Abstract
Plant extracts are used as cheap and environmentally friendly complexing and stabilizing agents for the green synthesis of nanoparticle with good properties. Zinc oxide nanoparticles were synthesized from Onopordum acaulon ethanol extract (zinc oxide nanoparticles–ethanol extract), aqueous extract (zinc oxide nanoparticles–aqueous extract), and dry plant powder (zinc nanoparticles–dry plant powder). The zinc oxide nanoparticles were characterized using ultra violet–visible spectroscopy, Fourier Transform Infrared spectroscopy, scanning electron microscopy, and X-ray diffraction which confirmed the formation of zinc oxide nanoparticles with sizes ranging from 11 to 17 nm and a Wurtzite crystalline phase. Cytotoxicity on Saccharomyces cerevisiae indicates that zinc oxide nanoparticles predominantly exhibit dose-dependent cytostatic effects with LC50 values exceeding 60 mg mL−1, suggesting good biocompatibility. Minimal inhibitory concentrations ranged from 0.312 to 2.5 mg mL−1. Zinc oxide nanoparticles from ethanol extract were most active against Bacillus subtilis and Escherichia coli, zinc oxide nanoparticles from aqueous extract exhibited the best antimicrobial activity against Staphylococcus aureus and Escherichia coli, while zinc oxide nanoparticles from dry plant powder had the lowest minimal inhibitory concentration against Bacillus subtilis. Nanoparticles synthesized with aqueous extract (zinc oxide nanoparticles–aqueous extract) showed highest biofilm inhibition against Bacillus subtilis, Escherichia coli, Salmonella typhi, and Candida albicans, while against Staphylococcus aureus, biofilm, the nanoparticles from hydroethanol extract (zinc oxide nanoparticles from ethanol extract) were most active. Zinc oxide nanoparticles from dry plant powder had the lowest antimicrobial activity against Chromobacterium violaceum 12472; it exhibited the best violacein inhibition. Zinc oxide nanoparticles from ethanol extract and zinc oxide nanoparticles from aqueous extract exhibited good quorum-sensing inhibition with anti-quorum-sensing zones of 21.5 ± 1.5 and 18.7 ± 0.9 mm, respectively, at minimal inhibitory concentration which reduced to 9.5 ± 0.1 and 8.3 ± 0.5 mm at minimal inhibitory concentration/8. All zinc oxide nanoparticles showed concentration-dependent inhibition of swimming and swarming motilities against Pseudomonas aeruginosa PA01. The green-synthesized nanoparticles exhibit promising antimicrobial properties and inhibited biofilm and quorum sensing, suggesting their application in reducing microbial virulence, pathogenicity, and resistance.

Keywords
Introduction
Antimicrobial resistance (AMR) occurs when bacteria adapt to and withstand the effects of antibiotics intended to eradicate them, usually due to misuse and overuse, making them persistently hazardous in agricultural and hospital settings.1,2 AMR is one of the crucial and global health burdens which endangers human life, with millions of annual deaths expected to substantially by 2050 if urgent measures are not taken. 3 There is a high demand for new antimicrobial targets and chemical entities that can function as antimicrobial medications since pathogenic microorganisms can adapt to resist the effects of some existing antibiotics.4,5 The world urgently needs new agents with novel modes of action and chemical structures to combat the pathogens that develop antimicrobial resistance or adopt combinations of multiple approaches to overcome AMR.6-8 Antimicrobial resistance as well as quorum sensing (QS) and biofilm formation are three components that interplay in microbial pathogenesis.9,10 QS and biofilm formation are believed to be some key mechanisms used by bacteria to develop resistance toward antibiotics since microbes make use of chemical molecules to emit and receive signals through QS that modulate biofilm development and trigger expression of resistant genes.10-12 Biofilm is a complex protective extracellular matrix that envelopes bacterial colonies, contributing to microbial virulence are aiding the pathogen in colonizing surfaces wile shielding them from antibiotics, stress and host immune system.13,14 QS involves the release and sensing of small signal which enable bacteria regulate the expression of some functions that coordinate behavior within the colony and provide information about cell density and certain gene expression. 15 Disrupting QS and biofilm formation using chemical substances from natural products, plant extracts and synthetic chemicals can be a suitable strategy to overcome AMR. 16 Nanoparticles have been shown to possess good antibiofilm and QS inhibition effects.17,18
Over the past decade, nanotechnology has experienced remarkable growth, driven by its wide-ranging applications in medicine, chemistry, and biotechnology. 19 This progress has opened new avenues in nanoscience, especially in areas such as drug and gene delivery, nanomedicine, and biosensing. 20 Recent breakthroughs have enabled the synthesis of nanoparticles with precise control over size and shape, leading to the development of powerful new biocidal agents. Often described as a “wonder of modern medicine” nanomaterials possess extraordinary antimicrobial efficacy, reportedly capable of killing up to 650 microbial cells, far surpassing the spectrum of conventional antibiotics. 21 Among these nanomaterials, zinc oxide nanoparticles (ZnO-Nps) have garnered significant attention due to their high specific surface area, biocompatibility, and multifunctional properties.22,23 Several methods have been used for the synthesis of ZnO-NPs with different properties and several applications. 24 The ability of ZnO-NPs to absorb and scatter ultraviolet light, along with potent antibacterial activity, makes them valuable in various domains including electrochemistry, medical devices, cosmetics, and the textile industry. ZnO can crystallize in three different structures, namely hexagonal wurtzite, cubic zinc blende, and rock-salt each associated with distinct physicochemical properties.25,26 Among these, the hexagonal wurtzite phase is the most stable and the most widely reported for antibacterial applications in addition to surface defects, high surface area-to-volume ratio, antimicrobial and antioxidant activities, along with its non-toxic nature and biocompatibility.22,27 However, despite their advantageous physicochemical and biological properties, ZnO-NPs may exhibit dose-dependent cytotoxicity toward mammalian cells, and their strong photocatalytic activity can induce the generation of reactive oxygen species, raising concerns about potential environmental toxicity when released into aquatic and terrestrial ecosystems.25,28 Given the increasing biomedical and environmental applications of ZnO-NPs, evaluating their cytotoxicity is essential to determine safe concentration thresholds and understand nanoparticle–cell interactions.27,29 The budding yeast Saccharomyces cerevisiae serves as an established eukaryotic model organism for toxicological studies due to its genetic conservation with higher eukaryotes, rapid growth, well-characterized stress-response pathways, and sensitivity to oxidative stress. 30 The spot dilution assay is a simple and reliable method for assessing growth inhibition and cell viability under stress conditions by comparing serially diluted cultures on solid media, allowing clear visualization of dose-dependent cytotoxic effects. 31 Although green synthesis approaches may modulate nanoparticle physicochemical properties through plant-derived capping agents, comparative cytotoxic profiling of ZnO-NPs synthesized using different extracts remains limited. Therefore, this study started with the evaluation of the cytotoxic activity of ZnO-Nps-dry plant (DP), ZnO-Nps-EE, and ZnO-Nps-aqueous extract (AE) on Saccharomyces cerevisiae using the spot dilution assay to determine differences in growth inhibition and toxicity patterns. One innovative approach to countering bacterial resistance involves targeting the QS mechanisms that regulate the expression of virulence factors in pathogenic microorganisms. Inhibiting these QS-mediated pathways offers a promising biocontrol strategy. 32
Onopordum acaulon is one of the plants traditionally used in Algerian medicine although it is understudied. Existing studies focus on its biological activities rather than exploring its potential value and application. Preliminary analyses confirmed that both ethanol and AEs of O. acaulon are rich in phenolic compounds and flavonoids, which can act as natural reducing and stabilizing agents in green nanoparticle synthesis and contribute to antimicrobial and QS inhibitory activity. This plant produces edible seeds and exhibits rapid propagation because a single flower bud can generate around 400 seeds, which are easily dispersed by the wind, enabling the plant to quickly colonize surrounding agricultural lands. 33 Since it is bountifully available, this study engaged on its valorization by using the plant powder and its extracts in synthesizing ZnO-Nps and using this nanoparticles as antibiofilm and QS inhibiting agents for possible application in eliminating AMR. The ZnO-Nps were synthesized using the plant dry powder, ethanol, and AEs and characterized using multiple methods.
Experimental
Plant collection and extraction
The aerial parts of O. acaulon were harvested from Tebessa, Algeria, in January 2024. After thorough washing, the plant materials were air-dried at room temperature, cut into small pieces, and ground using a blender. Part of the plant material (200 g) was used for direct synthesis of ZnO-Nps. In total, 200 g was extracted with 400 mL hydroethanol solution (70% ethanol) under ultrasonic conditions for 2 h. The process was repeated three times and the filtrates combined, filtered using Whatman No.1 filter paper, and evaporated to dryness using a Rota vapor to give the hydroethanol extract (EE). Water–ethanol mixture maximizes both yield and phytochemical diversity by leveraging the synergistic solvent properties. 34 The residues were boiled with distilled water, cooled, filtered, and evaporated to dryness using the rotary evaporator to afford the AE. Water is suitable for recovering water-soluble phenolics.
Green synthesis of zinc oxide nanoparticles
ZnO-Nps were synthesized using environmentally friendly approaches involving O. acaulon using dry powdered plant material, hydroethanol extract (EE), and AE. The synthesis followed the method reported elsewhere with specific modifications to accommodate both extract-based and dry plant-based procedures. 35 In each synthesis, a known amount of crude ethanol extract (100–1200 mg) or AE (100–600 mg) or dry plant powder (1–2 g) were dissolved in 20 mL of distilled water. Various concentrations were first used to determine optimal conditions for the synthesis. Separately, 40 mL of a 2 M zinc chloride (ZnCl2) solution was prepared as the zinc precursor because it is highly soluble, compatible with these procedures, and allows controlled formation of ZnO-NPs, as reported in the literature.27,35 The EE or AE solution at optimal concentration was added to the zinc chloride under constant stirring. The pH of the mixture was adjusted to 10 by the gradual addition of sodium hydroxide (NaOH), as recommended. The mixture was then heated under reflux in a water bath at 75 °C for 1 h under continuous magnetic stirring. After the reaction, the mixtures were allowed to cool and then centrifuged at 4000 rpm for 10 min to collect the solid nanoparticles. The resulting pellets were washed thoroughly with distilled water and centrifuged again. This washing step was repeated three times to ensure the removal of any residual salts or organic impurities. The purified nanoparticle samples were dried in an oven at 80 °C, followed by calcination in a muffle furnace at 500 °C for 2 h to obtain crystalline ZnO-NPs.
Characterization of the synthesized zinc oxide nanoparticles
To confirm the successful synthesis and evaluate the physicochemical properties of the ZnO-Nps, a set of complementary characterization techniques was employed. Each method provided specific insights into the structure, composition, and morphology of the nanoparticles.
UV–visible spectroscopy
UV-Vis spectroscopy was used to provide an initial indication of the possible formation of ZnO-NPs and to assess their optical properties by detecting characteristic absorption features in the UV region. This approach also allowed the comparison of extract concentrations by evaluating variations in peak intensity and wavelength, which may reflect differences in particle formation efficiency and size. 20
Fourier Transform Infrared Spectroscopy (FTIR)
Fourier Transform Infrared (FTIR) analysis was performed using a Bruker INVENIO-R spectrometer (serial number 329) and equipped with a Specac Quest™ multi-crystal accessory with a diamond ATR crystal. Spectra were collected over the range of 4000–400 cm−1 using OPUS software version 8.2 (Build 8.2.28, 2019-03-10). This analysis was conducted according to Jayachandran et al. 20 to identify the functional groups present on the surface of the nanoparticles. These functional groups, originating from the plant extracts, are likely responsible for the complexation of zinc ions and stabilizing the resulting nanoparticles. The presence of specific bonds (e.g. –OH, C=O, Zn–O) provided insight into the biomolecules involved in nanoparticle synthesis.
X-ray diffraction (XRD)
X-ray diffraction (XRD) was investigated using a Bruker D2 PHASER X-ray diffractometer. It was employed to investigate the crystalline structure, phase purity, and average crystallite size of the ZnO-NPs. In this study, diffraction patterns were recorded, and the average crystallite size was calculated using the Debye–Scherrer equation (1): 20
where D represents the crystallite size, λ is the X-ray wavelength, and β is the full width at half maximum (FWHM) of the diffraction peaks measured in radians; it should be noted that the peak broadening in X-ray diffraction patterns can also be influenced by lattice strains, crystal defects, and instrumental factors, in addition to crystallite size, and θ is the Bragg angle corresponding to the peak position.
Scanning electron microscopy (SEM)
Scanning electron microscopy (SEM) analysis was used to study the surface morphology and particle size distribution of the synthesized ZnO-NPs. The micrographs revealed the shape and surface texture of the particles and helped to visualize aggregation behavior and uniformity in different synthesis conditions. 36 SEM analyzed conducted using a Thermo Fisher Apreo 2 C scanning electron microscope, equipped with a field emission gun (FEG-Schottky cathode) and an Ultra High Resolution Gemini.
Cytotoxicity assay
The cytotoxic potential of green-synthesized ZnO-Nps namely ZnO-Nps-DP, ZnO-Nps-EE, and ZnO-Nps-AE was evaluated using the yeast spot dilution assay on S. cerevisiae to assess both cytostatic (growth-inhibitory) and cytocidal (cell-lethal) effects. Nanoparticle suspensions were prepared in sterile distilled water and sonicated briefly to ensure homogeneous dispersion. Serial dilutions were prepared to obtain final concentrations of 1.25, 2.5, 5, 10, 20, and 40 mg mL−1. Yeast cells were cultured overnight in YPD broth at 30 °C under constant agitation (150 rpm), harvested by centrifugation at 4000 rpm for 5 min, washed twice with sterile phosphate-buffered saline (PBS), and resuspended in sterile buffer to reach a final density of 2 × 107 cells/mL. Equal volumes of the yeast suspension and each nanoparticle dilution were mixed in sterile 96-well microplates and incubated at 37 °C for 30 min to allow direct interaction between ZnO-NPs and yeast cells. Untreated yeast suspensions mixed with sterile buffer served as the negative control, while cells exposed to hydrogen peroxide (optional positive control) may be used as a reference for cytotoxic stress. Following incubation, 3 µL of each treated suspension, including all nanoparticle concentrations and the control, was spotted in triplicate onto YPD agar plates supplemented with 2% glucose. The plates were incubated at 37 °C for 48 h, after which yeast growth was visually examined and documented. Spot size, colony density, and intensity were compared with the untreated control to estimate relative cell viability. A reduction in spot intensity without complete growth inhibition was interpreted as a cytostatic effect, whereas the absence of visible colony formation indicated a cytocidal effect. The classification of toxicity patterns was performed in accordance with previously described methodologies for yeast-based cytotoxicity screening. 37 In this assay, a nano-formulated version of the tested compound was employed to potentially enhance its bioavailability and cellular interaction. The compound was tested across six serially diluted concentrations: 80, 40, 20, 10, 5, and 2.5 mg mL−1. Following treatment and incubation, yeast growth was not only assessed visually on agar plates but also examined under a light microscope at 400× magnification. This microscopic evaluation enabled a more precise estimation of yeast cell proliferation compared with the untreated control, supporting the classification of observed effects as either cytostatic or cytocidal.
Microbial studies
The following microorganisms were used for the antimicrobial and antibiofilm assays: Staphylococcus aureus ATCC 25923, Bacillus subtilis, Escherichia coli ATCC 25922, Salmonella typhi ATCC 25922, and Candida albicans ATCC 10239. Pseudomonas aeruginosa PA01 was used in the swarming and swimming motilities inhibition assays while Chromobacterium violaceum CV12472 and C. violaceum CV026 were used in the violacein inhibition and anti-QS assays.
Determination of antimicrobial activity of nanoparticles
The various NPs were evaluated for their antimicrobial potential by determining their minimal inhibitory concentration (MIC) values through the broth dilution microtiter method. 38 The least concentration at which no visible microbial growth was observed was considered as the MIC and recorded in microgram per milliliter (µg mL−1). The test medium was Mueller–Hinton broth (MHB), and bacterial concentration of 5 × 105 colony-forming units (CFU)/mL density was considered. The wells of 96-well microtiter plates were filled with fresh bacterial cells cultured in MHB solutions and inoculated presence of fine powder of ZnO-NPs properly dispersed at final concentrations (5000, 2500, 1250, 625, 312.5, 156.25, 78.125, 39.0625, 19.53125, . . . µg mL−1). Wells containing media and bacterial culture without NPs served as controls. The plates were incubated for 24 h at 37 °C after which they were read for the determination of MIC.
Evaluation of antibiofilm activity of nanoparticles
The potential of the NPs to inhibit biofilms was evaluated at MIC and sub-MIC concentrations using 96-well microtiter combined with the crystal violet staining method.39,40 In total, 180 μL of Tryptose-Soy broth (TSB) supplemented with 0.25% glucose was introduced into wells of microplates followed by fresh cultures of S. aureus, B. subtilis, E. coli, S. typhi, and C. albicans (10 µL) and 20 µL of MIC and sub-MIC of NPs. The plates were inoculated at 37 °C for 48 h. Wells containing media and bacterial culture without NPs served as controls. At the end of the incubation period, the supernatant containing the planktonic colonies was carefully removed with a pipette leaving behind the sessile (biofilm) colonies which were further stained using 0.1% crystal violet solution for 10 min. The unabsorbed crystal violet solution was pipetted out, and 200 μL of ethanol or 33% glacial acetic acid was introduced into the wells and allowed for 30 min to dissolve the biofilms with absorbed crystal violet. The optical densities (OD) of the plates were read at 550 nm (Thermo Scientific Multiskan FC, Vantaa, Finland) and used in deducing the biofilm inhibition percentage using the formula:
Determination of violacein inhibition against C. violacium CV12472 by nanoparticles
Qualitative analysis of the reduction in violacein pigment production in C. violaceum 12472 by the NPs was evaluated using 96-well microplates. 16 In total, 10 µL of overnight-grown cultures of C. violaceum CV12472 adjusted to 0.4 OD at 600 nm and 20 µL of MIC and sub-MIC concentrations of NPs were introduced into each well of microtiter plates containing 170 µL of Luria–Bertani (LB). The plates containing LB broth and bacteria cells without NPs served as controls, and all plates were incubated for 24 h at 35 °C. The plates were visualized for the reduction in violacein pigment production followed by the measurement of the absorbance at 585 nm. The percentage inhibition of violacein production was calculated according to the following formula:
Determination of quorum-sensing inhibition (QSI) zones against C. violacium CV026 by nanoparticles
The QSI potential was evaluated as described previously. 41 Molten soft top agar was prepared by mixing 1.0 g sodium chloride, 2.0 g tryptone, and 1.3 g agar in 200 mL of deionized water. In total, 100 µL fresh cultures of overnight-grown C. violacium CV026 were seeded into 5 mL of the warm soft top agar, followed by exogenous addition of 20 µL of 100 µg mL−1 acylhomoserine lactone (AHL) as hormone source. The mixture was mixed and poured gently as an overlay over the surface of solidified Luria–Bertani Agar (LBA) plates. After solidification of the overlay, 5-mm-diameter wells were craved on the plates and filled with 50 µL of MIC and sub-MIC concentrations of the NPs and then incubated for 48 h at 35 °C. Formation of cream-colored halo around the wells on the purple lawn of normally grown CV026 bacteria indication of QSI. The diameters of were measured and reported in millimeters as QSI zones. Each experiment was done in triplicate.
Evaluation of swarming and swimming motilities inhibition against Pseudomonas aeruginosa PA01 by nanoparticles
The potential of the NPs in inhibiting bacterial movements was evaluated using swarming and swimming assays. The swarming inhibition was evaluated as described previously.5,17 Swarming plates were prepared with distilled water and 0.5% agar, 1% peptone, 0.5% NaCl, and 0.5% of filter-sterilized D-glucose together with different concentrations of NPs (100, 75, and 50 µg mL−1) solidified unto petri dishes. In total, 5 µL of overnight-grown fresh cultures of P. aeruginosa PA01 was point-inoculated at the center of each plate. Plates without NPs served as control. The plates were incubated in upright position at 35 °C for 18 h. The swarm fronts represented by the migration of bacterial cells were recorded and used to deduce the percentage swarming migration inhibition with respect to control plates. Swimming motility was evaluated as described elsewhere. 5 In total, 5 µL of P. aeruginosa PA01 was point-inoculated at the center of the swarming plates consisting of 1% peptone, 1.5% agar, 0.5% NaCl, and 0.5% D-glucose with different concentrations of NPs (100, 75, and 50 µg mL−1). The plates were incubated in upright position for 18 h at 37 °C. The reduction in swimming of bacterial cells in plates with NPs was recorded and used to deduce the percentage swarming migration inhibition with respect to control plates.
Results and discussion
Characterization of the zinc oxide nanoparticles
Using plant extracts in synthesizing NPs is considered as green synthesis since it is less costly and environmentally friendly, leading to nanoparticles with diverse structures and properties. The organic compounds present in the extracts primarily act as complexing, capping, and stabilizing agents during the synthesis process. The successful formation of ZnO-NPs using O. acaulon extracts suggests that the phytochemicals contained in the extracts interact with Zn2+ ions, facilitating controlled nucleation and growth of ZnO nuclei and preventing particle agglomeration, rather than inducing a reductive transformation to metallic zinc.
UV-Vis spectroscopy
UV-Vis spectroscopy was employed to monitor the optical properties of ZnO-Nps synthesized using dry powder and extracts of O. acaulon. This technique provides a rapid and non-destructive means to provide an initial indication of the possible ZnO-NP formation, particularly through the detection of characteristic absorbance peaks in the UV region. Variations in the position and intensity of these peaks reflect differences in nanoparticle size, crystallinity, and synthesis efficiency, and thus serve as key indicators for selecting optimal synthesis conditions. 42 All samples exhibited a characteristic absorption peak in the range of 310–370 nm (Figure 1), which indicates the possible formation of ZnO-NP. This aligns well with previously reported findings in the literature. 43
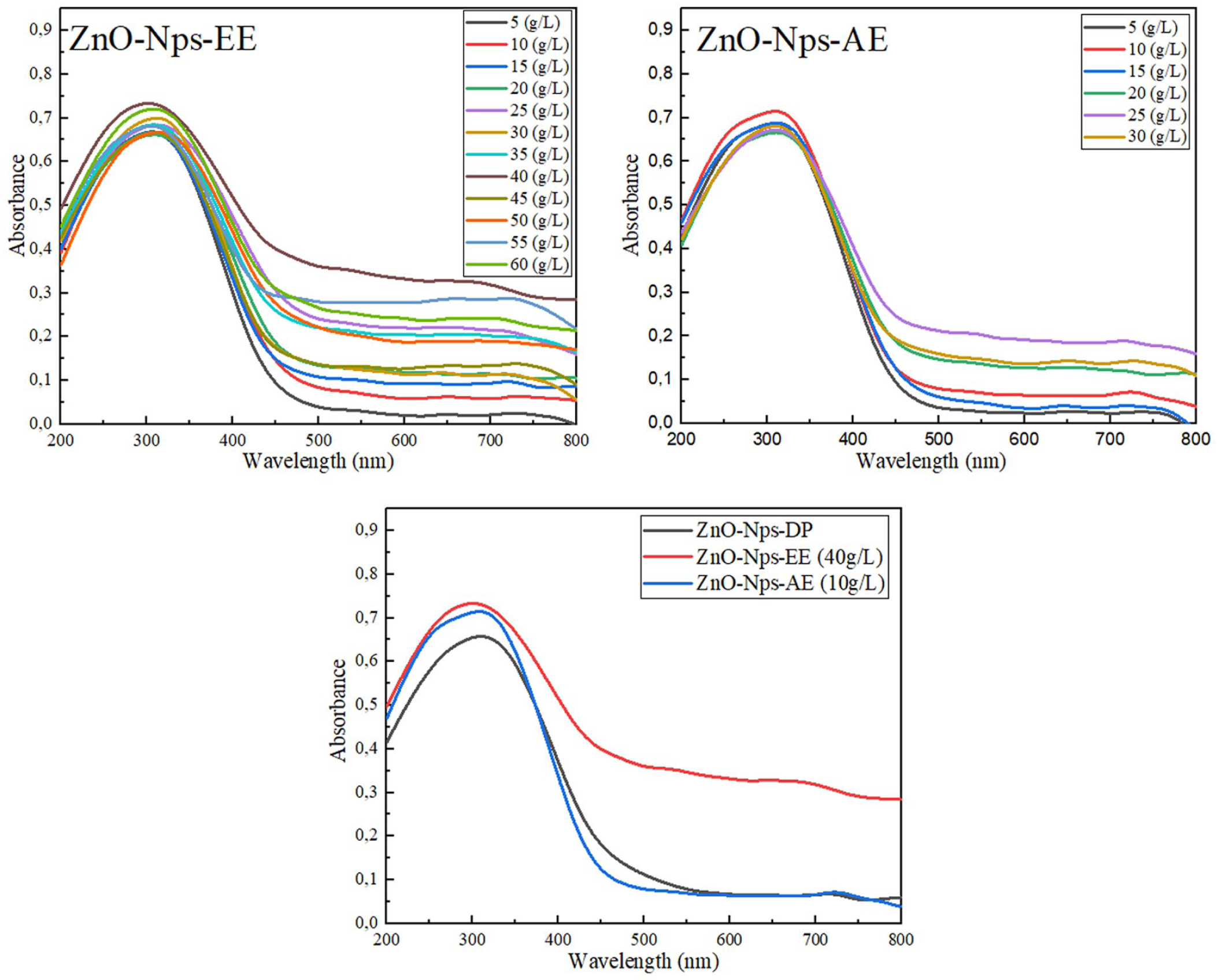
Ultra violet–visible absorption spectra of zinc oxide nanoparticles (ZnO-Nps) synthesized from Onopordum acaulon using hydroethanol extract (ZnO-Nps-EE), aqueous extract (ZnO-Nps-AE), and dry plant powder (ZnO-Nps-DP). ZnO-Nps-AE: Zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
In the extract-based synthesis, the ZnO-NPs synthesized using the ethanol extract (ZnO-Nps-EE) exhibited a pronounced and well-defined absorption peak around 373 nm, especially at the concentration of 10 g L−1. This sharp peak suggests high crystallinity and a more uniform particle size distribution. 44 Similarly, the aqueous extract (ZnO-Nps-AE) showed a peak in the same spectral region, with the best result also observed at 10 g L−1; however, its lower intensity indicates reduced synthesis efficiency or a smaller quantity of formed nanoparticles under equivalent conditions. The comparison between the three synthesis approaches, as illustrated in the third graph, showed that ZnO-Nps prepared from dry plant material (ZnO-Nps-DP) had the lowest absorbance, suggesting less effective nanoparticle formation under the same experimental setup. Overall, these UV-Vis findings indicate the successful synthesis of ZnO-NPs across all methods, with extract concentration significantly influencing both the yield and optical properties. Based on its sharp and intense absorbance peak, the EE at 10 g L−1 was selected as the optimal condition for further characterization and applications. Significantly, the spectrum showed no extra peaks, indicating that the ZnO-NPs produced by this quick and environmentally friendly method are highly pure. 22
Fourier Transform Infrared Spectroscopy (FTIR)
FTIR spectroscopy was employed to verify the presence of ZnO bonds in ZnO-NPs synthesized using dry powder and extracts of O. acaulon. The FTIR spectra for ZnO-Nps prepared with ZnO-Nps-EE, ZnO-Nps-AE, and ZnO-Nps-DP are presented in Figure 2. Distinct characteristic peaks were identified, indicating the involvement of bioactive compounds from the plant extracts in nanoparticle synthesis and stabilization. Peaks observed near 2900 cm−1 correspond to C–H stretching vibrations of aliphatic chains were negligible, indicating minimal contribution of aliphatic compounds from the plant. In the 1600–1650 cm−1 range, absorption bands corresponding to C=O and C=C stretching vibrations were observed with more pronounced and cleaner signals in the ZnO-NPs prepared using DP powder and EE, reflecting the effectiveness of these preparation methods in removing residual organic species. These bands are commonly associated with carbonaceous surface species or residual organic-derived structures formed during the thermal decomposition of plant biomolecules upon calcination. Before calcination, these phytochemicals likely contributed to the complexation of Zn2+ ions and the regulation of nucleation and growth of ZnO-NPs. Crucially, the sharp absorption band observed in the range of 400–600 cm−1 across all samples is attributed to the characteristic vibrational phonon modes of ZnO, confirming the successful synthesis of ZnO-NPs. 42 Variations in the intensity and position of these bands depending on the synthesis method suggest differences in surface chemistry and the nature of capping biomolecules. These FTIR findings are consistent with the UV-Vis absorption trends, which were employed as a complementary technique to compare ZnO-NP formation using different extracts, with the EE showing more pronounced features. Overall, the FTIR analysis indicates that phytochemicals present in O. acaulon extracts play a crucial role in the complexation of zinc ions and in the stabilization and growth control of the resulting ZnO-NPs.

Fourier Transform Infrared spectra of zinc oxide nanoparticles (ZnO-Nps) synthesized from Onopordum acaulon using ZnO-Nps-EE, ZnO-Nps-AE, and ZnO-Nps-DP. ZnO-Nps-AE: zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
X-ray diffraction
Figure 3 presents the XRD patterns of ZnO-Nps synthesized from O. acaulon aerial parts using ethanol extract, AE, and dry plant powder. The diffraction peaks observed confirm the formation of a crystalline hexagonal wurtzite ZnO structure, in agreement with standard JCPDS cards No. 01-089-7102, 01-089-7102, and 01-080-0074. Prominent peaks appear at 2θ values near 31°, 34°, 36°, 47°, 56°, 63°, 66°, 68°, and 69°, corresponding to the (100), (002), (101), (102), (110), (103), (112), (200), and (201) crystal planes, respectively, which are characteristic of pure ZnO.45,46 The absence of any extraneous peaks indicates that the synthesized nanoparticles are free of other crystals and exhibit high purity. The average crystallite sizes were calculated using the Debye–Scherrer equation, based on the FWHM of the most intense diffraction peaks. The resulting crystallite sizes were approximately 11.21, 14.60, and 17.84 nm for ZnO-Nps-EE, ZnO-Nps-AE, and ZnO-Nps-DP, respectively, as reported in Table 1. Since all values are below 100 nm, the results confirm the successful formation of ZnO-Nps. Interestingly, the slightly smaller crystallite size observed in the ethanol extract-derived nanoparticles (ZnO-Nps-EE) which may be attributed to a higher concentration of phytochemicals with strong capping and stabilizing capacities. Overall, these findings highlight the crucial role of extract composition in governing the nucleation and growth behavior of ZnO-NPs during green synthesis.

X-Ray diffraction patterns of synthesized zinc oxide using aerial parts of Onopordum acaulon.
The X-ray diffraction parameters and crystallite size of synthesized zinc oxide nanoparticles using Onopordum acaulon aerial parts.
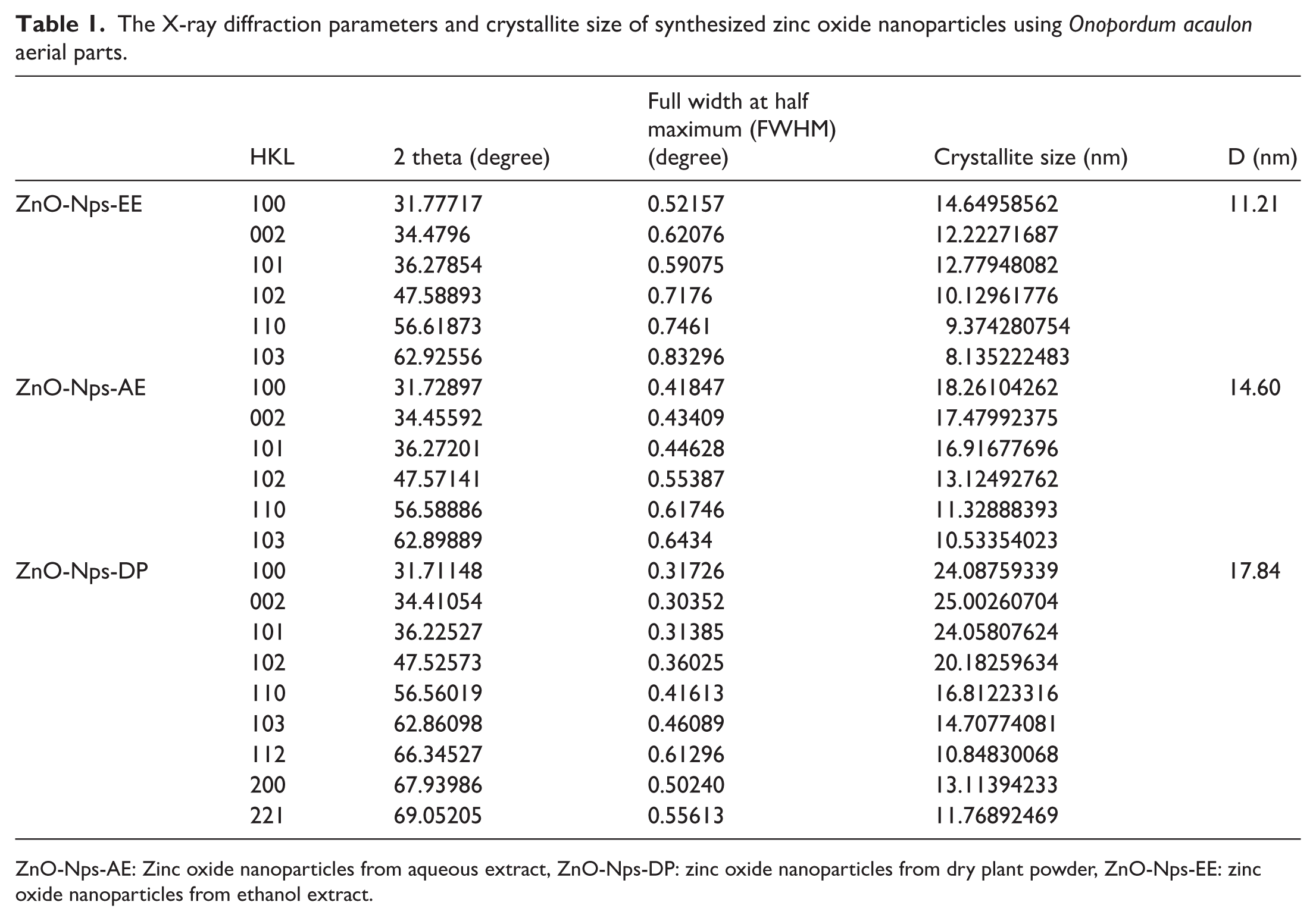
ZnO-Nps-AE: Zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
Scanning electron microscopy
Figure 4 presents SEM images and the corresponding energy-dispersive X-ray spectra (EDS) of ZnO nanoparticles from ethanol extract (ZnO-Nps-EE), aqueous extract (ZnO-Nps-AE), and dry plant powder (ZnO-Nps-DP). The SEM analysis reveals distinct morphological differences depending on the extraction method. ZnO-Nps-EE and ZnO-Nps-AE show predominantly quasi-spherical nanoparticles with noticeable degrees of aggregation. In contrast, the ZnO-Nps-DP sample displays well-organized rod-like and hexagonal crystalline structures, with significantly less agglomeration. The differences in morphology may be related to the nature of the bioactive compounds present in each extract and their interactions during the synthesis process, without making definitive claims about their exact role. The dry plant method, likely providing a more complex and sustained phytochemical environment, appears to facilitate the formation of anisotropic shapes like rods or plates. The EDS spectra for all samples display prominent peaks corresponding to zinc (Zn) and oxygen (O), indicating the formation of ZnO-NPs. Signals from organic components were not detected in EDS, which is consistent with the partial removal of organic matter during sample preparation. The atomic percentages of zinc and oxygen were relatively consistent across all three methods, supporting the reproducibility of the biosynthetic processes. Together, these findings affirm that not only the type of plant extract but also the physical form of the plant material plays a critical role in defining the morphology, purity, and potential functional properties of the synthesized ZnO-NPs.

Scanning electron micrographs and corresponding energy-dispersive X-ray spectra zinc oxide nanoparticles synthesized using Onopordum acaulon aerial parts.
Comparative analysis of plant-mediated ZnO-NP synthesis
To better contextualize the structural characteristics obtained in this study, a comparison with previously reported plant-mediated ZnO-NP syntheses is presented in Table 2. The comparison considers the plant source, crystalline phase, and particle size. As shown in Table 2, although several studies have reported plant-mediated ZnO-NP synthesis, most investigations have primarily focused on particle size and basic structural characterization. In contrast, this study not only confirms the formation of hexagonal wurtzite ZnO with controlled particle size but also introduces O. acaulon (a previously unexplored species) and compares both dry plant- and extract-mediated routes. Importantly, it further evaluates the synthesized nanoparticles for antibiofilm and QS inhibitory activities, highlighting the added functional value of this work.
Comparison of zinc oxide nanoparticles synthesized using different plant extracts reported in previous studies and the present work.

TEM: transmission electron microscopy; XRD: X-ray diffraction.
Cytotoxicity tests
The cytotoxic effects of green-synthesized ZnO-Nps-EE, ZnO-Nps-DP, and ZnO-Nps-AE were evaluated using a yeast model, and this has been described as a suitable means of evaluating cytotoxicity. 54 Yeast cells were used by employing both visual observation and microscopic examination at 400× magnification. Reference viability values were derived from prior calibration, providing a baseline for comparison (Table 3). Figure 5(a) shows the baseline yeast growth at different conditions, typical of a cytotoxicity assay, specifically a dose-gradient assay (dose–response assay) using cytotoxic cells.
The reference viability values.

Comparative cytotoxic effects and dose-dependent effect at serial dilutions (2−0 to 2−6) compared with untreated cells of (a) reference yeast viability used for calibration, (b) ZnO-Nps-DP, (c) ZnO-Nps-EE, and (d) ZnO-Nps-AE of ZnO-Nps-EE (CN, negative control). ZnO-Nps-AE: Zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
Yeast cells were treated with six serially diluted concentrations (80, 40, 20, 10, 5, and 2.5 mg mL−1) of each nano-formulated extract to assess dose-dependent effects, as shown in Figure 5.
ZnO-Nps-DP demonstrated a mild, dose-dependent cytostatic effect. Viability decreased to 84.33 ± 1.53% at 40 mg mL−1, while remaining above 97% at the lowest concentrations (Figure 5(b), Table 4). Microscopic examination confirmed the absence of cytocidal activity, and the extrapolated LC50 was >60 mg mL−1, reflecting low-to-moderate biological stress without lethal toxicity.
Effect of zinc oxide nanoparticles dry plant powder on cell viability at varying concentrations.

ZnO-Nps-EE exhibited minimal cytotoxicity. Even at the highest tested concentration (40 mg mL−1), yeast viability remained at 72.33 ± 1.53%, increasing to 97.00 ± 1.00% at 1.25 mg mL−1, approaching untreated control levels (100%) (Figure 5(c), Table 5). The observed growth inhibition was predominantly cytostatic, as no evidence of cell death was detected under microscopic observation. The extrapolated LC50 was estimated to be >100 mg mL−1, indicating high biocompatibility and negligible toxicological risk.
Effect of zinc oxide nanoparticles from ethanol extract on cell viability at varying concentrations.

ZnO-Nps-AE caused moderate growth inhibition at the highest concentration, with cell viability of 75.67 ± 2.52% at 40 mg mL−1 and ⩾88% at ⩽10 mg mL−1 (Figure 5(d), Table 6). The cytostatic nature of the effect was evident, as cells remained viable and capable of proliferation after incubation. The extrapolated LC50 of >80 mg mL−1 suggests limited biological impact under the experimental conditions.
Effect of zinc oxide nanoparticles from aqueous extract on cell viability at varying concentrations.

Comparative analysis across the three extracts indicates that ZnO-Nps-EE is the most biocompatible formulation, followed by ZnO-Nps-AE and ZnO-Nps-DP, with all formulations exhibiting dose-dependent inhibition of yeast growth without significant cytocidal activity (Table 7). This could be attributed to the well-documented antimicrobial mechanisms of ZnO-NPs including generation of reactive oxygen species and membrane depletion. 55 These findings are consistent with the literature reporting that green synthesis of ZnO-NPs mitigates cytotoxicity, likely due to the stabilizing and protective effects of biomolecules present in plant extracts, which reduce oxidative stress and enhance cell survival. 56
Estimated LC50 and toxicological interpretation of green-synthesized ZnO nanoparticles based on yeast cytotoxicity assay.

ZnO-Nps-AE: Zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
In summary, the yeast viability assay demonstrates that all tested ZnO-NPs are largely cytostatic rather than cytocidal, with high cell survival even at concentrations well above those typically used in biological applications. 57 This supports the potential for safe biomedical and environmental applications of these green-synthesized ZnO-NPs, while also providing quantitative LC50 benchmarks for future studies.
Antimicrobial activity
In this study, the green-synthesized ZnO-Nps using three different extracts of O. acaulon aerial parts are characterized as described above. The antimicrobial activity, reported in terms of MIC (mg mL−1) of the three different NPs, including ZnO-Nps-EE, ZnO-Nps-AE and ZnO-Nps-DP, are reported on Table 8. Overall, MIC values ranged from 0.312 to 2.5 mg mL−1. It is observed that ZnO-Nps-EE was most active against B. subtilis and E. coli with MIC of 0.156 mg mL−1. The ZnO-Nps-AE exhibited best antimicrobial activity against S. aureus and E. coli with MIC of 0.312 mg mL−1 while ZnO-Nps-DP had lowest MIC value of 0.312 mg mL−1 against B. subtilis.
Antimicrobial activity of zinc oxide nanoparticles.
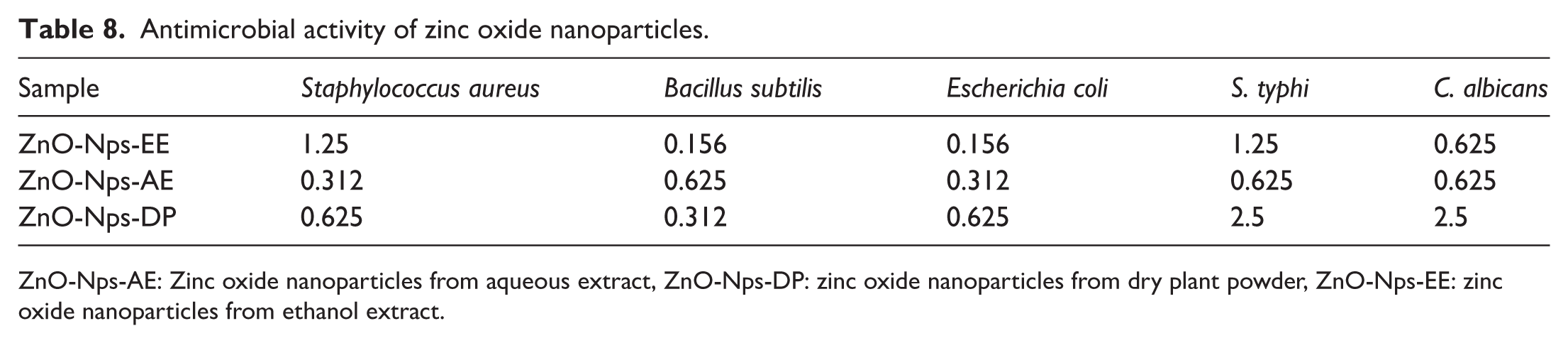
ZnO-Nps-AE: Zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
It should be noted that the shape, size, and properties of the ZnO-Nps are affected by the chemical constituents of the extract from which it is synthesized. This explains the differences in the antimicrobial properties of the ZnO-Nps. It is also believed that the antimicrobial activity of green-synthesized NPs involves a synergy between the metallic nanostructures and the phytomolecules from the plant extract used for the synthesis. 58 The plant extracts contribute to the determination of the antimicrobial properties of the resulting ZnO-Nps since they act as reducing, capping, and stabilizing agents. The results described here are in agreement with previous results which report the antimicrobial properties of ZnO-Nps. 59 ZnO-Nps are advantageous since they can exhibit effective antimicrobial activity against a wide range of microbes even at low concentrations (0.16–5.00 mmol L−1) and are low cost.60,61 It is reported that Gram-positive bacteria are generally less sensitive to ZnO-Nps than Gram-negative bacteria probably due to the lipopolysaccharides in their cell wall structure.61,62 In this study, this is observed for the Gram-positive bacteria B. subtilis and the Gram-negative bacteria S. typhi but the contrary is observed for S. aureus (Gram-positive) and E. coli (Gram-negative). ZnO has comparable effectiveness against S. aureus (Gram-positive) and E. coli (Gram-negative) and at the exponential growth phase. 63 However, the differences can be attributed in part to the differences in sizes and shapes of the synthesized ZnO-Nps which themselves depend on the chemical nature of the used extracts.64,65
Antibiofilm activity
The biofilm structure restricts the diffusion of conventional antimicrobials, making it difficult for them to reach the biofilm cells. However, ZnO-Nps may offer alternative to overcome this effect as their smaller sizes and modes of action may permeate biofilms. The percentage inhibitions of the various microbial biofilms by the synthesized ZnO-Nps are presented on Table 9. It can be observed that the ZnO-Nps from the dry plant powder (ZnO-Nps-DP) were the least active against biofilms. The nanoparticles from aqueous extract (ZnO-Nps-AE) showed be highest biofilm inhibitory potential ranging from 64.92 ± 1.07% (MIC) to 9.77 ± 0.36% (MIC/4) against B. subtilis, 79.12 ± 2.45% (MIC) to 8.78 ± 0.11% (MIC/8) against E. coli, from 63.33 ± 0.16% (MIC) to 14.60 ± 0.31% (MIC/4) against S. typhi and from 71.98 ± 0.87% (MIC) to 6.35 ± 0.41% (MIC/8) against C. albicans. Against S. aureus, the nanoparticles from the ethanol extract (ZnO-Nps-EE) exhibited the highest antibiofilm activity with percentage inhibitions ranging from 73.45 ± 0.99% (MIC) to 5.16 ± 0.43% (MIC/8). However, the antibiofilm activities of ZnO-Nps-EE and ZnO-Nps-AE remained relatively close. The results suggest that S. typhi formed the least susceptible biofilms.
Biofilm inhibition by nanoparticles.

MIC: Minimal inhibitory concentration, -: no inhibition, ZnO-Nps-AE: zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
The surface-to-volume of nanoparticles based on metals and their oxides makes them selective against animal and bacterial cells and able to provide antimicrobial and antibiofilm effects. 46 The antibiofilm of the synthesized ZnO-Nps can be therefore attributed in part to their surface-to-volume ratios resulting from their small sizes. Other mechanisms of action of ZnO-Nps against bacteria involve reduction in the expression of virulence genes and generation of reactive oxygen species (ROS) that trigger bactericidal effects by destroying physical structures, DNA synthesis, and metabolic pathways.61,66,67 It could be suggested that the ZnO-Nps have damaging effects on bacterial cell walls since ZnO-Nps usually generate ROS when in contact with bacterial proteins, causing cell membrane damage. 68 It should be noted that nanoparticles affect the bacterial cell walls seriously and interfere, therefore, in their capacity in establishing biofilms. Most metals and their nanoparticles can inhibit key microbial enzymes and generate ROS which results into the damage of cell membranes. 69 ZnO-Nps synthesized through green methods exhibited antibiofilm activities which were attributed to the production of reactive nitrogen intermediates that caused disruptions in cellular stress and metabolism. 70 The various nanoparticles in this study could be acting through various and multiple mechanisms against the tested microorganisms. The mechanisms of nanoparticles action on biofilms are thought to include the viability of individual microbial cells, including direct disruption of cell surface structures and oxidative stress linked to the production of ROS, as well as the impact on the synthesis of the exopolymer matrix and the QS system. 71 Because of their distinct physicochemical properties, NPs can enter biofilm matrices, enhance antibiotic delivery, and destroy structural integrity of the protective shields. 72 Summarily, most infections involved attached biofilms within protective matrices of extracellular polymeric materials which reduces access to antibiotics and making infections persistent and NPs are being engineered to address this problem.17,73 This indicates from the results that the nanoparticles synthesized in this research could be applied to overcome AMR involving biofilm formation.
Violacein inhibition and anti-QS activities
C. violaceum produces violacein pigment which is commonly used as an indicator in qualitative appraisal of bacterial cell-to-cell communication and QS. The percentage reduction in violacein production by the ZnO-Nps against C. violaceum CV12472 is reported in Table 10. Before the determination of violacein inhibition, MIC was determined against C. violaceum CV12472. ZnO-Nps-EE and ZnO-Nps-AE exhibited good antimicrobial activity against C. violaceum with MIC values of 19.53 and 39.06 µg mL−1, respectively. All ZnO-Nps showed 100% inhibition of violacein at MIC, and this reduced to 39.12 ± 1.00% and 24.56 ± 0.59% for ZnO-Nps-EE and ZnO-Nps-AE, respectively, at MIC/4 while percentage reduced to 21.23 ± 1.84% at MIC/8 for ZnO-Nps-DP. Although ZnO-Nps-DP had the lowest antimicrobial activity against C. violaceum 12472, it exhibited the best violacein inhibition compared with the other ZnO-Nps.
Inhibition of violacein production in C. violaceum CV12472 by nanoparticles.

MIC: Minimal inhibitory concentration, -: no inhibition, ZnO-Nps-AE: zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder; ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
The C. violaceum 12472 in nutrient-rich and aerated conditions produces violacein, whereas the mutant strain C. violaceum CV026 does not produce violacein under similar conditions unless an external N-AHL is supplied to it. If the presence of AHL and test sample the CV026 fails to produce violacein, then there is indication of QS inhibition by sample, and the diameter of the zones is measured. In this study, the QSI of the ZnO-Nps was measured and presented in Table 11. The MIC values against C. violaceum CV026 were 156.25 µg mL−1 (ZnO-Nps-EE), 312.5 µg mL−1 (ZnO-Nps-AE), and 625 µg mL−1 (ZnO-Nps-DP). The nanoparticles exhibited good QSI with inhibitory diameters at MIC of 21.5 ± 1.5, 18.7 ± 0.9, and 16.3 ± 1.2 mm for ZnO-Nps-EE, ZnO-Nps-AE, and ZnO-Nps-DP, respectively. ZnO-Nps-EE and ZnO-Nps-AE had higher QSI since they continued to inhibit QS with inhibitory zone diameters of 9.5 ± 0.1 and 8.3 ± 0.5 mm at MIC/8, respectively.
Quorum sensing inhibition zones in Chromobacterium violaceum CV026 by nanoparticles.

MIC: Minimal inhibitory concentration, -: no activity; ZnO-Nps-AE: zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles from dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
Microbial communication systems such as QS regulate many processes in bacteria, including the expression of antibiotic-resistant genes, which contribute to the development antimicrobial resistance. 74 This has motivated the search for new chemical entities that can interrupt QS processes in pathogenic bacteria. The effect of the ZnO-Nps on biofilm formation, violacein, and QSI indicates that they can disrupt cell-to-cell communication in bacteria and also prevent the establishment of recalcitrant biofilms. It should be noted that bacteria within established biofilms can continue to grow and reproduce and, as a result, re-establish communicative networks known as quorum sensing and nanoparticles have been proven to be effective in stopping this phenomenon. 75 A majority of traditional antibiotics treat the symptoms caused by the activities of planktonic bacteria, and those within biofilms continue to represent danger. The potential of the ZnO-Nps to reduce violacein and inhibit QS means that it can also stop many virulence factors. Inhibiting violacein production indicates inhibiting signal secretion, and inhibiting QS in the presence of AHL represents signal reception inhibition. This suggests that the ZnO-Nps can inhibit both signal production and reception. Biofilm formation, bioluminescence, secondary metabolite production, and virulence factors production in different bacteria are triggered by QS strategies. 76
Bacterial motility inhibition
Bacteria use various movements to displace themselves in media and on surfaces. Flagellated P. aeruginosa PA01 is a model organism for swarming and swimming motilities. The percentage inhibitions of swarming and swimming movements against P. aeruginosa PA01 by ZnO-Nps at 100, 75, and 50 µg mL−1 are reported in Table 12. The ZnO-Nps showed good concentration-dependent swarming and swimming inhibitions. In the swarming inhibition, the ZnO-Nps-EE was the most active with percentage inhibition of 90.8 ± 2.1% (100 µg mL−1) that reduced to 43.3 ± 0.8% (50 µg mL−1). In the swimming inhibition, ZnO-Nps-AE was the most active and exhibited percentage inhibition of 66.1 ± 1.7% (100 µg mL−1) that reduced to 42.5 ± 0.9% (50 µg mL−1).
Anti-motility effects against Pseudomonas aeruginosa PA01 by nanoparticles.

ZnO-Nps-AE: zinc oxide nanoparticles from aqueous extract, ZnO-Nps-DP: zinc oxide nanoparticles dry plant powder, ZnO-Nps-EE: zinc oxide nanoparticles from ethanol extract.
Swimming and swarming motilities are among the QS-mediated virulence factors of bacteria. Microbial movements and biofilm formation are both mediated by chemical sensing. 77 The inhibition of swarming and swimming movements by the ZnO-Nps indicates that they can reduce the incidence and spread of bacteria and this supplements biofilm inhibition as well. This is because before the establishment of biofilms, bacteria use various motilities to colonize surfaces. Swimming and swarming motilities are crucial in dispersal of bacteria and surface occupation in various environments and create a clinical problem because movements contribute to the resistance to antibiotics. 78 The development of chemical inhibitors of swarming and swimming including nanoparticles is, therefore, necessary. Swimming and swarming motilities aid bacteria to displace toward favorable environments and colonize desirable niches and contribute to symbioses, pathogenesis, initiation, and development of resistant biofilms. 79
Conclusion
Numerous studies have focused on the green synthesis of nanoparticles using different plants extracts and yielding NPs with various properties and applications. This work valorized the plant O. acaulon by applying its extracts in ZnO-NP synthesis. The synthesized ZnO-NPs (Wurtzite phase) were characterized using UV-Vis spectroscopy, FTIR, XRD, and SEM, providing insights into their optical properties, chemical structure, crystallinity, and morphology. In addition to the syntheses and structural characterization, assessment of cytotoxicity in a yeast model revealed that all ZnO-NPs exerted primarily cytostatic effects in a dose-dependent manner, maintaining high cell viability with LC50 values above 60 mg mL−1, highlighting their favorable biocompatibility. The ZnO-Nps also exhibited antimicrobial and antibiofilm properties against S. aureus, B. subtilis, E. coli, S. typhi, and C. albicans. Furthermore, the synthesized nanoparticles exhibited the ability to inhibit bacterial QS which involves cell-to-cell communication system linked to microbial virulence and pathogenesis. The ability of the ZnO-Nps in inhibiting microbial growth and QS-mediated processes such as violacein production, biofilm, swarming and swimming motilities indicates the suitable therapeutic potential of the biogenic ZnO-Nps. Taken together, these findings position O. acaulon as a promising, novel plant source for green nanotechnology. The study not only contributes to the sustainable development of nanoparticle synthesis but also opens up new perspectives for the biomedical and pharmaceutical applications of ZnO-based nanomaterials derived from underexplored natural resources.
Footnotes
Acknowledgements
The authors are grateful to Echahid Cheikh Larbi Tebessi University and Mugla Sitki Kocman University, for infrastructural, material, administrative, and technical support.
Ethical consideration
There is no research using human or animal subjects in this article.
Consent to participate
Every author has consented to take part.
Consent for publication
The final article has been seen by all authors, who have approved its publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The main text of the manuscript contains all of the data created or examined during this investigation, and the authors will make them available upon request.



