Abstract
Long-wavelength fluorescent sensors with large Stokes shifts show useful applications in chemical biology and clinical laboratory diagnosis. We have recently reported [4-(4-{[3-(4-boronobenzamido)propyl]carbamoyl}quinolin-2-yl)phenyl]boronic acid that can selectively recognize
Keywords
Introduction
As luminescent materials, organic compounds with good optical properties and biocompatibility1–3 are widely used in various research fields such as in light-emitting diodes, fluorescent sensors,4–6 and biosensors.7–9 Due to the hydrophobic conjugated system, traditional fluorescent molecules generally have a large emission wavelength and a high luminous efficiency in a single-molecule dispersion state. But when the molecules are in a state of aggregation, intermolecular interactions are enhanced, and the non-radiative transition is increased. The luminescence ability is weakened or even disappears, and this phenomenon is named “aggregation-caused quenching” (ACQ).10,11 ACQ tends to limit the applications of organic light-emitting materials. To avoid the ACQ phenomenon, the development of compounds with strong luminescence in the aggregated state as fluorescent sensors has become an interesting topic of research.2,12
In 2001, Tang reported 13 that a silicon heterocyclic pentadiene compound had weak fluorescence in the solution state, and that the fluorescence intensity increased sharply along with the turbidity due to a change of the solvent’s polarity leading to compound aggregation, and while the quantum efficiency increased remarkably. Subsequently, this phenomenon was named aggregation-induced emission (AIE).14,15 The restriction of intramolecular rotation (RIR) was identified as the main cause for enhancing the fluorescence. 16 When dissolved in a suitable solvent, the benzene ring undergoes intramolecular rotation with its single bond between the other rings as an axis, consuming excitation energy. However, when the internal rotation of the molecule is limited in the aggregate state, the benzene ring rotation amplitude is greatly reduced, and energy is released in the form of light. 17 Typical AIE compounds are mainly tetraphenylethylene, distyrylfluorene, hexaphenylsilole, and so on.18–21 Fluorescent sensors are usually constructed using these structures as the parent nucleus in order to recognize analytes and to generate biological images.
In addition to chemical synthesis intermediates and enzyme inhibitors,
22
boronic acids can be used as a biochemical tool for various purposes, including interference signaling pathways and cell delivery systems.
23
Moreover, due to the characteristics of the interaction of boronic acid with
2-(4-Boronophenyl)quinoline-4-carboxylic acid (PBAQA) (
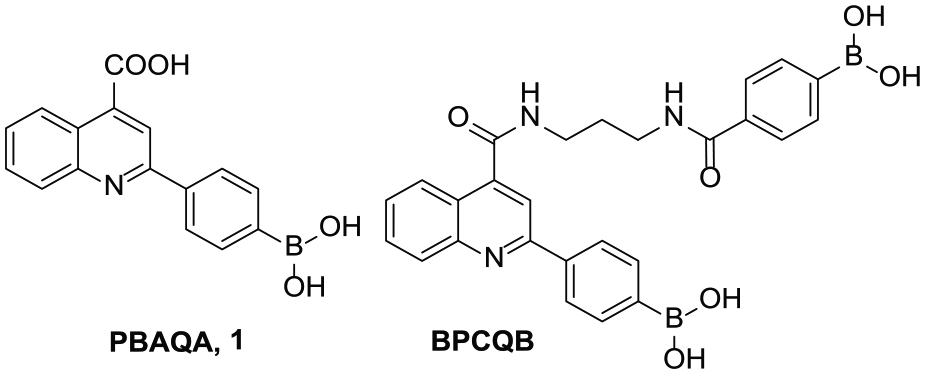
The structures of PBAQA and BPCQB.
In this paper, we describe the synthesis of novel diboronic acid compounds

Synthetic route to the target compounds: (i) DMT-MM, rt, 20 h; (ii) (Boc)2O; (iii) HCl, rt, 18 h; and (iv) DMT-MM, rt, 20 h.
Results and discussion
The synthetic procedure for 2-(4-boronophenyl)quinoline-4-carboxylic acid was based on the previously reported synthetic method.30,31 In this work, a highly efficient condensing agent, 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride (DMT-MM), was used as a condensing agent for the amide formation. This condensing agent is cheap and efficient. In addition, the low toxicity and low boiling point of the alcohol used as the solvent and the simple post-treatment of the reaction are advantages. There was no irritating odor during the reaction and fewer by-products formed. After the reaction was complete, water was added to the reaction mixture to produce a precipitate. The crude product was filtered and washed with water. Furthermore, direct recrystallization generally resulted in a product of higher purity. The method avoided the use of cumbersome column chromatography and overcame a major problem in the purification process of the diboronic acid.
Fluorescent properties of the diboronic acid compounds
The maximum emission wavelength of

Fluorescence spectra of compounds
Diboronic acid AIE effects
At present, the most common method for identifying whether an organic compound has an AIE effect is to add a poor solvent to a good solvent of a luminescent compound. The fluorescent compound is insoluble in the poor solvent. When the content of the poor solvent reaches a certain level, it will cause aggregation or even precipitation of the fluorescent compound. An organic luminescent compound with the AIE effect is weakly fluorescent in a good solvent. The fluorescence intensity was greatly enhanced when the compound aggregated. 32 PBAQA has a strong fluorescence in methanol and the fluorescence intensity did not change much with the change of water content. When the water content exceeded 50%, the fluorescence intensity decreased (Figure 3(a)). A solution of PBAQA with different water content could not be observed clearly aggregation or fluorescence with the naked eye under an ultra-violet (UV)-lamp (365 nm), which indicated that PBAQA did not have AIE phenomenon (Figure 3(b)).
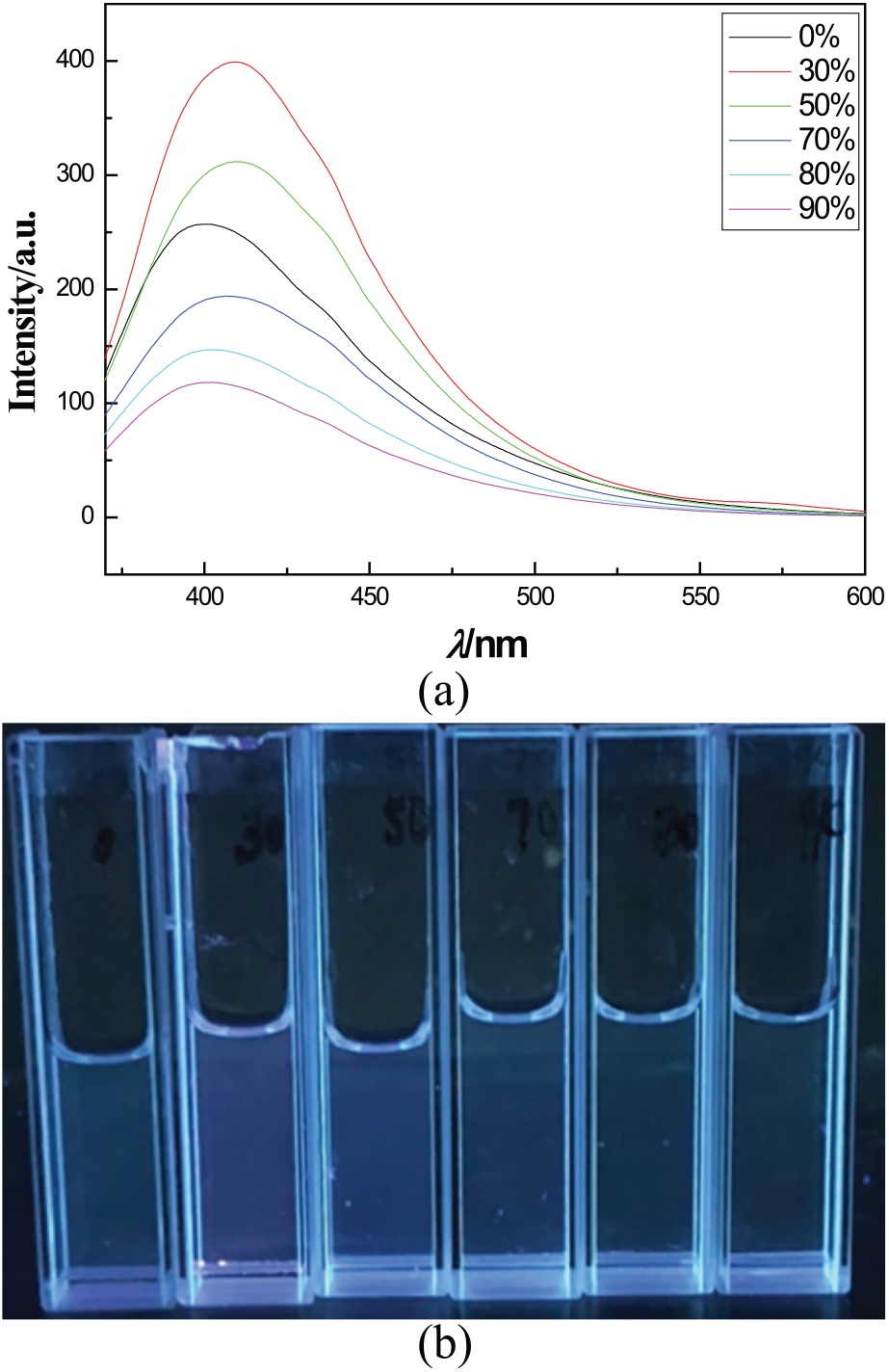
(a) Fluorescence spectra of
Compounds

Fluorescence spectra of

Photographic images of
The AIE of
Subsequently, time-resolved fluorescence measurements were introduced to investigate the AIE effect. The luminescence lifetimes of

Time-resolved fluorescence decay profile of
Time-resolved fluorescence decay parameters of 90% H2O–MeOH and pure MeOH solution. a

The solvent effect
The solvent effect of the compounds was further investigated. Taking

(a) Fluorescence spectra of compound
Stokes shifts of compound

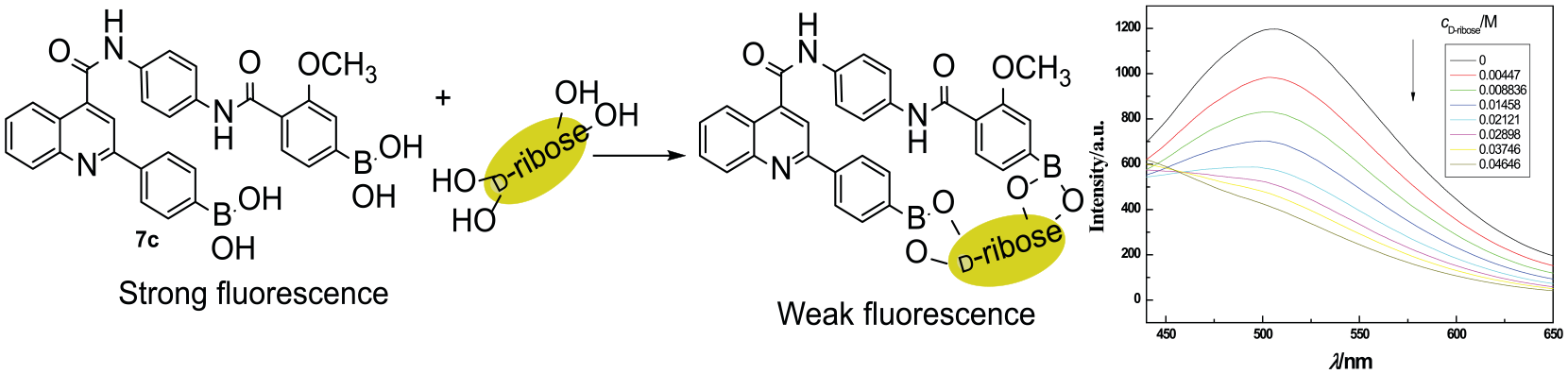
Selective fluorescence detection of carbohydrates
Boronic acids can combine with carbohydrates to produce different fluorescence changes. Therefore, boronic acids can be used for fluorescent sensors of carbohydrates. A stock solution (10–3 M) was prepared using methanol as the solvent. Then 1 mL of the stock solution was diluted to 20 mL using 0.1 M phosphate buffer (pH = 7.4) to give a blank sensor solution (5 × 10−5 M). A carbohydrate-sensor solution was prepared by adding the carbohydrate to the blank sensor solution (0.15 M). The blank sensor solution (1 mL) was added to a cuvette, and then different volumes of carbohydrate-sensor solution were added to obtain different concentrations of the carbohydrate-sensor solution. The fluorescence spectra were subsequently recorded.
In order to study the ability of compounds

Changes in the hydrodynamic diameters of
Interestingly, when the compounds were combined with

Variation of the fluorescence intensity of
The binding properties of

(a) Fluorescence response of
The reciprocal of fluorescence intensity quenching showed a good linear relationship with the reciprocal of
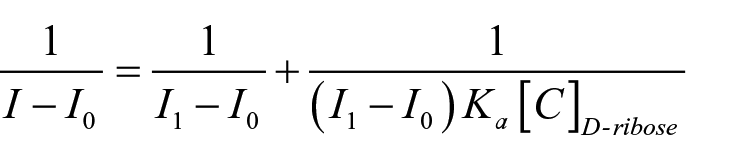
where
The binding constant (
Conclusion
Although their water solubility is good, PBAQA and its derivatives have a short emission wavelength of about 395 nm and demonstrate no AIE effects. Short emission wavelengths impose many limitations on future applications. In this work, novel diboronic acid compounds
Experimental
Materials
Apparatus
Fluorescence spectra were measured using a fluorescence spectrometer (F-280, China). Time-resolved fluorescence decay profile spectra were obtained using a high-resolution spectrofluorometer (FLS980; Edinburgh Instruments, UK) equipped with a 500 W Xenon lamp as an excitation source. The NMR spectra were recorded on a Bruker AM-600 spectrometer (Billerica, MA, USA) with tetramethylsilane as the internal standard. Mass spectra were obtained with an Agilent Trap VL LC/MS spectrometer (Santa Clara, CA, USA). High-resolution mass spectra (HRMS) were recorded on an Agilent 1290LC-6540 Accurate-Mass Q-TOF using electrospray ionization (ESI).
Synthesis
Supplemental Material
Supplementary_Material – Supplemental material for 2-(4-Boronophenyl)quinoline-4-carboxylic acid derivatives: Design and synthesis, aggregation-induced emission characteristics, and binding activity studies for D-ribose with long-wavelength emission
Supplemental material, Supplementary_Material for 2-(4-Boronophenyl)quinoline-4-carboxylic acid derivatives: Design and synthesis, aggregation-induced emission characteristics, and binding activity studies for D-ribose with long-wavelength emission by Guiqian Fang, Hao Wang, Zhancun Bian, Guimin Zhang, Min Guo, Zhongyu Wu and Qingqiang Yao in Journal of Chemical Research
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Innovation Project of Shandong Academy of Medical Sciences. This work was supported by the National Natural Science Foundation of China (grant no. 21801158), Shandong Academy of Medical Sciences Foundation (grant no. 2018-17), and the Graduate Instructor Guidance Ability Improvement Project of Jinan University (grant no. JDYY1804).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
