Abstract
Novel chiral fluorescent sensors are synthesized from a dibromide containing a tetraphenylethylene moiety and enantiomerically pure amino alcohols and an amine. The sensors are applied for the chiral recognition of a wide range of chiral carboxylic acids and related derivatives.
The synthesis of several novel aggregation-induced emission sensors and their application in chiral recognition are reported. The results suggest that sensors
Introduction
Chiral recognition is an important process in the natural world and plays a vital role in asymmetric synthesis, chiral drug discovery, catalyst screening, and many other aspects.1–6 However, conventional enantiomeric analyses not only usually need expensive instruments or complex chiral reagents, but they are also time-consuming in practice. Therefore, it is still important to develop simple, highly efficient, and low-cost methods for chiral recognition, such as chiral fluorescent sensors for the enantioselective discrimination of enantiomers.
In 2001, Tang and co-workers observed an uncommon and unconventional phenomenon in which some compounds with unique structures are almost non-fluorescent when dissolved in an organic solvent, but which become highly emissive in the aggregate state, a process known as aggregation-induced emission (AIE) or aggregation-induced emission enhancement (AIEE). 7 It is promising that AIE compounds not only solve the aggregation-induced quenching (ACQ) problem without causing any adverse effects,8,9 but are also stable and highly selective fluorescent sensors for proteins,10,11 DNA, 12 sugars, 13 metal ions,14,15 biological anions, 16 cyanide detection, 17 drug carriers, 18 cell imaging,19,20 and explosive detection.21,22 According to recent research achievements related to chiral recognition, the conclusion is that there is a simple and effective method to obtain excellent chiral sensors via the combination of one molecule having AIE character with another molecule having a chiral center.23–28 Unfortunately, for chiral carboxylic acids, very few sensors have been reported, and they are required to be tested at a large concentration.4,24,26
Herein, in continuation of our work on enantioselective discrimination,29–31 several new chiral sensors were de-signed, synthesized, and fully characterized. Their fluorescent response behaviors to various chiral substrates were analyzed by fluorescence spectroscopic methods. In addition, morphology studies for sensors were analyzed with various microscopic techniques, including transmission electron microscopy (TEM) and fluorescent inverted microscope analysis.
Results and discussion
Synthesis of the sensors
Phenylglycinol and α-methylbenzylamine are cheap and common quenchers that reduce the fluorescence intensity of fluorophores.
1
Tetraphenylethylene (TPE) and its derivatives are widely used as excellent fluorophores due to their facile synthesis and easy modification. We combined a known TPE derivative with two nitrogen-containing chiral auxiliaries in order to provide novel chiral sensors. Treatment of TPE derivative

Synthesis of sensors.
Photophysical studies
These chiral products were soluble in most organic solvents such as chloroform, tetrahydrofuran (THF), dimethyl sulfoxide (DMSO), and 1,2-dichloroethane, but were insoluble in petroleum ether and water. As expected, after
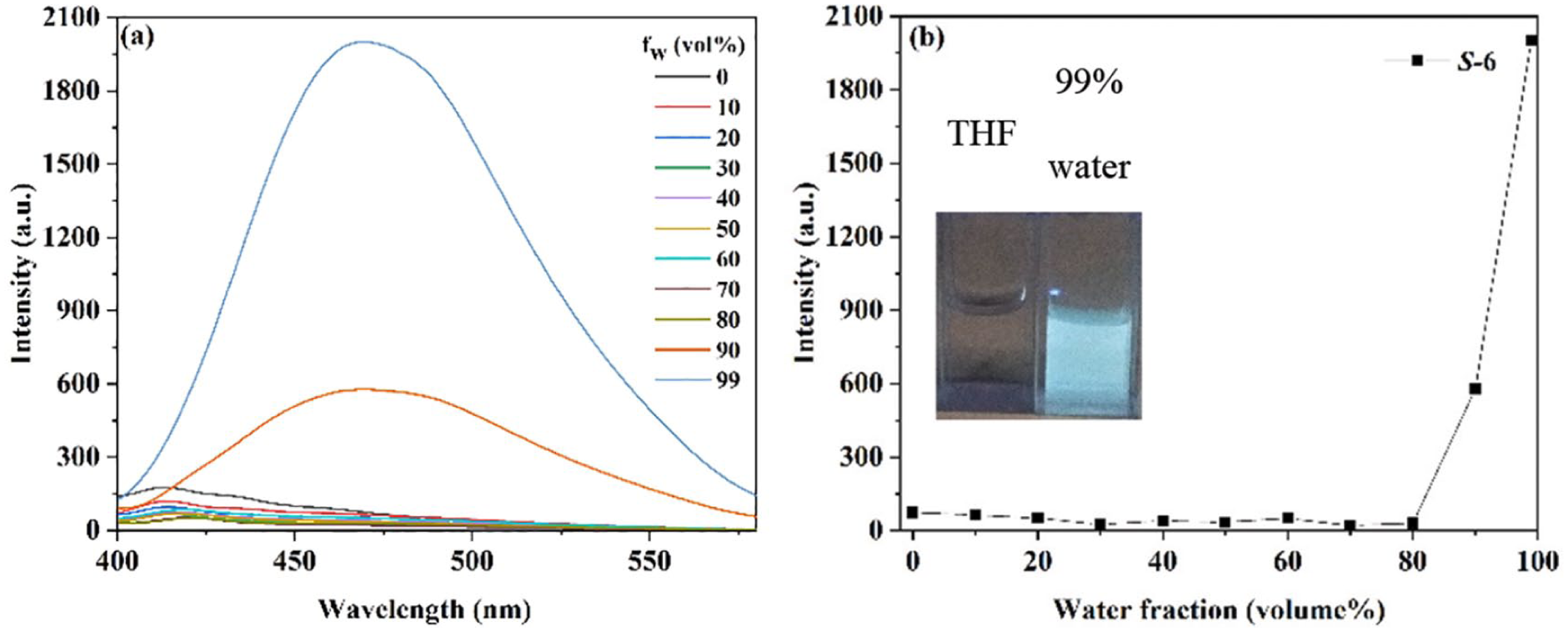
(a) Fluorescence emission spectra of
Studies of chiral recognition
The chiral recognition properties of
Fluorescence intensity ratios of mixtures of enantiomers of analytes with

Enantiomer 1/enantiomer 2.
Volume ratio of solvents, [sensor] = [analyte].
When

(a) Fluorescence spectra of a mixture of compound
The analytes in Table 1 were also tested with sensors
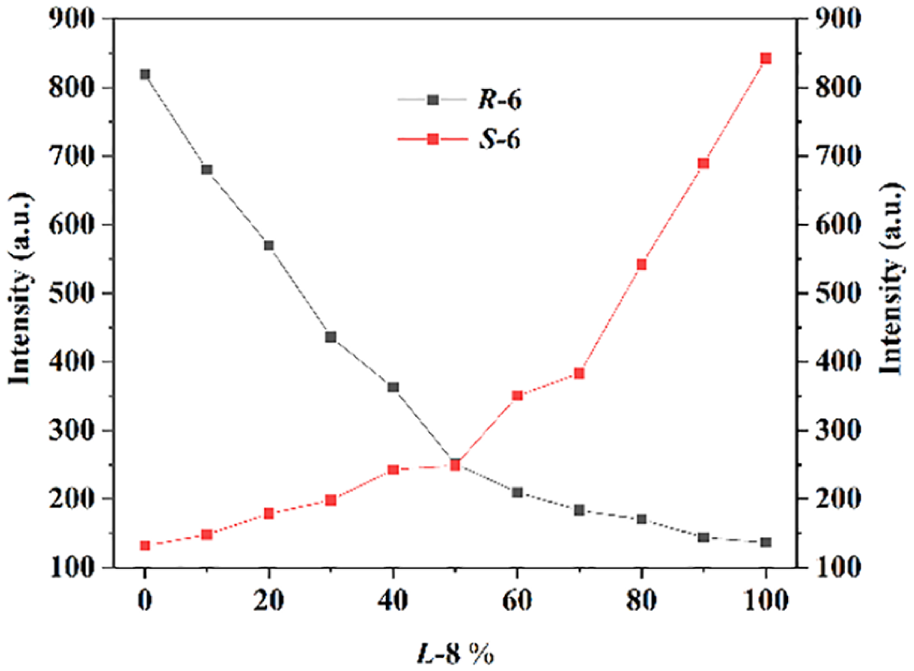
In addition, the chiral response for racemic acids was also investigated. For example, when racemate

Fluorescence spectra of mixtures in 1,2-dichloroethane (5 × 10−4 M). Mixtures: (a) compound
Change in fluorescence intensity of
Microscopic studies
To understand the morphology of aggregates in mixed solvents, TEM images were examined and the formation of aggregates of

TEM images of a suspension of

(a) TEM image of a suspension of
Experimental
Materials and measurements
All reagents and solvents were chemically pure (CP) or analytical reagent (AR) grade and were used as received without further purification. 1H NMR and 13C NMR spectra were measured on a Bruker AV 500 spectrometer at 303 K from sample solutions in CDCl3. Mass spectra were measured on a Waters Q-TOF microspectrometer. Optical rotations were measured at 25 °C on an Anton Paar MCP 500 polarimeter with a sodium lamp as the light source (589 nm). Fluorescence emission spectra were collected on a Perkin Elmer LS 55 Fluorescence Spectrometer. The fluorescence spectra for the AIE effect were measured after preparation and leaving the mixture to stand for 2 h at 298 K. To measure changes in the fluorescence intensity in the presence of chiral guests, all mixtures of sensors
Synthesis of ((S)-2-((2-(4-(1-(4-(2-(((R)-2-hydroxy-1-phenylethyl)amino)ethoxy)phenyl)-2,2-diphenylvinyl)phenoxy)ethyl)amino)-2-phenylethan-1-ol (
S-6
)
Compound
Synthesis of ((R)-2-((2-(4-(1-(4-(2-(((S)-2-hydroxy-1-phenylethyl)amino)ethoxy)phenyl)-2,2-diphenylvinyl)phenoxy)ethyl)amino)-2-phenylethan-1-ol (
R-6
)
The method was the same as that described for
Synthesis of 2-(4-(2,2-diphenyl-1-(4-(2-(((R)-1-phenylethyl)amino)ethoxy)phenyl)vinyl)phenoxy)-N-((S)-1-phenylethyl)ethan-1-amine (
R-7
)
To a flask was added
Conclusion
In summary, the combination of a TPE moiety and several optically pure molecules has provided several excellent novel chiral sensors. Sensors
Supplemental Material
SI_revised – Supplemental material for Synthesis of novel chiral fluorescent sensors and their application in enantioselective discrimination of chiral carboxylic acids
Supplemental material, SI_revised for Synthesis of novel chiral fluorescent sensors and their application in enantioselective discrimination of chiral carboxylic acids by Qiuhan Yu, Weiwen Lu, Zhiqiang Ding, Min Wei and Zhenya Dai in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We were thankful to the National Natural Science Foundation of China for financial support (no.: 21102180).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
