Abstract
Objective
We evaluated pressure-controlled ventilation (PCV) with multiple programmed levels of positive end expiratory pressure (programmed multi-level ventilation; PMLV) in patients with coronavirus disease 2019 (COVID-19)-related acute respiratory distress syndrome (ARDS).
Methods
We conducted a multicenter, retrospective study from November 2020 to February 2021. PMLV was used with PCV in all patients with intensive care admission until improvement in oxygenation (fraction of inspired oxygen [FiO2] ≤0.50 and oxygen saturation [SpO2] >92%). The observed outcomes were improvement of hypoxemia, length of mechanical ventilation, partial pressure of carbon dioxide (PaCO2) stability, and adverse events.
Results
Of 188 mechanically ventilated patients with COVID-19-related ARDS, we analyzed 60 patients treated with PMLV. Hypoxemia improved in 55 (92%) patients, as measured by the change in partial pressure of oxygen/FiO2 and SpO2/FiO2 ratios on day 3 versus day 1, and in 32 (66%) ventilated patients on day 7 versus day 3. The median (interquartile range) length of mechanical ventilation for survivors and non-survivors was 8.4 (4.7–14.9) and 6.7 (3.6–10.3) days, respectively.
Conclusions
PMLV appears to be a safe and effective ventilation strategy for improving hypoxemia in patients with COVID-19-related ARDS. Further studies are needed comparing the PMLV mode with the conventional ARDS ventilatory approach.
Keywords
Introduction
Since its emergence in late 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the cause of coronavirus disease 2019 (COVID-19), has led to a global pandemic that has placed a serious burden on health care systems worldwide. 1 The main cause of death among patients with COVID-19 infection is severe hypoxemic respiratory failure owing to acute respiratory distress syndrome (ARDS). Patients with ARDS have a high risk of mortality, and survivors experience substantial morbidity. 2 Despite emerging data regarding the pathogenesis and treatment of COVID-19, few studies have described detailed methods of mechanical ventilation in these patients.
One of the most debated topics in mechanical ventilation of patients with ARDS involves the selection of positive end expiratory pressure (PEEP). 3 Individual studies on higher versus lower PEEP have failed to show a decrease in mortality for these patients.4 –6 However, meta-analysis of the ALVEOLI, LOVS, and EXPRESS studies suggests a survival benefit with high PEEP in the subgroup with severe ARDS. 7 The problem with higher PEEP is that it contributes to ventilator-induced lung injury, especially in non-recruitable areas of the lungs. Rather than searching for the single best PEEP, which is only a part of the whole in terms of mechanical power delivered to the lungs, 8 we recently proposed that research is needed on multiple alternating PEEP levels. 9 The three potential benefits that we have pointed out are that ventilation with multiple PEEP levels might lead to better aeration and a decrease in ventilation inhomogeneities; that higher PEEP is not sustained and therefore, its negative consequences might not be very pronounced; and that this approach might lead to improved recruitment of the lungs. 9
Programmed multi-level ventilation (PMLV) is based on multiple levels of PEEP and optimization of expiratory time constants. Although this ventilation strategy has been used for non-homogeneous lung ventilation prior to the COVID-19 era,10 –12 the potential utility and benefits of the PMLV mode in patients with ARDS are largely unknown. Only one case report exists on PMLV used for mechanical ventilation in COVID-19-related ARDS, with no other related studies published to date.
Therefore, in this retrospective observational study, we evaluated the use of PMLV in critically ill patients with COVID-19-related ARDS as a pilot project to aid in the design of future studies on PMLV. We hypothesized that using multiple PEEP levels in non-homogeneous COVID-19-related ARDS might lead to improvement in oxygenation without serious adverse events.
Methods
Study design and participants
This retrospective, observational multicenter study involved data from eight intensive care units (ICUs) in Slovakia between November 2020 and February 2021. The study was first approved by the lead institutional ethics committee (East Slovak Institute for Cardiovascular Diseases, Ref: IEC No. EK - 02/2021, Chairperson: Stanislav Juhas, MD, PhD), followed by ethical approval received in each of the participating hospitals. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
To be included in the study, patients admitted to the ICU with COVID-19 pneumonia must have had a positive polymerase chain reaction result for SARS-CoV-2 via nasopharyngeal swab. All included patients were invasively mechanically ventilated and fulfilled the diagnostic criteria of moderate or severe ARDS (partial pressure of oxygen [PaO2] <200 mmHg with a PEEP ≥5 cmH2O), according to the Berlin definition 14 and had failed on either non-invasive ventilation or high-flow nasal cannula prior to intubation. Further inclusion criteria for study enrollment were age 18–80 years and PMLV mode used from admission until stabilization of oxygenation (defined as FiO2 <0.50 and oxygen saturation [SpO2] >92%). Patients were excluded from the study if they required cardiopulmonary resuscitation, had severe hemodynamic instability (cardiogenic or hemorrhagic shock) prior to admission, died within 24 hours of ICU admission, decided to have treatment withdrawn, or were lost to follow-up after transfer to another hospital.
Mechanical ventilation strategy
Mechanical ventilation was provided using pressure-controlled ventilation (PCV) or pressure-supported (PSV) along with PMLV. Aura V ventilators (Chirana Medical, Stará Turá, Slovakia) were used (Figure 1) for all patients from admission until stabilization (FiO2 <0.50 and SpO2 >92%). Then, PSV without the PMLV feature was used until liberation from mechanical ventilation.

Difference between PCV using one level of PEEP (top) and PCV with activated PMLV feature (bottom). Two or three levels of higher PEEP (PEEPh and/or PEEPh2) can be used with PCV when PMLV is used.
PMLV is not a ventilation mode per se but rather a feature that adjusts and modifies any ventilation mode used (e.g., PCV, PSV, volume-controlled ventilation, synchronized intermittent mandatory ventilation). 15 PMLV is based on the measured expiratory time constant (TauE) of the whole respiratory system. First, knowing the time for which 63% of the expiratory tidal volume is exhaled (TauE) will help to determine the optimal ventilation frequency (the longer the TauE, the lower the total frequency and vice versa) to avoid intrinsic PEEP. Second, cyclic changes in two or three PEEP levels (baseline PEEP and higher levels of PEEP—PEEPh and/or PEEPh2) may provide recruitment in lung areas with longer time constants. 12
For this study, the PMLV setting (i.e., recommended respiratory rate and combined alternating PEEP levels) was mathematically predicted using the ventilator software. 15 From among the options given by the software, treating clinicians then decided whether two or three levels of PEEP should be used. On the basis of standard institutional clinical care and the implemented local ICU protocol, baseline PEEP was selected according to the predicted body weight (1 cmH2O for each 10 kg of predicted body weight, with minimum 5 cmH2O and maximum 10 cmH2O), and this did not change during the observation period. According to the measured TauE, the optimal frequency was automatically selected, and based on measured respiratory system compliance, the ventilator added the pressure at higher level(s) of PEEP (PEEPh and/or PEEPh2). The possible ratios as well as pressures of PEEP, PEEPh, and PEEPh2 are shown in Table 1. All patients were initially placed in the prone position while on PMLV; later in the disease course, each patient’s position was at the discretion of the treating team.
Possible automatic PEEP selection combinations used in the study based on measured compliance of the respiratory system. Ventilator algorithm software automatically selected the PEEPh/PEEPh2 pressure, respecting pressure limits.

*Respiratory system compliance used to recommend PEEP levels.
PCV, pressure-controlled ventilation; PEEP, positive end expiratory pressure; PMLV, programmed multi-level ventilation; fPEEP ratio, ratio of alternating PEEP levels; h, higher PEEP levels.
A minute volume controller (automatic proportional minute volume, APMV) on the Aura V ventilator was used to provide constant minute ventilation. The APMV increases or decreases inspiratory pressure during PCV by ±50% in increments of 1 to 2 cmH2O of preset pressure to maintain the targeted minute volume set by the clinician. APMV is used to maintain minute volume with PMLV because different tidal volumes are obtained as peak inspiratory pressure is delivered at different PEEP levels.
Data collection
Data collected included demographics (age, sex, weight, height), comorbidities, arterial pH, arterial PaO2, arterial SpO2 measured using pulse oximetry, and arterial partial pressure of carbon dioxide (PaCO2) at admission and each day until liberation from invasive mechanical ventilation (successful weaning or death). PaCO2 stability, vasopressor requirement, need for continuous renal replacement therapy, and complications from mechanical ventilation were also recorded. Active PMLV was recorded daily until its discontinuation.
Outcomes
Our primary outcome was improvement of hypoxemia. Secondary outcomes were total time spent on the ventilator, hemodynamic stability, PaCO2 stability during invasive mechanical ventilation, and complications.
Ethical approval and consent to participate
This study was approved by the ethics committee of each participating hospital in accordance with the necessary regulations. Given the retrospective nature of the study, informed consent was waived.
Statistical analysis
Categorical data are expressed as number (percentage), and continuous data are expressed as mean ± standard deviation (SD) for normally distributed data or median with interquartile range (IQR) for non-normally distributed data. Paired t-tests and repeated measures one-way ANOVA (with Tukey and Dunnett correction for multiple comparisons) were used to compare the changes in pH, PaO2, PaCO2, SpO2, FiO2, PaO2/FiO2 (P/F ratio), and SpO2/FiO2 (S/F ratio) over time. Up to three between-day comparisons using ANOVA were considered rationale for adjustment, as described above. The means of arterial blood gas analytes are reported with 95% confidence intervals (CIs). Survivors and non-survivors were compared using the chi-square or Fisher’s exact test. We set a p-value <0.05 as the level of significance. Analyses were performed using IBM SPSS v. 25 (IBM Corp., Armonk, NY, USA) and GraphPad Prism v. 9 (GraphPad Software, San Diego, CA, USA).
Results
A total of 188 patients were admitted to the eight ICUs in Slovakia; of these, 60 (32%) were included in the analysis (Figure 2). The mean participant age was 58.5 ± 10.5 years, and 73% were men; mean BMI was 31.3 ± 6.2 kg/m2. The mean ± SD of baseline PEEP applied for patients in this study was 8 ± 2 cmH2O; higher-level PEEPs are presented in Table 1. Thirty (50%) patients presented with moderate ARDS, and 30 (50%) presented with severe ARDS. Baseline characteristics of survivors and non-survivors are summarized in Table 2. The survivors were significantly younger than non-survivors (p = 0.003), and fewer patients had asthma (p = 0.03) as a comorbidity among survivors. All other parameters were not significantly different between groups. The median (IQR) time spent on the ventilator was 8.4 (4.7–14.9) days for surviving patients. Figure 3 depicts the treatment outcomes on days 3, 7, 14, and 28 (i.e., mortality, ongoing mechanical ventilation, and discharge from the ICU).
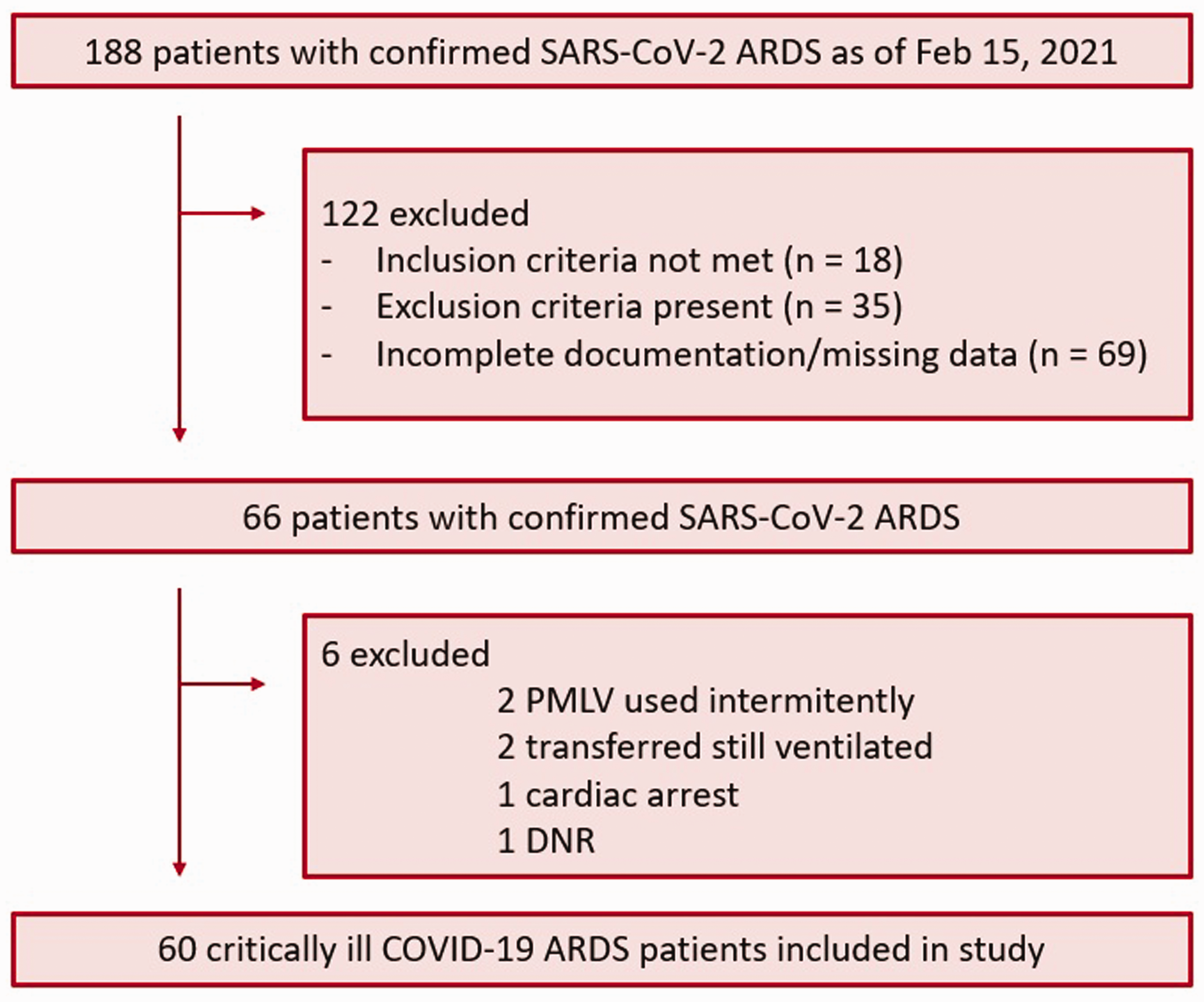
Study flow diagram.
Demographic and baseline characteristics of surviving and non-surviving patients.
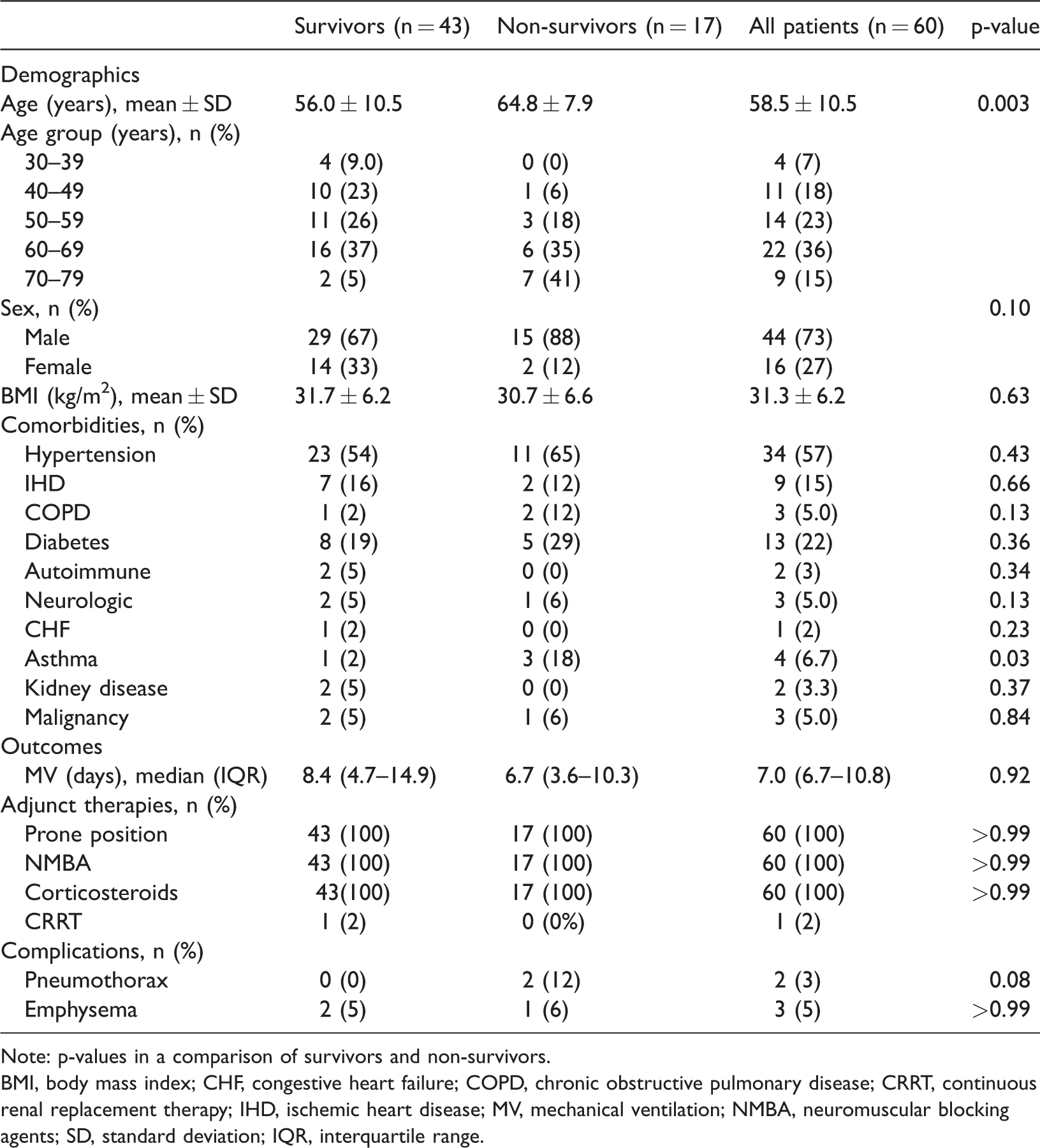
Note: p-values in a comparison of survivors and non-survivors.
BMI, body mass index; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; CRRT, continuous renal replacement therapy; IHD, ischemic heart disease; MV, mechanical ventilation; NMBA, neuromuscular blocking agents; SD, standard deviation; IQR, interquartile range.

Flowchart representing the clinical course of patients on days 1, 3, 7, 14, and 28.
Overall, hypoxemia was improved in 55 (92%) patients, as measured by the change in PaO2/FiO2 and SpO2/FiO2 ratios on day 3 (p < 0.001 for both), and in 32 (66%) ventilated patients on day 7 (p < 0.001 for both) in comparison with baseline and day 3, respectively. Compared with baseline, the values for pH, PaO2, SpO2, P/F ratio, and S/F ratio (p < 0.001, for all) were all increased at day 3 (Table 3). There was no difference for PaCO2. We then compared the results of arterial blood gas analyses only for the patients still on mechanical ventilation on day 7 (n = 44) (Table 4) and on day 14 (n = 17) (Table 5). Compared with day 3, patients on day 7 had increased PaO2, P/F ratio, and S/F and decreased FiO2. No differences were noted for pH, PaCO2, or SpO2 (Table 4). Even in patients who were still on invasive mechanical ventilation on day 14, improvement in hypoxemia was observed while on PMLV (Table 5).
Arterial blood gas analysis of 59 patients still on mechanical ventilation at day 3.
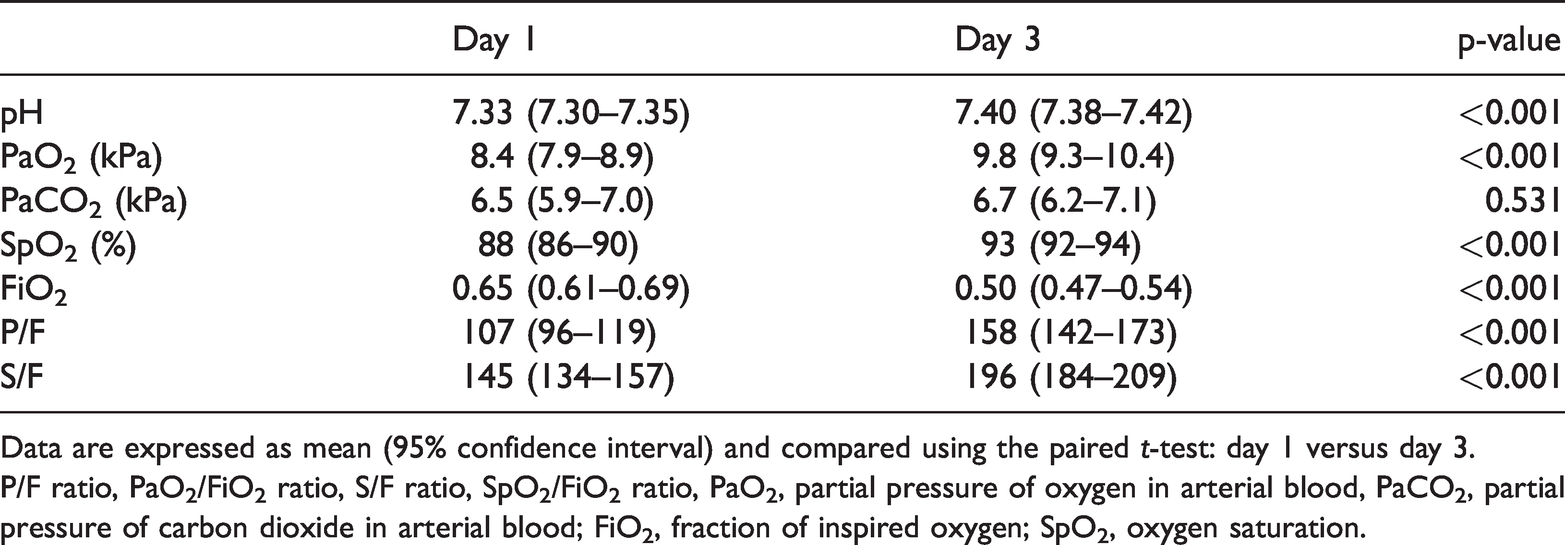
Data are expressed as mean (95% confidence interval) and compared using the paired t-test: day 1 versus day 3.
P/F ratio, PaO2/FiO2 ratio, S/F ratio, SpO2/FiO2 ratio, PaO2, partial pressure of oxygen in arterial blood, PaCO2, partial pressure of carbon dioxide in arterial blood; FiO2, fraction of inspired oxygen; SpO2, oxygen saturation.
Arterial blood gas analysis of the 44 patients still on mechanical ventilation on day 7.

Data are expressed as mean (95% confidence interval) and compared with repeated measures one-way analysis of variance (with Tukey correction for multiple comparisons): day 1 versus day 3, day 3 versus day 7, and day 1 versus day 7.
D1, day 1; D3, day 3; D7, day 7; P/F ratio, PaO2/FiO2 ratio; S/F ratio, SpO2/FiO2 ratio; PaO2, partial pressure of oxygen in arterial blood; PaCO2, partial pressure of carbon dioxide in arterial blood; FiO2, fraction of inspired oxygen; SpO2, oxygen saturation.
Arterial blood gas analysis in the 17 patients still on mechanical ventilation at day 14.

Data are expressed as mean (95% confidence interval) and compared with repeated measures one-way analysis of variance (with Dunnett correction for multiple comparisons): day 1 versus day 3, day 1 versus day 7, day 1 versus day 14.
D1, day 1; D3, day 3; D7, day 7; D14, day 14; P/F ratio, PaO2/FiO2 ratio; S/F ratio, SpO2/FiO2 ratio; PaO2, partial pressure of oxygen in arterial blood; PaCO2, partial pressure of carbon dioxide in arterial blood; FiO2, fraction of inspired oxygen; SpO2, oxygen saturation.
Thirty-three patients (55%) required norepinephrine infusion for some period while sedated and receiving mechanical ventilation with a median dose (IQR) of 0.15 µg/kg/minute (0.1–0.23). PaCO2 values did not show a significant deviation from baseline (median [IQR]: Day 1: 46.5 mmHg [38.2–51], Day 2: 48 mmHg [40.5–57], and Day 3: 46.5 mmHg [39–55.5]. This was expected as the minute ventilation was kept constant with the automatic minute volume controlled ventilation mode.
Discussion
The main finding of our study was that multiple levels of PEEP delivered via PMLV ventilatory mode could successfully improve oxygenation in patients with COVID-19-related ARDS, with no notable complications.
Marked improvement in oxygenation was observed between day 1 and day 3, and between day 3 and day 7, as measured with PaO2, FiO2. SpO2, and the P/F and S/F ratios. It is difficult to directly compare this improvement in oxygenation with other reports because many studies have not addressed the time of P/F measurement. However, in two recent reports, the P/F ratio in patients with COVID-19-related ARDS in the first 48 hours was reported to be 113 mmHg 16 and 107 mmHg, 17 which are both very similar to the P/F ratio on day 1 in our study (107 mmHg).
The median length of mechanical ventilation for survivors was 8.4 days in our cohort. In their systematic review and metanalysis, Chang et al. reported a similar duration of mechanical ventilation (i.e., between 7.7 and 10.1 days). 18 However, those authors included very early experiences with COVID-19-related ARDS and only included studies up to 1 May 2020. In comparison, several other studies from the same period or later have reported longer lengths of ICU stay, ranging from 10 to 15 days.19 –22 Whether these differences arise owing to the PMLV mode used in our study is an important area of further research. Any mode of ventilation that is closer to the physiological needs of the patient has the potential to reduce the length of stay in the ICU and would thus positively affect resource distribution, especially during periods of immense pressure on ICUs.
The 28-day mortality was 28% (n = 17) in our study. Survivors were significantly younger than non-survivors whereas all other parameters were similar, including the rate of complications. However, the incidence of pneumothorax among non-survivors might be significant with a larger sample size; we noted a p-value of 0.08. Mortality in our cohort (28%) coincided with the wide range of mortality reported in a systematic review and meta-analysis of 37,359 patients. 22 Although mortality in our cohort was relatively lower than that in other studies,23 –27 this finding cannot be generalized owing to the small number of patients and because of the collapsed state of the health care system when the COVID epidemic was at its peak in Slovakia. As a result, many patients were only stabilized using PMLV until such time as they could be transferred (while still on the ventilator) to other peripheral hospitals where weaning was commenced. Therefore, the three key factors influencing mortality are criteria for ICU admission (i.e., severity of illness, need for vasopressors, and oxygen requirements), criteria for intubation, and staffing standards in the ICU during a pandemic crisis. These factors should be considered in the design of future studies.
There is good evidence that patients with COVID-19-related ARDS on mechanical ventilation have heterogeneous respiratory mechanics at admission and during the first 24 hours after admission.28,29 There is ongoing debate regarding whether COVID-19 presents as typical or atypical ARDS, 30 with more recent studies suggesting that COVID-19 ARDS shows features similar to non-COVID ARDS. 31 Some reports have described increased static compliance in patients with COVID-19-related ARDS, although other multicenter studies have shown static compliance comparable to that of ARDS with a non-COVID etiology. 32 As supported by postmortem findings, the lungs in COVID-19-related ARDS show non-homogenous ventilation manifesting as diffuse alveolar damage. Moreover, perfusion is deranged and is associated with a high frequency of arterial thrombi.33,34 Nevertheless, both ventilation and perfusion show signs of a high degree of non-homogeneity.
There are three main physiological explanations why alternating multiple levels of PEEP might be beneficial in ventilating non-homogenous lungs. First, depending on the recruitable portion of the lungs in ARDS, a protective tidal volume is delivered at different PEEP levels, thereby ventilating a larger portion of the recruitable zone in ARDS-affected lungs. Second, high PEEP levels are not sustained; therefore, the negative consequences of high PEEP (i.e., dynamic hyperinflation and the impact on hemodynamics) may not be very pronounced. 9 Third, it has been shown in a mechanical five-compartmental model that part of the tidal volume is redistributed from a shorter to longer time constant compartment using multiple PEEP levels. 35 These physiological hypotheses, in combination with our clinical data showing improved oxygenation, provide solid grounds for further research on PMLV in patients with ARDS.
This study has several limitations. As a retrospective observational study, we were limited to the data recorded in each patient’s medical records. Thus, owing to the limited resources during the pandemic, we were unable to collect detailed data on illness severity and lung mechanics on each study day. The main limitation is the inability to obtain a comparable control group of patients being ventilated using conventional ventilation strategies. Another limiting factor in research on PMLV is that very few mechanical ventilators are equipped with the PMLV feature. Thus, further prospective studies with a control group are warranted to better assess the utility of PMLV (either prospective observational studies, or preferably, randomized controlled trials). These should include measurement of right ventricular function to better describe lung–heart interactions. Additionally, the small sample size limited our ability to perform multivariable analysis.
Using alternating PEEP levels in clinical practice presents some challenges. Different lung mechanics (most notably compliance) are present at each PEEP level; thus, the evaluation of pulmonary mechanics becomes problematic. This is owing to the different pressures applied during each PEEP; hence, the delivered tidal volumes differ at each PEEP level. Therefore, a minute volume controller is necessary to control minute ventilation via an automatic increase or decrease in the pressure delivered. At present, PEEP levels are mainly determined by measuring compliance during simple PCV. This could be an advantage but also a limitation in protective mechanical ventilation. The potential of TauE in advanced pulmonary diagnostics and PEEP titration should be also investigated. TauE is only slowly gaining interest in anesthesiology and intensive care and is thus an exciting area of further research.36,37
The outcomes of future studies comparing PMLV with conventional methods of ventilation should focus on the duration of (invasive) mechanical ventilation, lung compliance, right ventricular function, length of ICU and hospital stay, 28-day and 90-day mortality, and functional status at hospital discharge. The criteria for ICU admission, criteria for intubation, and staffing standards in the ICU should be reported. Also, studies focusing on lung recruitment visualization using electric impedance tomography with possible sequential computed tomography imaging would be of interest.
Conclusions
PMLV used to support patients with COVID-19-related ARDS led to improved oxygenation in this retrospective cohort study. This strategy appears to be safe and effective, as measured using gas exchange and complications. Additional studies are warranted to assess PMLV in a variety of patients with ARDS of different etiologies in comparison with conventional modes of mechanical ventilation.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221101970 - Supplemental material for Programmed multi-level ventilation in COVID-19-related acute respiratory distress syndrome: a multi-center retrospective observational study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221101970 for Programmed multi-level ventilation in COVID-19-related acute respiratory distress syndrome: a multi-center retrospective observational study by Filip Depta, Pavol Török, Andrew G. Miller, Peter Firment, Jozef Leškanič, Adam Porubän, Pavol Halaš, Stanislav Mandinec, Vladimír Filka, Henryk Zajac, Michael A. Gentile and Marko Zdravkovic: the DBDS Genomic Consortium in Journal of International Medical Research
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author contributions
All authors have participated in the concept of the study and critically revised the manuscript. All authors have approved the final version of the manuscript and agree with its publication.
Declaration of conflicting interests
FD and MG have been consultants at IPM Chirana Inc. All other authors report no competing interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
