stated otherwise.
P < 0.05
P < 0.01 (one-way analysis of variance).
CT, computed tomography; MRI, magnetic resonance imaging; HBV, hepatitis B virus.
Compared with laboratory parameters before IPA was diagnosed, after diagnosis the white blood cell count, proportion of neutrophils, blood glucose, serum creatinine and BUN were significantly increased, and globulin significantly decreased (all P < 0.05) (Table 2). These parameters might have been involved in the development of IPA in patients with AoCLF, therefore measuring them might be useful for monitoring the occurrence of IPA in such severely immunocompromised hosts.
Discussion
Aspergillus spp. are ubiquitous in soil and inhalation of the conidia into the respiratory tract can cause life-threatening disease. IPA is a key cause of morbidity and mortality in severely immunocompromised patients.1–4 Improvements in the diagnosis and treatment of IPA have been accompanied by an increase in the diagnosis of Aspergillus spp. infection in severely immuno -compromised patients. 23 The pathological features and natural history of IPA during the course of infection have been extensively studied.17,24 IPA is characterized by haemorrhagic infarction of lung tissue secondary to vascular invasion of Aspergillus spp., causing thrombosis of small arterioles and sometimes of larger pulmonary vessels. 25 On macroscopic examination, the characteristic early lesions of IPA are 1 – 3-cm nodules or target lesions that have a central grey area of necrosis surrounded by a red peripheral ring from haemorrhage or haemorrhagic infarction. 26 Liver failure is defined as fulminant hepatitis or prolonging of prothrombin time (INR > 1.5), occurs in severely immunocompromised patients and is accompanied by various infections, including IPA, that are frequently fatal. 27 It has been demonstrated that decompensated liver disease is an acquired immune deficiency state.28,29 Studies have shown that IPA frequently occurs in patients with decompensated cirrhosis and liver failure.13,16
A large number of patients with AoCLF were included in the present retrospective analysis, which is believed to be the first study to focus on the relationship between AoCLF and IPA in such a large cohort. The majority of patients with AoCLF and IPA were treated with prolonged antibiotic therapy (often involving more than one type), dexamethasone and plasma exchange, which could reduce the inflammation of the liver effectively and prevent possible bacterial infections. These treatments were, however, harmful to the host's immune function and did not cover all possible pathogens; therefore, patients were prone to many types of infection caused by opportunistic pathogens including Aspergillus spp. There are no specific symptoms or biomarkers for IPA; thus, the diagnosis of IPA is often made too late, especially in patients with AoCLF in combination with various severe complications such as hepatic encephalopathy, HRS, ascites, hydrothorax and haemorrhage, which ultimately affect prognosis. AoCLF patients with the above risk factors experienced fever, cough and chest pain, and were suspected of having IPA.
Imaging findings were important for IPA diagnosis. It has been reported that IPA may be identified by chest CT at an early stage, and that the initiation of specific antifungal treatment on the basis of CT findings may lead to better outcomes. 30 Imaging findings such as halo sign or air-crescent sign are useful for the diagnosis of IPA and could guide antifungal therapy at an early stage.20,26 Li et al. 16 have stated that early and effective antifungal treatment greatly improves the prognosis of these patients. Treatment of IPA in patients with AoCLF requires consideration of appropriate antifungal therapy, potential surgical management and immunomodulation. For high-risk groups, it is important to administer antifungal agents for IPA prophylaxis as early as possible. Although the current ‘standard' medication in invasive infection, amphotericin B (desoxycholate or lipid formulation) is used extensively to treat definite or probable IPA, its administration is hampered by serious side-effects. With the development of new azole derivates, amphotericin B has been replaced gradually by itraconazole and voriconazole, and the new echinocandins.
Early treatment regimens for IPA aggressively utilize surgical resection as adjunctive therapy; however, the utility of surgical resection has not been formally studied and is potentially associated with significant morbidity. Additional immunomodulatory therapies with cytokines (such as granulocyte macrophage colony stimulating factor and interferon-γ) and with granulocyte transfusions are under evaluation. Delayed diagnosis of definite or probable IPA, the deteriorating status in patients with AoCLF and antifungal therapy restricted to voriconazole, may have contributed to the high rate of mortality in the present study. The present findings have reinforced the importance of early diagnosis of definite or probable IPA in immunocompromised patients: it has been reported that early treatment with antifungal agents, such as voriconazole, significantly delay or prevent mortality in these patients. 31
As far as laboratory parameters were concerned, no specific biomarkers for IPA were available. AoCLF could change these parameters substantially in patients with or without IPA. Consistent with common infections in patients with different health statuses, white blood cell counts increased significantly in the present patients. Generally, patients with bacterial infections have a significant increase in the number and proportion of neutrophils, whereas patients with viral infections have a similar increase in lymphocytes. Patients with AoCLF and IPA included in the present study had a significant increase in the number and proportion of neutrophils. According to the course of AoCLF, liver function and coagulation deteriorated rapidly, and plasma exchange often did not lead to improvement. In spite of the occurrence of IPA, there were still no significant differences in parameters such as cholinesterase, total bilirubin, prothrombin time and INR before and after diagnosis of IPA. As one of the most important complications, HRS significantly deteriorated after IPA occurrence, with serum creatinine and BUN increasing rapidly. These conventional laboratory parameters might be used to monitor the progress and prognosis of patients with AoCLF and IPA.
In summary, the patients with AoCLF involved in this study were immuno -compromised hosts who were prone to IPA. Prolonged antibiotic therapy, dexa -methasone treatment and plasma exchange may have been predisposing factors for IPA in these patients. There are no specific biomarkers for IPA symptoms such as fever, cough and chest pain. Consequently, imaging findings might be useful for the diagnosis of definite or probable IPA in the early disease stage, and for monitoring the efficacy of antifungal therapy, which could improve prognosis.
Footnotes
Acknowledgements
This work was funded by the National Basic Research Program of China (973 program) Grant 2007CB513003 and partly supported by a grant from Zhejiang Provincial Science and Technology Bureau Project (No. 2006C23017).
The authors had no conflicts of interest to declare in relation to this article.
