Abstract
Antibiotic resistance is a major health risk, and biocompatible polymers like polyvinylpyrrolidone and their derivatives are used in developing suitable antimicrobials. In this study, polyvinylpyrrolidone was reacted with potassium hydroxide and later coupled with benzoyl chloride (C7H5ClO) to produce polyvinylpyrrolidone 2. Polyvinylpyrrolidone 2 was reacted with 3-(dimethylamino)-1-propylamine to obtain polyvinylpyrrolidone 3. The polyvinylpyrrolidone derivatives were characterized using nuclear magnetic resonance and Fourier transform infrared spectroscopy. Polyvinylpyrrolidone 2 and polyvinylpyrrolidone 3 inhibited the production of violacein in

Keywords
Introduction
Functionalized monomers undergo polymerization, while reactive polymers can be subjected to postpolymerization modification. Both processes are used to create chemically diverse and bioactive polymer libraries, with scientists concentrating on the first strategy and little attention being paid to the latter strategy. 1 Postpolymerization has proven to be more effective for the production of macromolecules with improved properties. Polymers can undergo postpolymerization modifications through ring-opening reactions to give access to various derivatives with grafted functionalities unto the original scaffold.2,3 Such postpolymerization modifications can be achieved either by copolymerization or by subsequently converting the functional groups and side chains of the original polymer, which are chemo-selective or inert toward the conditions of polymerization.4,5 Various properties of polymers can be improved through postpolymerization hemi-synthesis, and this approach imparts characteristic properties that are difficult to achieve during polymerization by adding functionalities that confer the desired properties.6,7 In addition to techniques of controlled polymerization, postpolymerization modification of already synthesized polymers leads to the preparation of a library of macromolecules with diverse structures and functions from a single polymer scaffold.8 –10 Polymer functionalization via postpolymerization provides an easy and efficient means of introducing new groups or chemical units into the polymer skeleton to offer desired properties. The original polymer main chain is not usually affected by postpolymerization modification, indicating that modified polymers only differ in the side chains or functional groups introduced, giving room for investigating their influence on the polymer properties. 11
It is necessary to avoid unwanted reactions during the functionalization and imparting of defects in the polymer backbone by selecting or using selective and efficient modification reactions. Poly(N-vinylpyrrolidone), also known as polyvinylpyrrolidone or povidone (PVP), is obtained by radical polymerization of N-vinylpyrrolidone and usually modified chemically by graft copolymerization or other techniques. PVP and most of its derivatives are pH-stable, temperature-resistant, biodegradable, biocompatible, physiologically inactive, nontoxic, and chemically stable with many desirable properties and are often used in delivery systems of nutraceutical, biomedical, and pharmaceutical products.12 –14 Its chemical surface modification prevents nonspecific protein adsorption, making its derivatives promising antifouling surface modifiers. 15 PVP and its derivatives have the advantage of being soluble in water and a variety of organic solvents, including butanol, chloroform, and dichloromethane. PVP and its derivatives offer remarkable advantages with a range of chemical properties, including enhanced solubility, increased bioavailability, and even the introduction of the desired swelling tract for control or sustained release. 16 By altering the methyl iodide amounts, N-methyl quaternized PVP derivatives were produced in high amounts with variable quaternization degrees, providing compounds with unexplored optical and solvation properties. 17 Postpolymerization of PVP in previous studies afforded novel macromolecular complex systems with thio-semicarbazone and thiourea sites through polymer-analogous transformation. 18
Sometimes, postpolymerization modification is used to confer biodegradability on the polymers and improve their mechanical, optical, and biological activities.19,20 Antimicrobial properties are easily introduced in polymers through postpolymerization functionalization. 21 The surface groups of PVP may be modified chemically and the resulting materials utilized to create antibacterial surfaces. 22 The antimicrobial property of polymers can be an intrinsic feature, but biocide activity can be imparted on polymers by grafting active substances and functionalization of the polymers.23,24 Antimicrobial polymers are a very promising class of medicines for treating microbial illnesses with a greater ability to successfully combat bacteria than traditional antibiotics. 25 In a variety of approaches including antifouling, anti-quorum-sensing (QS), efflux pumps, and biofilm inhibitors, the antimicrobial effects of polymers are described either as independent antibiotic substances or in combination with established active components.26 –28 Unchecked use of antibiotics results in multidrug-resistant bacteria, which is among the principal human health concerns. Antibiotic-resistant bacteria, fungi, viruses, and parasites are defined by specific alterations that enable them to withstand the effects of antibiotics. 29 Finding novel antimicrobial treatments is suitable but not sufficient, because microbial resistance against them can arise; therefore, new modes of action can complement this, for example, targeting virulence factors. Currently, the majority of antibacterial substances target the physiological processes in bacteria to kill them. This high selective pressure on bacteria encourages various mutations and the formation of widespread drug-resistant forms. 30 It has been demonstrated that the QS mechanism of interbacterial communication, which is regulated by signaling molecules known as "auto-inducers," governs several functions between bacterial colonies. 31 Individual cells can communicate with one another through the QS regulatory process, which also aids in coordinating collective behavior. 32 QS network mediates virulence gene expression, establishment of biofilm, bacterial motility, and resistance to antibiotics.33,34 The majority of pathogenic gene expression, antibiotic efflux pumps, swarming motility, toxin synthesis, and biofilm formation are examples of microbial virulence and resistance mechanisms. 35 Therefore, looking for quorum-quenching compounds that can stop microbiological resistance and employing them to combat infections resistant to drugs is a good strategy.
Most commercial polymers are biologically inert and require postpolymerization surface functionalization to render them bioactive and increase their applications. This work involves the postpolymerization chemical modification of commercial PVP, commonly called polyvidone or povidone. The antimicrobial, anti-QS, and antibiofilm effects of the obtained derivatives are also reported.
Results and discussion
The synthetic route for the PVP derivatives is presented in Figure 1. In this study, PVP was reacted with potassium hydroxide (KOH) and later esterified with benzoyl chloride (C7H5ClO) to produce PVP2. PVP2 was reacted with 3-(dimethylamino)-1-propylamine to obtain PVP3. The characterization of the derivatives of PVP obtained from postpolymerization was done through Fourier transform infrared (FTIR) spectroscopy and 1H nuclear magnetic resonance (NMR) experiments. The schematic FTIR spectra are presented in Figure 2, while detailed annotated FTIR spectra are provided in supplementary material as Figure S1 (PVP), Figure S2 (PVP1), Figure S3 (PVP2), and Figure S4 (PVP3). The appearance of a broad band at 3584.28 cm-1 and the modification of the nature of the peak at 1650.50 cm-1 on the FTIR confirm the formation of PVP1 from PVP. This was also substantiated by the appearance of the N–H proton signal on the 1H NMR at δ

Scheme for the chemical modification of PVP into PVP2 and PVP3.

FTIR spectra of the synthesized PVP derivatives.
QS and violacein production inhibition effects
Violacein inhibition against

-: no inhibition.
Anti-quorum-sensing effects against

-: No inhibition.
The purple violacein pigment is produced by the Gram-negative bacterium
Inhibition of swarming and swimming motilities
Flagella-dependent motilities represent an important virulence factor of pathogenic bacteria, and they are controlled by QS processes.
Motility inhibition against

-: No inhibition
Swimming and swarming are flagellum-dependent motilities that are exhibited by bacteria such as
Biofilm inhibition and antimicrobial activity
Antimicrobial (MIC values) and antibiofilm effects at subinhibitory concentrations of the polymer derivatives against
MIC and biofilm inhibition activity results of polymer derivatives.
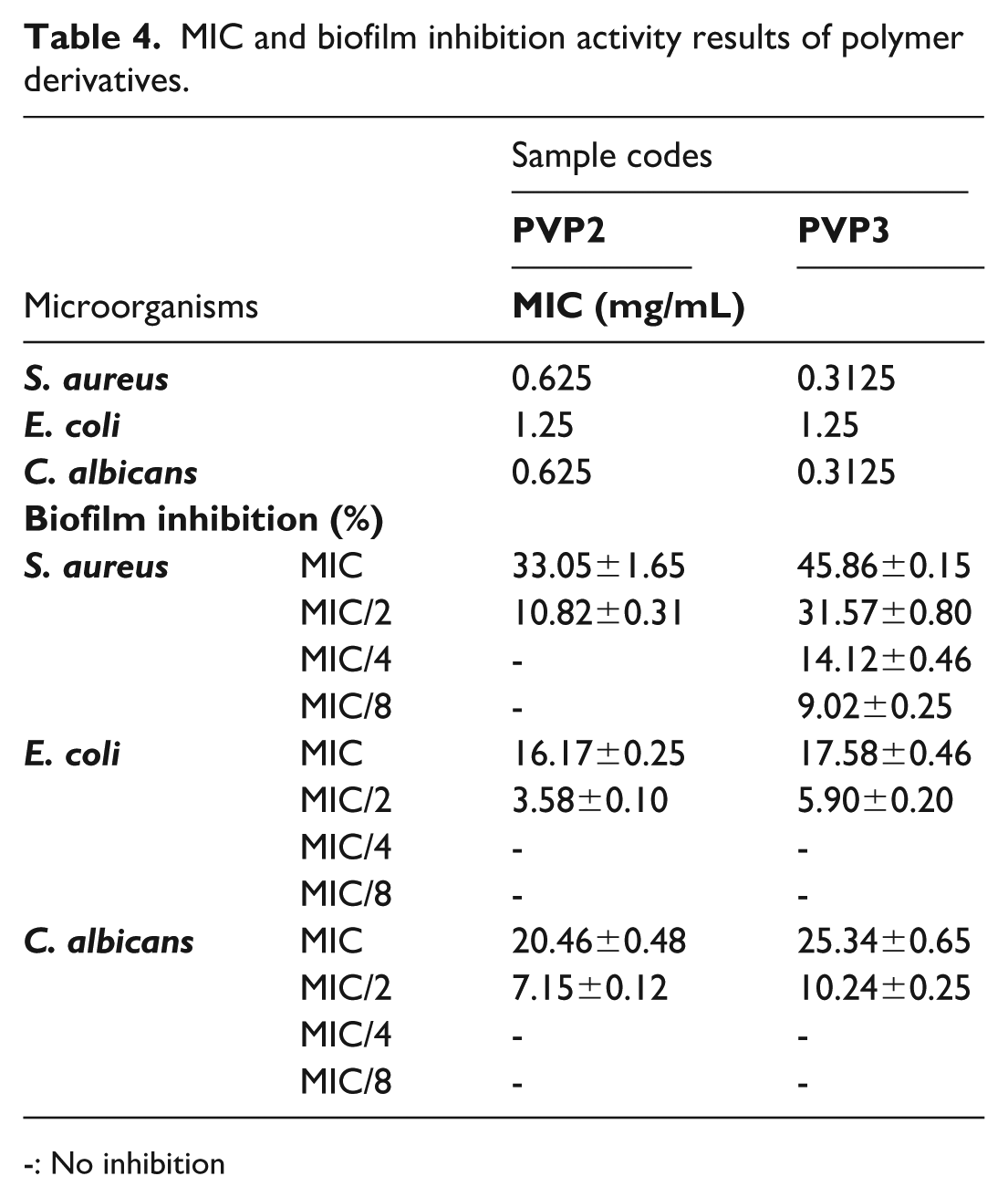
-: No inhibition
Motilities are crucial steps toward the passage from planktonic colonies to sessile ones and the establishment of biofilms. Biofilm is usually characterized as a cluster of cells of pathogenic microbes encased in a matrix predominantly composed of polysaccharides and usually adhering to a living or inert surface.44,45 PVP2 and PVP3 inhibited growth and biofilm formation against
Molecular docking results
The observed growth inhibition of
Hydrogen bonds, free binding energies, and number of closest residues to the docked compounds

From molecular docking results,
Figures 3–5 display the stable complexes showing 3D and 2D bonding interactions established between the docked

3D and 2D binding interactions of

3D and 2D binding interactions of

3D and 2D binding interactions of
Predicted absorption, distribution, metabolism, and excretion (ADME) and drug-likeness
The predicted ADME and drug-likeness properties of the monomeric units of

Bioavailability radars of

Boiled-egg model of

Predicted biological targets of
The bioavailability radar analysis on the polygon reveals that
Conclusion
The basis for copolymerization or postpolymerization of reactive polymer precursors is to obtain variable derivatives with different functional groups, properties, and applications. In this study, commercial PVP, which is a water-soluble, biocompatible, and biodegradable polymer, was reacted with KOH, leading to ring-opening, then esterified with benzoyl chloride to afford PVP2, and finally reacted with an amine to yield PVP3. PVP2 and PVP3 inhibited the growth of
Experimental Section
Chemicals and reagents
Chemical reagents such as anhydrous KOH (⩾99.95%), 4-(dimethylamino)pyridine (DMAP, ⩾99%), diisopropylcarbodiimide (DIC, 99%), and magnesium sulfate (anhydrous MgSO4, ⩾99.5%) were procured from Sigma-Aldrich. Solvents including dichloromethane (DCM, ⩾99.8%, Merck), N-N-dimethylformamide (DMF, ⩾99.8%, Merck), and anhydrous methanol (MeOH, ⩾99.8%, Sigma-Aldrich) of analytical grade together with triethylamine (TEA, ⩾99.5%, Merck), benzoyl chloride (⩾99%, Merck), 3-(dimethylamino)-1-propylamine (99%, Sigma-Aldrich), and PVP (MW 40000, CALBIOCHEM, USA) were used for the synthetic modifications. Luria-Bertani broth (LBA), tryptic soy broth (TSB), nutrient broth, Mueller-Hinton broth (MHB), Sabouraud dextrose broth, and agar were purchased from Merck. Ethanol (Merck), D-(+)-glucose (Merck), glacial acetic acid (Merck), and crystal violet (Merck) were used for biofilm inhibition assays. N-Hexanoyl-DL-homoserine lactone (C6-HSL, ⩾97%, Sigma-Aldrich), tryptone (Sigma-Aldrich), D-(+)-glucose (⩾99.5%, Sigma-Aldrich), kanamycin sulfate (Sigma-Aldrich), and sodium chloride (Sigma-Aldrich) were used in QSI and motility inhibition assays.
1H NMR and FTIR characterization
The polymer derivatives were synthesized as depicted in Figure 1. 1H NMR (CDCl3, 400 MHz) was recorded on a Bruker Avance 400 (AV400), and FTIR spectroscopy data were measured on a PerkinElmer FTIR spectrometer (UATR Spectrum II).
Chemical synthesis
Synthesis of PVP1
KOH (2.5 g) in 100 mL of distilled H2O was mixed with water-soluble PVP (2.5 g) dissolved in a two-neck flask, stoppered with septa, and placed under nitrogen in a Schlenk line. It was refluxed at a surface temperature of 140 °C for 15 h, cooled under tap water followed by the addition of DMF (95 mL), and then the solvents removed under vacuum at 80 °C with the aid of a rotary evaporator. 63 The product was dissolved in 50 mL of distilled H2O and extracted three times with 50 mL of DCM so that the polymer dissolves and leaves behind unreacted KOH and PVP in the aqueous phase. The organic layer was dried over anhydrous MgSO4, filtered, and DCM was evaporated on a rotary evaporator to afford PVP1 (2.3 g).
Synthesis of PVP2
PVP1 (2 g) was dissolved in 50 mL of DMF and mixed with TEA (10 mg) and DMAP (10 mg) and stirred for 2 h at 60 °C. 2.8 g of benzoyl chloride was added dropwise from a syringe for 5 min under nitrogen and the reaction mixture was kept for 16 h. The solvent was evaporated, and the product dissolved in distilled H2O (50 mL), and extracted with DCM three times. The resulting product was dried over anhydrous MgSO4, filtered, and DCM was evaporated on a rotary evaporator to afford PVP2 (1.8 g).
Synthesis of PVP3
PVP2 (1 g) in a flask was dissolved in DMF (40 mL), followed by the addition of a few drops of dilute HCl. 3-(Dimethylamino)-1-propylamine (4 mL) was introduced in the presence of DIC (200 µL) and reacted at 80 °C for 12 h. 20 mL of distilled H2O was introduced to the resulting reaction mixture and evaporated to dryness. The crude product was dissolved in distilled H2O (50 mL) and reextracted with DCM three times. The resulting product was dried over anhydrous MgSO4, filtered, and DCM was evaporated on a rotary evaporator to afford PVP3 (700 mg).
Strains of pathogenic microbes
The following microorganisms were used:
Measurement of MIC
Antimicrobial activity was expressed as MIC measured using the 96-well broth dilution method. 64 The MIC was considered as the lowest concentration of polymer derivatives at which no visible growth was observed. Fresh overnight cultures at 5×105 colony-forming units (CFU)/mL and MHB medium were employed. Microbial cultures were inoculated in microtiter plate wells in the presence of polymer derivatives at varying final concentrations (5, 2.5, 1.25, 0.625, 0.3125, 0.15625, 0.078125, 0.0390625, 0.01953, 0.009765 mg/mL). The microplates were incubated for 24 h at 37 ºC and read for MIC determination.
Biofilm inhibition measurement
The effect of the polymers at MIC and sub-MIC concentrations on biofilm formation against test microorganisms was determined by the microplate biofilm assay. 32 Overnight fresh bacteria cultures were seeded in TSB supplemented with glucose (0.25%) in the absence or presence of polymer derivatives at 37 ºC for 48 h. Wells without polymer derivatives (TSB+cells only) served as controls. Wells were rinsed with water to remove planktonic bacteria after incubation and stained for 10 min with 0.1% crystal violet solution. After removing the crystal violet, wells were filled with 200 μL of ethanol (for Gram-negative and Candida) or 33% glacial acetic acid (for Gram-positive) to dissolve the stained biofilms. 125 μL from each well was pipetted and transferred to sterile tubes, and the volume was adjusted to 1 mL with distilled water. Optical densities (ODs) were recorded at 550 nm (Thermo Scientific Multiskan FC, Vantaa, Finland). Percentage biofilm inhibition was deduced using the formula:

QSI against C. violaceum CV026
Determination of QSI was performed as described previously with little changes. 32 100 µL of overnight culture was added to 5 mL of warm molten Soft Top Agar (200 mL deionized H2O, 1.3 g agar, 1.0 g NaCl, 2.0 g tryptone). 10 µL of kanamycin sulfate and 20 µL of C6HSL AHL prepared at 100 µg/mL were introduced. The mixture was poured as an overlay on solidified LBA plates, and 5 mm diameter wells were made. Each well was filled with 50 µL of MIC and sub-MIC filter-sterilized polymer derivatives and incubated at 30°C for 3 days. A white or cream-colored halo around wells against a purple lawn of activated CV026 bacteria was an indication of QSI. Each experiment was done in triplicate, and the diameters of the QSI zones were measured.
Inhibition of violacein production against C. violaceum CV12472
The polymer derivatives were analyzed for their violacein inhibition potential against

Motility inhibition against Pseudomonas aeruginosa PA01
Inhibition of swimming and swarming motilities was evaluated as described elsewhere with minor changes.32,64 Swarming plates (0.5% of filter-sterilized D-glucose, 1% peptone, 0.5% agar, 0.5% NaCl) containing polymer derivatives (MIC, ½ MIC, ¼ MIC) were prepared and 5 µL of
Molecular docking study
The binding affinities of the monomeric repeating units of
ADME and drug-likeness properties
The predictions of ADME, drug-likeness, pharmacokinetics, and physicochemical properties of the monomeric units of
Statistical analysis
All experiments were repeated three times and results are mean values ± standard error of the means. The statistical differences between the test samples were analyzed through one-way ANOVA. Differences were considered statistically significant where
Supplemental Material
sj-docx-1-chl-10.1177_17475198251409909 – Supplemental material for Postpolymerization modification of polyvinylpyrrolidone and evaluation of anti-quorum-sensing, antimicrobial, and antibiofilm activities with molecular docking and absorption, distribution, metabolism, and excretion studies
Supplemental material, sj-docx-1-chl-10.1177_17475198251409909 for Postpolymerization modification of polyvinylpyrrolidone and evaluation of anti-quorum-sensing, antimicrobial, and antibiofilm activities with molecular docking and absorption, distribution, metabolism, and excretion studies by Alfred Ngenge Tamfu, Selahattin Bozkurt, Sameh Boudiba, Mehmet Kayhan, Ozgur Ceylan and El Hassane Anouar in Journal of Chemical Research
Footnotes
Acknowledgements
The Scientific and Technological Research Council of Turkiye (TUBITAK) is greatly acknowledged by A.N. Tamfu and S. Bozkurt for the grant (project code: 1059B212200092). The authors are grateful to the participating institutions, which are Usak University, Mugla Sitki Kocman University, Prince Sattam bin Abdulaziz University, and the University of Ngaoundere.
Ethical Considerations
Ethical approval is not applicable for the paper.
Statement of informed consent
There are no human subjects in this paper, and informed consent is not applicable.
Consent to Participate
The authors confirm that all of them participated in the research work of this paper.
Consent for Publication
The authors confirm that they give their full consent for publishing this paper.
Author contribution
Alfred Ngenge Tamfu: Conceptualization, Methodology, Investigation, Formal analysis, Data curation, Resources, Software, Visualization, Writing—original draft, Writing—review & editing. Selahattin Bozkurt: Conceptualization, Formal analysis, Data curation, Supervision, Resources, Visualization, Writing—original draft, Writing—review & editing. Sameh Boudiba: Formal analysis, Data curation, Writing—original draft. Mehmet Kayhan: Formal analysis, Data curation, Visualization, Writing-original draft. Ozgur Ceylan: Methodology, Formal analysis, Data curation, Supervision, Resources, Visualization. El Hassane Anouar: Investigation, Formal analysis, Data curation, Resources, Software, Visualization, Writing—original draft, Writing—review & editing. In addition, Alfred Ngenge Tamfu and Ozgur Ceylan performed the biological assays. All authors read, corrected, and approved the final copy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Data Availability Statement
The data generated in this study are available within the paper and its supplementary data files or upon request from the corresponding author.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
