Abstract
In the present study, via using the mixed-ligand synthesis approach, a new Mn(II)-containing coordination polymer (CP) with the chemical formula of {[Mn3(timb)2(SO3–IPA)2(H2O)2]·8H2O}
n
(
Introduction
Recently, transition metal complexes containing inorganic metal ions and organic ligands have become the focus of interest due to not only their structural versatility but also their wide range of applications such as antitumor and antibacterial activities, insulin mimetic activities, luminescence properties, catalytic activities, magnetic properties, DNA binding properties, and gas adsorption.1–6 It is well known that the structural versatility and activity properties of the metal–organic complexes are largely dependent on the choice of organic ligands and metal atoms used, as well as the reaction mechanism in which the complex is obtained.7–10 Consequently, the logical design of the organic ligands and the selection of suitable metal ions play a key role in obtaining the coordination complexes with the desired properties. Manganese is the second most abundant transition metal within the Earth’s crust. It is used to form oxygen in photosynthesis, an indispensable process for plant growth, and is involved in the absorption of nitrates in green plants and algae. In organisms having transactions with elemental oxygen, manganese enzymes are very imperative for the detoxification of superoxide free radicals.11–14 This metal carries out a variety of redox functions in living systems, like reduction of ribonucleotides to deoxyribonucleotides in coryneform bacteria and the disproportionation of hydrogen peroxide (catalase activity) in microorganisms. In biological system of higher animals, manganese(II) ions play a key role in the action of many enzymes and as a result there are a number of metalloproteins which necessitate manganese to execute their biological functions, and these have become important targets for synthetic modeling studies. Based on the above considerations, in the present study, via using the mixed-ligand synthesis approach, a new Mn(II)-containing coordination polymer (CP) with the chemical formula of {[Mn3(timb)2(SO3–IPA)2(H2O)2]·8H2O}
n
( The chemical drawings for the ligands.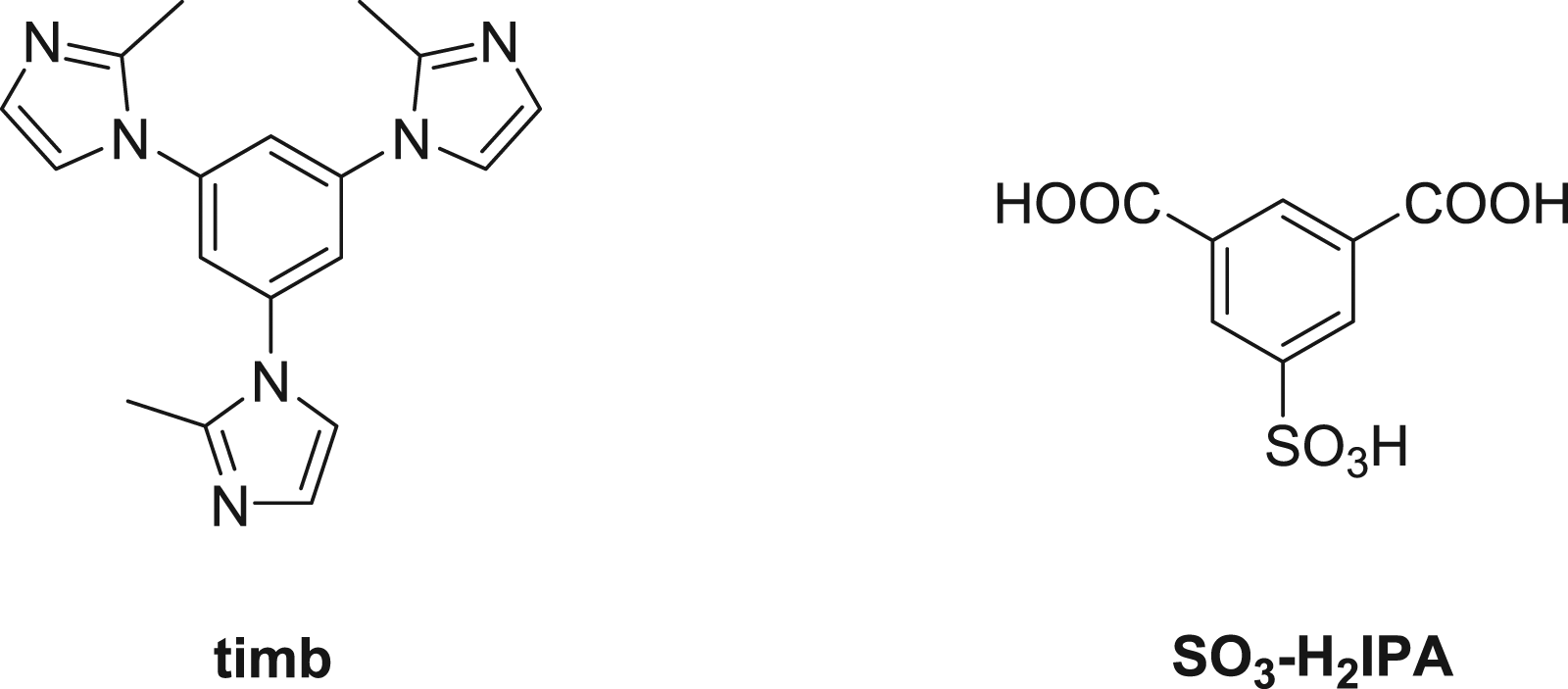
Experimental
Chemicals and measurements
The two ligands used in this work were purchased from Jinan Henghua Sci. & Tec. Co. Ltd. Other materials were commercially available and used without further purification. Elemental analyses for C, H, and N were carried out on a Vario MACRO cube elemental analytical instrument. IR spectra were performed on an FTIR–8400S Spectrometer within the 4000–400 cm −1 range in KBr pellets.
Preparation and characterization for {[Mn3(timb)2(SO3–IPA)2(H2O)2]·8H2O} n (1)
A mixture of MnCl2·4H2O (12 mg, 0.05 mmol), NaSO3–H2IPA (13.4 mg, 0.05 mmol), timb (15.9 mg, 0.05 mmol), DMA (1.5 mL), and H2O (1.5 mL) was stirred at room temperature for 15 min, then sealed in a 10 mL Teflon lined stainless steel vessel, and heated at 100°C for 2 days, followed by cooling to room temperature at a rate of 10°C·h−1. Light yellow block-shaped crystals of
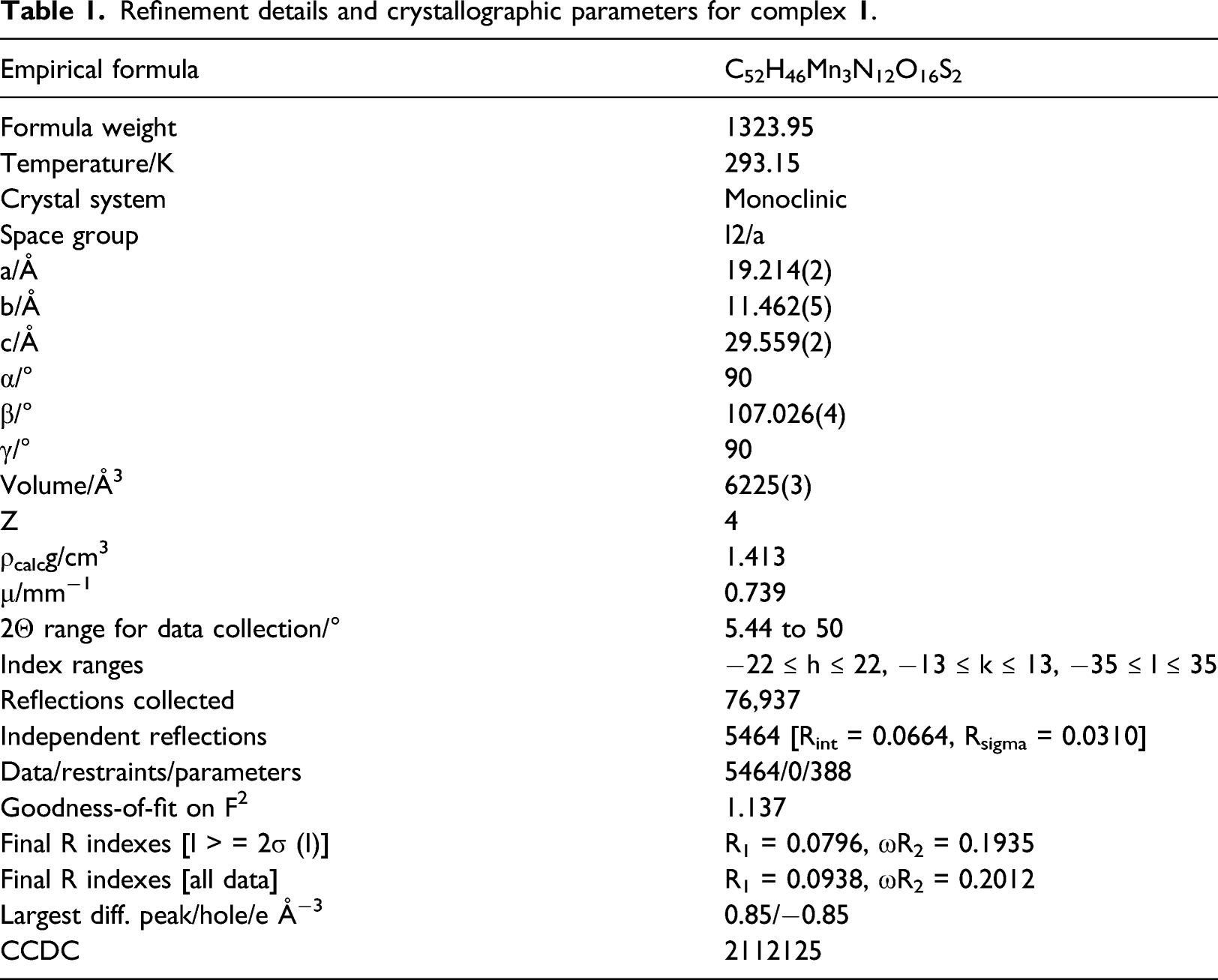
Refinement details and crystallographic parameters for complex
Calcium colorimetric assay
Calcium Colorimetric Assay was conducted in this present research to measure the treatment activity of the compound on the renal calculus mice through measuring the concentration of the Ca2+ in urine. This preformation was conducted totally under the guidance of the instructions with only a little change. In brief, 50 animals were used in this research, which were obtained from the Qingdao Municipal Hospital. Before the experiment, all the animals were cultured in the standard condition of 20–25°C, with free water and food. The animals were divided into five groups: the control group, model group, and compound treatment groups. The high-calcium diet containing oxalic acid was used to induce the renal calculus animal model, next the new compound was given for treatment at the concentrations of 1, 2, and 5 mg/kg. After that, the urine of all the mice was collected and the Ca2+ concentration in urine was determined with Calcium Colorimetric Assay with three repeats.
Western blotting assay
The western blotting assay was performed to evaluate the osteopontin expression levels in distal tubule. This conduction was carried out strictly in accordance with the manufacture instructions. In short, 50 animals were used in this research, which were obtained from the Qingdao Municipal Hospital. Before the experiment, all the animals were cultured in the standard condition of 20–25°C, with free water and food. The animals were divided into five groups: the control group, model group, and compound treatment groups. The high-calcium diet containing oxalic acid was used to induce the renal calculus animal model, next the new compound was given for treatment at the concentrations of 1, 2, and 5 mg/kg. After that, the distal tubule tissue was harvested and the Total Protein Extraction kit was used to extract the total protein in the cells. The BCA Protein Assay Kit was used to measure the total protein concentration. All the samples were separated by SDS-PAGE gel electrophoresis and electrophoretically transferred to a 0.22 mm polyvinylidene fluoride (PVDF) membrane. After incubated with primary antibody (OPN or GAPDH) and appropriate secondary antibody conjugated with horseradish peroxidase, the protein images were captured.
Simulation methods
The molecular docking simulation has been prepared by AutoDockTools 1.5.6 and performed by AutoDock 4.2. The biological antibody 23C3 has been employed as the receptor protein for investigating the possible binding interactions with the Mn complex, the structure of the antibody was downloaded from protein data bank, and the corresponding PDB ID is 3CXD. 17 The grid box that includes the docking pocket locates at the center of mass of the 3CXD structure, and detailed coordination of the center of mass is −8.314, 15.77, −29.053 (Å). The number of grid points in each direction is 60, and the spacing length is 0.375 Å. Only the polar hydrogens on the 3CXD protein are considered explicitly, during the docking simulation, the structure of the receptor is rigid, but the structure of the Mn complex is semi-flexible; thus, 9 torsional degrees of freedom are allowed for adjusting the conformation of Mn complex inside the docking pocket. The maximum allowed molecular docking run is set to 60 and the scoring method is Lamarckian Genetic algorithm (LGA).
Results and discussion
Structural characterization
Single crystal X-ray diffraction analysis reveals that (a) A view of the contents of the asymmetric unit. (b) 2D [Mn3(timb)2]
n
sheet in 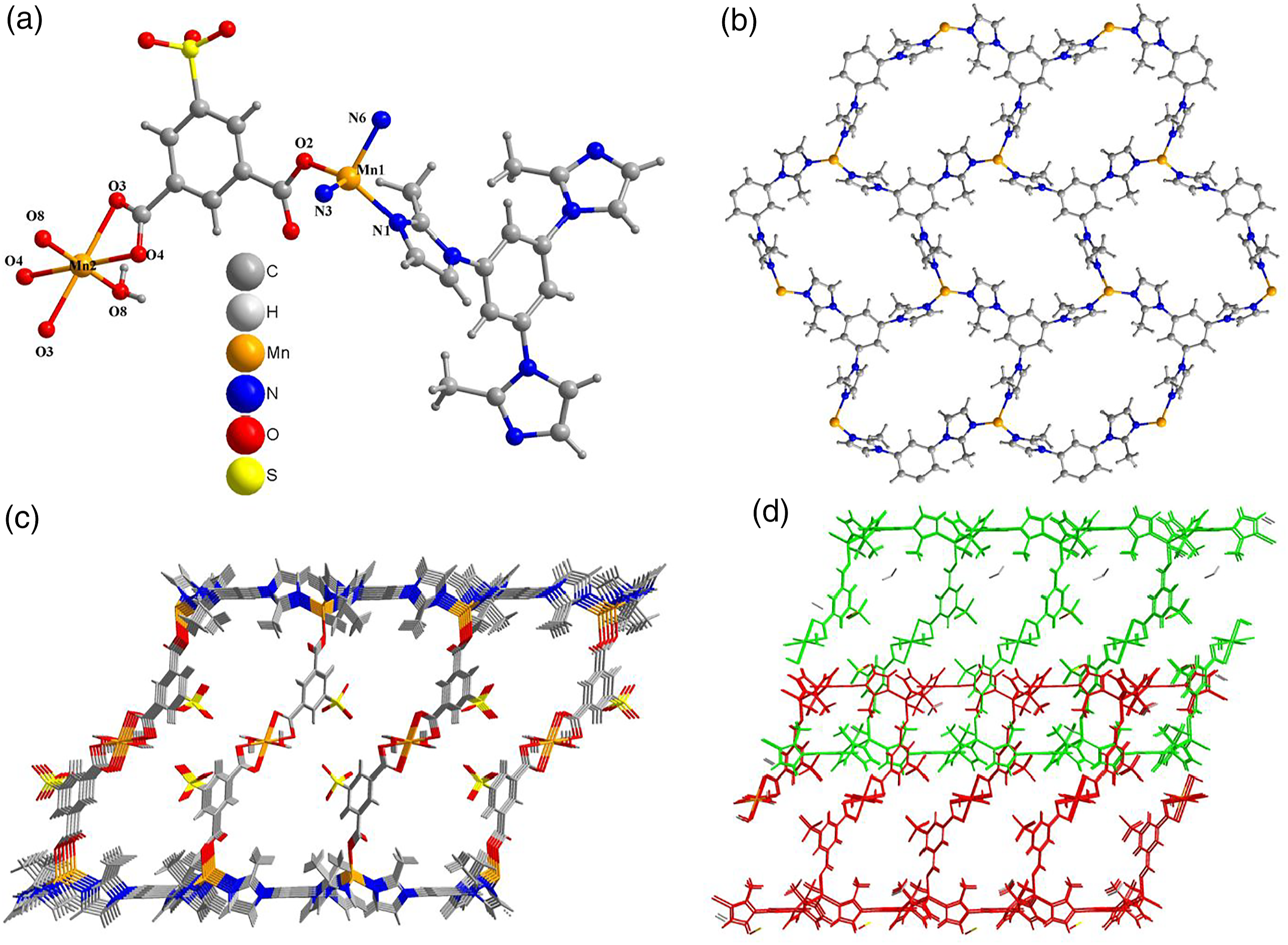
Reduced concentration of the Ca2+ in urine in a dose- and time-dependent manner after compound exposure
In the procession of renal calculus, the Ca2+ in the urine plays an important role. Thus, the Calcium Colorimetric Assay was firstly conducted in this present research to measure the concentration of the Ca2+ in urine after compound treatment. As the results showed in Figure 2, we can see that, in the model group, there was a much higher level of the Ca2+ concentration, which is significantly different from the control group. After the treatment of the new compound, the increased level of Ca2+ in urine was significantly reduced in a dose- and time-dependent manner, suggesting the excellent biological activity of the new compound on renal calculus therapy. Compound reduced the concentration of the Ca2+ in urine in a dose- and time-dependent manner. The high-calcium diet containing oxalic acid was used to induce the renal calculus animal model, next the new compound was given for treatment at the concentrations of 1, 2, and 5 mg/kg. Calcium Colorimetric Assay was used to measure the Ca2+ concentration in the urine.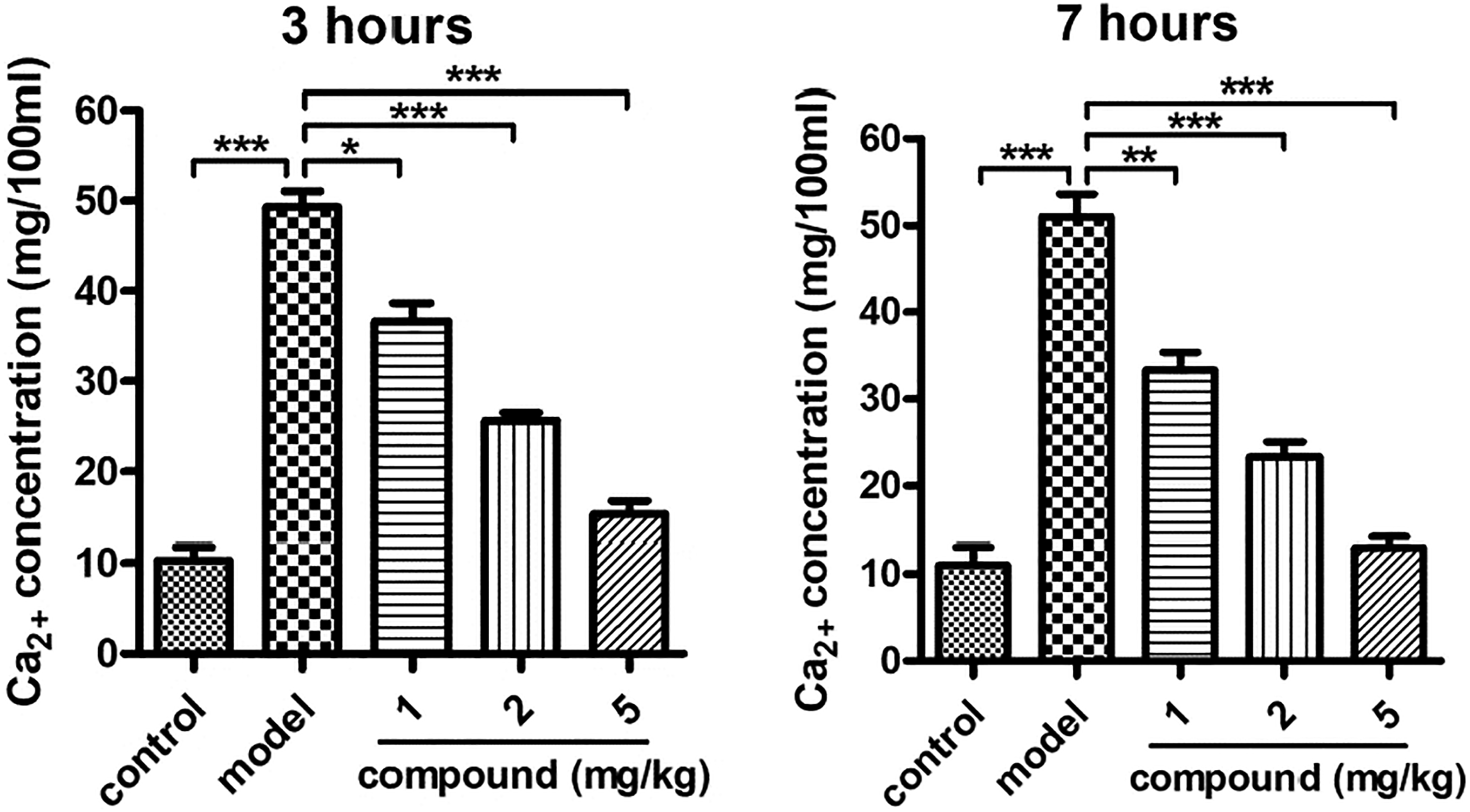
Inhibited expression levels of the osteopontin in distal tubule after compound treatment
As previously reported, the osteopontin plays an important role in cell matrix remodeling, tissue calcification, pro-inflammatory cytokines, and cell apoptosis regulation. In recent years, it has been confirmed that osteopontin is closely related to the formation of urinary stones. In the above research, we also proved that the new compound has excellent inhibitory effect on the Ca2+ concentration in the urine. Thus, in this research, western blotting assay was further conducted and the expression levels of the osteopontin in distal tubule were evaluated. In Figure 3, we showed that, the level of the osteopontin expression in the model group was much higher than that of the control group. After the treatment of the new compound, osteopontin expression was reduced in a dose-dependent manner. Compound inhibited the expression of the osteopontin in distal tubule. The high-calcium diet containing oxalic acid was used to induce the renal calculus animal model, next the new compound was given for treatment at the concentrations of 1, 2, and 5 mg/kg. Western blotting assay was conducted and the osteopontin expression evaluation in distal tubule was measured after compound exposure.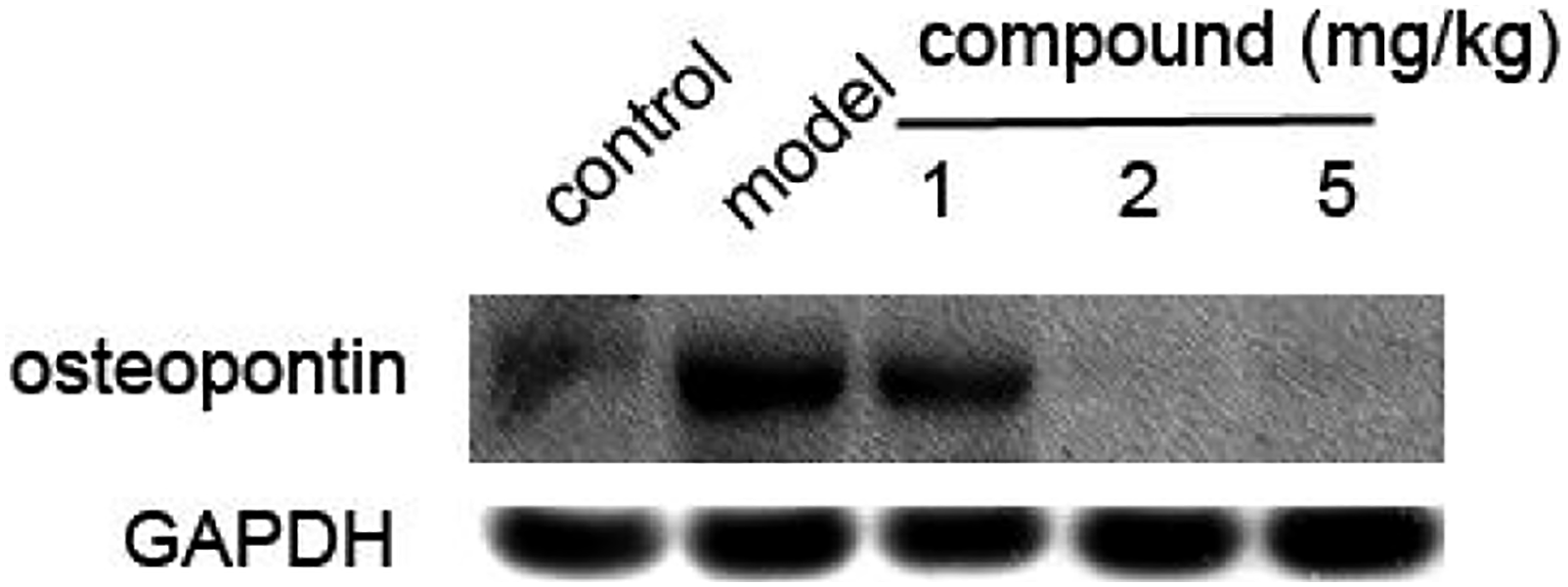
Molecular docking
It is well-known that osteopontin plays a key role in the development and perpetuation of related disease, and antibodies that are targeting the osteopontin have shown effective therapeutic benefits for the curing of related disease.
17
Thus, the biological antibody 23C3 has been chosen to be the receptor protein for probing the biological activity of the newly synthesized Mn complex. 60 binding poses have been generated randomly and scored by the LGA methods during the molecular docking simulation; the binding conformation that exhibits the lowest affinity energy (−14.6 kcal/mol) among the 60 binding poses is displayed in Figure 4. The binding conformation that exhibits the lowest affinity energy (−14.6 kcal/mol) among the 60 binding poses, and the estimated inhibition constant is 1.37 nM. Four active residues (pink) are involved in the binding interactions with the Mn complex, and five hydrogen bonds are formed.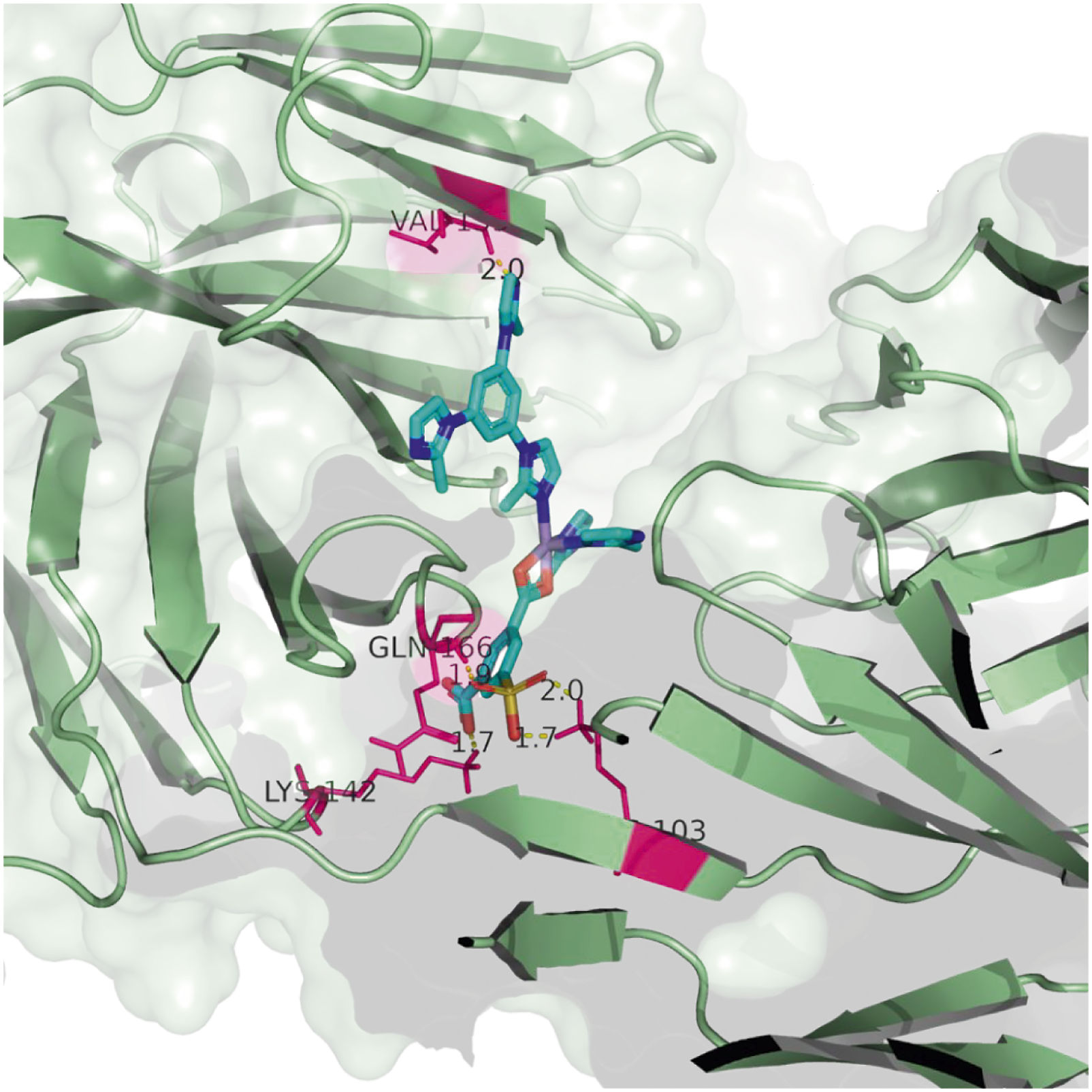
It can be seen from Figure 4 that five binding interactions have been formed between the Mn complex and the receptor protein. All types of polar groups on the Mn complex are able to form hydrogen bonding interactions with the active sites (polar hydrogens) inside the docking pocket. Explicitly, the carboxyl group interacts with residue LYS-142 with a hydrogen bond length of 1.7, the iminazole group interacts with residue VAL-155 and the hydrogen bond length is 2.0, the sulfonate group interacts with residues LYS-103 and GLN-166, interestingly, all three oxygen atoms on the sulfonate group are interacting with the active site, and the formed hydrogen bond lengths are 1.7 and 2.0 to residue LYS-103 and 1.9 to residue GLN-166. The above results suggest that the Mn complex has excellent biological activity and agree well with the experimental results.
Conclusion
In summary, we have prepared a two-dimensional Mn(II) coordination polymer via using the mixed-ligand synthesis approach by reaction of MnCl2·4H2O with the tripodal linker 1,3,5-tris(2-methylimidazol-1-yl)benzene and aromatic dicarboxylic acid ligand –SO3 group functionalized isophthalic acid ligand. The as-prepared coordination polymer
Supplemental Material
sj-pdf-1-prk-10.1177_14686783221090373 – Supplemental Material for A new mixed-ligand Mn(II) coordination polymer: Protective and nursing values on renal calculus via reducing the Ca2+ concentration in urine
Supplemental Material, sj-pdf-1-prk-10.1177_14686783221090373 for A new mixed-ligand Mn(II) coordination polymer: Protective and nursing values on renal calculus via reducing the Ca2+ concentration in urine by Yan Yang, Sheng-Mei Zhang, Yi-Qian Zhang, Liang Chen and Tao Xiong in Progress in Reaction Kinetics and Mechanism
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
