Abstract
Molecules that target quorum sensing and biofilm inhibition are useful antimicrobials. In this regard, a new diarylhydrazone was synthesized and characterized using infrared, high-resolution mass spectrometry and nuclear magnetic resonance experiments as

Introduction
Aryl hydrazones represent an important class of compounds for heterocyclic synthesis such as indoles and pyrazoles.1,2 These molecules have several applications in biology, organic/inorganic, and analytical chemistry3,4 and can act as antidepressants, anti-inflammatory, antimicrobial, antimalarial, antitumor, antioxidant, and antifungal agents.4–7 Drugs such as nitrofurazone, furazolidone, and nitrofurantoin bear either hydrazide or hydrazone functions and compounds of this nature are known to possess various biological activities. 8 Hydrazones are therefore attractive as synthetic compounds with biological activities, and some hydrazone-based drugs such as dihydralazine, mitoguazone, nifuroxazide, and ferimzone show clear evidence of bioactive relevance. 9 The structures of the hydrazone-based drugs mentioned here are given in Figure 1.

Examples of hydrazone-based drugs.
Resistance to antibiotics by pathogenic bacteria occurs when bacteria acquire the ability to overcome the effects of antibiotics that were used previously to inhibit them.10,11 Antimicrobial resistance is an emerging worldwide health challenge, causing morbidities and mortalities even in hospital settings. Despite the development of conventional antimicrobials as solutions, there is a multidrug-resistant pattern in Gram-positive and Gram-negative bacteria, as well as in fungi, resulting in difficult-to-treat or even untreatable infections.10,12–14 Millions of people suffer from microbial infections, some of which are caused by resistant strains, accounting for millions of deaths worldwide each year. 15 Bacterial biofilms are responsible for approximately 80% of severe and recurrent microbial infections in humans, and microbial cells living within biofilms maybe 10–1000 times more resistant to antibiotics than their planktonic counterparts. 16 Microbial pathogens can evolve and develop resistance, usually aided by quorum sensing (QS)-mediated virulence factors and biofilm formation, and this requires a multidisciplinary approach involving antimicrobial molecules of synthetic and natural origin that can act on the pathogens by various mechanisms involving biofilms and QS.11,17,18 Bacterial biofilms are aggregates of bacterial cells attached to a surface and coated with a polymeric layer.19–21 They protect bacteria and allow them to survive in harsh environmental conditions.22,23 They can resist immune response of the host and are much more resistant to antibiotics and disinfectants.24,25 Bacteria in the biofilm are less sensitive to antibiotics and can be very resistant because of the polymeric layer, which forms a barrier, reducing or preventing the diffusion of antimicrobials.26,27 The dissemination of antibiotic resistance is usually accompanied by genetic changes including genetic mutations, genetic transfer of resistance genes through plasmids, and mutations of target genes. 28 There are different strategies for inhibiting biofilm formation, such as inhibition of formation of the polymeric layer or its degradation, prevention of the initial microorganism adhesion, prevention of microbial growth, or the interruption of communication between bacterial cells (quorum sensing).24,29–31 The search for more effective and safer antibiotic alternatives, whether herbal or synthetic, as well as new therapeutic and nonpathogenic agents that might act as nontoxic inhibitors of QS, is increasing.31–35 Therefore, there is an urgent need to develop new therapies that can treat bacterial infections and overcome the emergence of drug-resistant strains and disrupt bacterial cell-to-cell communication, known as quorum sensing, and eliminate biofilm formation. 36
This study presents the synthesis and characterization of a new diarylhydrazone derivative and the evaluation of its effects on microbial biofilms and quorum sensing. To understand, determine, and visualize the most likely interaction of the hydrazone with the protein as a QS inhibitor and the
Results and discussion
Chemical synthesis and characterization
The new diarylhydrazone
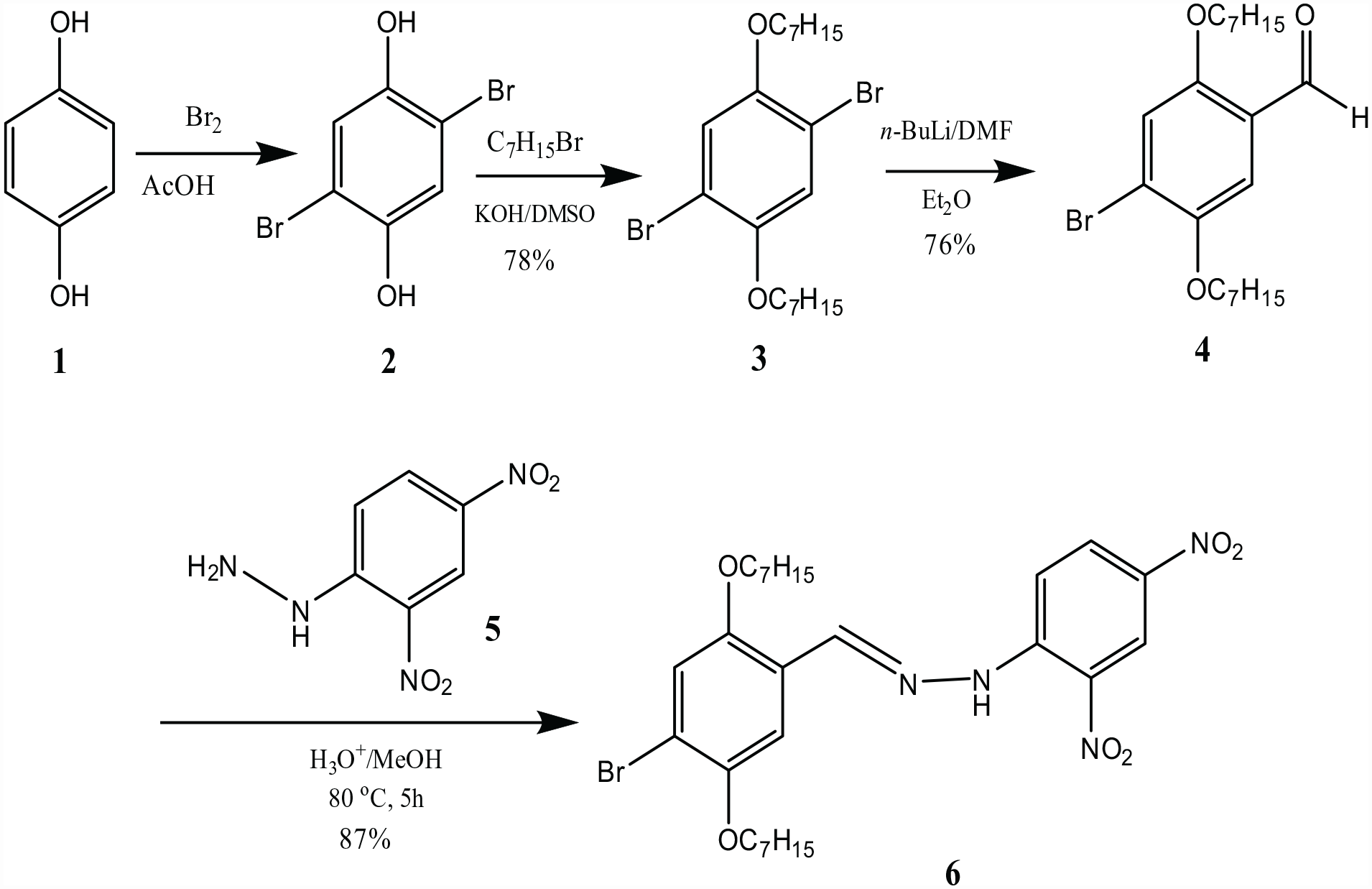
Synthesis of BHBANA (
Violacein inhibition and anti-quorum sensing activity of BHBANA
The molecule responsible for a pigment called violacein is a derivative of an indole that results from the condensation of two molecules of tryptophan, and it has numerous biological activities.
43
Quorum sensing inhibitory effects of BHBANA.

BHBANA:
Antimicrobial activity of BHBANA
The antimicrobial activity of BHBANA (
Antimicrobial and anti-biofilm activities of BHBANA.

BHBANA:
Anti-biofilm activity of BHBANA
Most pathogens within the host or in the environment are capable of undergoing an extracellular encapsulation, making them protected from and resistant to antibiotics, and their infections become chronic, persistent, and difficult to treat. Therefore, the design of new antibiotics capable of targeting and disrupting biofilms and also targeting all stages of biofilm formation within the drug discovery process is a useful strategy.
54
Compound
Swarming motility inhibition in P. aeruginosa PA01 by BHBANA
The early stages of biofilm establishment and the dispersion of cells from biofilms consist of bacterial cells moving toward the surfaces and then colonizing the surfaces through different movements or motilities such as swimming, swarming, and twitching.
58
Therefore, the inhibition of swarming motility could reduce the incidence of biofilm formation and surface colonization by bacteria and prevent contamination. The ability of compound
The positive effects of compound
Molecular docking analysis
Molecular docking contributes to improve the understanding of the interaction between the active sites of CviR and the BHBANA. This study targeted

A schematic 2D interaction diagram representing the ligand complexes with the

3D orientation of synthetic molecules: (a and c) the hydrogen bonds of the FAD and BHBANA (
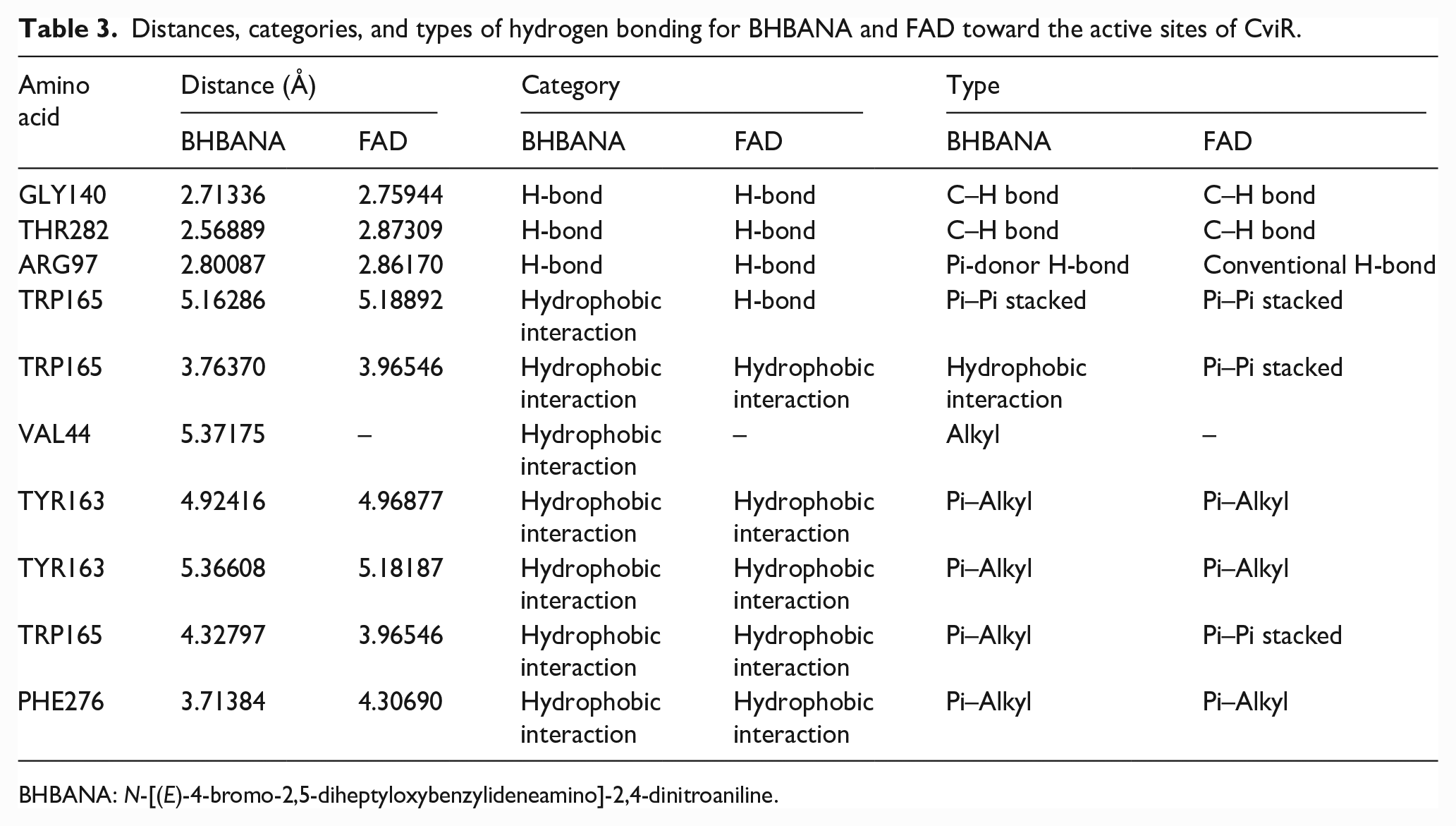
Distances, categories, and types of hydrogen bonding for BHBANA and FAD toward the active sites of CviR.
BHBANA:
As can be seen in Figure 2, the active sites of the target proteins were the Lys33, Leu40, Tyr192, Arg249, Arg97, Thr145, Arg105, and Phe251 amino acid residues, where the interactions with BHBANA were concentrated. The 3D orientation (Figure 3) reveals the hydrophobic sites and hydrogen bonds of both the BHBANA and FAD molecules. Similarities between the results reported in the literature for the FAD
63
and those for BHBANA are observed as strong hydrophobic interactions around Leu40, Val44, Val141, Tyr163, Tyr192, Ile202, Phe251, and Phe276. These interactions are attributed to the presence of the heptyl side chain in the BHBANA backbone. The results in Table 3 further show that the distances, categories, and types of hydrogen bonding for the two molecules under study (FAD and BHBANA) are convergent, if not similar. It should be noted that BHBANA has slightly smaller distances than FAD, which gives the protein–ligand complex it forms higher stability and robustness. In addition, hydrogen bonding and salt bridge interactions were detected in the active sites. It should be recalled that the interaction through hydrogen bonding is significant in inducing QS activity in the CviR protein.
64
From the previous docking visualization, the newly synthesized molecule adopts a very similar orientation in the active site to that revealed with FAD, which could play an important role in overcoming the QS system in
Conclusion
In this work, a new diarylhydrazone derivative was successfully synthesized in a good yield of 87%, and its structure was characterized using mass spectrometry and extensive NMR experiments. The newly synthesized hydrazine compound, BHBANA (
Experimental
Materials
For the synthesis, the reagents and solvents were obtained from Sigma-Aldrich or Fluka and were used without further purification. Thin layer chromatography (TLC) was performed on Merck 60 F254 silica gel plates. Luria-Bertani broth, nutrient broth, Mueller-Hinton broth, agar-agar,
Instrumentation
The melting points were determined on a Kofler-type apparatus. 1H and 13C NMR spectra were recorded in CDCl3 on a Gemini 300 MHz NMR spectrometer for
Synthesis and characterization
Synthesis of 1,4-dibromo-2,5-diheptyloxybenzene (3 )
KOH (48 g, 856 mmol) in anhydrous DMSO (300 mL) was stirred under nitrogen for 2 h at room temperature and 2,5-dibromohydroquinone
1
13
Synthesis of 4-bromo-2,5-diheptyloxybenzaldehyde (4 )
1,4-dibromo-2,5-diheptyloxybenzene
1
13
Synthesis of N-[(E)-4-bromo-2,5-diheptyloxybenzylideneamino]-2,4-dinitroaniline (BHBANA) (6 )
4-bromo-2,5-diheptyloxybenzaldehyde
1
13
Microbial strains
Two Gram-positive strains,
Determination of antimicrobial activity
MIC values of BHBANA (
Assays of the anti-biofilm activity of the test compounds
The anti-biofilm effect of BHBANA (

Bioassay for quorum sensing inhibition (QSI) on C. violacium CV026
The quorum sensing inhibition of BHBANA (
Evaluation of violacein inhibition in C. violacium CV12472
The test compound BHBANA was evaluated for its ability to inhibit the synthesis of violacein by

Inhibition of swarming motility on P. aeruginosa PA01
The determination of swarming movement in
Molecular docking studies and visualization
To explore the interaction between the targeted
Supplemental Material
sj-docx-1-chl-10.1177_17475198231184603 – Supplemental material for Synthesis of a new diarylhydrazone derivative and an evaluation of its in vitro biofilm inhibition and quorum sensing disruption along with a molecular docking study
Supplemental material, sj-docx-1-chl-10.1177_17475198231184603 for Synthesis of a new diarylhydrazone derivative and an evaluation of its in vitro biofilm inhibition and quorum sensing disruption along with a molecular docking study by Sameh Boudiba, Alfred Ngenge Tamfu, Karima Hanini, Ilhem Selatnia, Louiza Boudiba, Ibtissam Saouli, Paul Mosset, Ozgur Ceylan, Daniel Ayuk Mbi Egbe, Assia Sid and Rodica Mihaela Dinica in Journal of Chemical Research
Footnotes
Acknowledgements
The authors gratefully acknowledge Echahid Cheikh Larbi Tebessi University (Tebessa, Algeria), Larbi Ben M’Hidi University (Oum El Bouaghi, Algeria), Mugla Sitki Kocman University (Mugla, Turkey), and Johannes Kepler University (Linz, Austria), for supplying facilities to realize this investigation. Material, administrative, and technical support from the University of Ngaoundere, Cameroon; Mugla Sitki Kocman University, Turkey; and the Dunarea de Jos University, Romania are gratefully acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
