Abstract
Objective:
The records from a network of mobile wound clinics were analyzed to evaluate the efficacy of a human placental-based allograft compared with the standard of care for patients with chronic pressure ulcer wounds and multiple comorbidities.
Approach:
Real-world evidence from a mobile wound clinic (July 2022–June 2023) was used to analyze patients with pressure ulcer wounds, most of whom received home-based wound care and had multiple comorbidities that worsened their chronic wounds. Patients were grouped into standard of care with debridement or standard of care with debridement and grafting intervention. After each visit, the wound area and depth were measured and recorded. Wounds were scored for granulation and epithelialization and eschar and slough during each visit. The standard of care with debridement and grafting cohort was monitored for graft duration, and the number of graft applications per wound was recorded. Statistical analysis used a paired t-test and ANOVA for multiple comparisons.
Results:
The standard of care with debridement and grafting cohort demonstrated an average percent change in wound area of 46% reduction, compared to a 42% increase in the standard of care (p < 0.001). The mean percent change in wound depth was a 36% reduction in the graft recipients versus a 37% increase in the standard of care with debridement (p < 0.001). Graft recipients improved their positive wound score by 8% (granulation and epithelialization), and decreased their negative wound score decreased by 55% (eschar and slough) after the final graft visit, compared with patients receiving standard of care with debridement. The average duration of the graft was 147 days (±8 days), involving 22 applications (±1.7) per wound.
Conclusion:
We present real-world evidence demonstrating the use of placental-based grafts to reduce wound size in patients with chronic PU, multiple comorbidities and limited mobility. Our study highlights the importance of reviewing the current local coverage determination and making graft applications permissible based on patients’ wound-care responses, thereby benefiting patient care and quality of life.
Introduction
Comorbidities in a patient can complicate the normal healing response to a lesion, allowing it to develop into a chronic wound. A common comorbidity among chronic wound patients is limited mobility, which is a significant risk factor for the development of pressure ulcer (PU), 1 a condition resulting from forces that build up on the posterior side of the body and are exposed to the bed and pressure points that bear weight, such as the back, buttocks, heels, and tailbone of the patient. Limited mobility is an insidious comorbidity, making effective interventions, monitoring, and staging of PU wounds critical to preventing and mitigating the formation of PUs.
PU patients depend on the expertise of wound care nurses for preventive and treatment interventions provided in a community clinic setting. However, effective wound care necessitates regular monitoring and clinical attention, which can often discourage patients with multiple comorbidities and limited mobility from seeking help. For instance, chronic diabetic foot ulcer (DFU) patients cite many barriers to receiving care from a community-based clinic, such as time constraints, lack of mobility, financial insecurity, distance, lack of transportation, and lack of supportive assistance.2,3 Home-based mobile clinical care offers a lifeline for PU patients who face obstacles to travel and suffer from limited mobility; trained in wound care, licensed nurse practitioners travel to patients’ residences and provide twice-weekly wound care, which assists in timely wound monitoring. In this scenario, patients who avoid travel for much-needed wound treatment are visited by wound care nurses, preventing their PUs from worsening and festering; they initiate the healing process for difficult-to-treat wounds.
The standard of care (SOC) for PUs starts with off-loading pressure from the wound while minimizing shearing and friction to prevent further wound formation. Next, surgical and/or ultrasound debridement prepares a healthy wound bed; moisture control maintains a dry and clean wound using standard dressings, and vascular assessment and infection control are crucial. 4 In cases where chronic wounds fail to heal or show improvements, an advanced supplement to SOC is the application of human placental-based membrane allografts, which can benefit chronic wounds due to their superior moisture management and anti-inflammatory properties. Placental-based allografts outperform basic dressings because they are immunologically inert, acellular biological products that initiate healing by creating a barrier to the environment and providing a scaffold for re-epithelialization. 5 In addition, human placental-based membranes are classified as class I (Kumar system) 5 skin substitutes, which comprises of single or bilayer, temporary dressing material. Furthermore, some advantages of human placental-based membranes include pain relief, antibacterial properties, non-immunogenicity, which limits host rejection, reduced inflammation and scarring, and scaffolding for cell migration and proliferation. 5 Disadvantages of human placental-based membrane include its temporary durability and expense. 5 They have proven effective for many chronic wounds, including DFU 6 and venous leg ulcers (VLU),7,8 by promoting wound closure and reducing wound depth. Human placental-based allograft membranes also contain growth factors and cytokines that promote anti-inflammatory effects and aid in re-epithelialization, accelerating wound closures.9,10 Intractable chronic wounds, such as PUs, necessitate advanced interventions that the SOC alone often inadequately addresses.
Methods
Study design
The limited availability of peer-reviewed studies on the efficacy of human placental-based membranes in advanced treatment of PUs warrants further investigation. We present real-world evidence demonstrating the use of placental-based grafts to reduce wound size in PU patients with multiple comorbidities and limited mobility, who are unable to access community clinics for wound care. In our retrospective observational study, we used a sample of recorded patient data (n = 1303) covering 1 year from July 2022 to June 2023 from primarily home-based care (69%) visits in United States communities; we examined the effect of placental-based allograft on PU wound healing compared to SOC. SOC includes PU patients whose wounds responded to gauze dressing, either closing or shrinking in size by 40%–50% after 4 weeks. PU wounds that showed no improvement were treated with one set of human placental-based membrane allograft tissue products, applied weekly according to the manufacturer’s guidelines.
In our observational retrospective study, we examined 1303 total patient records from July 2022 to June 2023 with chronic wounds; 443 met our inclusion criteria, and 860 were excluded. Our inclusion criteria were focused on patients with chronic PU wounds who agreed to receive care from wound clinicians. The exclusion criteria for our study included patients with chronic wounds that were not PUs (such as DFU/VLU) and those who refused treatment explicitly, resulting in the exclusion of n = 860 patients. Patients with PU wounds were divided into two interventions: those who received SOC with debridement (n = 113) and those who received SOC with debridement and grafting (n = 330). The SOC of PUs included wound off-loading, adequate drainage, surgical and/or ultrasound debridement, and gauze dressings promoting a moist wound environment. Patients whose wound areas did not completely heal or contract by 40%–50% after 4 weeks were included in the SOC with debridement and grafting cohorts. This interventional group comprises PU patients who received SOC, which provides surgical and/or ultrasound debridement, wound off-loading, and infection control. In addition to SOC, their wounds were treated with human placental-based membrane allografts to protect against infection and water loss. Each patient, once assigned to a group, did not crossover and remained in the same group for the study. A summary of patient data and groups is provided in the chart shown in Figure 1. These patients were treated for at least one PU wound, undergoing initial SOC treatment before progressing to a placental-based graft. The study is observational in nature and retrospective, so real-time direct interventions were not administered during the study. The treatment for the patients was administered until the wounds healed and closed, unless the treatment was halted at the patient’s request. Each patient’s PU wounds were examined and measured for area size and depth, as well as wound characteristics such as positive and negative wound scores, and descriptive statistics on graft usage in recipients. However, for this study, the follow-up analysis had a maximum timeframe of 395 days, from the initiation of the first treatment to the final visit for both the SOC with debridement and the SOC with debridement and grafting groups. Patients whose wound care recovery exceeded this timeframe were excluded from the study.

A chart of treatment groups and the total number of Pts in the retrospective study. The PU Pts are grouped between the SOC with debridement and the SOC with debridement and grafting treatments. Eligibility for the study was determined based on Pt inclusion and exclusion criteria.
Wound scoring
Data parameters measured at all visits included wound area size, depth, and the presence of granulation and epithelialization (G + E, positive score), as well as eschar and slough (E + S, negative score). To assess the efficacy of the interventions, we calculated the change in the percentage of area and depth of the wound across the first visit to the final visit for the SOC with debridement and the SOC with debridement with grafting cohorts. A positive value of the wound area percent change signifies the percentage of shrinkage of the wound area, while a negative value indicates an increase in wound area. This calculation method was similarly applied to wound depth measurements.
Aside from wound size measurements, wound scores were tracked at the initial, first, and final graft visits. This dataset was examined for evidence of change in outcome between SOC and graft treatment. Wound healing was assessed by measuring the change in wound area as treatment progressed, as well as by scoring the presence of G + E, a positive indicator of healing, or the presence of E + S, a negative indicator of healing. The granulation progression of a PU was evaluated from 0 to 100, and the re-epithelialization progression was evaluated from 0 to 100. For instance, if the wound exhibited complete granulation it would be given 100, and similarly for re-epithelialization. The average of these two numbers is the positive wound score. Similarly, eschar progression is assessed with sloughing progression, with the average of these scores being the negative wound score. These populations were also compared internally based on how tissue graft application affected wound healing and outcome rates. Wound assessments were measured and recorded with a metric ruler and a cue tip. The duration of graft treatment and the number of applications per wound were closely monitored in SOC with debridement and grafting recipients. The duration was determined using the initial graft dates and final visit dates. The number of grafts used and the number of chronic wounds were recorded to calculate the number of grafts applied per wound for SOC with debridement and grafting recipients (N = 330).
Statistics
The analysis of wound parameters and their descriptive statistics, such as mean, standard error of the mean (SEM), median, and the number of n, was completed with Prism (v9; GraphPad Software, Boston, MA, USA). The p value was calculated using Student’s t-test or one-way ANOVA.
Results
The patient population in our study had various comorbidities contributing to their PU’s initial size and condition. Our PU patients presented with larger baseline wound areas, averaging 34.47 cm2 and a median of 9.5 cm2. Published studies have reported baseline average PU (±SEM) sizes of 5.8 cm2 (±6.4 cm2; n = 16), 11 which aligns with another study that has a baseline average PU area of 3.4 cm2 (±0.56 cm2; n = 10). 12 The smaller initial sizes of PU wounds are strong indicators of the likelihood of healing. This demonstrates that our patient population significantly deviates from the smaller baseline found in other studies and presents with severe comorbidities. This patient dataset was analyzed to evaluate the impact of graft treatments on patient outcomes, specifically with respect to wound-healing characteristics and comorbidity management.
Table 1 summarizes the demographics of patients with PU in our observational study. A total of 443 patients met the inclusion criteria: 113 in the SOC with debridement group, 330 in the SOC with debridement and grafting group, 195 males (44%), and 248 females (56%). Most patients were over 65 years old (82%), with an average age (±SEM) of 78 (.76) years. The majority of patients were White (29%), followed by Latino (10%), Black (7%), and Asian (5%). Many individuals had unknown or unrecorded racial identities (49%). At the mobile health clinic, most care was provided in patients’ homes (69%), whereas the remainder received care at a clinic (31%). Many patients had multiple comorbidities, with mounting ailments exacerbating their hard-to-heal chronic wounds; the incidence of each comorbidity was determined for each patient. In our study, 224 patients (51%) had limited mobility, leaving many relying on supportive travel assistance. In addition, neurodegenerative diseases were observed in 183 of all patients (41%), such as Alzheimer’s disease, Parkinson’s disease, dementia, and multiple sclerosis. In many cases, other comorbidities found in PU patients include incontinence or kidney disorders (31%), heart disease (26%), diabetes (24%), and spinal cord injuries (5%). The presence of multiple comorbidities heightens additional risks, leading to larger PUs and longer healing times.
Summary of the demographics of the pressure ulcer patients.

SEM: standard error of the mean; SOC: standard of care.
The study summarized the characteristics of the PUs in patients in Table 2. There were 1135 total PU wounds recorded from 443 total patients, 338 PU from SOC with debridement group, and 797 PU from the debridement and grafting group. In terms of the location of PU, the highest frequency was found in the sacrum, butt, and heel of the patient, in the following ratios 20.2%, 18.8%, and 14.4%, respectively. The rest of the locations of the PUs were <10%, at the hip (9.4%), foot (6.1%), coccyx (5.5%), leg (4.6%), ankle (4.3%), back (3.6%), ischial (3.2%), and toe (2.3%). The PU wounds were also staged during the initial visit as 1–4, deep tissue, and unstageable, as described previously. 13 Most PU wounds were stageable, with stage 3 (41.3%) the most frequent, followed by stage 2 (18.6%), unstageable (15.6%), and stage 4 (12.8%), as shown in Table 2. A minority of the PU wounds were deep tissue (4.4%), N/A (4.3%), stage 1 (1.9%), and healed (1.1%). Some patients were recorded as healed, although their other wounds were at various stages of healing.
Summary of the characteristics of the pressure ulcers in patients.

PU: pressure ulcer; SOC: standard of care.
The study collected outcome parameters related to the wound-healing response. The percentage change in healed wound area and the mean differences between the SOC with debridement and the SOC with debridement and grafting cohorts were compared. As shown in Figure 2(a), the mean percent change in wound area (±SEM) for SOC with debridement and grafting recipients reflected a mean reduction of 46% (±4%) compared to SOC with debridement, who had a mean increase of 42% (±24%); the mean difference was statistically significant (***p = 0.0002). Another recorded parameter was the wound depth, measured during the initial visit, the first graft application, and the final visit. In Figure 2(b), we illustrate that the mean wound depth for SOC with debridement and grafting recipients decreased by 36% (±3.7%) compared to an increase of 37% (±19%) in SOC with debridement recipients during treatment; the mean difference was statistically significant (***p = 0.0001).

The mean percent change of wound area and mean wound depth in PU wound patients after treatment. The PU patients’ wound metrics included wound area and depth, measured after the initial visit, first graft visit, and final visit. These values were transformed into a percentage of the healed wound compared to its initial value at the first visit. The percentage of reduction of wound area and depth reduction was calculated for each group on the SOC with debridement (n = 113) and the SOC with debridement and grafting (graft; n = 330). The mean’s statistical significance between these groups was assessed using Welch’s t-test: (a) the comparison shows that patients whose pressure wounds received a graft have increased healing or mean (±SEM) wound area reduction of 46% (±4.0%) compared to SOC, a mean increase of 42% (±24%; ***p = 0.0002) and (b) analysis of mean (±SEM) wound depth for PU patients who received graft showed a 36% (±3.7%) reduction overall versus a mean increase of 37% (±19%) for SOC (***p = 0.0001).
Another essential characteristic recorded was the wound quality of PU, based on positive and negative wound scores for each patient during the initial visit, first grafting, and final visit. The positive wound score (±SEM) for PU patients changed slightly from an initial value of 76.24 (±1.42) to 81.15 (±1.43; p < 0.001) after the first graft application. The positive wound score, as shown in Figure 3(a), increased from the initial visit to the final graft visit, from 76.24 (±1.42) to 86.86 (±1.31; p < 0.0001). Similarly, the positive wound score rose from the first graft visit to the final graft visit, from 81.15 (±1.43) to 86.86 (±1.31; p < 0.001; Figure 3(a)). In addition, analysis of the negative wound score, as shown in Figure 3(b), displayed no change from the initial visit to the first graft, from 25.69 (±1.46) to 26.52 (±1.36; p > 0.05, NS), respectively; however, by the final graft visit, the score decreased to 15.90 (±1.25; p < 0.0001), representing a percent decrease of 38% compared to the initial visit. The initial value of the negative wound score remained unchanged after the first graft visit; however, by the final graft visit, the value had decreased below its initial level.

The mean positive wound scores (G + E) and negative wound scores (E + S) for graft recipients. After the initial visit, at the time of first graft, and the final visit, PU patients’ positive and negative wound scores were collected for each wound. PU patients who received graft treatment (n = 331) were scored for positive and negative wound scores. Scores were statistically compared for each time point using a one-way ANOVA with Bonferroni’s multiple comparisons: (a) the positive mean wound scores (±SEM), measurement of G + E, increased from baseline of the SOC to the first graft visit from 76.24 (±1.42) to 81.15 (±1.43; ****p < 0.0001), and increased from SOC to second graft visit from 76.24 (±1.42) to 86.86 (±1.31; ****p < 0.0001), respectively. In addition, the positive wound score increased from the first graft to the final graft from 81.15 (±1.43) to 86.86 (±1.31; ***p < 0.001), respectively and (b) the negative wound scores, which quantified the presence of E + S, did not change significantly from SOC baseline to the first graft visit, 25.69 (±1.46) versus 26.52 (±1.36; ****p < 0.0001), respectively. However, upon final graft visit, mean negative scores were reduced to even below initial visit, from 25.69 (±1.46) to 15.19 (±1.25; ****p < 0.0001).
SOC with debridement and grafting recipients were also monitored for the total recovery time needed to fully heal the chronic wounds and the average number of graft applications required to complete the treatment. The average (±SEM) recovery time of graft treatment lasted 147 days (±8 days), or 21 weeks, as shown in Figure 4(a). In addition, the number of graft applications used per wound was recorded for each SOC with debridement and grafting recipient (n = 330), and the mean number of graft applications required per wound was determined to be 22 (±1.7), as shown in Figure 4(b). The mean treatment time for the SOC with debridement cohort was 28 days, and the mean number of visits was 6.
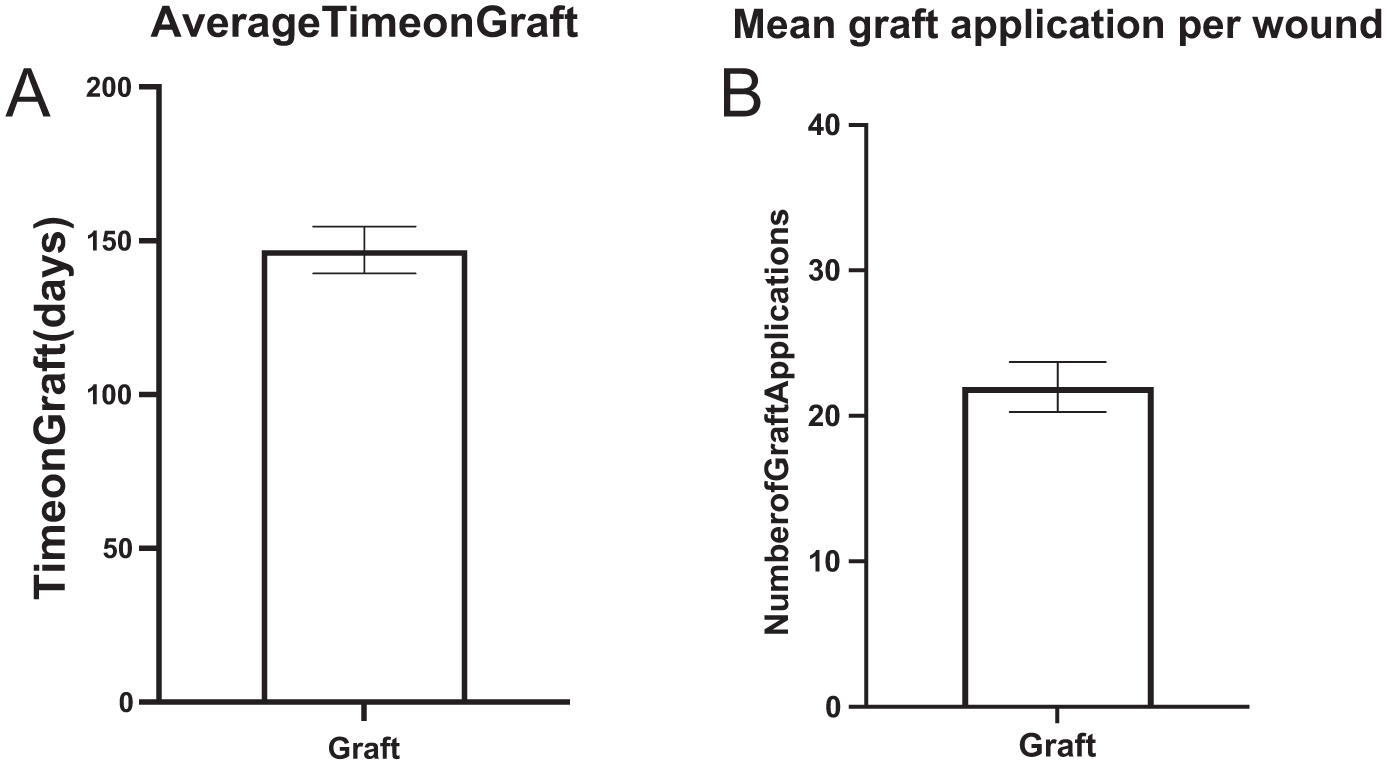
Descriptive statistics of graft recipients: (a) the average duration of graft treatment per PU patient. The dates of visits of grafted patients (n = 331) for the first graft application and the last graft visit were recorded, and the total duration was determined and reported in days. Each patient’s mean (±SEM) graft treatment duration was 147 days (±8 days) and (b) the mean graft applications per wound. The number of grafts applied was recorded for each patient (n = 331), and the number of chronic wounds in each patient; the computed mean (±SEM) is 22 graft applications per wound (±1.7).
Discussion
Our study examined patients undergoing wound care for PU and compared the treatment effects of SOC with debridement versus SOC with debridement and grafting. Patients who underwent placental-based grafting exhibited improved wound-healing responses and enhanced closure characteristics, as evidenced by reduced wound size. The reduction in wound area for SOC patients with debridement and grafting exceeded 46%; by contrast, SOC patients with debridement experienced a 42% increase. The larger PU wounds in our mobile patient population, averaging 34.47 cm2 with a median of 9.5 cm2, suggest comorbidities such as decreased mobility (51%), diabetes (24%), heart disease (26%), incontinence (31%), neurodegenerative disease (41%), and spinal cord injury (5%) can lead to hard-to-heal wounds that require advanced interventions. In each SOC with debridement and grafting case, before advanced treatment, chronic PU wounds failed to close completely, starting with initial wound sizes ranging from six to 10 times larger than the published wound area baselines for PU (±SEM): sizes of 5.8 cm2 (±6.4 cm2; n = 16) 11 and 3.4 cm2 (±0.56 cm2; n = 10). 12 This aligns with a randomized clinical study indicating that grafting with amniotic membrane allograft reduced wound-healing times and effectively managed wound closures in PU patients. A prospective randomized clinical trial enrolled 24 patients with PU and was randomized to receive either amniotic membrane or control treatment; the control group received application of Dilantin powder. The duration of healing and the success rate were compared. 14 The skin-grafted group exhibited a faster healing time, achieving results in 20 days compared to 54 days for the control group, and the graft group’s partial healing of PUs was significantly higher. 14 Our study provides additional data that supports the positive effects of skin allografts on wound healing for recalcitrant PUs.
Despite comorbidities that prevented most patients from receiving care in a facility, they received PU care regardless of location. A comparison of wound area outcomes showed that mobile versus facility (±SEM) had a 12.5% (±15%) increase and a 21.4% (±15.9%) reduction, respectively. The comparison of mobile versus clinic for wound area reduction showed no statistically significant difference between the two populations. The data is not shown. This report examines the use of placental-based grafts among patients with chronic PUs, regardless of treatment location. The absence of a statistically significant difference in outcomes suggests that the grafting treatment drives wound closure rather than the location of care.
Outcomes for stage 3 and stage 4 PUs were comparable to those observed with SOC with debridement and grafting, regardless of staging. Stage 3 PUs showed a 34% reduction in wound area in the debridement and grafting group, compared with a 25% increase in the SOC with debridement alone group (data not shown). Among exclusively stage 4 PUs, there was a 37% reduction in wound size, compared with a 16% increase in wound area for SOC with debridement; this difference was not statistically significant (data not shown). Interestingly, the reduction in wound area for stage 3 PUs was statistically significant, whereas it was not for stage 4, despite a similar trend; SOC with debridement and grafting improved wound healing.
Furthermore, in addition to improving wound closure rates in the SOC with debridement and grafting group, these patients also experienced enhanced wound depth closures. We observed that, on average, wound depth in hard-to-heal wounds worsened under SOC with debridement; however, this effect was reversible and improved significantly with grafting. A completely healed wound will have no depth, achieving a 100% reduction in wound depth. In contrast, the average PU treated with SOC with debridement showed an increased wound depth, with a mean increase of 37% change. In comparison, SOC with debridement and grafting treatment resulted in a 35.8% reduction in wound depth (p = 0.0001), indicating a significant improvement. The application of the placental-based graft contributed to reducing the depth of PUs, consistent with other studies demonstrating increased healing and better closure rates following graft treatments. Consequently, compared to the SOC with debridement, which merely delays wound closure and increases wound depth in chronic wounds, effective healing is achieved in patients receiving SOC with debridement and grafting treatments.
An essential means of preventing PU is the proper diagnosis of PUs, which can be confused with other comorbid skin conditions. Incontinence is a significant comorbidity (31%) in our patient population, characterized as the involuntary loss of bodily fluids, typically urine or stool. Incontinence-associated dermatitis (IAD) is skin damage when in contact with urine or feces, often due to incontinence. 15 It is formed in similar locations of PU, at their posterior near the genital area, buttocks, and perineal area. 15 Damage caused by IAD can result in redness, inflammation, and skin peeling, covering a wide area of skin that may be exposed to excess moisture. However, observing key differences between PU and IAD can help prevent misdiagnosis; these include the location of the PU, which often forms at bony prominences, particularly the sacrum, heels, elbows, and the back of the head. 15 Another critical distinction is that the margins of the wound are very well-defined in PU, and in IAD, they are not usually well-defined. They can form over a wider area, blending into healthy skin. Preventive measures for IAD include effectively addressing each incontinence incident and maintaining clean and dry skin at the point of contact with feces or urine. This constant vigilance of maintaining a clean wound bed helps prevent exposure to infectious material. Another important consideration for patients with IAD is monitoring bowel movement and urinary conditions in graft recipients. Maintaining cleanliness and proper wound care requires cleaning and redressing wounds with fresh, placenta-based grafts when they become soiled. Frequent bowel movements and urinary incontinence may necessitate changing grafts at sacral locations more often than the usual weekly replacement schedule. As a result, more grafts may be required to fully heal a sacral or posterior wound in patients with comorbidities related to incontinence. Correctly diagnosing PU and IAD helps clinicians address the wound with appropriate care and treatment and helps limit complications that can worsen the skin condition.
While many factors can contribute to PUs, including comorbidities, we will outline four critical factors. The four crucial factors contributing to the development of PUs are shearing forces, pressure, friction, and moisture. 16 Shearing force and friction relate to the outer forces that disturb the skin and can cause a wound opening. Pressure is another factor, resulting from the patient’s weight, which restricts circulation in the area. Finally, moisture is a factor that can be either excessive or insufficient without proper wound monitoring. Maintaining a clean, dry wound promotes healing. PU tends to form over bony prominences of the skin, on the sacrum, the coccyx, and the ischial tuberosities. Other important risk factors for PU formation are prolonged immobilization, sensory deficit, circulatory interruptions, and poor nutrition. 16 Understanding these factors contributing to PUs enhances our approach and methods for preventing PU formation.
The SOC of PUs requires wound off-loading, adequate drainage of infected areas, debridement of dead tissue, and wound care that supports the wound-healing process. Off-loading pressure refers to the practice of frequently turning or repositioning the patient to relieve pressure at the wound site. This can be accomplished with onlays or pads over beds that help relieve pressure points for patients. 17 Also, automatic beds with air circulation help distribute pressure while the patient is lying on the bed. These pressure-relieving methods can be monitored manually or mechanically using specialized beds. Thus, specialized beds with air circulation can relieve pressure at sites vulnerable to PUs.
Aside from relieving pressure to treat PUs, controlling infection is also an essential aspect of treatment. The wound area must be cleaned, evaluated for infection, and treated with antibiotics as needed. These treatments could be locally applied antiseptics, such as povidone-iodine, silver sulfadiazine, hydrogen peroxide, and sodium hypochlorite. 4 The application of antibiotics will directly kill bacteria that are within the PUs. The clearance of bacteria promotes healthy wound healing and prevents the infection from worsening. In some cases, when necrotic tissue envelops the PU, further debridement and drainage of the wound would be required before local application of antiseptics.
The presence of necrotic tissue at the site of PU and excessive fluid accumulation are detrimental to healthy wound healing and must be cleared. The debridement of the wound area removes devitalized tissue, which can harbor biofilms and abscesses filled with fluids. The drainage of abscesses becomes necessary to clear the wound of excess fluids that can retard a healthy wound-healing process. Several procedures can be employed to perform debridement, including surgical, acoustic debridement using ultrasound, hydrotherapy, and enzymatic debridement of wounds. The purpose of debridement is to allow the formation of well-granulated tissue within the ulcer to re-epithelialize the PU.
The process of wound healing of PUs includes cleaning the wound area and clearance of infection by antibiotics, debridement of necrotic tissues, drainage of excess fluids, and application of topical agents and dressings. The dressings that can be used to protect the wound from the environment, excessive moisture, and physical disturbances such as friction or shearing are diverse; they include gauze dressings, alginate dressings, foam dressings, hydrocolloid dressings, hydrogel dressings, silver-containing dressings, honey-containing dressings, transparent film dressings, negative wound therapy, and bio-sourced human placental-based dressings. 4 Various dressings are available to protect the wound from excessive moisture and physical friction to prevent disturbances. If soiled by urine or feces, dressings should be replaced to avoid unnecessary moisture or exposure to infectious material. SOC consists of cleaning, debridement if needed, and using non-graft dressings. Our study parallels others that show that the application of human placental-based membrane graft, when appropriate, is superior to SOC when it comes to improving wound closures of PUs based on wound area and depth characteristics. 14
Allograft was superior to SOC in accelerating wound-healing, as evidenced by improvements in wound area and depth. Human placental-based grafts serve as an effective barrier, helping to retain moisture and acting as a biodegradable scaffold. Other benefits of this biologic dressing include growth factors that induce re-epithelialization and pro-angiogenic factors.4,14 An in vitro study demonstrated the healing effects of placental-based epithelial cells in a rodent PU model, which produced significantly reduced inflammatory infiltration, more pronounced collagen fibrosis, and scar formation compared to the control group, which received only a gauze dressing. 18 Our study parallels many studies that have utilized placental-based allografts in other types of ulcers, such as DFU19,20 and VLU7,8,21 to help improve wound healing and prevent adverse events. 22 The benefits of using placental-based allograft treatment compared to SOC are significant and reduce wound-healing time.
U.S. Medicare and Medicaid Service coverage guidelines allow only eight graft applications per wound. 23 We show that PU patients, on average, used 22 graft applications for wound recovery. Therefore, expanding the allowable maximum number of grafts per patient would provide proper wound care, enhance wound healing, and improve overall patient care. The current coverage guidelines were likely based on studies with patients with greater and more frequent access to care. This small demographic is not representative of the general population. In our practice, our patients differ significantly from the general population, necessitating the provision of more than eight grafts per wound for their wound care.
Limitations
This retrospective study was based on patient data collected by a regional wound-health clinic, whose patient population may not be representative of the general U.S. population. The data collection and methodology conformed to established guidelines practiced by this specific clinic and are not necessarily widely adopted by all wound clinics in the United States; therefore, this limitation applies to the study. Since the outcome of interest has already occurred alongside the intervention, this is an inherent limitation of a retrospective study, as it can predispose the study to selection bias. Furthermore, the sample size selected for the study did not undergo calculations for statistical power since the study was retrospective, and was based on patients who underwent treatment within a given timeframe, and therefore is a limitation of the study. Additionally, confounding variables are challenging to identify and control, especially in a patient population with multiple comorbidities that may or may not be diagnosed during the study. Finally, while vigorous efforts were made to keep records updated, unidentified parameters were not collected, which is also a study limitation.
Conclusions
In this retrospective study, we demonstrated that PU patients benefit from improved wound healing when placental-based allografts are applied more extensively compared to SOC alone. We observed a statistically significant improvement in wound characteristics, as indicated by reductions in wound area and depth. In our study, PU patients utilized an average of 22 graft applications for complete wound closure, underscoring the need to review the local coverage determination regarding allowable graft applications based on patient wound response. This consideration is in the best interest of patient care and quality of life.
Footnotes
Ethical considerations
IRB exemption not granted.
Consent to participate
Patients granted informed consents. This retrospective study is exempt from informed consent. The analyzed data is recorded information that did not require direct interaction to perform the analysis. The human subjects were de-identified to comply with HIPAA Privacy rules.
Author contributions
The general idea and direction of the article was provided by D.K., M.S.-C., and H.H.M. The collection and analysis of the data were done by H.H.M. The research, writing, and figures of the article were completed by H.H.M. D.K., M.S.-C., and H.H.M. proofread and provided suggestions for the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
