Abstract
We systematically reviewed contemporary literature to create an evidence-informed framework for research studies involving children and adolescents who can assent to participate. We searched seven citation indices to locate peer-reviewed research published in English language journals between 2000 and 2012. After screening 1,231 titles and abstracts for relevance, we assessed levels of evidence, extracted information, and analysed content from 87 articles. Most articles narrowly focused on paediatric assent barriers and facilitators for decision-making about research participation. No articles provided a single, comprehensive ethical framework to guide the development and review of research assent protocols. We developed a 6-step framework that provides guidance to: prepare the child for the assent process; assess the child’s readiness to engage in decision making; discuss the elements of informed consent to the greatest extent possible; seek an initial assent decision; monitor and affirm assent; and respect the child’s role as a research participant. The PAeDS-MoRe framework also supports the creation of process models that address the unique, developmental needs of paediatric sub-groups, and guides the operationalization of jurisdictional requirements for ethical research involving children who are unable to provide free, informed and ongoing consent.
A clinical research team based at a children’s rehabilitation hospital plans a 3-year randomized controlled trial designed to examine the efficacy of an investigational drug therapy on hyperactivity and aggression in school-age children with autism spectrum disorders. Eligibility criteria include children who are verbal, between the ages of 7 and 10 years, and have a confirmed diagnosis of autism. The researchers propose to randomize 40 children to one of two experimental arms (active drug and placebo) in a 1:1 fashion. Each child will be asked to stay on the study drug (or placebo) for 24 weeks. Outcome measures include baseline and follow-up psychological tests and magnetic resonance imaging scans, and safety measures involving blood work, electrocardiogram tests and biweekly physical exams. The institutional research ethics board requires the team to submit a detailed research assent protocol as part of its initial ethics submission. The lead researcher asks the research ethics board to provide direction on the elements to include in an assent protocol.
Background
The Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans − 2010 (
Although free, informed and ongoing consent of research participants is the overarching tenet for the ethical conduct of research involving humans, individuals who are incapable of consenting to research participation are more vulnerable and require additional consideration and protections. Children and adolescents (hereafter ‘children’) who lack or have an emerging capacity to consent may not understand and appreciate essential elements of informed consent – including the goals of research, their involvement in a study, the possible risks and benefits of participation, issues of privacy and confidentiality, and alternatives to participation. Because children may appreciate what is involved in participating in research and at least some elements of informed consent, researchers must afford them the opportunity to assent to participate during the consent process (TCPS 2, 2010, Article 3.10). The TCPS 2 calls for researchers to respect the wishes of children by involving them in decision-making about research participation to the greatest extent possible.
Assent can be defined as ‘an affirmative agreement to participate in research’, and ‘mere failure to object should not be construed as assent’ (Beigay, 2007: 55). Conversely, dissent may be considered an expression or indication of a desire not to participate in research (TCPS 2, 2010, Article 3.10). It is important for institutional review and research ethics boards (REBs) and researchers to operationalize assent and dissent indicators because of the emerging competence and unique vulnerabilities of children. These vulnerabilities can include varying capacity, developing autonomy, and an evolving ability to make mature and informed decisions (Johnston, 2006; Ross, 2003; Simpson, 2003).
The TCPS 2 and other international regulatory standards are consistent in their requirement that researchers seek child assent and respect dissent to participate in research (TCPS 2, 2010, Article 3.10; Declaration of Helsinki, 2008, Article 28). Yet, the standards are silent on the breadth and expected features of child assent protocols.
We conducted a systematic review of contemporary peer-reviewed literature to inform the development of a structure for paediatric assent protocols for researchers and REBs. Specifically, our review was designed to answer the research question: ‘What evidence-informed framework can be used to guide the development and ethical review of proposed research assent protocols involving children who are unable to consent?’
Methods
Search strategy
Peer-reviewed articles published in English journals between January 2000 and June 2012 were identified using seven on-line citation indices: Medline, CINAHL, Scopus, Cochrane, BIOSIS, EMBASE, and Web of Science. A keyword and database search strategy were selected in consultation with a senior health sciences librarian who was based at a children’s teaching hospital and had extensive experience conducting searches for narrative and systematic reviews of biomedical and allied health research literature. The selected search strategy used the key words ‘assent’ and (‘child*’, ‘paediatric*’, ‘consent’, ‘informed consent by minors’ or ‘disability research’). We chose the latter term because our institutional REB reviewed submissions involving children and youth with developmental disabilities.
Study selection
Articles were eligible for inclusion if they were peer-reviewed articles that described paediatric research assent processes for children under the age of 18 years. We excluded articles that solely considered child assent processes for clinical rather than research purposes. The lead author screened article titles and abstracts for relevance. From an initial pool of 1,231 non-duplicate abstracts, we included 87 full articles that met eligibility criteria for our analysis (Figure 1).
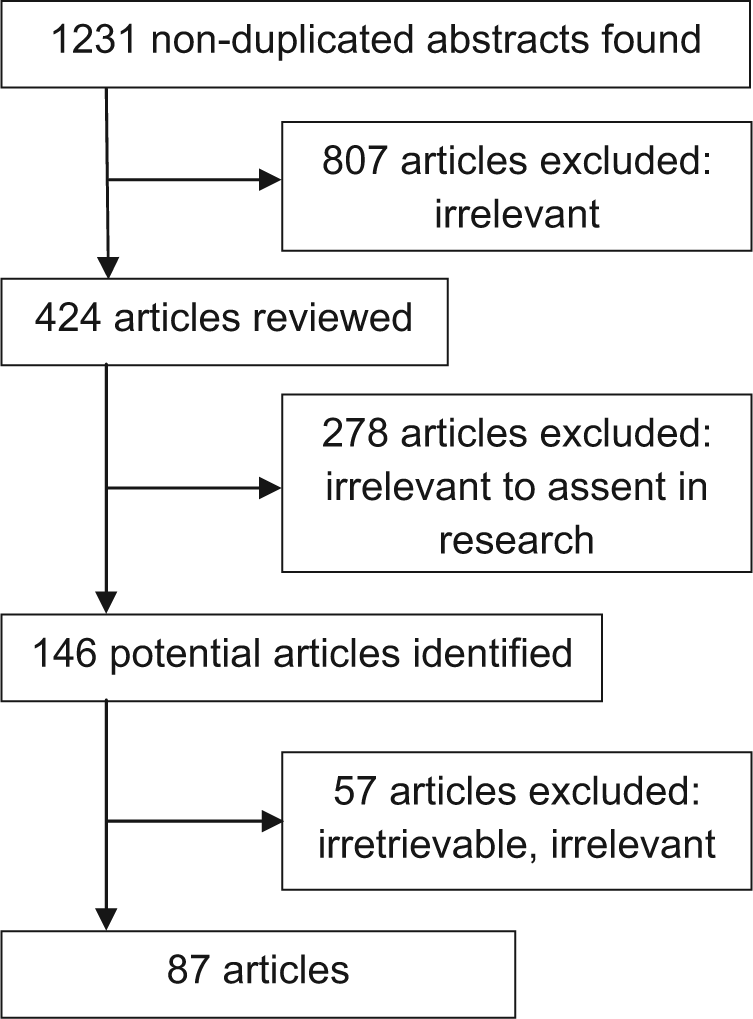
Screening summary for relevant articles on research assent involving children and adolescents.
Data extraction and reporting
Data related to elements of the paediatric assent processes were mined from eligible articles by the lead author. Extracted information was summarized on a data collection form developed for the review. The form logged the article source, the nature, level, and quality of evidence, and the age range or developmental levels of children addressed. We adopted Guyatt and colleagues’
Eligible articles included in the review by jurisdiction, methodology, and paediatric context (n=74) (NB: Refer to References for full article citations.).

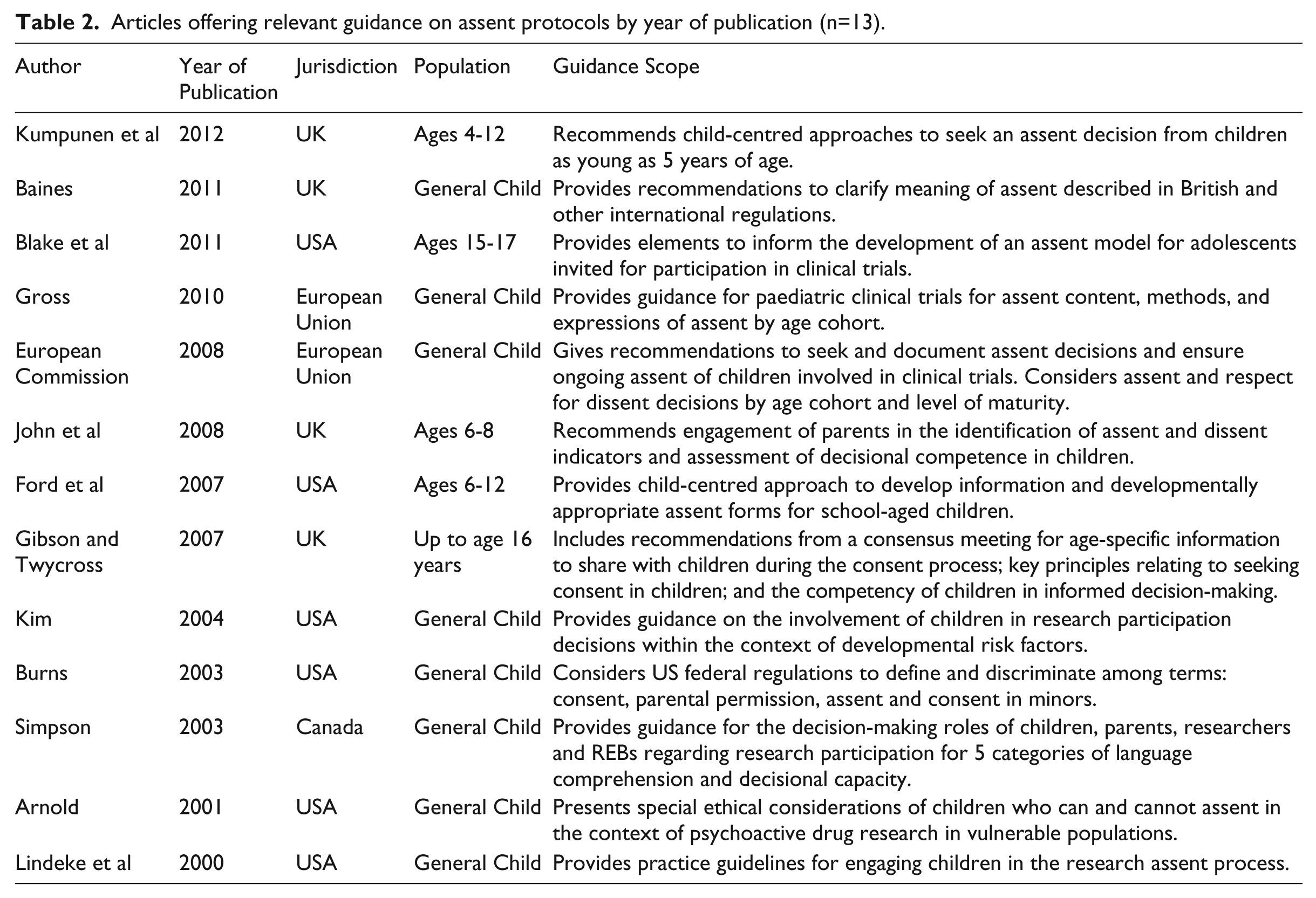
Articles offering relevant guidance on assent protocols by year of publication (n=13).
We identified emergent themes after reviewing the article content and assessing its quality. Articles that covered relevant aspects of the research assent process were selected, reviewed and collated. Studies with higher levels of evidence and document reviews of more recent regulations and policies assumed greater significance and priority during the distillation of converging themes.
Results
The majority of original research and review articles originated from the US, followed by contributions from the UK, the European Union, Canada and Australia (Table 1). Selected articles included qualitative research designs (including surveys, interviews and focus groups), narrative and systematic reviews, document reviews of regulations and policies, and practice statements. Only three articles employed quantitative research methodologies, including two randomized control trials (O’Lonergan et al., 2011; Tait et al., 2007) and one a retrospective quasi-experimental study (Kimberly et al., 2006). Application of the quality assessment guidance resulted in 68 percent of the articles being assigned a low to medium quality and the remaining articles being assigned a high quality grade.
Thirteen articles provided explicit guidance on child assent processes in research (Table 2). However, authors focused mainly on specific elements of the assent discussion with children and lacked a holistic framework to guide the development and review of research protocols over the proposed assent continuum. Although article authors acknowledged obtaining a child’s decision regarding research participation as a key element in the ethical rigor of an assent protocol, they consistently identified challenges in operationalizing the entire child assent process in the context of international regulations and ethical principles.
Analysis
PAeDS-MoRe assent framework
We reviewed, consolidated and synthesized research assent evidence, reviews and recommended practice into a process framework intended to guide research assent protocols involving children. We conceived the PAeDS-MoRe mnemonic (‘peeds more’) to aid recall of the six action steps within the proposed research assent protocol framework: Prepare for Assent, Assess Readiness, Discuss the Study, Seek Decision, Monitor Decision and Respect Role.
Each step of the PAeDS-MoRe assent framework is elucidated below through a referenced narrative and checkpoint that summarizes its key action elements as informed by source literature and confirmed in relevant chapters and articles of the TCPS 2.
Parents and researchers should partner to reinforce this distinction by reassuring the child that participation is voluntary and no one will mind or be upset if he refuses to take part in the study at any time (TCPS 2, 2010, Article 3.1). In preparing the child for the study-specific information sharing and initial discussion about participation, researchers should consider prospective threats to free decision-making by consulting with parents to understand personal factors, cultural influences, and family relationships and preferences (Johnson and Nelson, 2000; Lambert and Glacken, 2011).
Researchers and REBs must consider the appropriateness and influence of the magnitude, timing, recipient, and type of compensation in assent and consent decision-making (TCPS 2, 2010, Articles 3.1 and 4.7). Compensation may take the form of either payments to defray participant expenses associated with travel, time and inconvenience, or payments intended to motivate participation (Kimberly et al., 2006). The latter is a controversial and less defensible form of payment in children who may otherwise not participate in a research study (Diekema, 2006). Regardless, researchers should share REB-approved compensation details with the child and parent before seeking a decision about study participation (Kimberly et al., 2006).
Reinforce the distinction between clinical treatment and research intervention to avoid therapeutic misconception
Seek parent awareness, advice and support for the assent discussion
Consider and plan to mitigate threats to free and informed decision-making of both child and parent
Share approved compensation details before seeking an assent decision.
Researchers should ready the child for the discussion and decision about participation by introducing the child and family to the research team, facility, and the general scope of the study in a child-friendly manner. Following initial child−parent−researcher interactions, the researcher can reflect on the child’s receptiveness to the research discussion and environment, and assess the child’s cognitive ability, emotional maturity, and physical readiness to engage in discussion about research participation (Blackmer, 2003; Lambert and Glacken, 2011; Masty and Fisher, 2008).
Assessing a child’s readiness for participation may be challenging in special paediatric populations. For example, prospective participants may have expressive language disorders and use augmentative or alternative systems for communication. Special accommodations such as mediation by a communication specialist and support from a familiar communication partner may need to be arranged to allow the child to participate meaningfully in the decision-making process (Blackmer, 2003).
The researcher should also have or seek the support of others who have the skills to assist children and parents in shared decision-making. REBs should consider the following: the researcher’s ability to assess the child’s readiness to take part in discussions about the potential consequences of and alternatives to participation; the training and experience to respond to questions in a child-friendly manner; and awareness of factors that may influence decision-making, including child development level, family dynamics, emotional state and the context-specific ability to make a reasoned choice (Lambert and Glacken, 2011).
Introduce self and research team members
Discuss purpose and uncertainty of research with both child and parent
Assess child’s cognitive, emotional and physical readiness to engage in a discussion about research participation
Identify and implement appropriate accommodations to support the discussion and decision-making process
Consider the ability of the researcher to share study information in a suitable way and assist the child in making a reasoned decision about participation.
Researchers should provide written consent and assent information in advance of the assent discussion to allow time for the child and parent to consider and discuss the research opportunity. Written assent information in large font using short sentences or bullets that engage the child using one main idea at a time is recommended to improve understanding (Gibson et al., 2007; Kumpunen et al., 2012; Vitiello, 2003).
The researcher should share key information with the child, including: the study purpose, how the child is involved, who the researchers are, the possible benefits, possible harmful effects, voluntary participation, free withdrawal at any time, and confidentiality of the study (Gibson and Twycross, 2007). These topics resonate with information generally required for informed consent involving prospective participants who have the capacity to understand and appreciate these elements (TCPS, 2010, Article 3.2). Of note, the literature suggests that children may have difficulty understanding concepts such as research goals and confidentiality, so researchers should take additional care in describing these concepts in terms that the child can understand, but avoid overly simplistic explanations (Hurley and Underwood, 2002; Unguru et al., 2010).
Employing multimedia information sharing strategies (videos) rather than text-based approaches have been shown to enhance family comprehension during the assent process (O’Lonergan and Forster-Harwood, 2011). Furthermore, researchers should be receptive to cues from the child that could indicate boredom, distraction, feelings of fear, and/or signs of poor understanding, and reformulate or adjust the delivery of key study messages to improve comprehension and avoid misunderstandings (Blackmer, 2003; Bray, 2007).
Be clear, concise, and use developmentally appropriate language
Be conscious of non-verbal communication
Focus on the child’s unique needs
Provide sufficient time to explain the key information, particularly the research goals and confidentiality
Involve the child in the consent discussion with the parent
Encourage questions and provide honest and clear answers
Reformulate and adjust the delivery of the message if the child is distracted, bored, scared, or does not understand.
Documenting assent via the signature of the child is generally recommended where possible (Mattison et al., 2002). Inviting a child to sign or print his name to indicate assent is empowering and may serve to reinforce autonomy and the voluntary nature of participation (Helseth and Slettebo, 2004). Because the absence of dissent does not infer assent, researchers are encouraged to identify child dissent indicators (both verbal and non-verbal) within their research assent protocols – particularly when inviting children to take part in research where no prospect of direct therapeutic benefit exists (Gross, 2010; John et al., 2008; Roth-Cline et al., 2011).
Help child to understand and appreciate research purpose, voluntariness, risks, benefits and confidentiality
Promote parental involvement but mitigate undue influence
Decide how child dissent will be assessed before seeking a decision about research participation
Identify possible verbal and non-verbal dissent indicators
Seek assent or dissent decision and document this appropriately
Confirm verbal assent by inviting child to print or sign assent form where possible.
Confirm child’s desire to continue to participate at key points in the research process
Assess evolving capacity and autonomy
Share new information that may be relevant to the child’s ongoing assent and parent’s ongoing consent.
Share study results with child and parent in an accessible way to reaffirm child’s role, reinforce the trust relationship and promote confidence in research
Discussion
The proposed PAeDS-MoRe framework operationalizes research assent protocols involving children who are unable or do not have the capacity to consent. We encourage researchers and REBs to consider the needs of special paediatric populations they engage when employing the framework.
Because the core principles of the TCPS 2 are consistent with international standards for research ethics involving children, the proposed framework also provides a universal, evidenced-informed structure that can underpin the conceptual development of specific models for child assent protocols involving vulnerable or marginalized sub-populations engaged in different types of research. Children with complex communication needs, mental health illnesses, different developmental needs and/or behavioural problems may require researchers to use the proposed conceptual framework to develop tailored assent models for unique needs to ensure greater utility and relevance for the intended application. Further, the research community must consider child development levels and ensure compliance with local legal and regulatory requirements when applying the PAeDS-MoRe framework to guide assent protocol development and review.
Article selection and quality assessment were conducted by the lead author and restricted to English language publications, so this may have added bias to our findings. However, the search strategy, article abstraction process, data collation and interpretation, and framework development were conducted collaboratively among all members of the investigative team. We encourage further empirical studies that yield higher levels of evidence to inform and improve research assent protocols as our review demonstrated a paucity of high quality empirical evidence.
Future research goals associated with the PAeDS-MoRe framework include assessing the validity and utility of the framework for the development of research assent protocols and review of research ethics submissions that propose involving children who are unable or do not have the capacity to consent. These quality improvement activities will continue to advance the ethical conduct of research for children and other vulnerable populations who rely on others to consent to research participation.
Conclusion
The PAeDS-MoRe framework provides a general structure for paediatric assent protocols informed by peer-reviewed literature and framed by the fundamental ethical principles of the TCPS 2 and other international standard and regulations. It provides a broad conceptual process that supports the application of relevant policies and regulations within local, federal and international jurisdictions. Importantly, the framework consolidates contemporary thinking about assent processes that optimize decision-making, respect human rights, and promote fairness and equity in research endeavours involving children and adolescents who are unable to provide free, informed and ongoing consent.
Appendix 1 provides a sample research assent protocol that the lead researcher could include in an REB submission for the proposed clinical trial involving children with autism.
Footnotes
Appendix
Sample child assent protocol applying the PAeDS-MoRe framework.
| Step | Element | Description |
|---|---|---|
| 1 | Prepare for Assent | The researcher will follow the REB-approved script and recruitment protocol when speaking with the parent for the first time. She will inform the parent about the assent process during the screening phone call with the parent regarding their child’s potential participation in the clinical trial. The researcher will explain that both parent consent and child assent will be required before the child is enrolled. |
| The researcher will employ a list of verbal and nonverbal indicators of dissent prior to recruitment. This list includes cues such as crying, head shaking, saying ‘no’ or ‘I don’t like this’. She will add to this list by asking the parent if the child has any other verbal or behavioral indicators of discomfort, disagreement, and unwillingness to participate in discussion. | ||
| The researcher will send a copy of the approved informed consent and assent forms 2 weeks in advance of a phone call to discuss elements of the study. She will ask the parent to help to prepare the child by reviewing the information in the assent form with the child before meeting the researcher. The researcher will follow the proposed script during the screening phone call with the parent: | ||
| ‘We want to learn whether the study drug helps to reduce aggression and hyperactivity in children with autism. As you know, this is a research study not clinical care, and therefore we do not know whether or not this study drug will benefit your child. Therefore, we must confirm that both you and your child are willing to participate in the study. | ||
| As your child is young, we understand that he may not be able to understand those parts of the study needed for informed consent. Therefore, if you are interested, we would like to meet with both you and your child to discuss the study. If you would like to take part in the study after we meet, I will ask for your consent and your child’s assent. Please understand that if your child says or acts in a way that suggests he does not want to participate, we will not be able include him for the study. It is very important for your child to know that no one will be upset with him if he decides he does not want to take part in this study. We will tell your child this when we meet to talk about what’s involved in the study. | ||
| Please speak with your child before coming to meet with me. During the assent process, I will speak with you and your child about the study including why we are studying the drug, what will be involved if he participates, what are the known risks and benefit, and alternatives to participation. | ||
| As you know your child best, please let me know if you can suggest any techniques we can use to help him understand the nature of the study and how he will be involved. Also, can you give me any advice about verbal or nonverbal behaviors that your child may use to indicate that he understands the information and is interested or not wanting to take part in the study? | ||
| If you or your child has questions about the study before your visit, please do not hesitate to contact me.’ | ||
| 2 | Assess Readiness | When the participant arrives for the first visit, a researcher who is unknown to the child will greet the family and introduce herself by first name. The researcher will introduce the child and his parent to other research members by their first names, explain the research role of each, and show the family around the lab. |
| The researcher will meet the child and parent in a private room where consent and assent discussion will take place. She will ask the child the following questions to develop rapport and assess his cognitive, emotional and physical readiness. | ||
| - How old are you? / What grade are you in? | ||
| - What is your favourite color/sport/animal/movie? | ||
| - Do you know why you are here? | ||
| - Do you know what the study is about? | ||
| - Have you ever been part of a research study before? | ||
| - Do you want to be here? | ||
| The researcher will move on to discuss the study if the child appears engaged, gives reasonable responses to the questions, and the parent agrees to proceed. | ||
| If the child answers questions with hesitation or demonstrates signs that suggest he does not want to participate in the study, the researcher will try to understand why he is feeling uncomfortable and remind him that participation is voluntary. She will tell the child that if he does not have to participate and no one will be upset. She will seek guidance from the parent to help understand the child’s feelings when these are unclear. | ||
| If the researcher feels the child is cognitively, emotionally and physically ready to assent, she will explain that the purpose of this research is to find out whether the study drug will help calm children with autism. She will tell the child and parent that no one knows for sure whether this study drug will work and that is why she is doing this study. | ||
| 3 | Discuss Study | The researcher will use the REB-approved assent form to help guide her discussion of the study with the child. She will review each section of the assent form slowly and clearly, take breaks when needed, and leave time for the child and parent to ask questions (see below for details). During this time, the researcher will observe the body language of the child and adjust the conversation accordingly. She will reword sections that the child does not seem to understand. She will show how the test equipment works to familiarize the child with the different aspects of involvement. The following information will be discussed in detail, keeping in mind the child’s developmental level: |
| 1. What the child will do if they participate in the study including: | ||
| • Specific details about the study drug he will take (how they will take it, when they will take it); | ||
| • The concept of randomization (50-50 chance he will receive a sugar pill that looks and tastes like the study drug, but should not change his behaviour in any way); | ||
| • The concept of blinding (no one will know whether he is took the sugar pill or study drug); | ||
| • Specific details about each outcome measure involved in the study; and, | ||
| • Specific details about medical procedures required for the study (blood draw, electrocardiogram tests, magnetic resonance imaging scans). Where possible the equipment set-up and operation will be shown to both the child and parent. | ||
| 2. The positive aspects of being in the study: | ||
| • The study drug may make him feel happier and more relaxed; | ||
| • The study drug may help him to concentrate better at home and school; | ||
| • The child and parent will find out what the research team learned from the study; and, | ||
| • The study drug may help him and other kids with autism. | ||
| 3. The negative aspects of being in the study: | ||
| • It may be hard for him to swallow the pills; | ||
| • The study drug may give him a headache or upset stomach; | ||
| • The blood draws may hurt a bit; | ||
| • The psychological testing may take a long time and be boring; | ||
| • The magnetic resonance imaging scanner is very loud and he may have to lie still for a long time; and, | ||
| • The researchers may find important things about his health that needs to be shared with his family doctor. These findings could be worrying, but the risk of finding something will be very small. | ||
| 4. Confidentiality | ||
| • No one else will know that he is taking part in the research study. | ||
| 5. Voluntariness of participation | ||
| • Participation in this study is completely voluntary and if he does not want to try the study drug, it is okay. No one will be mad or upset if he decides not to take part in the study; and, | ||
| • If he decides to participate but changes his mind later that is also okay. | ||
| 6. Compensation | ||
| • He will receive a small ‘thank you’ gift each time he comes in for a study visit; | ||
| • If he and his parent decide that he should stop taking part at any time, the researcher will still give him the gift when he comes. | ||
| The researcher will encourage the child to ask questions at this time or at any point during the study. She will also explain to the child that he does not have to make a decision on the spot. He can take time to think about it and call her or have his mom call when he decides. | ||
| 4 | Seek Decision | The researcher will ask a few questions about the study to ensure that the child has a general understanding about the study and knows what the alternative is to participation. She will take necessary steps to avoid undue influence by the parent and others during the decision-making stage. |
| The researcher will ask the following questions: | ||
| - What do you have to do to be part of this study? | ||
| - What are the good things about being in this study? | ||
| - What are the bad things about being in this study? | ||
| - Do you have to take part in this study? | ||
| - What will happen if you do not take part in this study? | ||
| - Who can you ask if you have questions? |
||
| Once the child has had time to think about participation, the researcher will ask if he would like to take part in the study. If yes, then the researcher will ask him if he would like to sign the assent form to say that he would like to do the study. The researcher reminds the child that he can change his mind at any time. He only needs to tell his parent or the researcher that he does not want to do the study any more. | ||
| 5 | Monitor Decision | As this study is 24 weeks long, the child’s autonomy and decision making abilities will likely not change over the course of the study. However, during each study visit, the research team members will assess the child’s readiness to discuss and willingness to continue to participate in the study. |
| If at any point a researcher feels that the child shows signs of dissent, she will speak with the child and ask if he would like to continue to take part. The researcher will remind the child that he does not have to continue to participate and no one will be upset with them if he decides not to continue. The parent will be available during this discussion so the child can get advice from others before deciding. The child will be offer the a nominal value gifts (under CDN$5) from the researcher’s ‘treasure box’. | ||
| 6 | Respect Role | At the end of research participation, the lead researcher will meet with the child and parent to explain what she learned. The researcher will give the child a picture from the magnetic resonance imaging scan to take home. |
| The researcher will tell the child and parent that she will be unable to share whether the child received drug or placebo until the entire study is over. She will tell the child and parent that they will find out what the research team learned once the study is over. The researcher will tell the family when to expect these results. | ||
| Annually, the researcher will mail updates about the study progress to the child and parent using a child- and family-friendly approach. At the end of the study, she will email/mail a letter to both the parent and child to share what the research team learned. The summary will be written at a developmentally appropriate level with pictures to reinforce what it is that the child did during the study. | ||
| The lead researcher will thank the child for taking part in the study, say how the child’s involvement helped his team to understand more about how the study drug works with children who have autism, and what new research is planned. The researcher will provide her office phone number so the child and parent can contact her any time if they have questions about the study findings. |

Acknowledgements
We gratefully acknowledge conceptual advice on research assent protocols from Dr Barbara Gibson, Dr Cherisse McKay and Ms Maria McDonald, and search strategy counsel from Ms Pui-Ying Wong.
Conflict of interest statement
The authors declare no conflicts of interest.
Funding
This work was supported by the Holland Bloorview Kids Rehabilitation Hospital Foundation and the David and Anne Ward Family Research Summer Studentship Award. We received no grant from any other funding agency in the public, commercial or not-for-profit sectors.
