Abstract
Objectives:
The authors invented the shoulder balance support device aiming to prevent skin complications and neck pain in surgical patients operated in lateral decubitus position. This study aimed to compare skin complications and neck pain between patients with shoulder balance support device and traditional positioning instruments and to assess surgeons’ and anesthesiologists’ satisfaction in using the device.
Methods:
A randomized controlled trial, which followed Consolidated Standards of Reporting Trials(CONSORT) statement, was conducted in patients who underwent laparoscopic upper urinary tract surgery in the lateral decubitus position from June 2019 to March 2020. The shoulder balance support device was used in 22 patients and 22 other patients were in the control group. The area of skin erythema, bruising, or abrasion resulting from the pressure effect of the lateral decubitus position was measured and the pain score for the neck and shoulder area after the operation was assessed. Furthermore, the satisfaction of the medical personnel taking care of the patients and using the shoulder balance support device was investigated.
Results:
A total of 44 patients were included. No patient in the intervention group reported neck pain. Skin erythema was found in six patients in each group and the median area of skin erythema was significantly smaller in the intervention group. Most of the medical personnel reported satisfaction with use of the device.
Conclusion:
This device is an innovative tool with the aim of ultimate care for surgical patients.
Clinical Trial Registration Number:
Thai Clinical trials registry ID TCTR 20190606002
Introduction
Upper urinary tract surgeries, including nephrectomy, pyeloplasty, and ureterolithotomy are among the most common procedures performed in the field of urology. These operations can be performed using an open or robot-assisted laparoscopic approach. 1 The kidneys and ureters are retroperitoneal organs that are located quite deep into the skin. As a result, the surgical approach to these organs requires proper positioning that widens the space between the lower ribs and iliac bone. The lateral decubitus position is the most common position for this purpose and is used worldwide. 2 Although this position is considered safe, it can still be associated with complications such as skin abrasions, pressure sores, or neck and shoulder pain, especially in patients with a body mass index (BMI) of >25 kg/m2. A medical engineer assisted this study to design a novel shoulder balance support (SBS) device to assist the medical team in placing the patients in an appropriate and safe position. The primary objective of the present study was to compare skin and shoulder complications and neck pain between patients who underwent surgery using the SBS and traditional positioning instruments. The secondary objective was to assess the surgeons’ and anesthesiologists’ satisfaction with using the SBS device.
Methods
Study protocol
This study was ethically approved by the Institutional Committee on Human Rights Related to Research Involving Human Subjects (MURA2018/903). The study protocol was registered and conducted in accordance with CONSORT guidelines, good clinical practice guidelines, and the principles of the Declaration of Helsinki. A randomized controlled trial was conducted from June 2019 to March 2020 to assess complications of and satisfaction with our novel SBS device in patients who underwent laparoscopic upper urinary tract surgery in the lateral decubitus position versus standard positioning at Ramathibodi Hospital (Supplemental Figure 1). The inclusion criteria were kidney or ureter surgery in the lateral decubitus position, male or female patients aged from 20 to 70 years, and full consciousness with the ability to communicate well in Thai language. The exclusion criteria were chronic back and shoulder pain before the surgery; ulcers or wounds in the head, neck, or shoulder area; and refusal to participate in the study. The patients were informed of the possible adverse events secondary to the intervention, both verbally and in written form during the enrollment period. After the patients were admitted for laparoscopic upper urinary tract surgery, they provided written informed consent with preservation of their anonymity. The patients were allocated into the intervention group or control group at a ratio of 1:1. The randomization sequence was generated using a permuted block randomization technique with random sequence generation using a randomization plan generator (https://www.sealedenvelope.com). The sequential randomization numbers were sealed in opaque envelopes prepared by an independent research assistant. The envelopes were opened in the exact and correct order of numbers by the research team. The sample size estimation was calculated by the area of skin complication in patients from a pilot study with the result of 22 patients in each group.
Outcome measurements
The patients’ baseline characteristics (age, sex, BMI, and underlying diseases) and perioperative variables were collected by the research assistant. The primary outcomes of the study were the calculated area of skin erythema, bruising, or abrasion resulting from the pressure effect of the lateral decubitus position and the pain score for the neck and shoulder area after the operation (Figure 1). Upon completion of the operation, the position of the patient was changed to supine and transferred to the recovery room. The patient’s shoulder, neck, and back areas were inspected, and a photograph was taken 1 hour after arrival in the recovery room. The size of the skin complication was calibrated by placing a ruler scale in the photograph. The affected area was outlined and calculated twice using ImageJ (National Institutes of Health, Bethesda, MD). The mean area of the skin complication was compared between the intervention and control groups. The patients were asked to use a visual analog scale at 24 h postoperatively to assess pain in the neck and shoulder area. The patients, data collectors, and data analysts were blinded to the intervention. All possible adverse events were monitored throughout the study. The secondary outcome was the satisfaction of the medical personnel taking care of the patients and using the SBS device. The questionnaire was developed comprising of six items: (1) the SBS device enhances patient safety in terms of skin complications and shoulder dislocation; (2) the SBS device is easy to use and helps in placing the patient in the lateral decubitus position; (3) the SBS device does not impede the anesthetic procedure; (4) the SBS device can cause injury to patients; (5) the SBS device is safe, durable, and environment friendly; and (6) overall, I am satisfied with this device. Each item was graded on a five-point scale ranging from “strongly agree” to “strongly disagree.” After the questionnaire was built, the questionnaire was pilot tested in about 10% of the total population.

Skin complications. Panel (a) showed skin complication from pressure effect after surgery in the lateral decubitus position. Panel (b) demonstrated how the shoulder of a patient with a high BMI falls downward with gravity, resulting in an improper shoulder position.
Instrument details
The SBS device was designed and invented by Ms. Nicha, a registered nurse cooperating with the medical engineer under the concept of lever balance. The device comprises of two metallic stands of 52.5 cm in length connected to the surgical bed, two medical-grade metallic arms of 49 cm in length, and a curved metallic support size of 10 cm in width and 12.5 cm in length for support at the shoulder area. When the patient was on the lateral decubitus position, two metallic arms were attached to the surgical bed, the first arm was just below the shoulder level and another arm was at the head level. The metallic support with sponge will be at the shoulder level. Because the SBS device has two arms, the force that helps to support the shoulder extends in two directions, upward and caudally (Figure 2). The force from the device prevents gravity-induced back falling of the shoulder. In addition, a small soft sponge between the metallic support and skin minimizes the pressure where the skin contacts the device. As a result, the shoulder remains in a balanced position without the use of tape.
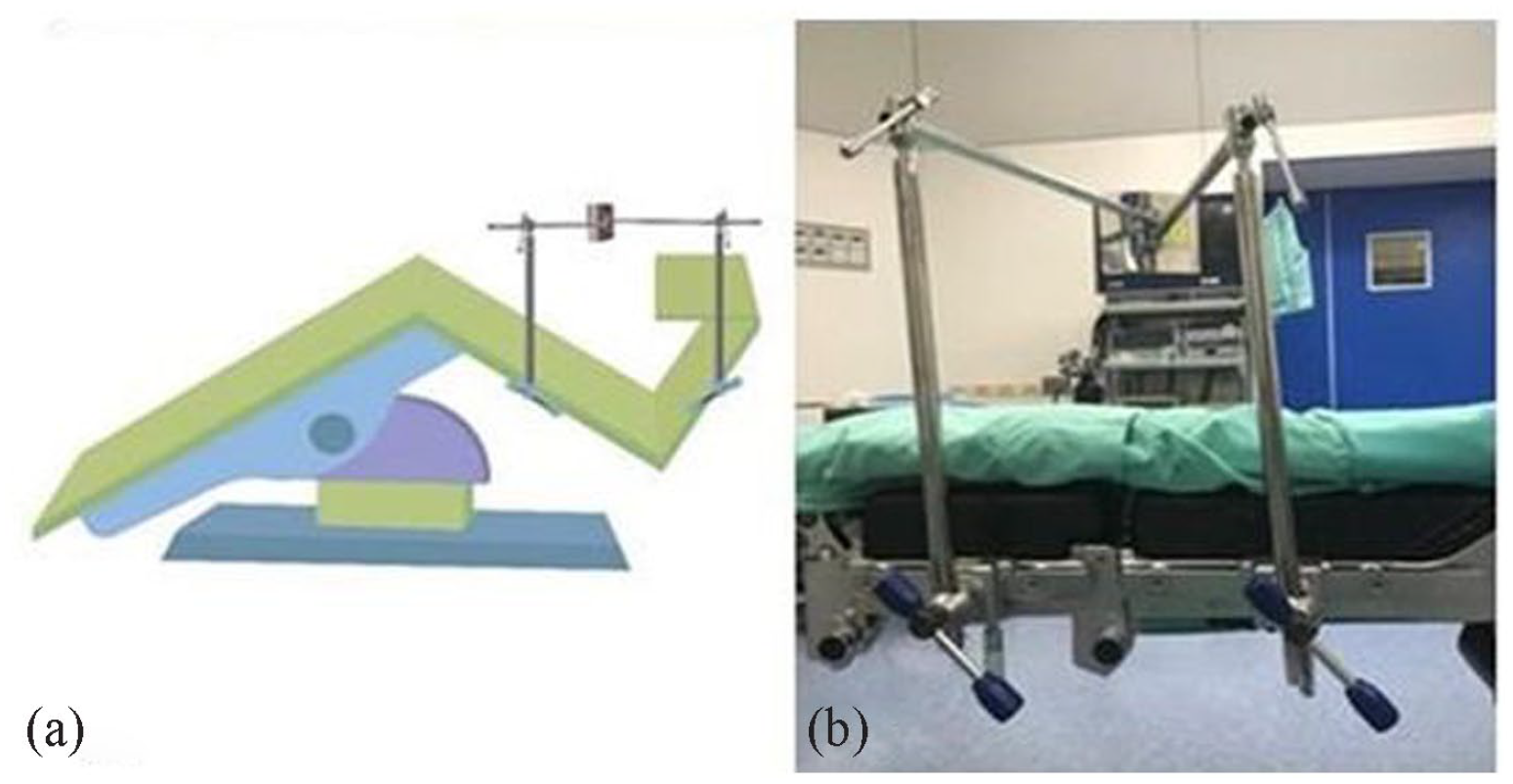
SBS device. Panel (a) showed the graphic picture when the SBS device was attached to the surgical bed. Panel (b) demonstrated that the real device is easily attached to the surgical bed before the bed is flexed. The first arm was just below the shoulder level and another arm was at the head level. The metallic support with sponge was at the shoulder level.
Statistical analysis
The patients’ baseline characteristics were presented as frequency and percentage for categorical data and mean ± standard deviation or median (interquartile range) for continuous variables. The area of the skin complication and the pain score were compared using an independent
Results
Forty-four patients were eligible for our study, among whom 22 used the SBS device. Among the 44 patients, 17 (38.6%) were men and 27 (61.4%) were women as shown in Table 1. Twenty-one patients (47.7%) were from 20 to 50 years of age. The mean age in the intervention group and control group was 24.70 ± 3.80 years and 25.60 ± 4.50 years, respectively. Six patients (13.6%) had diabetes and 14 patients (31.8%) had hypertension. With respect to the operation type, 42 patients (95.5%) underwent kidney surgery, and 2 patients (4.6%) underwent ureter surgery. Mean ± SD of operative time in SBS group was 193.4 ± 54.8 min and in control group were 176.8 ± 54.8 min (
Patients’ baseline characteristics.

SBS, shoulder balance support; SD, standard deviation; BMI, body mass index. Data are presented as
Study outcomes.

SBS, shoulder balance support; IQR, interquartile range. Data are presented as
Statistical significance.
In total, 17 surgeons and 13 scrub nurses answered the questionnaire regarding satisfaction of the medical personnel with the SBS device. Among these 30 staff members, 96.7% expressed overall satisfaction with use of the SBS device (Supplemental Table 1).
Discussion
Kidney and ureter surgeries are among the most common procedures in urology. The approaches for these surgeries can be open or robot-assisted laparoscopic. The lateral decubitus position is commonly used for most open and minimally invasive procedures such as robotic pyelolithotomy, extended pyelolithotomy, nephrolithotomy, and anatrophic nephrolithotomy. 3 Minimally invasive stone surgery, including ultrasonography-guided percutaneous nephrolithotomy, can also be performed in this position, which is preferred especially for patients with a higher BMI.4,5 This position is further applied to other uncommon procedures, such as retroperitoneal lymph node dissection and nephrectomy for horseshoe kidney. 6
Although this position is considered safe, some complications can occur, such as brachial plexus injury, pressure sores or ulcers, and pain in the neck, shoulder, arm, or hip area. Not only anterior and posterior support is important in this position, but proper padding and arm rest are also essential. The overall incidence of pressure sores after surgery is reportedly around 12%. 7 However, no specific report has focused on the incidence of pressure sores in the lateral decubitus position. Ulcers in this position are believed to be caused by a decrease in blood flow in the affected area during surgery. 8 The factors associated with skin complications include advanced age, malnutrition, preexisting medical conditions such as diabetes mellitus, and an operative time of >2 h. 9 Perioperative pressure sores are not only painful but are also associated with higher costs, a higher mortality rate, and a longer hospital stay.10,11
In the authors’ experience, patients with a high BMI may be placed in an improper shoulder position, resulting in postoperative neck and shoulder pain. Generally, the problem of a falling shoulder has previously been solved by using a tape with an arm support in an attempt to lift the shoulder. However, as a result, the incidence of skin complications such as skin blebs and abrasions increase. Our novel SBS device was developed in cooperation with a medical engineer to resolve this problem during routine patient care. Our results indicate that this novel device facilitates support of the shoulder and decreases the incidence of skin complications compared with standard lateral decubitus positioning. This device is feasible, safe, and pragmatic, and the medical team is satisfied with its use.
The main strength of our study is its randomized controlled design, which is ideal for investigating an intervention with minimal bias. In addition, not only the outcomes of the patients, but also the satisfaction of the device users were assessed. However, our study also has several limitations. Firstly, the number of patients was quite small. This may have prevented the detection of rare complications that can occur in the lateral decubitus position. Secondly, the pain score is a subjective outcome and can be subject to interference from other factors such as surgical wound pain or pain control medication. Thirdly, it was impractical to blind the doctors and nurses. The patients, data collectors, and data assessors were blinded. Finally, the study’s questionnaire has not been validated yet. However, the study’s questionnaire was pilot tested and has already completed pilot testing in 10% of the total population.
Conclusion
Our SBS device is an effective innovation for lateral decubitus positioning which was invented with the aim of ultimate care for surgical patients. This device can significantly decrease skin complications and shoulder and neck pain caused by improper positioning. Furthermore, the results of this study demonstrate the feasibility of using this device and the satisfaction of the medical personnel.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231167966 – Supplemental material for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial
Supplemental material, sj-docx-1-smo-10.1177_20503121231167966 for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial by Nicha Piyasoontrawong, Pokket Sirisreetreerux and Wachira Kochakarn in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121231167966 – Supplemental material for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial
Supplemental material, sj-docx-2-smo-10.1177_20503121231167966 for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial by Nicha Piyasoontrawong, Pokket Sirisreetreerux and Wachira Kochakarn in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121231167966 – Supplemental material for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial
Supplemental material, sj-docx-3-smo-10.1177_20503121231167966 for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial by Nicha Piyasoontrawong, Pokket Sirisreetreerux and Wachira Kochakarn in SAGE Open Medicine
Supplemental Material
sj-pdf-4-smo-10.1177_20503121231167966 – Supplemental material for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial
Supplemental material, sj-pdf-4-smo-10.1177_20503121231167966 for Complications prevention of shoulder balance support device for lateral decubitus position: A randomized controlled trial by Nicha Piyasoontrawong, Pokket Sirisreetreerux and Wachira Kochakarn in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Mr. Anusorn Adirekkittikun, medical engineer, for helping us develop the shoulder balance support.
Authors’ contributions
NP did the provision of study materials, data collection, and was a major contributor in writing the article. PS contributed to the conception and design, data analysis, interpretation, and article writing. WK provided the administrative support. All authors read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Institutional Committee on Human Rights Related to Research Involving Human Subjects in Mahidol University No. MURA2018/903
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Thai Clinical trials registry ID: TCTR20190606002
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
