Abstract
A new 1,3,4-oxadiazole derivative (BPOXD-B8) was synthesized. The emission properties of BPOXD-B8 in different solutions were studied. BPOXD-B8 could form organogels in different solvents, especially in methanol. The gelation and self-assembly properties of BPOXD-B8 were investigated by SEM, X-ray diffraction (XRD), temperature-dependent FTIR, and UV-vis. The main driving forces during self-assembly are π–π interaction and the van der Waals force. In addition, the BPOXD-B8 solution showed fluorescence sensing properties for Fe3+ ions. BPOXD-B8 with a relatively low detection limit (0.079 μM) and relatively small standard deviations (<5%) indicate that the present system is indeed suitable for real-life sample analysis.

Introduction
In the past two decades, 1,3,4-oxadiazole derivatives have received wide attention as an important class of materials.1–3 1,3,4-Oxadiazole derivatives are derivatives containing one or more 1,3,4-oxadiazole rings, which have been extensively studied in the fields of organic light-emitting diodes (OLEDs)4,5 and liquid crystals. 6 In addition, due to the π structure and electron-withdrawing ability of oxadiazole ring, the research of 1,3,4-oxadiazole in organogels,7,8 self-assembly, 9 and fluorescent materials10,11 has also received attention.
Low molecular weight organogels (LMWGs) are usually formed by self-assembly of small organic molecules (molecular weight is usually less than 3000) through non-covalent bonds.12,13 In recent years, as a kind of important soft material, LMWGs have attracted the attention of researchers.14–16 Typical LMWGs mainly include amide,17,18 cholesteric,19,20 dendrons derivatives, 21 and so on. The research of 1,3,4-oxadiazole derivatives as a kind of LMWG was relatively few.9,22 For example, El-Metwaly’s group 7 synthesized three kinds of structurally similar, asymmetric fluorine substituted 1,3,4-oxadiazole derivatives. Among them, the compound with a hexoxy group at the end could form gel in different solvents. The π-π interaction provided by aromatic groups and the van der Waals force provided by long alkyl chains were the main driving forces in the self-assembly process. Li’s 8 group synthesized three pyridine substituted 1,3,4-oxadiazole derivatives with different length alkyl chains. The correlations between the gelation of 1,3,4-oxadiazole derivatives and the solvents were established.
Recently, the use of fluorescent materials as probes for the detection of cations has received widespread attention.23–32 As an important cation, iron ion is abundant in higher life bodies (including plants and animals), and it plays a particularly important role in the human environment. For example, it can be used as nutrients to promote metabolism. 33 In addition, iron ions can protect the human body from pathogens and promote the body’s immune response. 34 Excessive or insufficient iron ions will lead to a variety of diseases in the human body.35,36 Therefore, the recognition and detection of iron ions is a very important topic. However, in general, the detection of iron ions is easily affected by interference from other metal ions.37–39 Therefore, it is necessary to develop new fluorescent Fe3+ indicators.
Here, a novel 1,3,4-oxadiazole derivative named 2-(biphenyl-4-yl)-5-(3,4-bis(octyloxy)phenyl)-1,3,4-oxadiazole (BPOXD-B8) was synthesized (Scheme 1) and its photophysical, gelation, self-assembly. BPOXD-B8 showed Fe3+ fluorescence sensing properties in methanol solvent. Furthermore, an uncommon gel–gel transition with an obvious fluorescence quenching phenomenon was observed, which made BPOXD-B8 different from other optical sensors. The research of this molecule enriches the example of 1,3,4-oxadiazole derivatives as gel material and fluorescent sensing material, and provides a certain reference for the synthesis of new functional materials in the future.

Molecular structure of compound BPOXD-B8.
Results and discussion
Photophysical properties of BPOXD-B8 in solutions
The UV-vis absorption and fluorescence emission spectra of BPOXD-B8 in different polar solvents (1.0 × 10−5 mol L −1 ) were measured, and the results are shown in Figure 1, with specific information presented in Supplemental Table S1.

Normalized (a) UV-vis absorption and (b) fluorescence emission spectra of BPOXD-B8.
As shown in Figure 1(a), the maximum UV-vis absorption peak of BPOXD-B8 in cyclohexane (a non-polar solvent) is at ca. 315 nm. With the increase of polarity, the position of the maximum absorption peak appeared redshift. The maximum absorption peak in DMSO (polar solvent) is at ca. 322 nm, and the position of the peak is only 7 nm relative to that of cyclohexane (non-polar solvent). Such a small peak shift indicated that there is no significant change in the electronic and structural properties of the ground state and the Frank Condon excited state.10,40
In contrast, the fluorescence emission spectra of BPOXD-B8 varied greatly in solvents of different polarities. As shown in Figure 1(b), three fluorescence emission peaks were observed for BPOXD-B8 in cyclohexane, with the main peak position occurring at 376 nm. The peak of BPOXD-B8 in toluene appeared at 386 nm, in THF at 400 nm, in N,N-Dimethylformamide (DMF) at 422 nm, and in Dimethyl sulfoxide (DMSO) at 432 nm. As a whole, there is a large shift (about 56 nm) in the position of the peak as the polarity increases. Such a large peak shift (redshift) indicated that due to intramolecular charge transfer (ICT), the dipole moment of the ground state is much smaller than that of the excited state.41,42 Moreover, the fluorescence quantum yields (Φf) of BPOXD-B8 in these five different polar solutions were listed, as shown in Supplemental Table S1. For instance, the Φf of BPOXD-B8 is 83.5% in cyclohexane and 41.6% in DMSO, exhibiting a typical positive solvato-kinetic effect.43,44
Gelation properties of BPOXD-B8
The gelation properties and the critical gelation concentrations (CGCs) of BPOXD-B8 were tested in 10 typical organic solvents and listed in Table 1. The results showed that BPOXD-B8 could form a gel in three solvents: methanol (CGCs is 3.9 mg mL −1 ), acetonitrile (CGCs is 4.1 mg mL −1 ), and DMSO (CGCs is 12.0 mg mL −1 ). While BPOXD-B8 can only form solution in toluene, chloroform, THF, and DMF, precipitate in cyclohexane, and partly gel in ethanol and acetone. In the following, as a typical example, we will test the gelation properties of BPOXD-B8 gel in methanol. Because BPOXD-B8 has the lowest CGCs (3.9 mg mL −1 ) in methanol, in the following, we choose methanol as a typical solvent and study the properties of BPOXD-B8 methanol gel.
Gelation properties of BPOXD-B8 at room temperature.

P, precipitate; S, solution; PG, partly gel; G, gel.
Numbers in parentheses represent the critical gelation concentrations (CGCs, mg mL −1 ) of the gel.
The relationship between gel–sol transition temperature (Tgel) and concentration of BPOXD-B8 methanol gel was tested, and its function is shown in Figure 2. With the increase of concentration, Tgel increases until it reaches the platform area, and the value of the platform area is ca. 57 °C.

Concentration-dependent gel-sol transition temperature (Tgel) of BPOXD-B8 gel in methanol.
In order to research the micro-aggregation morphology of BPOXD-B8 methanol gel, its xerogel in methanol was prepared and observed by field emission scanning electron microscopy (FE-SEM). As shown in Figure 3, long fibers are observed in methanol xerogel. The width of fibers is in the range of 0.5–2 μm, and their length can be as long as dozens of microns or more. These long fibers intertwine to form a typical three-dimensional gel network.

The SEM image of xerogel formed by BPOXD-B8 in methanol.
In order to reveal the molecular stacking mode of BPOXD-B8 methanol gel at the molecular level, we obtained the X-ray diffraction (XRD) pattern of BPOXD-B8 xerogel, as shown in Figure 4. It was found that several sharp diffraction peaks in the small angle region indicate its crystalline feature. Furthermore, as shown in Supplemental Table S2, the diffraction peaks found in the low-angle range have d-spacing of 43.2 Å, 38.3 Å, 21.7 Å, 18.9 Å, and 12.6 Å, respectively. We believe that these peaks belong to a columnar rectangular structure, with a = 52.3 Å and b = 76.6 Å.

X-ray diffraction pattern of BPOXD-B8 xerogel from methanol.
Self-assembly properties of BPOXD-B8
To investigate the driving force during the self-assembly process, xerogel of BPOXD-B8 from methanol was prepared, and its temperature-dependent FTIR spectra are shown in Figure 5. The bands of νs(CH2) and νas(CH2) of BPOXD-B8 were located at 2851 and 2919 cm −1 at 25 °C, while they red-shifted to 2853 and 2924 cm −1 with the temperature gradually increased to 135 °C. The red shift of these bands indicated a strong van der Waals force between the alkyl groups, which reduces the flowability of the alkyl chains. 45

Temperature-dependent FTIR spectra of BPOXD-B8 methanol xerogel.
In addition, UV-vis absorption spectra of BPOXD-B8 in solution and gel state were also tested to explore more in-depth information about the aggregation state of BPOXD-B8 molecules. The π–π* absorption maximum of BPOXD-B8 (Figure 6) is at 319 nm in the dilute solution (1.0 × 10−5 mol L −1 ) state and blue shifts to 313 nm in the gelation state. The blue shift indicated that H-aggregation was formed and π-π interaction played a role during the self-assembly process. 7

Normalized UV-vis absorption spectra of BPOXD-B8 under solution (1.0 × 10−5 mol L −1 ) and gelation (1.0 × 10−2 mol L −1 ) state in methanol at room temperature.
According to the above discussion, the possible self-assembled stacking mode and the gelation process of BPOXD-B8 in methanol can be inferred as below. As shown in Scheme 2, BPOXD-B8 molecules first self-assemble through π–π interaction to form one-dimensional supramolecular chains with columnar rectangular structure (a = 52.3 Å and b = 76.6 Å), known as elemental fibrils. Second, through van der Waals interaction, the element fibrils become longer and thicker and entangle to form a typical three-dimensional gel network.
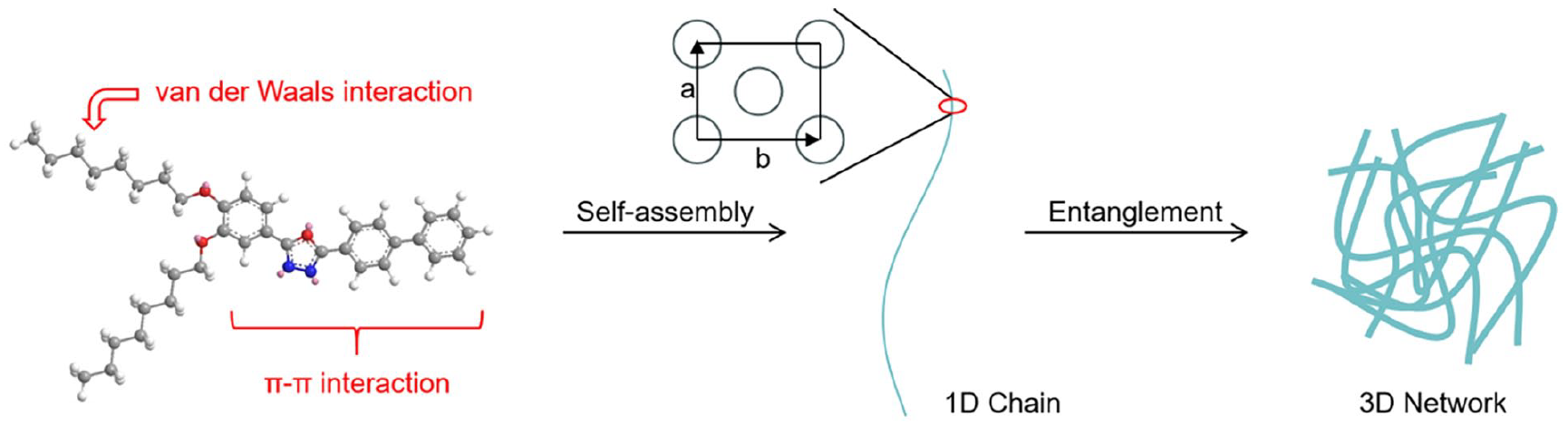
The possible self-assembled stacking mode and the gelation process of BPOXD-B8 in methanol.
Fluorescence selectivity of BPOXD-B8 toward Fe3+
The fluorescence changes of BPOXD-B8 methanol dilute solution (1.0 × 10−5 mol L −1 ) after adding different cations were studied using fluorescence spectroscopy. As shown in Figure 7, after adding a cation other than iron ions (such as Ca2+ and Cd2+), the fluorescence intensity of BPOXD-B8 showed relatively little or almost no change. However, with the addition of Fe3+, the fluorescence intensity of BPOXD-B8 decreased obviously, indicating that BPOXD-B8 has significant fluorescence sensing properties toward Fe3+. Moreover, the UV-vis absorption spectra of BPOXD-B8 in the presence of Fe3+ was different from that of other metal ions (Supplemental Figure S5), indicating an interaction between BPOXD-B8 and Fe3+.

Fluorescence spectra of BPOXD-B8 in methanol after adding different types of metal ions (10 equiv).
In addition, the fluorescence titration spectra of BPOXD-B8 solution with Fe3+ were also tested, as shown in Figure 8. With the gradual addition of Fe3+, the fluorescence intensity of BPOXD-B8 gradually decreased, and its fluorescence peak showed a certain red shift (from 428 nm to 443 nm). According to literature report, Fe3+ was prone to interact with electron withdrawing group in the ICT molecule, thereby promoting the “push–pull” electronic system, accompanied by a decrease in fluorescence intensity and a redshift in fluorescence peak position. 46 We studied the FTIR spectra of BPOXD-B8 before and after the addition of Fe3+ ions (as shown in Supplemental Figure S6). The peak of C=N is at 1611 cm−1, and the peak of C=N–N=C is at 1500 cm−1.47–49 After adding Fe3+ ions, the peak of C=N shifts to 1608 cm−1, while the peak of C=N–N=C shifts to 1508 cm−1. This indicates that Fe3+ ions do indeed interact with the 1,3,4-oxadiazole ring. Furthermore, we also studied the 1H NMR spectra of BPOXD-B8 before and after the addition of Fe3+ ions. As shown in Supplemental Figure S7, after the addition of Fe3+ ions, the peak of BPOXD-B8 widens, and the peak on the benzene ring near the 1,3,4-oxadiazole ring shifts toward a higher field. This once again indicates that Fe3+ ions indeed interact with 1,3,4-oxadiazole. Based on the above analysis, it was inferred that Fe3+ interacts with the electron-withdrawing group (1,3,4-oxadiazole ring) of BPOXD-B8 (ICT molecule), causing a significant decrease in the fluorescence intensity of the system, thereby enabling BPOXD-B8 molecule to possess fluorescence sensing properties for Fe3+. As illustrated in Supplemental Figure S8, it is clear that the Stern–Volmer curve deviates from the straight line, suggesting that both dynamic quenching and static quenching mechanisms appear in the process. 50 We also tested the fluorescence ratio before and after adding 10 equivalents of Fe3+ ions in the gel state (1 × 10−2 mol L−1), its fluorescence was significantly quenched (Supplemental Figures S9 and S10), but the gel did not collapse, which may be caused by Fe3+ ions only interacting with the electron-withdrawing group, but not destroying its self-assembled structure.51,52
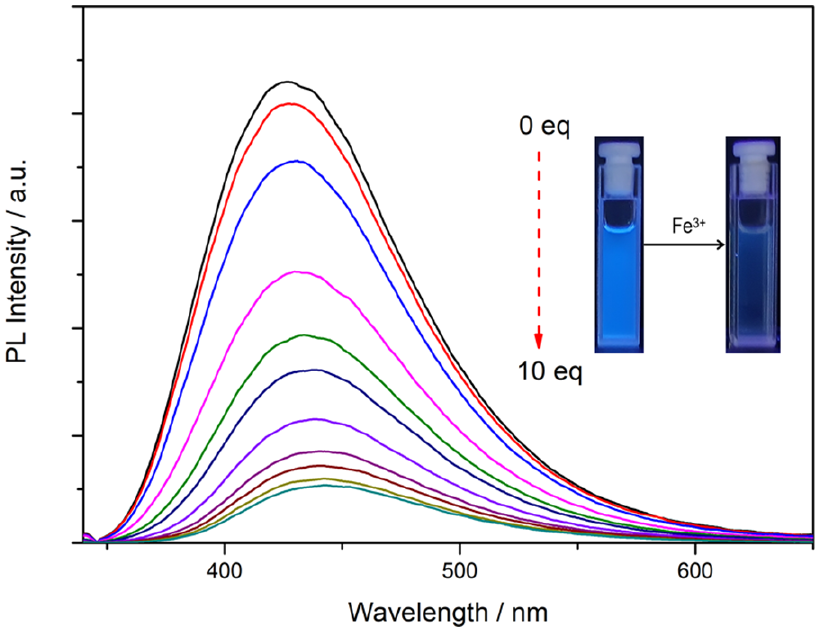
The fluorescence spectra of BPOXD-B8 methanol solution after gradually adding different equivalent (0–10 equiv) Fe3+.
In order to evaluate the Fe3+ selectivity of BPOXD-B8, the fluorescence spectra of the BPOXD-B8 system were tested under the interference of other metal ions. It was found that in the presence of Fe3+, even with the interference of other metal ions, the fluorescence intensity of BPOXD-B8 remained almost unchanged (Supplemental Figure S11). This phenomenon meant that BPOXD-B8 molecules have high fluorescence selectivity and sensitivity to Fe3+, and have high anti-interference performance against external cations. Meanwhile, as an important index of chemosensors, the fluorescence detection limit for sensing Fe3+ in solution was found to be 0.079 μM in methanol solution (Supplemental Figure S12).53,54 The BPOXD-B8 sensor has a good linearity for Fe3+ ions in the range of 10–40 μM (R2 = 0.997). We used sensor BPOXD-B8 to prepare test strips for detecting Fe3+ ions. The sensor BPOXD-B8-containing filter paper displayed bright fluorescence under 365 nm light (Supplemental Figure S13(b)), the fluorescence of written “Fe” disappeared and “Fe” letter was clearly observed. As a comparison, the filter paper without sensor BPOXD-B8 did not present fluorescence (Supplemental Figure S13(a)). This result indicated sensor BPOXD-B8 can be used as test strips to monitor the existence of Fe3+ ions. Finally, the proposed method was applied to real sample, including tap water, nearby pond water, and nearby river water, and the results were shown in Supplemental Figure S14 and Supplemental Table S3. A relatively small standard deviations (<5%) indicate that the present system is indeed suitable for real-life sample analysis. 26
Conclusion
A new 1,3,4-oxadiazole derivative (BPOXD-B8) was synthesized. We investigated the photophysical properties of BPOXD-B8 in solvents with different polarities using fluorescence spectroscopy and UV-vis spectroscopy. The BPOXD-B8 molecule exhibited ICT properties. BPOXD-B8 could gelate different solvents, especially in methanol. It was found that π-π interaction and van der Waals interaction played an important role in the self-assembly process of forming the 3D network structure of gel. BPOXD-B8 molecule showed remarkable fluorescence selective detection performance for Fe3+ in methanol solvent. Besides, a gel-gel transition was observed with obvious fluorescence quenching. FTIR and 1H NMR spectra proved that Fe3+ interact with the electron-withdrawing group (1,3,4-oxadiazole ring) of BPOXD-B8 (ICT molecule), causing a significant decrease in the fluorescence intensity of the system, thereby enabling BPOXD-B8 molecule to possess fluorescence sensing properties for Fe3+. BPOXD-B8 has a relatively low detection limit (0.079 μM) and a relatively small standard deviation (<5%) for iron ions, indicating that it is indeed suitable for sample analysis in real life.
Experimental
Materials and methods
The chemicals mentioned in the article are all analytical reagent grade and have not been further purified when used. The metal ions we have chosen are derived from CaCl2, Cd(NO3)2·4H2O, Co(NO3)2·6H2O, CuCl2·2H2O, FeCl3·6H2O, KOH, LiCl, MgCl2, MnCl2, NaOH, Ni(NO3)2·6H2O, and ZnCl2.
Mercury-300BB spectrometer (300 MHz) was used to record 1H NMR spectra using tetramethylsilane (TMS) as internal chemical shift reference. High Resolution Mass Spectrometry (HRMS) was performed by Bruker solanX 70. FTIR spectra were measured by Perkin-Elmer Spectrum one B. Field emission scanning electron microscope (FE-SEM) observations were performed using the JSM-6700F device. UV-vis absorption spectra were recorded on the Shimadzu UV-2550 spectrometer, and fluorescence emission spectra were determined on the Perkin-Elmer LS55 spectrometer.
Synthesis of 2-(biphenyl-4-yl)-5-(3,4-bis(octyloxy)phenyl)-1,3,4-oxadiazole (BPOXD-B8)
The synthesis of compound BPOXD-B8 was similar to that of L-OXD, 55 which was shown in Scheme 3. 1.10 g (0.0028 mol) 3,4-bis(octyloxy)benzohydrazide 56 was weighted and dissolved in 100 mL THF. 0.6 g (0.0028 mol) biphenyl-4-carbonyl chloride was dissolved in 10 mL THF. The THF solution of biphenyl-4-carbonyl chloride was added to the THF solution of 3,4-bis(octyloxy)benzohydrazide and stirred for 12 h at room temperature. At the end of the reaction, a portion of THF was distilled, and the remaining reactants were poured into 500 ml of distilled water, and white solid of 1 was separated out. The unpurified dried 1 was dissolved in a phosphorous oxychloride (POCl3) solvent, and the reaction was performed by heating and refluxing for 30 h. Finally, the reactants in this step were poured into ice water to separate the precipitate. The precipitate was recrystallized with ethanol for several times to obtain pure BPOXD-B8. (1.12 g, 72%). Characterization data and scanned copies of BPOXD-B8, including 1H NMR, 13C NMR, FTIR spectra, and mass spectra, were displayed in SI.
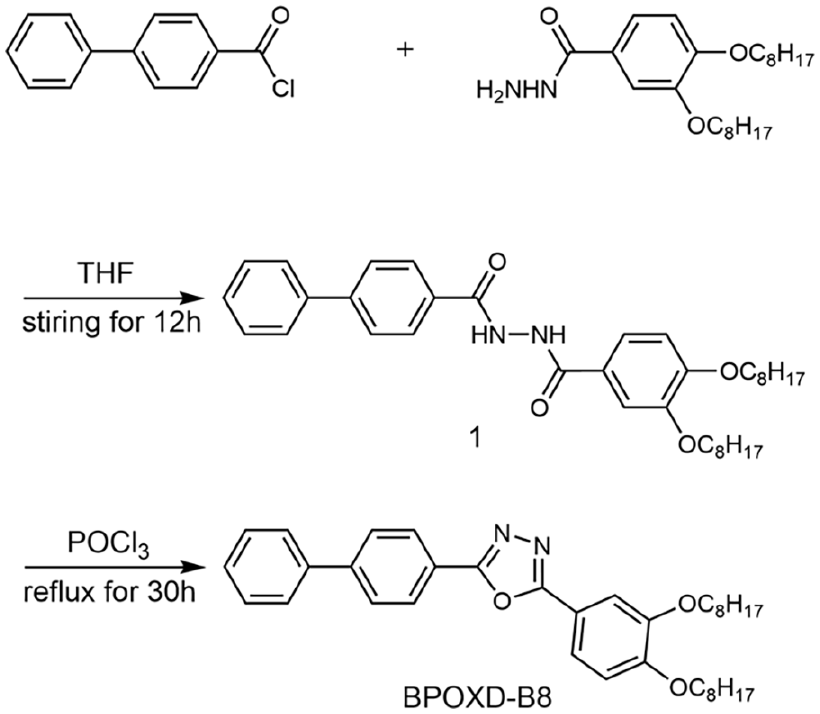
Synthesize route for BPOXD-B8.
Gelation test
A certain amount of gelator and organic solvent were successively put into a vial (3.5 cm × 0.5 cm) with a lid, and heated until the system became transparent and clear solution. Then, without disturbing the system, the obtained thermal solution was cooled at room temperature for 1 h. Finally, the gel formation was determined by “falling drop” method. 57
Supplemental Material
sj-doc-1-chl-10.1177_17475198241255789 – Supplemental material for 1,3,4-Oxadiazole derivative: Photophysical, gelation, self-assembly, and Fe3+ fluorescence sensing properties
Supplemental material, sj-doc-1-chl-10.1177_17475198241255789 for 1,3,4-Oxadiazole derivative: Photophysical, gelation, self-assembly, and Fe3+ fluorescence sensing properties by Tianren Zhang, Sitong Jiang, Guizhi Wang, Youpeng Zuo, Shuwen Yu, Qing Zhang and Xueqing Hu in Journal of Chemical Research
Footnotes
Acknowledgements
We thank Prof. Min Li, Prof. Haitao Wang, and Chengxiao Zhao at Jilin University for their guidance in the synthesis of 1,3,4-oxadiazole derivative.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (22206145), the Excellent Youth Research Project of Anhui Province (2022AH030135), the Natural Science Research Project of Anhui Education Department (KJ2021A1104 and 2022AH051381), the Provincial of Natural Science Foundation of Anhui (2208085QB65), the Ph.D. Research Funding of Suzhou University (2020BS019, 2021BSK009, and 2021BSK011), the Scientific Research Platform Project of Suzhou University (2021XJPT08 and 2021XJPT09), and the Medical Science and Technology Innovation Project of Xuzhou Municipal Health Commission (XWKYHT20230075).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
