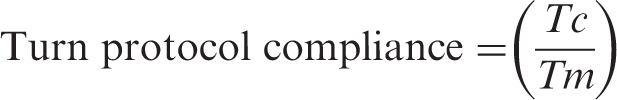Abstract
Purpose
Hospital-acquired pressure ulcers are a significant cause of morbidity and consume considerable financial resources. Turn protocols (repositioning patients at regular intervals) are utilized to reduce incidence of pressure ulcers. Adherence to turn protocols is particularly challenging for nursing teams, given the high number of interventions in intensive care unit, and lack of widely available tools to monitor patient position and generate alerts. We decided to develop and evaluate usefulness of a continuous patient position monitoring system to assist nurses in improving turn protocol compliance.
Methods
We conducted a prospective, non-randomized, multiphase, multicentre trial. In Phase I (control group), the function of the device was not revealed to nurses so as to observe their baseline adherence to turn protocol, while Phase II (intervention group) used continuous patient position monitoring system to generate alerts, when non-compliant with the turn protocol. All consecutive patients admitted to one of the two intensive care units during the study period were screened for enrolment. Patients at risk of acquiring pressure ulcers (Braden score < 18) were considered for the study (Phase I (N = 22), Phase II (N = 25)).
Results
We analysed over 1450 h of patient position data collected from 40 patients (Phase I (N = 20), Phase II (N = 20)). Turn protocol compliance was significantly higher in Phase II (80.15 ± 8.97%) compared to the Phase I (24.36 ± 12.67%); p < 0.001.
Conclusion
Using a continuous patient position monitoring system to provide alerts significantly improved compliance with hospital turn protocol. Nurses found the system to be useful in providing automated turn reminders and prioritising tasks.
Introduction
Hospital-acquired pressure ulcers (HAPUs) are injuries caused by prolonged pressure on the skin and its underlying tissues.1,2 Blood supply is disrupted under the influence of pressure, shear, and friction acting alone or in combination. This has detrimental effects on skin and its underlying tissues, which breaks down due to insufficient blood circulation. Pressure ulcer injuries are prevalent in all healthcare settings throughout the world. For example, the reported prevalence of pressure ulcers in hospitalized patients in India ranges from 3.1% to 7.8%,3–5 while in United Kingdom (UK) it ranges from 3.45% to 4.49%. 6 In addition to the increased morbidity, pressure ulcers have a significant cost impact, 7 imposing a huge financial burden on both individuals and healthcare systems in developing and developed countries. The mean cost to treat pressure ulcer injuries in India is significantly higher than its per capita income ($2460 versus $1670),8,9 while in developed countries like the UK, it ranges from £1214 (stage 1) to £14,608 (stage 4). 10
The risk of HAPUs has traditionally been minimized by turning the patient once every 2 h, alternating lateral and supine positions, commonly referred to as turn protocol. 11 Compliance with turn protocol, however, is variable in intensive care units (ICUs) and have been found to be suboptimal. 12 This could be because adherence to turn protocols is particularly challenging in a busy ICU environment, given the high number of routine nursing interventions required. Furthermore, while use of continuous monitoring for vital signs is routine, tools to monitor patient position and generate alerts are not widely prevalent. To address this, several technologies are being developed to enable continuous patient position monitoring, which can alert nurses when the patient is due for a turn. Depth sensor,13–15 wearable sensor,12,16 and pressure sensing mat17–19 are some of the sensing methodologies that have been evaluated for continuous monitoring of patient position. We decided to use a wearable sensor to continuously monitor patient’s position and in collaboration with an academic research institution developed a continuous patient position monitoring system (PRESENSE). PRESENSE was built as a readily deployable solution that could be used without any infrastructure upgrades. 16 The system was tested extensively before clinical immersion. 16 We also investigated the effectiveness and challenges faced in implementing such systems in regular care practices.
Materials and methods
Participants
The study was conducted in two adult ICUs consisting of 18 beds in total, located in Chennai and Trichy in the state of Tamil Nadu, India. The units were combined medico-surgical ICUs admitting all patients requiring intensive care, except those needing coronary care or undergoing cardiothoracic surgery. The study was approved by hospital’s Institutional Review Board (IRB) and registered with Clinical Trails Registry – India (CTRI); CTRI/2018/02/011713). All consecutive patients admitted to either of the two ICUs were screened for enrolment. Eligible participants satisfied the following inclusion criteria: (1) expected to stay in ICU for a minimum period of 24 h, (2) considered at risk of acquiring pressure ulcers assessed using Braden scale (Braden score < 18). 20 Participants were excluded from the study if they (1) for any reason could not have the wearable sensor attached to their chest in the midline, (2) had an implantable cardiovascular-defibrillator or pacemaker. Research nurses (RNs) assessed the eligible participants for their ability to participate in the study and obtained informed consent from their next of kin.
Interventional technology
The PRESENSE uses an ultra-low-power wearable sensor containing an inertial measurement unit (IMU) to capture a patient’s movement and position. IMU consists of multiple micro-electro-mechanical system (MEMS) sensors including a three-axis accelerometer, a three-axis gyroscope, and a three-axis magnetometer in a single package. Using novel sensor fusion algorithms, 21 the wearable sensor can represent its orientation in 3D space in Euler angle representation (roll, tilt, or upright angle). When attached to the patient’s chest in the sternum area, it determines the rotation of the torso in the transverse plane with respect to the bed surface to detect the patient’s position. These data are continuously transmitted from the wearable sensor and received wirelessly by a gateway device (Android-based tablet). The gateway device concurrently pushes the position data to the cloud and alerts the nurses visually via the screen display according to the hospital’s turn protocol. 16 The gateway device is placed next to the patient’s bedside adjacent to the vital signs monitor. Doctors and hospital administrators can access the data using a cloud-based web application. PRESENSE is designed to interact with multiple stakeholders and inform/alert them hierarchically to ensure that non-compliance with the protocol is captured and acted upon.
Details of intervention
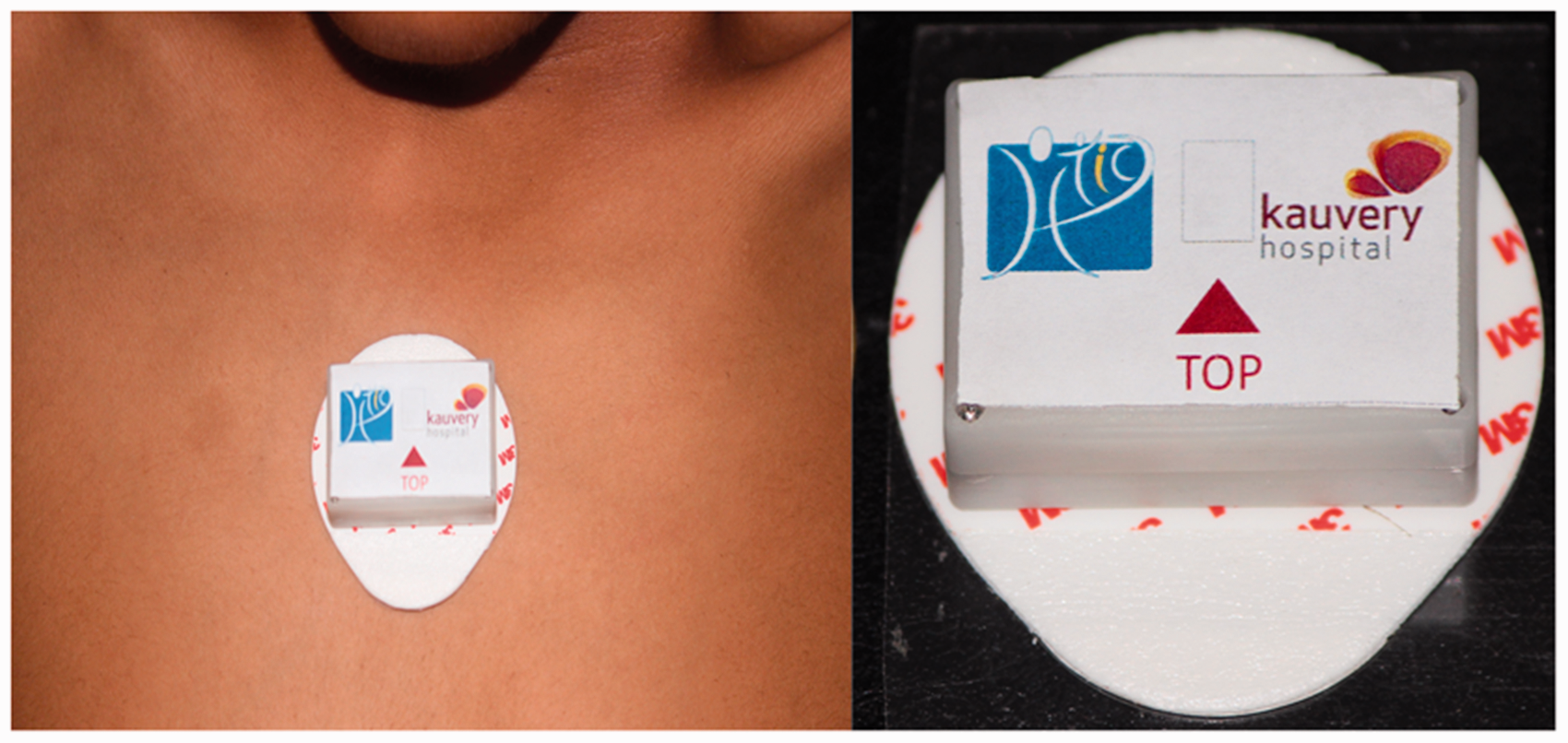
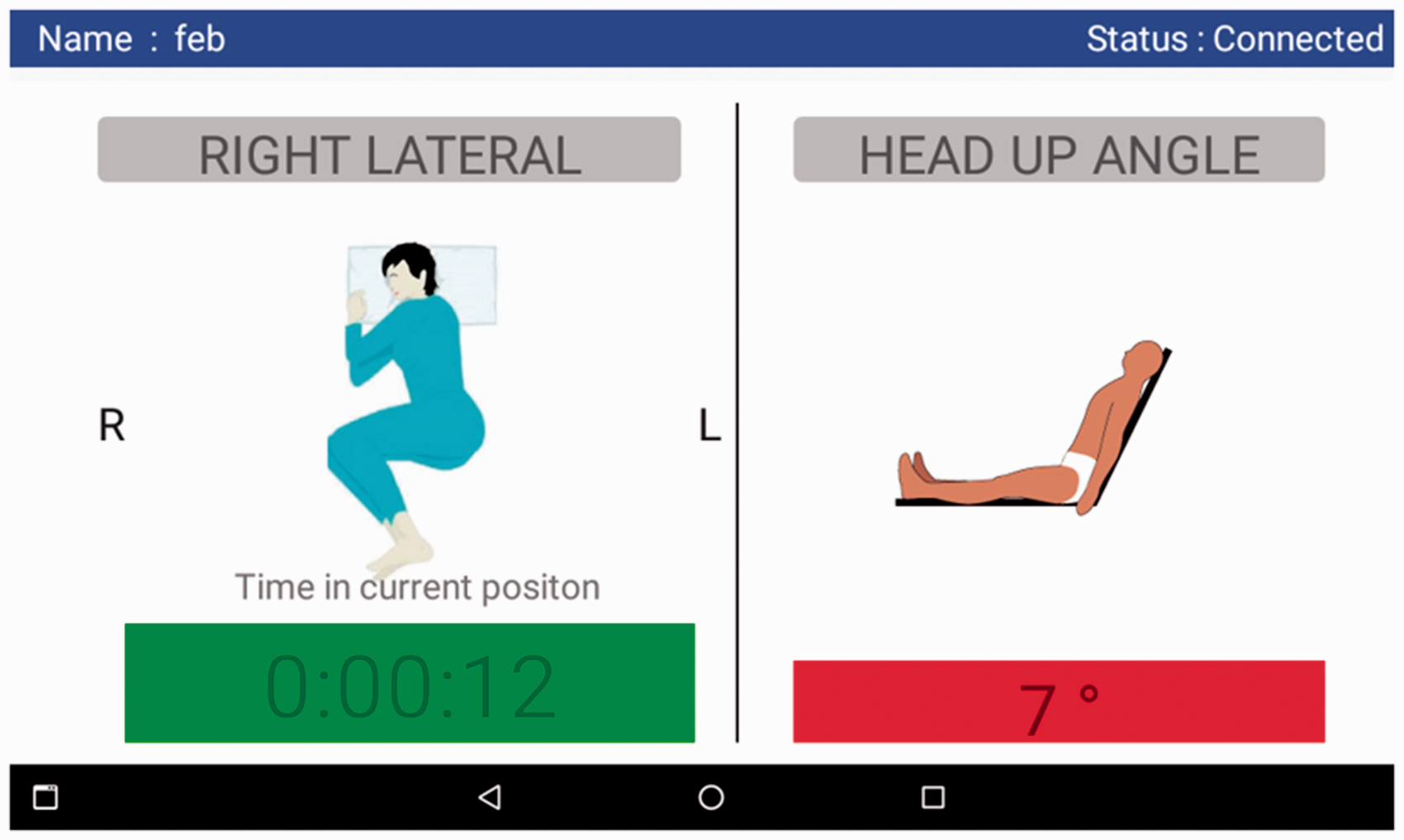
On admission, PRESENSE’s wearable sensor (of size: 29 × 39 × 10 mm) is attached to the patient’s sternum area using a disposable ECG electrode which acts as the adhesive skin interface. Figure 1 shows the wearable sensor attached to a patient’s chest and separately. It connects and transfers the data once every 30 s to the tablet (gateway device). The patient’s position and time in current position is displayed to the nurses through the interface as shown in Figure 2. The gateway device alerts the caregiver at regular intervals of time with visual cues as presented below:
Green colour signifies compliance with turn protocol Yellow colour signifies compliance with turn protocol and the patient is due for turn soon (less than 15 min). This helps nurses prepare themselves for the upcoming patient turn. Red colour signifies non-compliance with the turn protocol and the need to turn the patient immediately.
Wearable sensor attached to a patient’s chest (left) and separately (right). A sample screenshot of android tablet used for visual display (best when reproduced in colour).
Nurses comply with the turn protocol if, (1) the patient is repositioned at least once every 2 h (turn period), (2) on repositioning, the patient stays in new position for a minimum time period of 15 min (decompression interval) to sufficiently decompress the tissues. Any change in patient position which fails to meet the above criteria will not be considered as a valid turn and time elapsed in the new position will be added to the previous patient position. For example, consider a patient who has been in the supine position for 2 h, then repositioned to right lateral position for 10 min, and then returns to supine position again. As the patient has now returned to a supine position without spending 15 min in the new position, the timer value is updated as 2 h and 10 min in supine position and will not register the short (less than 15 min) positional changes as a valid turn.
The two-hourly turning protocol was programmed into PRESENSE system as this was the local protocol in both units taking part in the study at the time of commencement of the project.
Outcomes
Primary outcome
To assess the change in average turn protocol compliance in Control group (Phase I) and Intervention group (Phase II).
Measures used to assess the study outcomes are
Turn angle: Degree of rotation of the patient’s torso in the transverse plane with respect to the bed surface. In this study, the turn angle threshold was set as 20 degrees. When turn angle is between +20 and –20 degrees, patient position is considered as supine, beyond +20 degrees as left lateral position and beyond –20 degrees as right lateral position. Turn protocol compliance: Turn protocol compliance is the ratio of total time the patient was compliant with the turn protocol (Tc) to the total time monitored (Tm).
Data collection methods
The data collected from the wearable sensor is stored as a digital log both in the gateway device and in the cloud. Data from the cloud and the gateway was exported and processed using a custom computing tool developed using Python language. To ensure consistent quality of recorded data, the wearable sensor needs to be placed and oriented correctly on the patient’s chest – the mid sternum was the location chosen for attachment of the device. In both phases, nurses recorded the position and the orientation of the device during the nursing handover. If any deviations were recorded, the data collected from that nursing shift were excluded from the final analysis.
Validated instruments
PRESENSE was validated for its accuracy using a professional angle gauge as suggested by Schutt et al. 12 The device also received a full functionality validation test 16 before being put to clinical use.
Assignment method
All consecutive patients admitted to either of the two ICUs were considered for admission to the study once they met the inclusion criteria.
Sample size
We conducted a sample size calculation a priori. As there were no data on turn protocol compliance in Indian ICUs, we hypothesized that the PRESENSE system would improve turn protocol compliance by 50%. Based on this hypothesis and an alpha error of 10% we had to recruit 20 patients in each phase of the study to reject the null hypothesis with a power of 90%.
Blinding
In Phase I, to reduce Hawthorne effect, although PRESENSE’s wearable sensor was visibly attached to the patient’s chest, nurses were unaware of the real functionality of the system and information collected by it was not made available to them. The tablet display was turned off.
In Phase II, nurses were told about the purpose of PRESENSE and the visual display (Figure 2) was made available to nurses to visually alert them.
Statistical analysis
Turn protocol compliance was expressed in percentage with values ranging from 0% to 100%. Hence, we considered it as a parametric value. Data were checked for normality using Shapiro-Wilk Test. Student paired t-test was used for normal data to assess if a statistically significant difference exists between study groups, while Mann-Whitney U test was used for non-normal data. All study results are presented in the form of mean (M) ± standard deviation (SD).
Correlation between any baseline characteristics and turn protocol compliance was also assessed. To compare the difference between two categorical data, two-sample proportion test was used. All tests are deemed to be statistically significant when p < 0.001. Average hourly compliance to turn protocol was computed by dividing the sum of “time in compliance with turn protocol per patient for a given hour interval each day” by “total time monitored per patient for a given hour interval each day”. 12 Microsoft Excel software package was used for all statistical analyses.
Results
Participant flow
Figure 3 shows the participants’ flowchart. Out of all participants who were screened for eligibility (Phase I: N = 47, Phase II: N = 43), we excluded those who did not meet the inclusion criteria (Phase I: N = 25, Phase II: N = 18) and participants declining to participate in the study (Phase I: N = 1, Phase II: N = 3). Among those who were allocated to the study (Phase I: N = 21, Phase II: N = 22), some participants were excluded from final analysis for not complying with the study protocol (Phase I: N = 1, Phase II: N = 2). Deviations from the study protocol included: sensor fell off and was replaced by the nurses (N = 2), sensor was misplaced on the participant’s arm (N = 1), sensor transiently removed from the participant while shifted out of ICU for procedure/scan and reattached following return to ICU, but data collection continued recording data from the unattached sensor (N = 3). Overall, 40 participants’ position data were considered for the final analysis with 20 participants’ data used for each phase.
The participant flowchart.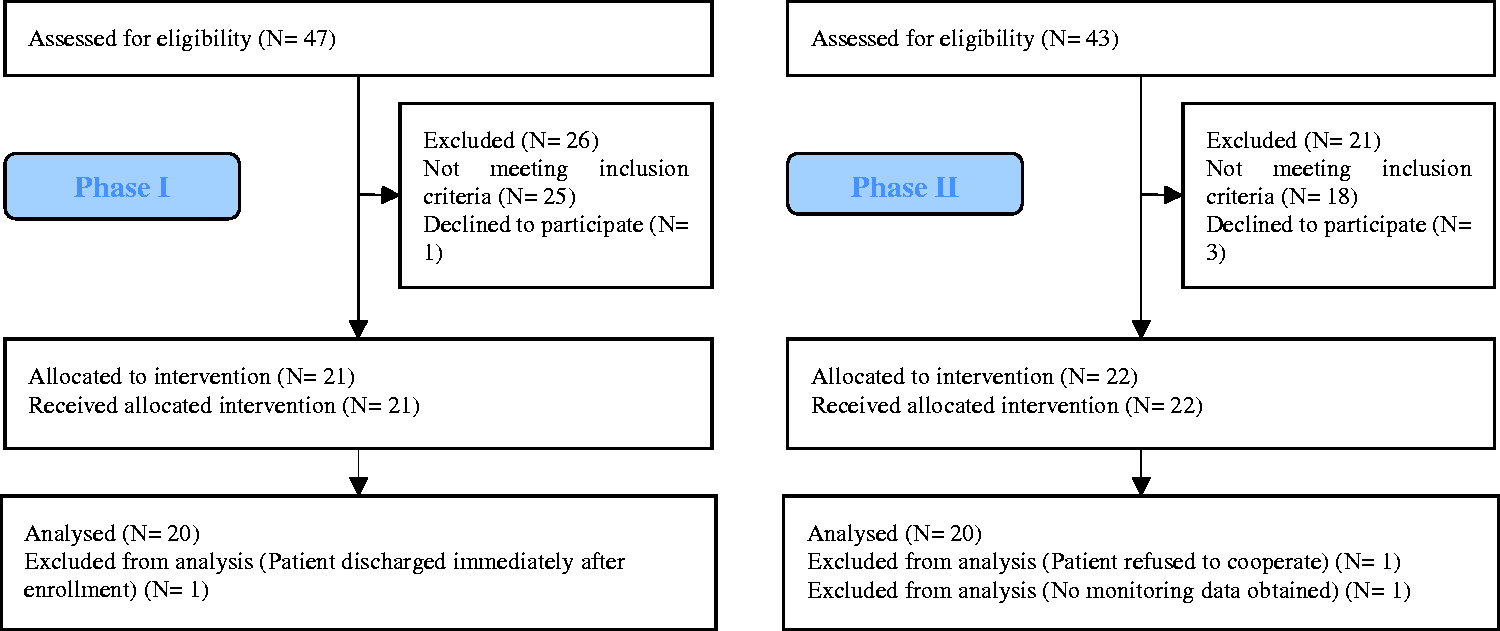
Recruitment
The study was approved in the last week of December 2017. Recruitment started in the second week of February 2018 and was completed in the fourth week of April 2018.
Baseline data
Baseline characteristics.
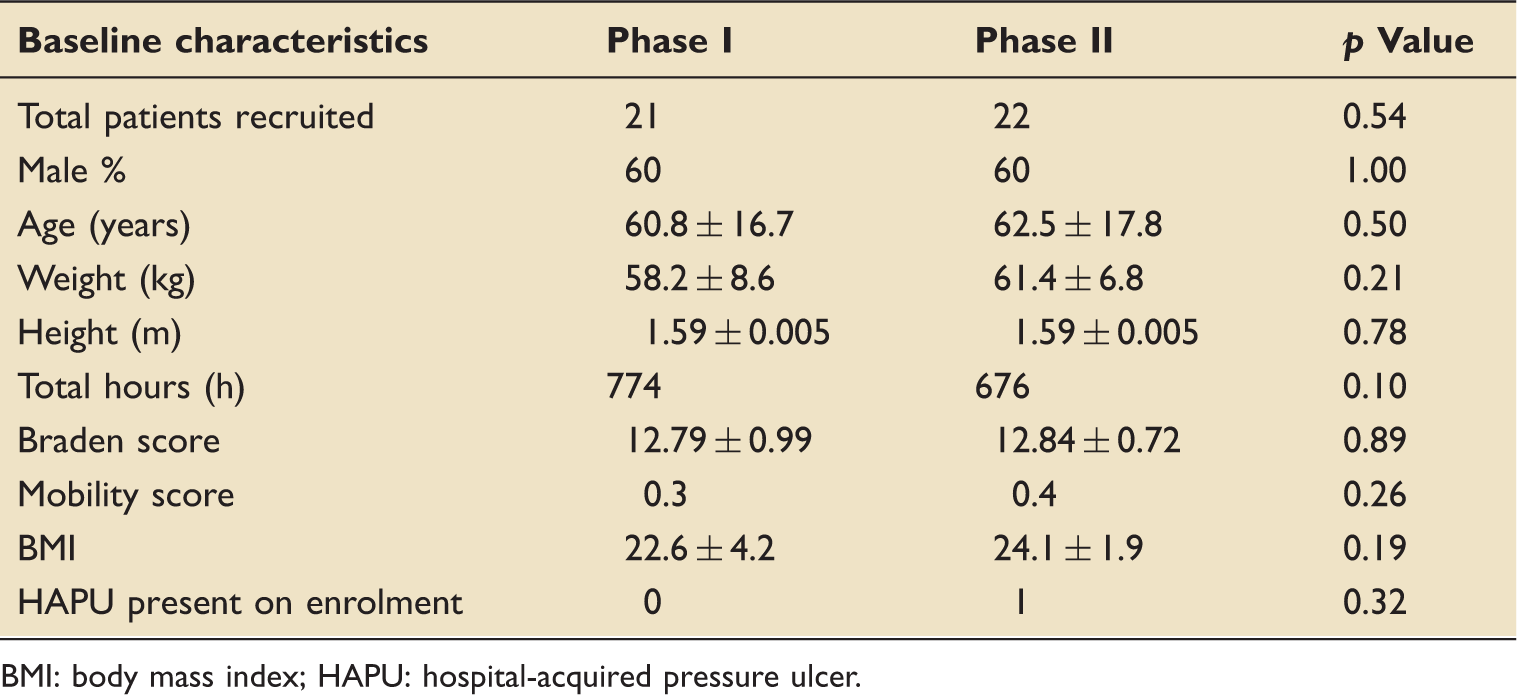
BMI: body mass index; HAPU: hospital-acquired pressure ulcer.
Outcomes and estimation
Compliance data from both phases were normally distributed. Student-paired t-test showed a statistically significant difference exists in turn protocol compliance in Phase II (80.15 ± 8.97%) compared to Phase I (24.36 ± 12.67%); t(38) = –16.07, p < 0.001. The effect size calculated using Cohen’s d test (5.07) was found to be larger. No adverse events were recorded during the conduct of the study. Further, none of the patients in both phases developed any pressure ulcer injuries during their participation in the study.
Ancillary analyses
No statistically significant correlation was observed between any of the baseline characteristics and the turn protocol compliance. The average hourly compliance to turn protocol for both phases is represented in Figure 4. Compliance in Phase II was relatively higher than the compliance in Phase I during all times of the day.
The average hourly compliance to turn protocol.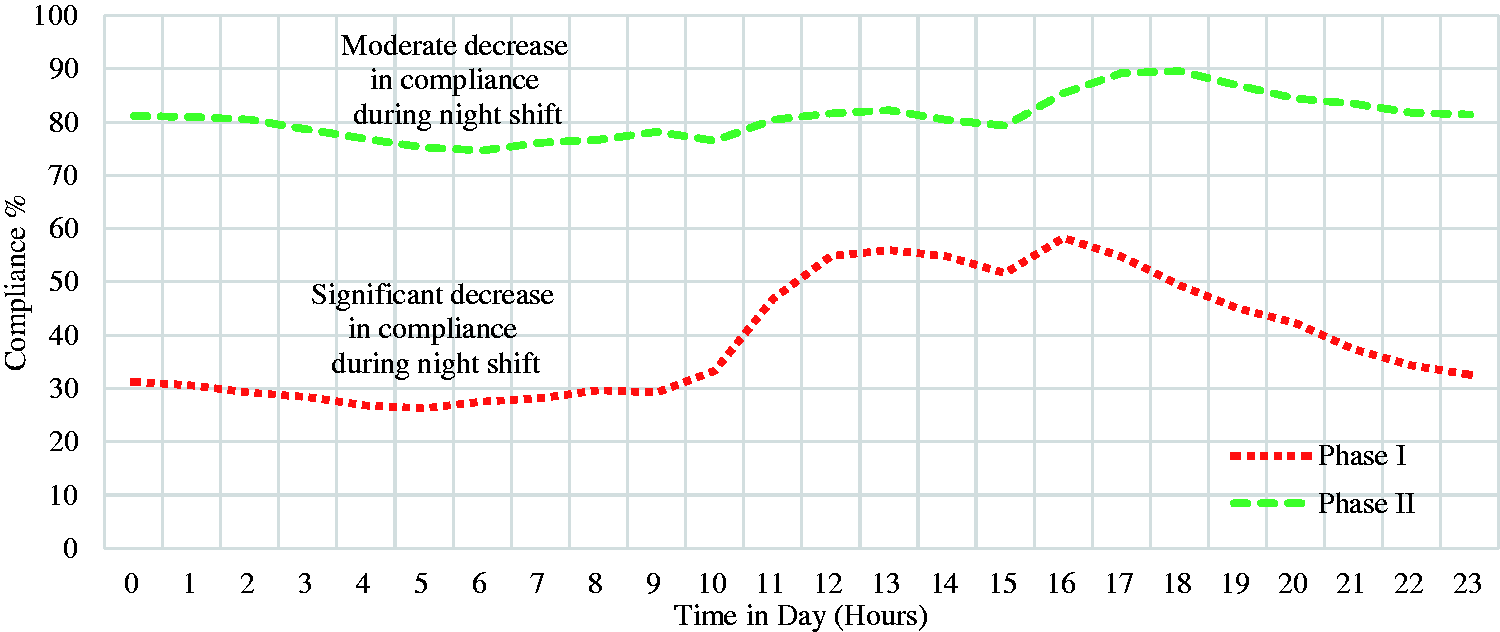
Discussion
The study showed a statistically significant difference in turn protocol compliance in Phase II compared to Phase I. The mean compliance achieved in Phase II (80.15%, N = 20) was found to be within the range of compliances previously reported by the other studies – pragmatic trial (67%, N = 659) 22 and pilot study (98%, N = 63). 12 The pragmatic trial analysis was performed with the intention to treat, 22 while the pilot study was not. The analysis in our study was not done with an intention-to-treat strategy as there were incidences of the sensor not recording the turn performed due to various reasons (non-cooperative patient, misplaced sensor, sensor not worn). Delays in turning the patient due to other reasons (medical intervention, emergency procedure) were included and no grace period was offered beyond the turn period to obtain a realistic turn protocol compliance.
A minimal decompression time period of 15 min was considered, failing which leads to rejecting the patient’s repositioning as a valid turn. Without sufficiently arresting the patient’s movement upon repositioning to a lateral position, they occasionally slide back to the supine position within the decompression period. Sometimes, patients could reposition themselves back to their previous position within the decompression interval. In the former case, with the correct training, the nurses repositioned the patient providing sufficient cushioning on either side to arrest their movement. In the latter case, the decompression algorithm scrutinized the nurses when the patient repositioned themselves within the decompression interval. This increased the non-compliance rate while they were temporarily compliant (time elapsed in patient’s new position) with the protocol. A better solution would be to reduce the turn period according to the time elapsed in the decompression interval. 22 This may have influenced the compliance results reported here.
Nurses’ opinions were gathered using an extended Technology Assessment Model (TAM)-based questionnaire 23 and interviewed to understand their perceived usefulness, ease of use, intention to use the system, and compatibility of the system with their current clinical practices. We recorded a response rate of 70.5% of all nurses who were part of the study. Most of the responses were recorded using a seven-point Likert scale, ranging from 1 “strongly disagree” to 7 “strongly agree”. More than 75% of them found PRESENSE to be more useful than manual turn reminders. Though everyone welcomed the use of new technology in their clinical practice, 25% disagreed that the system was easy to learn and 91.7% said that they would require technical assistance to use the system regularly. Nurses were guided on how to use the system before the start of phase II of the study, though no subsequent training was conducted. All of them believed that use of such systems promotes good clinical practices, and improves their performance. Of the nurses interviewed, 90% also believed that the use of such systems would imply major changes to their clinical practices.
During the system implementation, we found a few drawbacks in using a wearable sensor to continuously monitor the patient position. The wearable sensor could not recognize whether it is attached to a patient’s skin or not, hence it could be moved or turned even without being attached to a patient and still be recognized as a valid turn. Additional sensing units (for example: temperature sensor) would be required to check if the sensor is placed on the skin. When attached to a patient’s chest, it can only monitor their upper torso movement accurately and could not capture the movement of other body parts including heels and the lower back region (for example: Gluteus Maximus). In such cases, more than one device is needed to capture the entire body movement and accurately assess the quality of a patient’s repositioning. Hence, a wearable sensor attached to a patient’s chest can estimate the patient’s position but not accurately determine the quality of patient’s repositioning.
Through this study, we were able to (1) clinically evaluate PRESENSE which was previously validated only in laboratory settings, (2) understand the constraints in using continuous patient position monitoring system, and (3) explore the utility of such systems in ICU care management.
Conclusion
When presented with visual alerts generated by a continuous patient position monitoring system in the ICU, nurses were able to significantly improve their turn protocol compliance. Further clinical validation needs to be carried out in multiple ICUs, with different baseline rates of turn protocol adherence, before it can be ascertained whether such systems would universally increase turn protocol compliance.
Supplemental Material
Supplemental material for Effectiveness of a continuous patient position monitoring system in improving hospital turn protocol compliance in an ICU: A multiphase multisite study in India
Supplemental material for Effectiveness of a continuous patient position monitoring system in improving hospital turn protocol compliance in an ICU: A multiphase multisite study in India by BS Renganathan Sridhar Nagaiyan, SP Preejith, Shameer Gopal, Susovan Mitra and Mohanasankar Sivaprakasam in Journal of the Intensive Care Society
Footnotes
Acknowledgement
We thank Dr Senthil Kumar, Ganesh Arvind, Varalakshmi, Mahalakshmi, Suriyakumar, and Justin for their outstanding efforts in the conduct and monitoring of the study. We thank all centres and staff involved in this study, and the patients and their families.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.