Abstract
In a five-step procedure, 18 new esters (

Introduction
Oxadiazole is a general name referring to a five-membered ring that contains an oxygen atom and two nitrogen atoms.
1
The position of heteroatoms on the ring results in the specific name of isomers starting from oxygen, including 1,3,4-, 1,2,6-, and 1,2,4-oxadiazole (

The structures of 1,2,4-oxadiazole
In pharmaceutical chemistry and drug development, esters are often used as prodrugs with the purpose of enhancing some properties of parent drugs, such as solubility, oral adsorption, membrane permeability, duration of action, and reducing side effects.10–14 Some typical examples given approve the structural diversity of esters in commercially available medicines in the world, including aspirin (
A new era of selective chemotherapeutic hits has emerged with the advent of targeted therapies, specifically the signaling-pathways-targeting chemotherapeutic agents. These agents can target cancer cells or the tumor microenvironment that promotes tumor growth, leading to increased efficacy and minimal side effects on healthy cells. One of the most principal signaling pathways was the tyrosine kinase (TK) epidermal growth factor receptor (EGFR) which belongs to the TK receptor family and is present in the epithelial cell membrane. 21 Its overexpression has been proven to play an important role in the proliferation of tumor cells, invasiveness, and metastasis in various cancer types, including breast, colon, and ovarian subtypes. 22 Conformational alterations brought on by the endogenous EGFR ligand’s binding contacts with the EGFR extracellular binding domain activate the EGFR. Consequently, a number of tyrosine residues undergo autophosphorylation, which triggers the mitogen-activated protein kinase (MAPK) pathway by phosphorylating the subsequent proteins in the route. Profound mutations in several MAPK pathway proteins, including EGFR, have been connected to a number of cancer forms, including non–small cell lung cancer, colorectal cancer, hepatocellular carcinoma, pancreatic cancer, and breast cancer. This clarifies the reason why searching for new EGFR inhibitors is one of the most effective pathways for cancer therapy. 23
Recently, computer-aided drug design (CADD) has been widely used to decrease both time and cost for discovering potent compounds that are able to inhibit the biological function of a specific enzyme. 24 In the CADD, the binding-free energies (ΔG) can be computed over atomistic simulations. This metric discloses the binding affinity between ligand and protein through docking simulation, and their potential binding conformations were investigated. In this work, the physical insights into the EGFR protein target and synthesized ester binding were clarified using AutoDock4. The achieved results are significant contributions toward seeking new candidates for targeted cancer therapy and providing valuable guidance for further drug development studies.
Results and discussion
The esters

The synthesis of esters
The structures of esters
Biology
The synthesized esters
Cytotoxicity of the prepared esters

DHA: dihydroartemisinin.
Data represent the mean ± standard deviation of three independent wells.
Ellipticine was used as a positive control.
(–): No activity.
The IC50 in Table 1 showed that most esters
Among the esters in the two series, esters
Docking studies
Re-docking validation
EGFR is critical for controlling the growth and survival of epithelial cells; thus, EGFR inhibitors have been used for the treatment of various cancer patients, for example, gefitinib for non–small lung cancer, erlotinib for pancreatic cancer, lapatinib and neratinib for breast cancer, and so on. However, it is reported that patients treated with these drugs might respond to treatment initially, but most of them will develop resistance within a year. 30 Due to this burden, there is still an urgent need for scientists to find novel compounds with significant inhibition activities. Recent studies have shown that tyrosine kinase inhibitors (TKIs) associated with mutations in EGFR are used to treat initial lung cancer. In this context, molecular docking simulation was conducted to clarify the synthesized esters against the inactive EGFR TK domain (PDB ID: 4HJO). It is reported by Gohlke et al. 31 that ligand-calculated partial charge with the B3LYP method has been shown to greatly increase docking accuracy and cluster population of the most accurate docking.
The co-crystallized ligand, erlotinib, was re-docked to the binding site of the targeted enzyme to validate the accuracy of the docking procedure. Molecular docking is a useful tool to quickly find, for each protein interacting with the ligand, the optimal value of the score function. The objective of any docking calculation is to find the best pose, which corresponds to the lowest energy. 32 The docking result is considered reliable when the root-mean-square deviation (RMSD) value does not exceed 2.0 Å. In this study, the docked pose of erlotinib with the lowest binding-free energy was compared with its native structure based on its RMSD value, which was 0.655 Å (Figure 2). This result suggests that the procedure and the setting parameters were reproducible and appropriate for further docking simulation.

Dock pose of the studied erlotinib (green) and native erlotinib (magenta) in the active site of EGFR protein (PDB ID: 4HJO).
Docking results
We further investigated the EGFR inhibitory effect of the studied compounds by revealing how they interact with the active site of this protein through molecular docking studies. AutoDock4 is one of the most common docking tools that has been widely used with more than 6400 citations. 33 According to the ranking criteria of AutoDock, the more negative docking energy suggests a higher binding affinity of the compound toward the targeted receptor. 34 The binding-free energies, interaction type, and residues participating in interaction are tabulated in Table 2.
Binding-free energy and residue interactions of the studied compounds against EGFR protein.
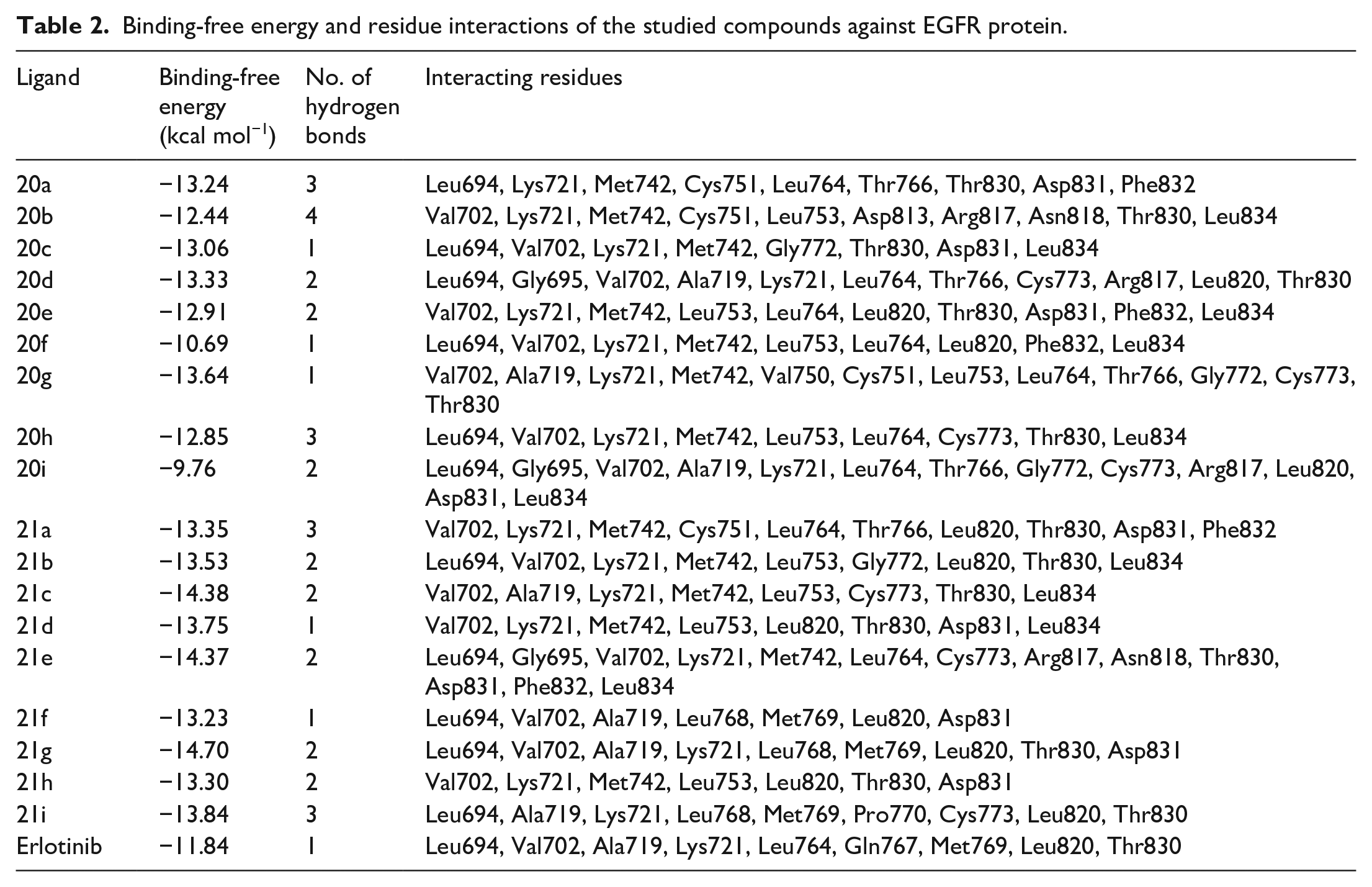
In this study, the obtained dock score of the reference compound erlotinib was −11.84 kcal mol−1; thus, any ligands with docking energy close to this threshold would be assumed to exhibit high binding affinity toward the targeted protein. The docking results showed that most of the synthesized esters exhibit higher binding affinity toward EGFR than the reference compound, with binding-free energies varying within the range of −12.44 to −14.70 kcal mol−1, except for compounds
In Figure 3 and Supplemental Material 2, the docking pose of the synthesized esters within the active site of the protein under study was shown. It has been reported in previous studies that the active site of the targeted receptor was mainly composed of residues Phe699, Gly700, Lys721, Met769, Arg817, Thr830, and Asp831.35,36

Docking conformation of studied compounds in the binding site of the inactive EGFR TK domain suggested by molecular docking studies.
The hydrophobic pockets formed with compound
Compound
Conclusion
A five-step procedure was applied for the synthesis of esters
Experimental
Chemistry
DHA and zerumbone were prepared as described in our previous study by Tran et al. 16 Other chemicals were purchased from Sigma Aldrich (Singapore) and used without further purification. 1H NMR and 13C NMR spectra were recorded at ambient temperature on a Bruker Avance 600 MHz spectrometer (BioSpin, Germany) in DMSO-d6. Chemical shifts δ are quoted in parts per million (ppm) referenced to the residual solvent peak (DMSO at 2.50, 3.32, and 39.5 ppm) relative to TMS. Mass spectra were recorded using an Agilent LC/MSD Trap SL. Thin-layer chromatography was performed on precoated Silica Gel 60 F254 aluminum sheets (Merck, Germany), and products were visualized under a UV lamp at 254 nm. Column chromatography was carried out on silica gel (40–230 mesh).
The esters
The 2-(bromo)ethyl ether of DHA
General procedure for the hydrolysis of esters 14a–i
A suspension of each ester
3-(5-(3,4,5-trimethoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoic acid 15i
Yield 84%. 1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.63 (t, J = 1.5 Hz, 1H, H-2′), 8.33 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.17 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.74 (t, J = 7.8 Hz, 1H, H-5′), and 7.47 (s, 2H, H-3′′′, H-5′′′).
General procedure for the synthesis of esters 20a–i and 21a–i
Compounds
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-methyl-1,2,4-oxadiazol-3-yl)benzoate (20a)
Yield 52%, colorless oil; Rf = 0.41 (n-hexane/acetone 2.5:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.61 (t, J = 1.5 Hz, 1H, H-2′), 8.31 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.20 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.78 (t, J = 7.8 Hz, 1H, H-5′), 5.27 (s, 1H, H-12), 4.82 (d, J = 3.0 Hz, 1H, H-10), 4.69 (m, 1H, 10-O-CH2a-), 4.48 (m, 1H, 10–O-CH2b-), 4.11 (m, 1H, 10-O-CH2-CH2a-), 3.75 (m, 1H, 10-O-CH2-CH2b-), 2.74 (s, 3H, 5′′-CH3), 2.43 (m, 1H, H-9), 2.14 (m, 1H, H-41), 1.95 (m, 1H, H-42), 1.67 (m, 2H, H-81, H-51), 1.58 (m, 1H, H-82), 1.33 (m, 1H, H-8a), 1.30 (s, 3H, H-15), 1.24 (m, 1H, H-71), 1.14 (m, 1H, H-52), 1.06 (m, 1H, H-5a), 0.87 (d, J = 7.2 Hz, 3H, H-13), 0.76 (m, 2H, H-6, H-72), 0.65 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 177.8 (C-5′′), 166.9 (C-3′′), 164.8 (1′-COO-), 131.8 (C-6′), 131.4 (C-4′), 130.6 (C-1′), 129.9 (C-5′), 127.4 (C-2′), 126.9 (C-3′), 103.2 (C-3), 100.6 (C-10), 86.8 (C-12), 80.3 (C-12a), 65.0 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-isopropyl-1,2,4-oxadiazol-3-yl)benzoate (20b)
Yield 53%, colorless oil; Rf = 0.41 (n-hexane/acetone 2.5:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.57 (t, J = 1.5 Hz, 1H, H-2′), 8.28 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.16 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.74 (t, J = 7.8 Hz, 1H, H-5′), 5.23 (s, 1H, H-12), 4.78 (d, J = 3.0 Hz, 1H, H-10), 4.66 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.06 (m, 1H, 10-O-CH2-CH2a-), 3.69 (m, 1H, 10-O-CH2-CH2b-), 3.39 (m, 1H, H-1′′′), 2.38 (m, 1H, H-9), 2.12 (m, 1H, H-41), 1.90 (m, 1H, H-42), 1.63 (m, 2H, H-81, H-51), 1.54 (m, 1H, H-82), 1.40 (d, J = 7.2 Hz, 6H, H-2′′′, H-3′′′), 1.29 (m, 1H, H-8a), 1.26 (s, 3H, H-15), 1.18 (m, 1H, H-71), 1.09 (m, 1H, H-52), 1.01 (m, 1H, H-5a), 0.83 (d, J = 7.2 Hz, 3H, H-13), 0.77 (m, 1H, H-6), 0.70 (m, 1H, H-72), 0.60 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 184.5 (C-5′′), 166.7 (C-3′′), 164.8 (1′-COO-), 131.8 (C-6′), 131.4 (C-4′), 130.6 (C-1′), 129.9 (C-5′), 127.4 (C-2′), 127.0 (C-3′), 103.2 (C-3), 100.6 (C-10), 86.8 (C-12), 80.3 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-phenyl-1,2,4-oxadiazol-3-yl)benzoate (20c)
Yield 63%, white powder, m.p. 75 °C–77 °C; Rf = 0.43 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.66 (t, J = 1.5 Hz, 1H, H-2′), 8.37 (dt, J1 = 1.5 Hz, J2 = 7.5 Hz, 1H, H-4′), 8.21 (m, 3H, H-6′, H-2′′′, H-6′′′), 7.77 (m, 2H, H-5′, H-4′′′), 7.69 (m, 2H, H-3′′′, H-5′′′), 5.26 (s, 1H, H-12), 4.79 (d, J = 3.0 Hz, 1H, H-10), 4.68 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.09 (m, 1H, 10-O-CH2-CH2a-), 3.71 (m, 1H, 10-O-CH2-CH2b-), 2.39 (m, 1H, H-9), 2.08 (m, 1H, H-41), 1.80 (m, 1H, H-42), 1.57 (m, 3H, H-81, H-51, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.16 (m, 1H, H-71), 1.08 (m, 1H, H-52), 0.98 (m, 1H, H-5a), 0.83 (d, J = 7.2 Hz, 3H, H-13), 0.78 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.57 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.7 (C-5′′), 167.6 (C-3′′), 164.8 (1′-COO-), 133.5 (C-4′′′), 132.0 (C-6′), 131.5 (C-4′), 130.6 (C-1′), 129.9 (C-5′), 129.6 (C-3′′′, C-5′′′), 127.9 (C-2′′′, C-6′′′), 127.5 (C-2′), 126.7 (C-3′), 123.1 (C-1′′′), 103.2 (C-3), 100.5 (C-10), 86.8 (C-12), 80.2 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl)benzoate (20d)
Yield 45%, white powder, m.p. 75 °C–77 °C; Rf = 0.41 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.65 (t, J = 1.5 Hz, 1H, H-2′), 8.37 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.25 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′′′), 8.20 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.82 (m, 1H, H-4′′′), 7.78 (t, J = 7.8 Hz, 1H, H-5′), 7.56 (ddd, J1 = 1.5 Hz, J2 = 2.4 Hz, J3 = 7.8 Hz, 1H, H-3′′′), 7.50 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-5′′′), 5.25 (s, 1H, H-12), 4.78 (d, J = 3.0 Hz, 1H, H-10), 4.66 (m, 1H, 10-O-CH2a-), 4.44 (10-O-CH2b-), 4.07 (m, 1H, 10-O-CH2-CH2a-), 3.69 (m, 1H, 10-O-CH2-CH2b-), 2.38 (m, 1H, H-9), 2.08 (t, J = 3.9 Hz, 1H, H-41), 1.81 (m, 1H, H-42), 1.57 (m, 3H, H-81, H-51, H-82), 1.28 (m, H-8a), 1.23 (s, 3H, H-115), 1.17 (m, 1H, H-71), 1.07 (s, H-52), 0.98 (m, 1H, H-5a), 0.82 (d, J = 7.8 Hz, 3H, H-13), 0.77 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.57 (d, J = 6.0 Hz, 1H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 172.8 (C-5′′), 167.3 (C-3′′), 164.8 (1′-COO-), 160.0 (d, J = 255.0 Hz, C-2′′′), 135.9 (d, J = 9.0 Hz, C-4′′′), 132.1 (C-6′), 131.6 (C-4′), 130.9 (C-6′′′), 130.7 (C-1′), 130.0 (C-5′), 127.5 (C-2′), 126.6 (C-3′), 125.5 (d, J = 3.0 Hz, C-5′′′), 117.3 (d, J = 21.0 Hz, C-3′′′), 111.6 (d, J = 10.5 Hz, C-1′′′), 103.2 (C-3), 100.5 (C-10), 64.9 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(3-methoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (20e)
Yield 67%, white powder, m.p. 72 °C–74 °C; Rf = 0.36 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.66 (t, J = 1.5 Hz, 1H, H-2′), 8.37 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.20 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.79 (m, 2H, H-5′′′, H-6′′′), 7.68 (m, 1H, H-2′′′), 7.60 (t, J = 7.8 Hz, 1H, H-5′), 7.33 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′′′), 5.26 (s, 1H, H-12), 4.79 (d, J = 3.0 Hz, 1H, H-10), 4.68 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.09 (m, 1H, 10-O-CH2-CH2a-), 3.90 (s, 3H, 3′′′-OCH3), 3.70 (m, 1H, 10-O-CH2-CH2b-), 2.38 (m, 1H, H-9), 2.08 (m, 1H, H-41), 1.80 (m, 1H, H-42), 1.58 (m, 3H, H-81, H-51, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.15 (m, 1H, H-71), 1.08 (m, 1H, H-52), 0.98 (m, 1H, H-5a), 0.83 (d, J = 7.2 Hz, 3H, H-13), 0.78 (m, 1H, H-6), 0.69 (m, 1H, H-72), 0.56 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.6 (C-5′′), 167.6 (C-3′′), 164.8 (1′-COO-), 159.7 (C-3′′′), 132.1 (C-6′), 131.5 (C-4′), 130.9 (C-5′′′), 130.6 (C-1′), 130.0 (C-5′), 127.5 (C-2′), 126.7 (C-3′), 124.5 (C-1′′′), 120.2 (C-6′′′), 119.5 (C-4′′′), 112.6 (C-2′′′), 103.2 (C-3), 100.5 (C-10), 86.8 (C-12), 80.3 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(4-methoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (20f)
Yield 64%, white powder, m.p. 121 °C–123 °C; Rf = 0.37 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.64 (t, J = 1.5 Hz, 1H, H-2′), 8.34 (dd, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.18 (d, J = 7.8 Hz, 1H, H-6′), 8,15 (m, 2H, H-2′′′, H-6′′′), 7.77 (t, J = 7.8 Hz, 1H, H-5′), 7.20 (m, 2H, H-3′′′, H-5′′′), 5.26 (s, 1H, H-12), 4.79 (d, J = 3.0 Hz, 1H, H-10), 4.67 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.09 (m, 1H, 10-O-CH2-CH2a-), 3.89 (s, 3H, 4′′′-OCH3), 3.70 (m, 1H, 10-O-CH2-CH2b-), 2.39 (m, 1H, H-9), 2.08 (m, 1H, H-41), 1.81 (m, 1H, H-42), 1.57 (m, 3H, H-81, H-51, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.17 (m, 1H, H-71), 1.08 (m, 1H, H-52), 0.98 (m, 1H, H-5a), 0.83 (d, J = 7.2 Hz, 3H, H-13), 0.78 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.57 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.5 (C-5′′), 167.4 (C-3′′), 164.8 (1′-COO-), 163.2 (C-4′′′), 131.9 (C-6′), 131.5 (C-4′), 130.6 (C-1′), 130.0 (C-2′′′, C-6′′′), 129.9 (C-5′), 127.5 (C-2′), 126.9 (C-3′), 115.5 (C-1′′′), 115.0 (C-3′′′, C-5′′′), 103.2 (C-3), 100.5 (C-10), 86.8 (C-12), 80.2 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(m-tolyl)-1,2,4-oxadiazol-3-yl)benzoate (20g)
Yield 57%, white powder, m.p. 73 °C–75 °C; Rf=0.40 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.65 (t, J = 1.5 Hz, 1H, H-2′), 8.36 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.19 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 8.00 (m, 2H, H-2′′′, H-5′′′), 7.77 (t, J = 7.8 Hz, 1H, H-5′), 7.56 (m, 2H, H-4′′′, H-6′′′), 5.26 (s, 1H, H-12), 4.79 (d, J = 3.0 Hz, 1H, H-10), 4.68 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.09 (m, 1H, 10-O-CH2-CH2a-), 3.71 (m, 1H, 10-O-CH2-CH2b-), 2.45 (s, 3H, 3′′′-CH3), 2.39 (m, 1H, H-9), 2.08 (m, 1H, H-41), 1.82 (m, 1H, H-42), 1.58 (m, 3H, H-81, H-51, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.17 (m, 1H, H-71), 1.08 (m, 1H, H-52), 0.99 (m, 1H, H-5a), 0.84 (d, J = 7.2 Hz, 3H, H-13), 0.78 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.57 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.8 (C-5′′), 167.5 (C-3′′), 164.8 (1′-COO-), 139.1 (C-3′′′), 134.1 (C-2′′′), 132.0 (C-6′), 131.5 (C-4′), 130.6 (C-1′), 129.9 (C-5′), 129.4 (C-5′′′), 128.1 (C-4′′′), 127.5 (C-2′), 126.8 (C-3′), 125.1 (C-1′′′), 123.1 (C-6′′′), 103.2 (C-3), 100.6 (C-10), 86.8 (C-12), 80.2 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(p-tolyl)-1,2,4-oxadiazol-3-yl)benzoate (20h)
Yield 60%, white powder, m.p. 78 °C–80 °C; Rf = 0.40 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.65 (t, J = 1.5 Hz, 1H, H-2′), 8.37 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.20 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 8.11 (d, J = 7.8 Hz, 2H, H-2′′′, H-6′′′), 7.78 (t, J = 7.8 Hz, 1H, H-5′), 7.49 (d, J = 7.8 Hz, 2H, H-3′′′, H-5′′′), 5.26 (s, 1H, H-12), 4.79 (d, J = 3.0 Hz, 1H, H-10), 4.68 (m, 1H, 10-O-CH2a-), 4.44 (m, 1H, 10-O-CH2b-), 4.09 (m, 1H, 10-O-CH2-CH2a-), 3.70 (m, 1H, 10-O-CH2-CH2b-), 2.45 (s, 3H, 4′′′-CH3), 2.38 (m, 1H, H-9), 2.08 (m, 1H, H-41), 1.80 (m, 1H, H-42), 1.57 (m, 3H, H-81, H-51, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.16 (m, 1H, H-71), 1.07 (m, 1H, H-52), 0.99 (m, 1H, H-5a), 0.82 (d, J = 7.2 Hz, 3H, H-13), 0.76 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.56 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.8 (C-5′′), 167.5 (C-3′′), 164.8 (1′-COO-), 144.0 (C-4′′′), 132.0 (C-6′), 131.5 (C-4′), 130.6 (C-1′), 130.1 (C-2′′′, C-6′′′), 130.0 (C-5′), 127.9 (C-3′′′, C-5′′′), 127.5 (C-2′), 126.8 (C-3′), 120.4 (C-1′′′), 103.2 (C-3), 100.5 (C-10), 86.8 (C-12), 80.3 (C-12a), 64.9 (10-O-CH2-), 64.0 (10-O-CH2-
2-(((3R,5aS,6R,8aS,9R,10S,12R,12aR)-3,6,9-trimethyldecahydro-12H-3,12-epoxy[1,2]dioxepino[4,3-i]isochromen-10-yl)oxy)ethyl 3-(5-(3,4,5-trimethoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (20i)
Yield 58%, white powder, m.p. 186 °C–188 °C; Rf = 0.34 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.65 (t, J = 1.5 Hz, 1H, H-2′), 8.38 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.20 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.78 (t, J = 7.8 Hz, 1H, H-5′), 7.47 (s, 2H, H-2′′′, H-6′′′), 5.25 (s, 1H, H-12), 4.79 (d, J = 3.6 Hz, 1H, H-10), 4.67 (m, 1H, 10-O-CH2a-), 4.45 (m, 1H, 10-O-CH2b-), 4.08 (m, 1H, 10-O-CH2-CH2a-), 3.94 (s, 6H, 3′′′-OCH3, 5′′′-OCH3), 3.80 (s, 3H, 4′′′-OCH3), 3.70 (m, 1H, 10-O-CH2-CH2b-), 2.38 (m, 1H, H-9), 2.09 (m, 1H, H-41), 1.82 (m, 1H, H-42), 1.60 (m, 2H, H-81, H-51), 1.53 (m, 1H, H-82), 1.28 (m, 1H, H-8a), 1.24 (s, 3H, H-15), 1.16 (m, 1H, H-71), 1.07 (m, 1H, H-52), 0.99 (m, 1H, H-5a), 0.83 (d, J = 7.2 Hz, 3H, H-13), 0.79 (m, 1H, H-6), 0.68 (m, 1H, H-72), 0.58 (d, J = 6.0 Hz, 3H, H-14). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 175.6 (C-5′′), 167.6 (C-3′′), 164.8 (1′-COO-), 153.5 (C-3′′′, C-5′′′), 141.9 (C-4′′′), 132.0 (C-6′), 131.6 (C-4′), 130.6 (C-1′), 130.0 (C-5′), 127.5 (C-2′), 126.7 (C-3′), 118.2 (C-1′′′), 105.4 (C-2′′′, C-6′′′), 103.2 (C-3), 100.6 (C-10), 86.8 (C-12), 80.3 (C-12a), 65.0 (10-O-CH2-), 64.0 (10-O-CH2-
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-methyl-1,2,4-oxadiazol-3-yl)benzoate (21a)
Yield 57%, colorless oil; Rf = 0.42 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.54 (t, J = 1.5 Hz, 1H, H-2′), 8.27 (m, 1H, H-4′), 8.16 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.74 (t, J = 7.8 Hz, 1H, H-5′), 6.05 (t, J = 5.7 Hz, 1H, H-3), 5.97 (d, J = 16.2 Hz, 1H, H-10), 5.91 (d, J = 16.2 Hz, 1H, H-11), 5.59 (t, J = 8.1 Hz, 1H, H-7), 4.97 (s, 1H, H-15a), 4.80 (s, 1H, H-15b), 2.67 (s, 3H, 5′′-CH3), 2.59 (m, 2H, H5a, H-8a), 2.48 (m, 1H, H-4a), 2.28 (m, 2H, H-5b, H-4b), 2.03 (m, 1H, H-8b), 1.55 (s, 3H, H-12), 1.23 (s, 3H, H-13 or H-14), 1.09 (s, 3H, H-14 or H-13). 13 C NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.5 (C-1), 177.9 (C-5′′), 166.8 (C-3′′), 164.9 (1′-COO-), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.0 (C-6), 131.9 (C-6′), 131.5 (C-7), 131.4 (C-4′), 130.6 (C-1′), 130.0 (C-5′), 127.4 (C-2′), 126.94 (C-11), 126.88 (C-3′), 61.8 (C-15), 41.6 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 20.7 (3′′′-CH3), 11.9 (5′′-CH3), 11.6 (C-12). ESI-HRMS calculated for C25H29N2O4: [M+H]+ (m/z): 421.2122, found: 421.2105.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-isopropyl-1,2,4-oxadiazol-3-yl)benzoate (21b)
Yield 54%, colorless oil; Rf = 0.42 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.55 (t, J = 1.5 Hz, 1H, H-2′), 8.28 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.16 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.75 (t, J = 7.8 Hz, 1H, H-5′), 6.05 (t, J = 5.4 Hz, 1H, H-3), 5.97 (d, J = 16.2 Hz, 1H, H-10), 5.92 (d, J = 16.2 Hz, 1H, H-11), 5.60 (t, J = 8.1 Hz, 1H, H-7), 4.98 (s, 1H, H-15a), 4.81 (s, 1H, H-15b), 3.38 (m, 1H, H-1′′′), 2.58 (m, 2H, H5a, H-8a), 2.47 (m, 1H, H-4a), 2.28 (m, 2H, H-5b, H-4b), 2.03 (m, 1H, H-8b), 1.55 (s, 3H, H-12), 1.23 (s, 3H, H-13 or H-14), 1.10 (s, 3H, H-14 or H-13). 13 C NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.5 (C-1), 184.5 (C-5′′), 166.7 (C-3′′), 164.9 (1′-COO-), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.0 (C-6), 131.9 (C-6′), 131.54 (C-7), 131.49 (C-4′), 130.6 (C-1′), 130.0 (C-5′), 127.4 (C-2′), 126.96 (C-11), 126.94 (C-3′), 61.8 (C-15), 41.6 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 26.8 (C-1′′′), 24.4 (C-4), 23.6 (C-14 or C-13), 19.7 (C-2′′′, C-3′′′), 11.7 (C-12). ESI-HRMS calculated for C27H33N2O4: [M+H]+ (m/z): 449.2435, found: 449.2415.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-phenyl-1,2,4-oxadiazol-3-yl)benzoate (21c)
Yield 60%, white powder, m.p. 68 °C–70 °C; Rf = 0.39 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.64 (t, J = 1.5 Hz, 1H, H-2′), 8.37 (dt, J1 = 1.5 Hz, J2 = 7.5 Hz, 1H, H-4′), 8.20 (m, 3H, H-6′, H-2′′′, H-6′′′), 7.77 (m, 2H, H-5′, H-4′′′), 7.68 (m, 2H, H-3′′′, H-5′′′), 6.06 (t, J = 5.7 Hz, 1H, H-3), 5.98 (d, J = 16.2 Hz, 1H, H-10), 5.93 (d, J = 16.2 Hz, 1H, H-11), 5.61 (t, J = 8.1 Hz, 1H, H-7), 5.00 (s, 1H, H-15a), 4.82 (s, 1H, H-15b), 2.61 (m, 2H, H5a, H-8a), 2.48 (m, 1H, H-4a), 2.29 (m, 2H, H-5b, H-4b), 2.05 (m, 1H, H-8b), 1.56 (s, 3H, H-12), 1.24 (s, 3H, H-13 or H-14), 1.11 (s, 3H, H-14 or H-13). 13 C NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.5 (C-1), 175.8 (C-5′′), 167.5 (C-3′′), 164.9 (1′-COO-), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.0 (C-6), 133.5 (C-1′′′), 132.1 (C-6′), 131.6 (C-4′), 130.6 (C-1′), 130.1 (C-5′), 129.6 (C-3′′′, C-5′′′), 128.0 (C-2′′′, C-6′′′), 127.5 (C-2′), 126.9 (C-11), 126.8 (C-3′), 123.2 (C-1′′′), 61.8 (C-15), 41.7 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 11.7 (C-12). ESI-HRMS calculated for C30H31N2O4: [M+H]+ (m/z): 483.2278, found: 483.2254.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl)benzoate (21d)
Yield 28%, colorless oil; Rf = 0.38 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.64 (t, J = 1.5 Hz, 1H, H-2′), 8.38 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.26 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′′′), 8.21 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.82 (m, 2H, H-4′′′, H-5′), 7.57 (t, J = 7.8 Hz, 1H, H-3′′′), 7.52 (m, 1H, H-5′′′), 6.06 (t, J = 5.4 Hz, 1H, H-3), 5.98 (d, J = 16.5 Hz, 1H, H-10), 5.93 (d, J = 16.5 Hz, 1H, H-11), 5.61 (t, J = 8.1 Hz, 1H, H-7), 5.01 (s, 1H, H-15a), 4.82 (s, 1H, H-15b), 2.62 (m, 3H, H5a, H-8a, H-4a), 2.30 (m, 2H, H-5b, H-4b), 2.05 (m, 1H, H-8b), 1.56 (s, 3H, H-12), 1.24 (s, 3H, H-13 or H-14), 1.11 (s, 3H, H-14 or H-13). 13C NMR (125 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 212.0 (C-1), 176.8 (C-5′), 174.4 (C-3′), 169.5 (d, J = 257.5 Hz, C-2′′′), 168.9 (1′-COO-), 158.4 (C-10), 147.1 (C-3), 145.4 (C-4′′′), 143.6 (C-6), 141.7 (C-6′), 141.2 (C-4′), 141.0 (C-7), 140.4 (C-1′′′), 140.2 (C-1′), 139.7 (C-5′), 137.1 (C-2′), 136.5 (C-11), 136.1 (C-3′), 135.0 (C-5′′′), 126.8 (d, J = 21.15 Hz, C-3′′′), 121.1 (C-6′′′), 71.4 (C-15), 51.2 (C-8), 46.5 (C-9), 44.2 (C-6), 38.4 (C-13 or C-14), 33.9 (C-4), 33.3 (C-14 or C-13), 21.2 (C-12). ESI-HRMS calculated for C30H30FN2O4: [M+H]+ (m/z): 501.2198, found: 501.2176.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(3-methoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (21e)
Yield 55%, white powder, m.p. 82 °C–84 °C; Rf = 0.36 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.61 (t, J = 1.8 Hz, 1H, H-2′), 8.34 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.18 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.77 (m, 2H, H-5′′′, H-6′′′), 7.65 (t, J = 1.8 Hz, 1H, H-2′′′), 7.58 (t, J = 7.8 Hz, 1H, H-5′), 7.31 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′′′), 6.05 (t, J = 5.4 Hz, 1H, H-3), 5.97 (d, J = 16.2 Hz, 1H, H-10), 5.92 (d, J = 16.2 Hz, 1H, H-11), 5.60 (t, J = 8.4 Hz, 1H, H-7), 4.99 (s, 1H, H-15a), 4.80 (s, 1H, H-15b), 3.89 (s, 3H, 3′′′-OCH3), 2.60 (m, 2H, H5a, H-8a), 2.49 (m, 1H, H-4a), 2.29 (m, 2H, H-5b, H-4b), 2.03 (m, 1H, H-8b), 1.57 (s, 3H, H-12), 1.24 (s, 3H, H-13 or H-14), 1.11 (s, 3H, H-14 or H-13). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.4 (C-1), 175.6 (C-5′′), 167.5 (C-3′′), 164.9 (1′-COO-), 159.7 (C-3′′′), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.0 (C-6), 132.1 (C-6′), 131.6 (C-4′), 131.5 (C-7), 130.8 (C-1′), 130.6 (C-5′), 130.0 (C-5′′′), 127.5 (C-2′), 127.0 (C-11), 126.7 (C-3′), 124.3 (C-1′′′), 120.3 (C-6′′′), 119.5 (C-4′′′), 112.5 (C-2′′′), 61.8 (C-15), 55.5 (3′′′-OCH3), 41.7 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 11.7 (C-12). ESI-HRMS calculated for C31H33N2O5: [M+H]+ (m/z): 513.2384, found: 513.2360.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(4-methoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (21f)
Yield 54%, white powder, m.p. 109 °C–111 °C; Rf=0.36 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.60 (t, J = 1.5 Hz, 1H, H-2′), 8.33 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.17 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 8.13 (m, 2H, H-2′′′, H-6′′′), 7.76 (t, J = 7.8 Hz, 1H, H-5′), 7.19 (m, 2H, H-3′′′, H-5′′′), 6.04 (t, J = 5.7 Hz, 1H, H-3), 5.97 (d, J = 16.2 Hz, 1H, H-10), 5.91 (d, J = 16.2 Hz, 1H, H-11), 5.59 (t, J = 8.4 Hz, 1H, H-7), 4.98 (s, 1H, H-15a), 4.80 (s, 1H, H-15b), 3.88 (s, 3H, 4′′′-OCH3), 2.60 (m, 2H, H5a, H-8a), 2.49 (m, 1H, H-4a), 2.29 (m, 2H, H-5b, H-4b), 2.04 (m, 1H, H-8b), 1.55 (s, 3H, H-12), 1.23 (s, 3H, H-13 or H-14), 1.10 (s, 3H, H-14 or H-13). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 201.9 (C-1), 175.4 (C-5′′), 167.2 (C-3′′), 164.7 (1′-COO-), 163.1 (C-4′′′), 158.7 (C-10), 148.3 (C-3), 137.3 (C-2), 133.8 (C-6), 131.6 (C-6′), 131.3 (C-4′), 131.2 (C-7), 130.5 (C-1′), 129.7 (C-2′′′, C-6′′′), 129.6 (C-5′), 127.3 (C-2′), 126.8 (C-11), 126.7 (C-3′), 115.4 (C-1′′′), 114.8 (C-3′′′, C-5′′′), 61.6 (C-15), 55.4 (4′′′-OCH3), 41.5 (C-8), 36.7 (C-9), 34.6 (C-5), 24.1 (C-4), 11.3 (C-12). ESI-HRMS calculated for C31H33N2O5: [M+H]+ (m/z): 513.2384, found: 513.2367.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(m-tolyl)-1,2,4-oxadiazol-3-yl)benzoate (21g)
Yield 45%, white powder, m.p. 88 °C–90 °C; Rf = 0.38 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.60 (s, 1H, H-2′), 8.33 (d, J = 7.8 Hz, 1H, H-4′), 8.17 (d, J = 7.8 Hz, 1H, H-6′), 7.97 (m, 2H, H-2′′′, H-5′′′), 7.76 (t, J = 7.8 Hz, 1H, H-5′), 7.54 (m, 2H, H-4′′′, H-6′′′), 6.05 (t, J = 5.4 Hz, 1H, H-3), 5.97 (d, J = 16.5 Hz, 1H, H-10), 5.93 (d, J = 16.5 Hz, 1H, H-11), 5.60 (t, J = 8.4 Hz, 1H, H-7), 4.99 (s, 1H, H-15a), 4.80 (s, 1H, H-15b), 2.60 (m, 2H, H5a, H-8a), 2.48 (m, 4H, H-4a, 3′′′-CH3), 2.29 (m, 2H, H-5b, H-4b), 2.03 (m, 1H, H-8b), 1.57 (s, 3H, H-12), 1.24 (s, 3H, H-13 or H-14), 1.11 (s, 3H, H-14 or H-13). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.4 (C-1), 175.8 (C-5′′), 167.4 (C-3′′), 164.8 (1′-COO-), 159.7 (C-3′′′), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.1 (C-2′′′), 134.0 (C-6), 132.0 (C-6′), 131.54 (C-7), 131.51 (C-4′), 130.6 (C-1′), 130.0 (C-5′), 129.4 (C-5′′′), 128.2 (C-4′′′), 127.5 (C-2′), 127.0 (C-11), 126.8 (C-3′), 125.1 (C-1′′′), 123.1 (C-6′′′), 61.8 (C-15), 41.7 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 20.7 (3′′′-CH3), 11.6 (C-12). ESI-HRMS calculated for C31H33N2O4: [M+H]+ (m/z): 497.2435, found: 497.2417.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(p-tolyl)-1,2,4-oxadiazol-3-yl)benzoate (21h)
Yield 48%, white powder, m.p. 118 °C–120 °C; Rf = 0.38 (n-hexane/acetone 2:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.61 (t, J = 1.5 Hz, 1H, H-2′), 8.33 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.17 (dt, J1 = 1.5 Hz, J2 = 7.8 Hz, 1H, H-6′), 8.08 (d, J = 7.8 Hz, 2H, H-2′′′, H-6′′′), 7.77 (t, J = 7.8 Hz, 1H, H-5′), 7.47 (d, J = 7.8 Hz, 2H, H-3′′′, H-5′′′), 6.05 (t, J = 5.7 Hz, 1H, H-3), 5.97 (d, J = 16.2 Hz, 1H, H-10), 5.93 (d, J = 16.2 Hz, 1H, H-11), 5.60 (t, J = 8.4 Hz, 1H, H-7), 4.99 (s, 1H, H-15a), 4.81 (s, 1H, H-15b), 2.60 (m, 2H, H5a, H-8a), 2.43 (m, 4H, H-4a, 4′′′-CH3), 2.29 (m, 2H, H-5b, H-4b), 2.04 (m, 1H, H-8b), 1.56 (s, 3H, H-12), 1.24 (s, 3H, H-13 or H-14), 1.11 (s, 3H, H-14 or H-13). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.4 (C-1), 175.8 (C-5′′), 167.4 (C-3′′), 164.9 (1′-COO-), 159.3 (C-10), 148.9 (C-3), 137.6 (C-2), 134.0 (C-6), 132.0 (C-6′), 131.6 (C-4′), 131.5 (C-7, C-4′′′), 130.6 (C-1′), 130.1 (C-2′′′, C-6′′′), 130.0 (C-5′), 127.9 (C-3′′′, C-5′′′), 127.5 (C-2′), 127.0 (C-11), 126.8 (C-3′), 120.4 (C-1′′′), 61.8 (C-15), 41.7 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 11.7 (C-12). ESI-HRMS calculated for C31H33N2O4: [M+H]+ (m/z): 497.2435, found: 497.2414.
((1Z,5E,8E)-4,4,8-trimethyl-7-oxocycloundeca-1,5,8-trien-1-yl)methyl-3-(5-(3,4,5-trimethoxyphenyl)-1,2,4-oxadiazol-3-yl)benzoate (21i)
Yield 50%, white powder, m.p. 154 °C–156 °C; Rf = 0.33 (n-hexane/acetone 3:1 v/v);1H NMR (600 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 8.60 (t, J = 1.5 Hz, 1H, H-2′), 8.34 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-4′), 8.17 (dt, J1 = 1.8 Hz, J2 = 7.8 Hz, 1H, H-6′), 7.76 (t, J = 7.8 Hz, 1H, H-5′), 7.42 (s, 2H, H-2′′′, H-6′′′), 6.05 (t, J = 5.7 Hz, 1H, H-3), 5.97 (d, J = 16.8 Hz, 1H, H-10), 5.91 (d, J = 16.8 Hz, 1H, H-11), 5.59 (t, J = 8.1 Hz, 1H, H-7), 4.99 (s, 1H, H-15a), 4.80 (s, 1H, H-15b), 3.92 (s, 6H, 3′′′-CH3, 5′′′-OCH3), 3.80 (s, 3H, 4′′′-OCH3), 2.59 (m, 2H, H5a, H-8a), 2.49 (m, 1H, H-4a), 2.29 (m, 2H, H-5b, H-4b), 2.04 (m, 1H, H-8b), 1.58 (s, 3H, H-12), 1.23 (s, 3H, H-13 or H-14), 1.10 (s, 3H, H-14 or H-13). NMR (150 MHz, DMSO-d6, 25 °C, TMS, δ (ppm)): 202.4 (C-1), 175.5 (C-5′′), 167.5 (C-3′′), 164.9 (1′-COO-), 159.2 (C-10), 153.4 (C-3′′′, C-5′′′), 148.9 (C-3), 141.8 (C-4′′′), 137.6 (C-2), 134.0 (C-6), 132.0 (C-6′), 131.7 (C-4′), 131.4 (C-7), 130.6 (C-1′), 130.0 (C-5′), 127.5 (C-2′), 127.0 (C-11), 118.1 (C-1′′′), 105.3 (C-3′′′, C-5′′′), 61.8 (C-15), 60.2 (4′′′-OCH3), 56.2 (3′′′-OCH3, 5′′′-OCH3), 41.6 (C-8), 37.0 (C-9), 34.7 (C-5), 28.9 (C-13 or C-14), 24.4 (C-4), 23.6 (C-14 or C-13), 11.7 (C-12). ESI-HRMS calculated for C33H37N2O7: [M+H]+ (m/z): 573.2595, found: 573.2571.
Biology
The in vitro cytotoxic evaluation of the synthesized compounds against Hep-G2 and MCF-7 cancer cell lines was carried out according to the described protocols.37,38
Molecular docking
The three-dimensional structures of compounds were prepared using MarvinSketch version 19.27.0 and PyMOL version 1.3r1. 39 Energy minimization of the studied ligands was conducted using the MM2 force field, and quantum chemical calculations were performed at the B3LYP/6-31g(d, p) level implemented in Gaussian 09. 40 Erlotinib, a well-known inhibitor of the EGFR protein, was selected as a reference ligand. 41
The X-ray crystal structure of the EGFR protein was obtained from the Protein Data Bank archive (PDB) with entry ID: 4HJO. 42 It is assumed that the protein is rigid, and the conformational space of the ligands to analyze the inductive effect of the compounds is considered. The Graphical User Interface program named AutoDock Tools 1.5.6rc3 (ADT) was employed to set up input data. 33 Details of the molecular docking simulation are given in Supplementary Material 2.
Supplemental Material
sj-pdf-1-chl-10.1177_17475198231219451 – Supplemental material for Dihydroartemisinin and zerumbone esters of ataluren and its analogs as anticancer agents and EGFR inhibitors
Supplemental material, sj-pdf-1-chl-10.1177_17475198231219451 for Dihydroartemisinin and zerumbone esters of ataluren and its analogs as anticancer agents and EGFR inhibitors by Duc Quan Tran, Ngoc Hung Truong, Thi Hoang Anh Nguyen, Thi Thuy Trinh, Thi Cham Ba, Thi Thuy Linh Nguyen, Van Tu Ngo, Thi Inh Cam, Minh Quan Pham and Van Chinh Luu in Journal of Chemical Research
Supplemental Material
sj-pdf-2-chl-10.1177_17475198231219451 – Supplemental material for Dihydroartemisinin and zerumbone esters of ataluren and its analogs as anticancer agents and EGFR inhibitors
Supplemental material, sj-pdf-2-chl-10.1177_17475198231219451 for Dihydroartemisinin and zerumbone esters of ataluren and its analogs as anticancer agents and EGFR inhibitors by Duc Quan Tran, Ngoc Hung Truong, Thi Hoang Anh Nguyen, Thi Thuy Trinh, Thi Cham Ba, Thi Thuy Linh Nguyen, Van Tu Ngo, Thi Inh Cam, Minh Quan Pham and Van Chinh Luu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This study was financially supported by the Vietnam Academy of Science and Technology under the project with the code: KHCBHH.02/21-23.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
