Abstract
Background
Tidal hyperinflation can still occur with mechanical ventilation using low tidal volume (LVT) (6 mL/kg predicted body weight (PBW)) in acute respiratory distress syndrome (ARDS), despite a well-demonstrated reduction in mortality.
Methods
Retrospective chart review from August 2012 to October 2014. Inclusion: Age >18years, PaO2/FiO2<200 with bilateral pulmonary infiltrates, absent heart failure, and ultra-protective mechanical ventilation (UPMV) defined as tidal volume (VT) <6 mL/kg PBW. Exclusion: UPMV use for <24 h. Demographics, admission Acute Physiology and Chronic Health Evaluation II (APACHE II) scores, arterial blood gas, serum bicarbonate, ventilator parameters for pre-, during, and post-UPMV periods including modes, VT, peak inspiratory pressure (PIP), plateau pressure (Pplat), driving pressure, etc. were gathered. We compared lab and ventilator data for pre-, during, and post-UPMV periods.
Results
Fifteen patients (male:female = 7:8, age 42.13 ± 11.29 years) satisfied criteria, APACHEII 20.6 ± 7.1, mean days in intensive care unit and hospitalization were 18.5 ± 8.85 and 20.81 ± 9.78 days, 9 (60%) received paralysis and 7 (46.67%) required inotropes. Eleven patients had echocardiogram, 7 (63.64%) demonstrated right ventricular volume or pressure overload. Eleven patients (73.33%) survived. During-UPMV, VT ranged 2–5 mL/kg PBW(3.99 ± 0.73), the arterial partial pressure of carbon dioxide (PaCO2) was higher than pre-UPMV values (84.81 ± 18.95 cmH2O vs. 69.16 ± 33.09 cmH2O), but pH was comparable and none received extracorporeal carbon dioxide removal (ECCO2-R). The positive end-expiratory pressure (14.18 ± 7.56 vs. 12.31 ± 6.84 cmH2O), PIP (38.21 ± 12.89 vs. 32.59 ± 9.88), and mean airway pressures (19.98 ± 7.61 vs. 17.48 ± 6.7 cm H2O) were higher during UPMV, but Pplat and PaO2/FiO2 were comparable during- and pre-UPMV. Driving pressure was observed to be higher in those who died than who survived (24.18 ± 12.36 vs. 13.42 ± 3.25).
Conclusion
UPMV alone may be a safe alternative option for ARDS patients in centers without ECCO2-R.
Introduction
Mechanical ventilation in acute respiratory distress syndrome (ARDS) can induce ventilator-induced lung injury (VILI) from barotrauma, hyperinflation, and cyclical recruitment and de-recruitment of diseased lung. 1 Lung protective ventilation adopting low tidal volume (LVT) using 6 mL/kg of predicted body weight (PBW) while limiting plateau pressures (Pplat) to <30 cm H2O, demonstrated a 22% mortality reduction when compared to using tidal volumes 12 mL/kg PBW.2,3 However, tidal hyperinflation may still occur leading to hypothetical considerations to use tidal volume <6 mL/kg PBW.4,5 A nonrandomized trial in 2009 used tidal volumes that ranged from 3.7 to 4.6 mL/kg PBW in conjunction with extra-corporeal carbon dioxide removal (ECCO2-R) noted improved inflammatory markers and lung morphology. 6 Bein et al. 7 compared LVT to ultra-low tidal volumes <3 mL/kg PBW in combination with ECCO2-R and reported that survivors with greater hypoxemia in the ECCO2-R group had less ventilator days. A physiologic study using tidal volumes 4 mL/kg PBW and compensatory tachypnea to maintain minute ventilation (MV) revealed a reduction in cyclic atelectasis and hyperinflation. 8 In a systematic review, limited by significant technological and practice variations in the use of ECCO2-R across the few studies (2 randomized controlled trials and 12 observational studies), a mortality benefit favoring ECCO2-R was not observed. 9 We performed a chart review to examine the outcomes of ARDS patients ventilated with tidal volumes <6 mL/kg of PBW without the use of ECCO2-R (ultra-protective mechanical ventilation (UPMV)). The intent of this review is to ascertain whether tidal volume can be safely lowered in hypoxemic patients with Pplat >30 cmH2O.
Methods
At this institution, we routinely use LVT for mechanical ventilation in ARDS patients targeting Pplat <30 cmH2O per ARDS-net protocol; however, changes can occur per physician discretion. In patients with persistent Pplat >30 cmH2O, in an attempt to mitigate lung injury, we reduced the tidal volume to <4 mL/kg if oxygenation, acceptable acid base, and hemodynamic status could be maintained in the presence of hypercapnia. Our medical intensive care unit is not equipped with ECCO2-R capabilities. We selected the interval between August 2012 and October 2014 for chart review, because of the abundance of patients with ARDS during this period. 10 The University of Missouri Institutional Review Board (IRB) approved this study (IRB#1213901HS). The local instance of Informatics for Integrating Biology and Bedside (i2b2) identified patients aged 18–64 years in the electronic medical records (EMR) database, diagnosed with ARDS according to Berlin definition, who were placed on invasive mechanical ventilation for the study period. Data on 1310 patients were loaded into the Research Electronic Data Capture (REDCap) application hosted at the University of Missouri, a secure web-based application designed to support data capture for research studies, providing (1) an intuitive interface for validated data entry; (2) audit trails for tracking data manipulation and export procedures; (3) automated export procedures for seamless data downloads to common statistical packages; and (4) procedures for importing data from the external sources. 11
The following filters were then applied to the EMR to identify the patients. Inclusion: Age >18 years, ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (PaO2/FiO2) <200 with bilateral pulmonary infiltrates and no evidence of heart failure, and use of an UPMV strategy defined by tidal volume <6 mL/kg PBW. Of note, all patients had a PEEP level ≥5 cmH20. The patients in whom UPMV was used for <24 h were excluded. Demographics, Acute Physiology and Chronic Health Evaluation II (APACHE II) scores at admission and serial data of arterial blood gas, serum bicarbonate, and ventilator parameters for pre- and post-implementation of UPMV including mode, set tidal volume, peak inspiratory pressure (PIP), Pplat, etc. were gathered. Data were compiled on an excel spreadsheet and we performed descriptive statistics. Continuous variables were expressed as mean ± standard deviation (SD) or median and range as appropriate. As we did not have a control group and because of the relatively low numbers and retrospective nature of our study, we did not perform further comparative statistical analysis.
Results
Demographic and baseline characteristics at admission.
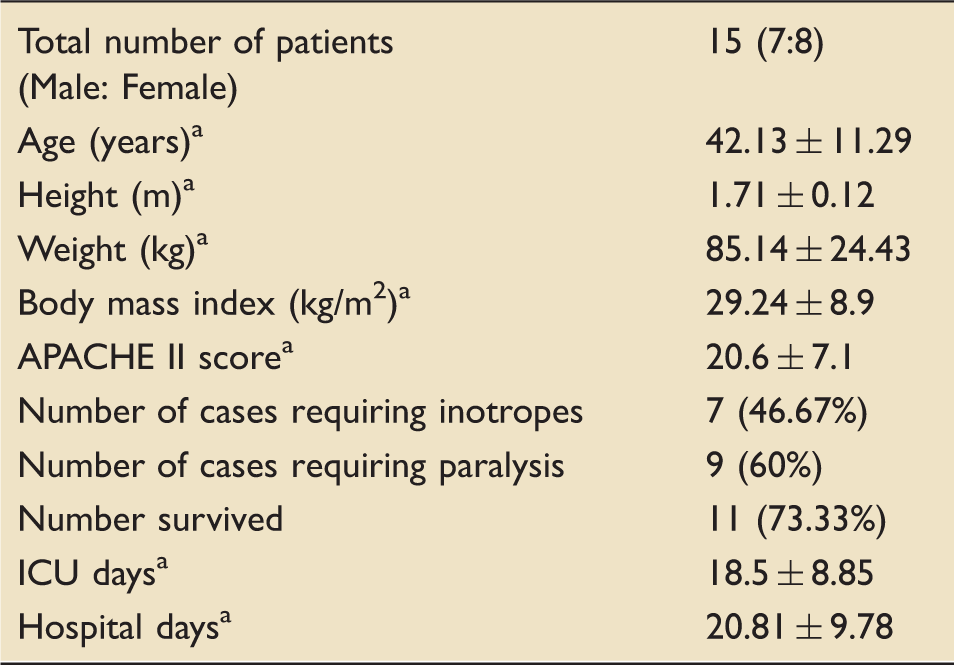
Data expressed as mean ± standard deviation (SD).
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICU: intensive care unit.
Comparison of ventilatory and laboratory parameters prior to, during, and after ultra-protective ventilation.
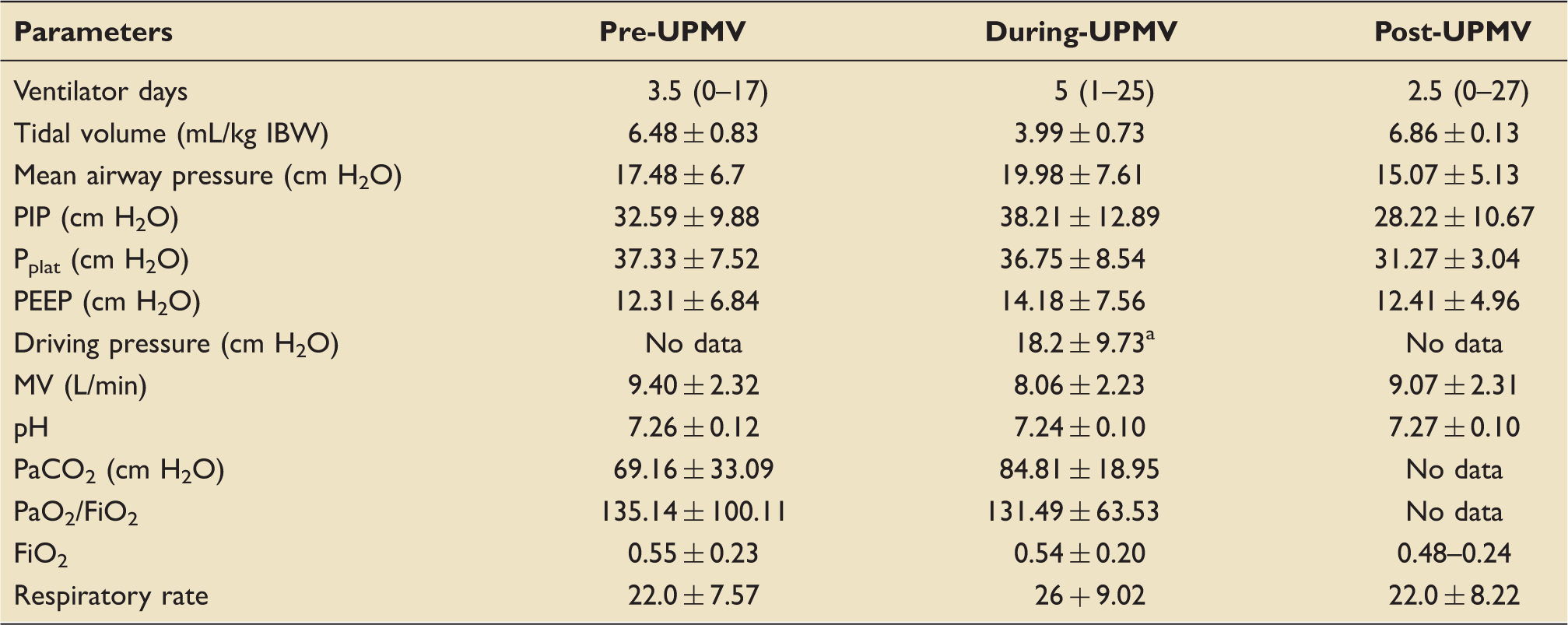
All data are expressed as mean ± SD or median (range) as appropriate.
All data from paralyzed patients. Driving pressures were lower in those who survived (N = 5, 13.42 ± 3.25) than who died (N = 4, 24.18 ± 12.36). FiO2: fraction of inspired oxygen; MV: minute ventilation; Pplat: plateau pressure; PaO2: arterial partial pressure of oxygen; PEEP: positive end-expiratory pressure; PIP: peak inspiratory pressure; UPMV: ultra-protective mechanical ventilation.
Discussion
Mechanical ventilation using LVT, higher PEEP, and lower Pplat improved the survival in ARDS patients in randomized trials, the benefit primarily attributed to less VILI from reduced tidal stretch (with lower VT) and atelectrauma (by using higher PEEP).2,3,12–14 Utilizing LVT invariably results in hypercapnia, which is often feared to worsen acidosis and cause deleterious systemic effects, especially in patients with myocardial and central nervous system affection, leading to a therapeutic dilemma. In our study, during-UPMV, the lowest tidal volume was 2 mL/kg (approaching dead space ventilation), but acidosis (pH) did not worsen despite a rise in the arterial PaCO2, mitigating the need for ECCO2-R. Although our center has limited capabilities (number of trained staff and equipment) for extra-corporeal membrane oxygenation strictly within the division of cardiothoracic surgery and mainly for cardio-pulmonary bypass surgery purposes, it is unavailable for MICU due to nonavailability of a dedicated perfusionist. All prior studies, where UPMV was employed, have used ECCO2-R to ameliorate worsening hypercapnic acidosis, but from our data, it is evident that UPMV may be carefully and safely employed in ARDS patients without the use of ECCO2-R.6,7 With a corresponding rise in the respiratory rate to maintain reasonable minute ventilation, most patients tolerated UPMV without the need for a buffer or ECCO2-R. Interestingly, there is further evidence from animal studies for direct beneficial physiological effects of hypercapnia on the immune system, and possibly lung compliance and ventilation-perfusion ratio (V/Q).15–19 This has not been confirmed in the clinical trials involving human subjects and the direct effect of hypercapnia in patients with ARDS remains unclarified. 20 Like pH, oxygenation and Pplat were indifferent in our study, which is most likely from the use of higher PEEP during-UPMV that may have mitigated atelectrauma. Current evidence for guiding physicians towards an optimal strategy for balancing hypercapnic acidosis and specific ventilator strategies remains controversial and hence ventilator strategies must be individualized to patient characteristics e.g. the presence or the absence of myocardial or central nervous system co-morbidities, severity of ARDS, observed effects of hypercapnic acidosis and its severity, etc.21–23
As mentioned, of the 11 patients who had echocardiography (at admission or within the first few days), 7 (63.64%) had evidence of either or both right ventricular volume or pressure overload. A prospective observational study involving 11 adult subjects with severe ARDS, keeping a constant Pplat (within 20–25 cm H2O), compared various ventilator strategies for an hour each as follows: low or high PEEP with high respiratory rate (compensating for low VT) or high PEEP and low respiratory rate (compensating for dead space), and evaluated cardiac function using trans-esophageal echocardiography along with alveolar dead space and recruitment. Blood pH was lower and PaCO2 higher in the groups with high PEEP, who also demonstrated right ventricular dilation, left ventricular deformation, and a decline in the cardiac index. 24 In our study, none had echocardiography performed both in the pre-UPMV and during-UPMV periods to ascertain if right ventricular function changed in response to the lower tidal volumes.
Overall mortality for this study population was 26.67% and was comparable to the ARDSnet data, even among those who received UPMV at LVT ≤ 4 mL/kg (4 deaths in 12 subjects; 33.33%). Driving pressures reflect the tidal increase in the static trans-pulmonary pressure, and have been associated with improved mortality when kept minimal (<15 cm of H2O or changes <7 cm of H2O). 25 In our cohort, driving pressures were higher in those who died than those who survived and appears to be concordant with the current level of evidence in literature, notably those who survived had lower driving pressures. We are unable to derive any further conclusions from this observation, as we suspect that driving pressure was not used by our providers for clinical management during the study period because the concept of driving pressure was still evolving and not prevalent in common practice at that time.
The feasibility and safety of ECCO2-R was assessed by Faneilli et al. 26 in a multi-center trial involving 15 patients with moderate ARDS and demonstrated that ECCO2-R is safe and efficient, but noted that 27% had worsening hypoxemia and required prone positioning. There are ongoing multi-center trials to determine if ECCO2-R will improve outcomes and provide further insight into its safety and efficacy in the treatment of patients with ARDS: pRotective vEntilation with veno-venouS lung assisT in respiratory failure (REST, ClinicalTrials.gov Identifier: NCT02654327) 27 and Strategy of UltraProtective Lung Ventilation With Extracorporeal CO2 Removal for New-Onset Moderate to severe ARDS (SUPERNOVA, ClinicalTrials.gov Identifier: NCT02282657). Although the results of these trials may shed light into the efficiency and safety of ECCO2-R to enable UPMV, our study, likely the first and largest retrospective series to date to display results on the successful use of UPMV without the use of ECCO2-R in a series of ARDS patients, demonstrates that an UPMV strategy may be adopted safely in ARDS patients without the use of ECCO2-R.
The obvious limitations to our study include the low power, retrospective design, absent controls prohibiting comparative statistics, and the paucity of documentation of specific reasons behind decisions that led to further reducing the VT to UPMV levels in every case. Prospective randomized controlled trials comparing standard care versus UPMV with and without ECCO2-R are needed to confirm our results. Given the heterogeneity of disease process in ARDS, stemming from variations in clinical and biochemical variables, future trials should consider measurement of plasma biomarkers of lung injury and inflammation, and serial radiological assessment between the subgroups. 28 This will enable identification of various ARDS phenotypes and their comparative response to the different treatment arms of such trials.
Conclusions
Our results demonstrate that UPMV may be well tolerated provided the respiratory mechanics and hemodynamic status allow for permissive hypercapnia, and ECCO2-R may not be necessary in all cases. This approach may serve as a potential alternative strategy for patients with ARDS, especially in centers where ECCO2-R is unavailable or when transfer to a qualified facility may be delayed. Such a personalized approach has also been proposed by others.22,28,29
Footnotes
Authors’ note
One of the cases in this series was presented as a poster in American Thoracic Society Conference 2014, Colorado, USA. 10
Acknowledgements
The authors thank Diane E Johnson for assisting with the use of i2b2 and RedCap for generating the patient list from EMR using the inclusion and exclusion criteria.
Authors’ contribution
Mohammed Alnijoumi, Jonathan Collins, and Troy Whitacre conceived the idea, designed this study, and obtained IRB approval. Nathanial Moulton and Daniel Woolery collected most of the data, performed preliminary descriptive analysis, and first draft of results and discussion. Hariharan Regunath then reviewed the protocol and preliminary data, guided Nathanial Moulton and Daniel Woolery to collect and compile further additional data and descriptive statistics. Hariharan Regunath then revised and compiled the results, created tables with data, and restructured the entire paper. Troy Whitacre, Jonathan Collins, and Alnijoumi reviewed this final paper and added further edits. Hariharan Regunath and Mohammed Alnijoumi will be the guarantors of this paper taking responsibility for the integrity of the work as a whole, from inception to publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
