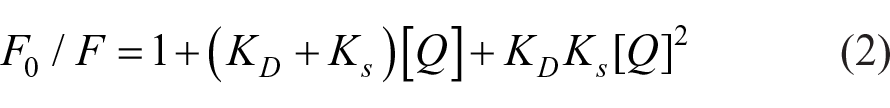Abstract
The detecting and screening of explosives has become a global consideration in dealing with potential terrorism threats and the misuse of high explosives. Several detection techniques have been developed, but their disadvantages include the requirement of expensive equipment, complicated pre-treatment, and prolonged testing. So far, fluorescence-based sensors have been of great interest as they overcome the limitations of other techniques. BODIPY, which is a fluorophore, shows excellent fluorescence features, and it can be used as a sensor material for explosives detection. However, to date, BODIPY-based explosive sensors have not been explored in detail. This work reviews the recent developments on explosive sensors based on BODIPY and its analogs.
Keywords

Introduction
Explosives are reactive materials containing a great amount of potential energy that can cause detonations, accompanied by the formation of more stable materials. Explosives can be classified in different ways, depending on their chemical aspects (nitro compounds, acid salts, peroxides), their applications (for military uses or commercial uses), and their power release (low, primary high, secondary high). There are nearly 16 common explosives (Figure 1). Nitro-aromatic compounds, which are high explosives, such as 2,4,6-trinitrotoluene (TNT), dinitrotoluene (DNT), and triazacyclohexane (RDX), are usually used as military explosives and are major components in unexploded landmines. Mines and solid rockets for industrial use are mainly made of ammonium salts like ammonium nitrate (AN) and ammonium phosphate (AP).1,2 Besides, improvised explosive devices have also been developed by using peroxide-based explosives such as triacetone triperoxide (TATP) and hexamethylene triperoxide (HMTD), as they are commercially available and inexpensive materials.
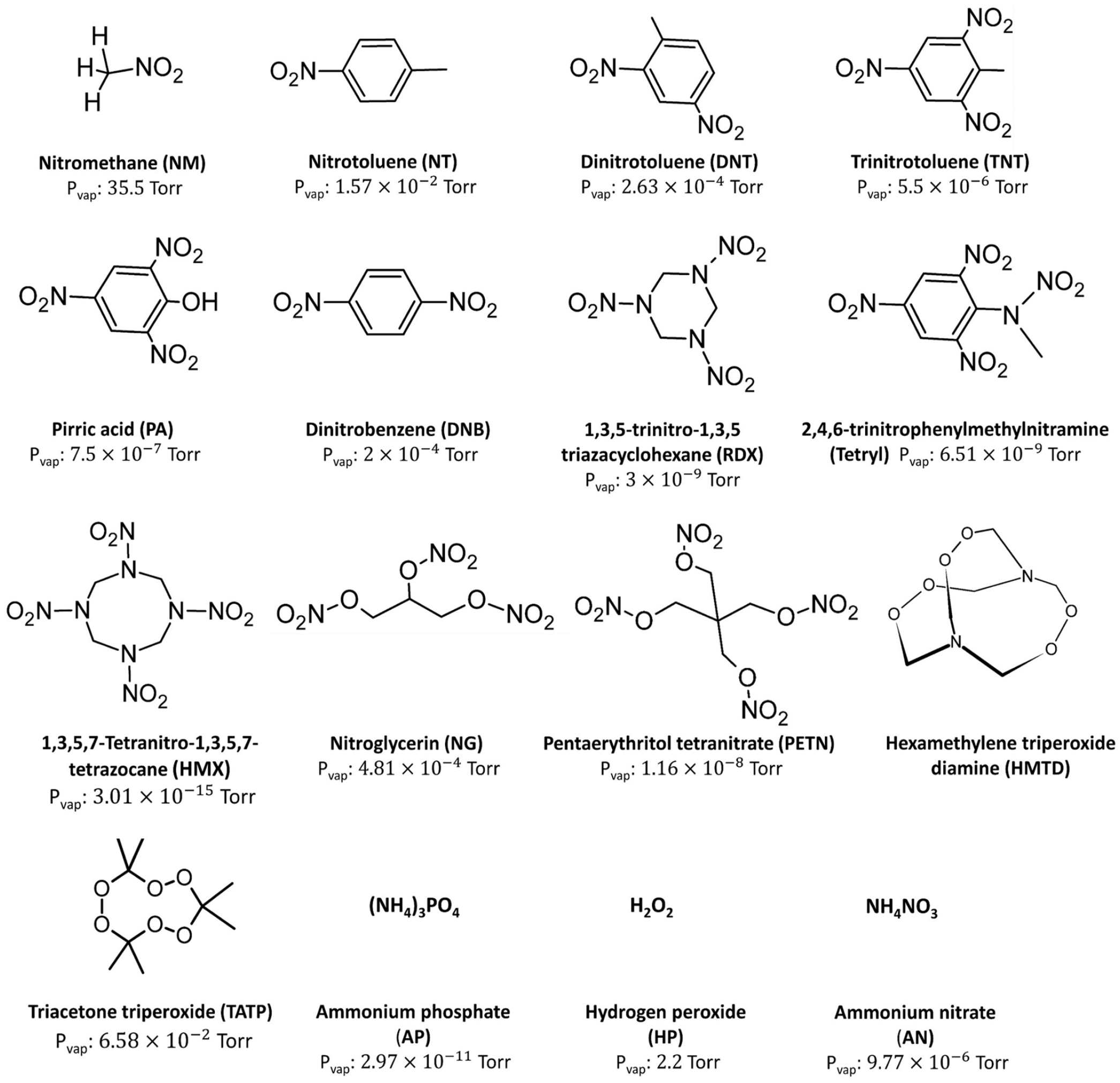
Common explosives and their vapor pressure at 25 °C (Ewing et al. 3 and Haz-Map).
The growth of terrorism worldwide has heightened global concern over methods for the sensitive, selective, and rapid detection of explosives. For terrorist attacks, explosives have been the preferred weapons, being used in more than 80% of worldwide attacks related to firebombs and explosives since 2015 (Global Terrorism Index 2022). Besides, there are also environmental and health risks related to explosive residues released from military sites and ammunition plants (GICHD Annual Report 2021).4,5 Over-exposure to TNT, for example, can cause anemia, liver problems, and even cataracts because this compound can penetrate through the skin and enter the bloodstream directly. 6 Consequently, the reliable detection of explosives is one of the pressing concerns in anti-terrorism efforts and environmental pollution control. In particular, rapid, selective, and sensitive explosive-detecting techniques have been an important issue of global concern.
Over the decades, several techniques have been developed for the detection of explosives, such as X-ray imaging, gas chromatography coupled with mass spectroscopy, 7 ion mobility spectroscopy (IMS),8,9 and surface-enhanced Raman scattering (SERS).10,11 Such methods, however, need expensive instrumentation and a complicated set-up followed by a long response time. Furthermore, they are sensitive to contaminants, or may not be amenable to stand-off detection. To date, fluorescence-based sensing technology represents one of the most promising approaches for tracing explosives due to the several advantages of this technique, such as short response times, excellent sensitivity, instrumental simplicity, and low cost.12–14 Different fluorescent sensory materials (probes) have been developed for sensing explosives and have shown high efficiency. Several such examples are conjugated polymers, supramolecular systems, small-molecule fluorophores, bio-inspired materials, aggregation-induced emission-active (AIE) materials, and so on. Such sensory materials, which have shown excellent results in the sensing of explosives, mainly rely on two sensing mechanisms: fluorescence quenching and/or turn-on fluorescence. 15 Modifications of small-molecule fluorophores, such as conjugated fluorophores and self-assembling small fluorophores, have led to even greater potential in explosives sensing since these molecules can enhance binding and/or exciton migration. 16 BODIPY is among the class of small-molecule fluorophores with great versatility, and their photophysical properties could be fine-tuned over wide ranges using straightforward synthetic protocols. 17 They display both turn-on and turn-off fluorescence in sensing explosives, depending on the polarity of the working media and the type of analyte. Thus, using BODIPY as a fluorescent sensory material holds promise in explosives detection tasks. However, there has not been a lot of research done on developing BODIPY as fluorescence probes for the detection of explosives. Therefore, in this short review, we summarize the research on BODIPY as optical sensory probes for the detection of explosives.
Recent fluorescence-based explosives sensing techniques
Explosives can be detected by using fluorescent sensors, as explosive materials are not fluorescent. The interaction between analytes and probes would result in fluorescence changes, including enhanced or quenched fluorescence intensities, lifetimes, wavelengths, and anisotropy. Although these changes can be used to detect explosives, fluorescence quenching and fluorescence turn-on have been preferred over other mechanisms. The subsequent paragraphs give information on quenching-based detection and turn-on fluorescence detection.
Fluorescence-quenching-based detection
Fluorescence-quenching is the result of either diffusive encounters (collisional quenching) or complex formation processes (static quenching). Collisional quenching refers to the radiationless deactivation of an excited state fluorophore in collisions with quenchers. This process is attributed to photo-induced electron transfer (PET), charge-transfer intersystem crossing (ISC), or Förster resonance energy transfer (FRET) processes. Meanwhile, static quenching refers to the formation of a non-fluorescent complex between a fluorescent probe and analytes.
18
The prevalent way to examine whether the quenching is a static or a collision process is based on fluorescence lifetime changes in the absence or presence of explosive quenchers, given by the ratio of lifetime
where
where
In terms of explosives detection ability, static quenching-based sensors show a larger Stern–Volmer quenching constant (KSV) and a higher sensitivity in general. Fluorescent sensors based on the collisional quenching process have a smaller KSV, showing a lower sensitivity but faster and more reversible detection. 20 The two quenching processes sometimes occur at the same time with the same analytes, allowing for a greater sensing range. For example, the conjugated copolymer PFPy can detect TNT at femtomolar (fM) level (4.94 fM) due to the combination of both collisional and static quenching processes. 21 In addition, the sensitivity toward explosives detection based on fluorescence quenching depends on different types of sensory platforms and the sensing phase (the detection environment) (Table 1). Conjugate polymers show excellent detection sensitivity at pM and fM levels.21,22 Small-molecule fluorophores also show excellent sensing ability, and their analogs, such as conjugated fluorophores, showed even greater sensitivity.23,24
Recent efficient explosive-sensing techniques based on fluorescence quenching.

Fluorescence turn-on-based detection
To date, the fluorescence turn-on approach has received more attention, as it is believed that this approach combines the higher relative intrinsic sensitivity with the higher chemical selectivity and represents an emerging frontier for explosive sensing. 30 Despite this, there have not been many studies done on explosive sensors based on fluorescence turn-on, especially for the detection of nitro-aromatics. A recent report introduced a series of supramolecular ensembles that acted as turn-on fluorescence sensors for various nitroaromatic explosives. However, the sensitivity, response time, and sensing mechanism have not been proposed. 30 Besides, Yu et al. proposed an enhanced-fluorescence sensor based on a blend consisting of perylene-3,4,9,10-tetracarboxylic acid derivative nanofibers and amberlyst-15 particles. This sensing system showed rapid turn-on fluorescence responses (ca. 5 s) and incredible sensitivity (0.1 ppm) for TATP detection (vapor phase). 31
BODIPY and its application in explosives sensing
Synthesis and design of BODIPY fluorophores
BODIPY (4,4-difluoro-4-bora-3a,4a-diaza-s-indacene) dyes are a class of small-molecule fluorophores. 32 They are fascinating compounds with multiple modification sites on their structure. Also, they have remarkable features, including small Stokes shifts, narrow absorbance bands, sharp emissions with high-fluorescence quantum yields, and excellent stabilities.31,33–35 BODIPY dyes can be readily made from pyrrole-based materials, often in high yields and in multigram quantities. The synthesis of the BODIPY core can be conducted in different ways, including the following:
Dipyrrin chemistry; 36
The acylation of pyrrole followed by condensation and complexation;37,38
One-pot condensation–decarbonylation of pyrrole-2-carbaldehydes. 39
Although the chromophoric core of BODIPY is the key to its success and versatility, novel analogs of BODIPY can easily be made by functionalization at the 8-(meso) positions as well as the pyrrole rings (

Different methods for the synthesis of the BODIPY core and functionalization approaches at the preferential sites of attack.
BODIPY in explosive sensing
BODIPY fluorophores have shown versatile spectroscopic and photophysical properties, depending on their chemical structure functionalization and the conjugated system.32,47 Furthermore, as proposed in several research studies, analogs of the fluorophore, such as conjugated fluorophores and AIE-based fluorophores, displayed an excellent detection sensitivity for explosives based on fluorescence sensing.23,29,48,49 Perhaps, BODIPY fluorophores and their analogs, such as AIE-based BODIPYs and conjugated BODIPYs, could hold promise as they probably show intrinsic sensitivity, simplicity in design and synthesis, easy set-up, and recognition. The use of BODIPY and the design of its analogs as sensory materials for explosives detection, nevertheless, have not appeared often in the literature. To date, BODIPY-based explosive sensors have only been introduced for the tracing of picric acid (PA), RDX, and peroxide. These BODIPY sensors exhibit dynamic fluorescence in various media and interact with analytes based on the modified sites and attached moieties (refer to Table 2). Among these, PA has attracted more research into BODIPY-based sensors over other analytes, with the detection mechanism based on fluorescence turn-on or fluorescence quenching.50–55 Later, BODIPY-based sensors for peroxide and RDX were also introduced.54,56
Photo-spectroscopic data of the introduced BODIPY probes.
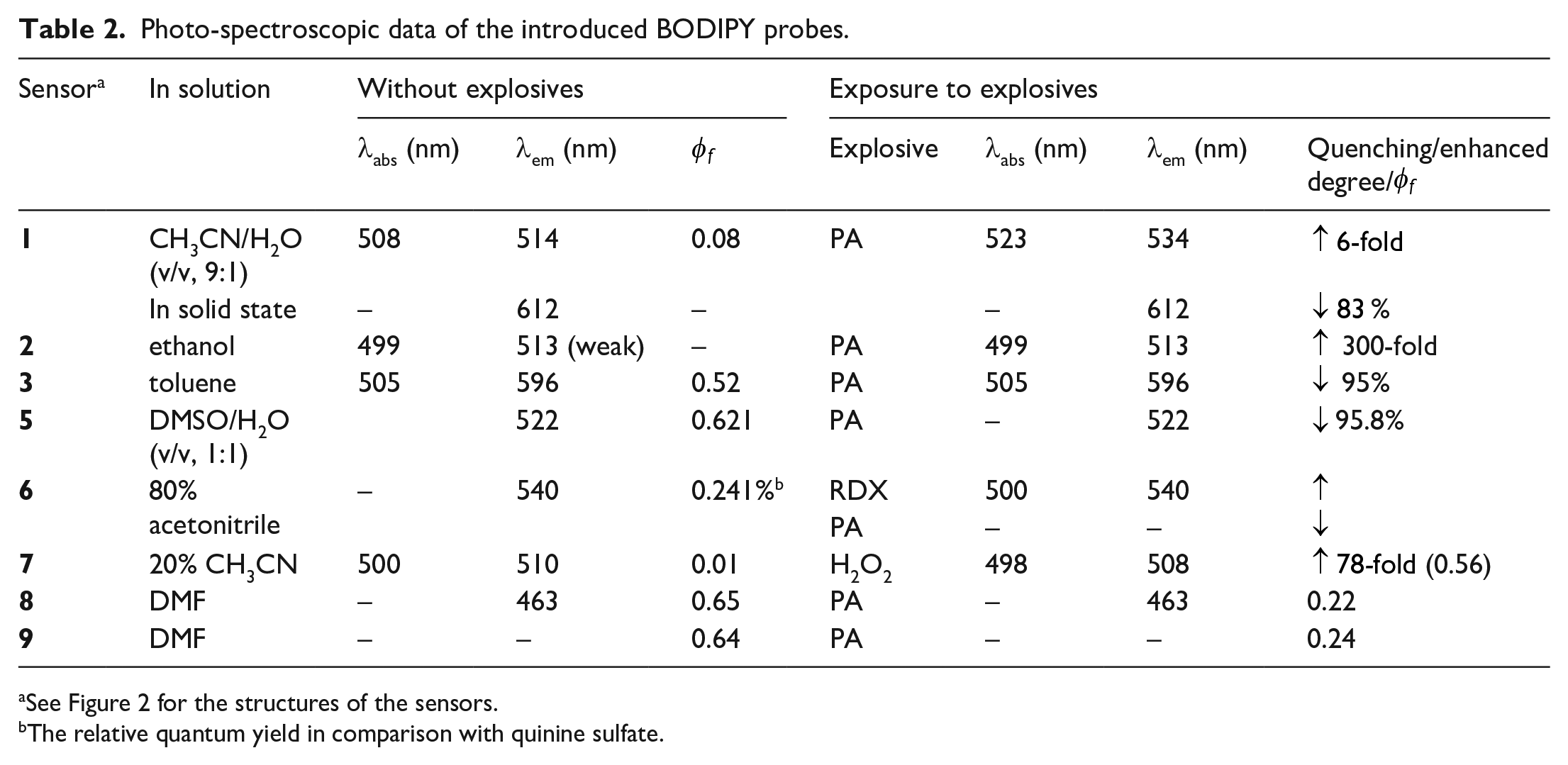
See Figure 2 for the structures of the sensors.
The relative quantum yield in comparison with quinine sulfate.

BODIPY probes used for explosives detection.
Detection of PA
Based on fluorescence turn-on mechanism
3,5-Bis(acetal) BODIPY (
In other research, fluorescent meso-diaminophenyl-1,3,5,7-tetramethyl BODIPY dye (
Based on the fluorescence quenching mechanism
The triphenylamine-substituted BODIPY derivative (
P-Pyridine BODIPY (
A hydrazine-substituted BODIPY (
Two macromolecular chemo-sensors based on BODIPY (
Detection of other explosives
The detection of peroxide-based explosives by using a BODIPY-based fluorescence sensor, m-NBBD (
Interestingly, compound
Discussion
In the above examples, BODIPY probes for the detection of explosives have been discussed. The detection mechanism of these BODIPY probes refers to two fluorescence processes, either fluorescence quenching (mainly involved in collisional quenching via PET) or fluorescence turn-on. It can be seen from the described BODIPY compounds that designing BODIPY probes for explosives detection consists of three tasks: (1) extending the π-conjugation, (2) tuning the photospectroscopic properties, and (3) designing the explosive recognition site (Figure 3). To achieve the first task, most of the introduced BODIPY probes were prepared by functionalizing the BODIPY core at its meso-site with aromatic substituents. These substitutions extend the π-conjugation of the BODIPY dyes, allowing them to absorb and emit light at longer wavelengths and achieve higher quantum yields. For instance, compound
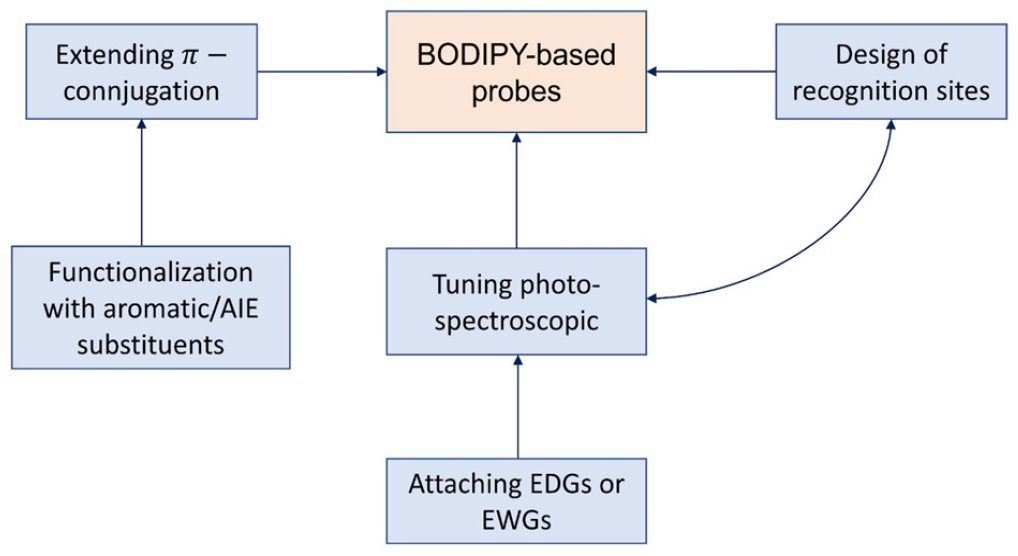
Schematic approach for designing a BODIPY-based probe for explosives detection.
Although some research has examined the ability of BODIPY probes in real detection, thereby revealing their versatility and potential use in actual case analysis,53,54,56 additional studies are needed to integrate these probes into a portable device for practical applications.
Conclusion
In this short review, we have summarized the recent approaches in explosive sensing and the possibilities of using BODIPYs. As a class of small fluorophore molecules, BODIPY and its analogs are promising candidates for explosives detection. Although there have not been many research studies focused on this subject, the available data have revealed the versatility and possibility of using BODIPY for explosives detection in both laboratories and actual cases.
Footnotes
Author contributions
All the authors contributed to the literature search as well as the writing of the manuscript. All the authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Research Foundation of Korea (NRF) grant funded by the Korea government (No. 2021R1I1A3060405) and Basic Science Research Program to Research Institute for Basic Sciences (RIBS) of Jeju National University through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2019R1A6A1A10072987) and by the Big Issue Program funded by KITECH, Republic of Korea (Project No. EO23013).