Abstract
Research ethics committees (RECs) have a crucial role in protecting children in research. However, studies on REC members’ perspectives on paediatric research are scarce. We conducted a qualitative study to explore Swedish scientific REC members’ perspectives on ethical aspects in applications involving children with severe health conditions. The REC members considered promoting participation, protecting children and regulatory adherence to be central aspects. The results underscored the importance of not neglecting ill children’s rights to adapted information and participation. REC members supported a contextual and holistic approach to vulnerability and risk, which considers the child’s and parents’ psychological wellbeing and the child’s integrity, both short and long term. The ethical complexity of paediatric research requires continuous ethical competence development within RECs.
Keywords
Introduction
Research with children is essential, but raises ethical issues. Most children cannot provide legal consent and may have difficulties protecting their rights in research, making them more susceptible to harm (Council for International Organizations of Medical Sciences, 2016; World Medical Association, 2013). Vulnerability is a central concept in research ethics. However, the concept of vulnerability is underdefined by research ethics guidelines and its application contested (Bracken-Roche et al., 2017). Children’s vulnerability in research is increasingly approached in contextual terms, as opposed to the traditional group-based categorical labelling of children as vulnerable (Racine and Bracken-Roche, 2019). Contextual approaches focus on interactions between the individual child’s characteristics and situation, and the research context, to unveil the actual sources and reasons for children’s vulnerability (Hurst, 2015; Luna, 2009; Nuffield Council on Bioethics, 2015; Racine and Bracken-Roche, 2019). With this contextual understanding of vulnerability in mind, children diagnosed with a chronic or severe health condition, for example cancer, can be particularly vulnerable in the context of research as they are already burdened by their disease. This vulnerability can be related to the child’s medical, psychological and social circumstances as well as their dependency on healthcare, and warrants specific ethical considerations in research with paediatric patients (Alahmad, 2018; Barned et al., 2018; Boles and Daniels, 2019).
The legal requirements for research with children differ between countries (European Union Agency for Fundamental Rights, 2019). In Sweden, ethical review of research involving humans is governed by the national Swedish Ethical Review Authority, under the Ethical Review Act (2003:460). The Ethical Review Act prescribes respect for human dignity and the right to protection from physical, psychological, and integrity related risks (Swedish Ministry of Education and Research, 2004; The Swedish Ethical Review Authority, 2023). With regard to minors, the Ethical Review Act regulates informed consent in that it states that research with children under 15 years requires permission from both parents/custodians. However, researchers must, in addition to parental consent, also obtain assent from children below 15 years for the parts of research that the children are able to understand. According to law and international research ethics guidelines, children’s dissent must always be respected, regardless of parents’ consent. However, international ethics guidelines states that overriding the child’s dissent may exceptionally be justified when a treatment that is accessible only through research participation provides an important direct medical benefit to the child (Council for International Organizations of Medical Sciences, 2016). From 15 years, children can consent themselves if they are mature enough to understand what their participation entails. Children of all ages should always be informed as far as possible. According to the law, specific consideration should be given to informed consent in cases where participants are dependent on the researcher or may have difficulties asserting their rights. For clinical drug trials, which are regulated by The Medicinal Products Act (2015:315), parental consent is required until the child is 18 years (Ministry of Social Affairs, 2015).
Parental consent differs from regular informed consent, which is based on respect for autonomy, as parental consent primarily serves to protect the interests of the child rather than the parent’s autonomy. Seeking assent from children is motivated by respect for children as persons (moral recognition of children’s right to active participation, and to dissent, in support of their developing autonomy) and beneficence (promoting the child’s best interests) (Miller and Nelson, 2006; Spriggs, 2023). The United Nations Convention on the Rights of the Child (UNCRC), which was recently adopted as law in Sweden, further asserts the rights of every child to dignity, to self-determination, to be heard, and to have their best interests considered by adult decision-makers (The United Nations, 1989). This requires healthcare professionals and researchers to develop child-centred practices that enable and encourage children to express themselves (Darcy et al., 2019).
Medical settings constitute a unique context for conducting research with children. Research and care are often integrated practices, which can make it difficult to maintain the conceptual and ethical distinctions between them (Barned et al., 2018). In paediatric oncology, for example, a large proportion of children participate in research during treatment, enabling rapid advancements in the medical field (Bond and Pritchard, 2006; de Vries et al., 2011). However, families recruited in medical settings may have difficulties appreciating the differences between research and care during informed consent, known as the ‘therapeutic misconception’ (Henderson et al., 2007). Informed consent in paediatric settings is complex, and parents and children may have opposing views. Further, clinical researchers have described recruitment to genetic studies which may impact the whole family, and experimental drug research with terminally ill children, as particularly ethically challenging. The number of studies that children with severe or chronic diseases may be invited to can create information and decision burden on families (Norbäck et al., 2023). Regardless of paediatric sub-speciality, research ethics committees (RECs) play a key role in ensuring the protection and respect of children’s rights in research. Despite this, few studies have explored the perspectives of REC members on the specific ethical issues raised by research involving children (Angell et al., 2010; Taplin et al., 2022).
The importance of exploring ethical concerns related to paediatric research, including regulatory perspectives, is recognized by international paediatric societies (Vassal et al., 2014). REC members have specialized expert knowledge and are mandated a regulatory function in research with humans. Understanding REC members’ perspectives on ethical values 1 and decision-making about paediatric research can promote transparency towards the research community and the public’s trust in biomedical research (Resnik, 2011; Taplin et al., 2022). Ethical values governing medical research are broadly upheld by research ethics guidelines and codes. However, guidelines are not exhaustive, and challenges may arise in the interpretation, implementation and merging of these guidelines (Eriksson et al., 2007). Previous research from Switzerland on this topic has reported concerns of an ethical, legal, and scientific nature in paediatric applications; for example, there are shortcomings in age-adapted information, informed consent forms, and insufficient data protection (Bergstraesser et al., 2020).
The aim of this study was to explore Swedish REC members’ perspectives on ethical values, challenges and potential for improvement of ethical aspects in paediatric research applications. Children with severe health conditions encompass a heterogeneous group that may be subjected to a wide range of research designs and methods. This study sought to explore and describe perspectives of REC members on research involving children with severe health conditions, without specific reference to diagnoses, designs, or methods. In line with this study’s descriptive and exploratory aim, the purpose was to generate new knowledge by exploring relatively unexplored perspectives on a topic, rather than providing normative solutions (Rendle et al., 2019). Empirical ethics is valuable as it generates context-sensitive ethical knowledge, for example about people’s beliefs, attitudes, and ethical reasoning (Musschenga, 2005). This study is relevant to anyone interested in the ethical conduct and oversight of paediatric research, and to our knowledge, it is the first study of its kind in the Swedish context.
Method
Study design
An explorative qualitative study was conducted using semi-structured interviews. The study was guided by naturalistic enquiry, implying that the REC members’ perspectives and experiences were analysed inductively without any a priori theoretical commitments or hypotheses (Guba and Lincoln, 1982; Lincoln and Guba, 1985, 1986; Sandelowski, 2000). An exploratory and inductive approach was chosen based on the scarcity of studies on this topic (Rendle et al., 2019). The present report of the study was prepared in accordance with the
Study setting
The Swedish Ethical Review Authority has six operational regions consisting of RECs vetting medical and non-medical research applications. Each REC is led by a current or former judge, and consists of 10 scientific members and five representatives from the public (The Swedish Ethical Review Authority, 2023). There are requirements that competence in paediatrics, psychiatry, and geriatrics should be represented within each medical REC. REC decisions result in either approval, approval with conditions, a request for additional information, rejection, or an advisory statement. Advisory statements can be granted when ethical permission is not a legal requirement, but a formal statement is desired; for example, for publication purposes. Decisions are appealable to a central board (The Ethics Review Appeals Board, 2023).
Participant recruitment
A list of all 250 scientific members was obtained from the Swedish Ethical Review Authority. Members (
Characteristics of the participants (

Data collection
Individual semi-structured interviews, each lasting 47 minutes on average (range 37–53 minutes) were conducted via Zoom between June 2021 and October 2021. All interviews were conducted by the first author (KNW), who had no prior relationship with the participants. KNW is a PhD candidate in clinical ethics and research ethics, with an MSc in psychology as well as qualitative research training. The interview guide was pilot tested with two REC members; no amendments were made, and the pilot interviews were included in the analysis. Interviews covered the topics presented in Table 2, with additional prompting questions. The participants also responded to a brief demographic questionnaire before the interview (see Table 1).
Interview topics.

Data analysis
Interviews were audio recorded and transcribed verbatim by a professional transcriber. KNW checked the transcript against the audio files to ensure accuracy, and removed identifying details. A reflexive thematic analysis was performed, following Braun and Clarke (Braun and Clarke, 2006, 2021b). Thematic analysis is a useful method for uncovering perspectives and identifying meaningful patterns in qualitative data (Braun and Clarke, 2006). The analysis was guided by an experiential framework and conducted inductively in line with the study’s exploratory aim. The analysis focused primarily on semantic content (Braun and Clarke, 2021b). KNW and TG led the analysis. TG is a registered nurse and associate professor in caring sciences, specialized in clinical and research ethics. First, KNW and TG separately familiarized themselves with three transcripts each through reflexive reading and open coding (i.e. noting down reflexive comments and short phrases summarizing text segments). They then discussed the data and generated initial codes for these six interviews, with the aim of gaining richer insights about the data collaboratively. KNW then performed open coding on the remaining interviews in a similar manner. Initially, 234 open codes were generated from the 15 interviews, but this number was significantly reduced by merging and grouping codes with similar or related content. Next, an initial thematic map was developed by KNW and TG, and then refined iteratively through discussions among all co-authors. KNW attempted to maintain authenticity and consistency by comparing the developing thematic map to the initial codes and transcripts. Version 1.3 of the NVivo software package (QSR International 2021) and Microsoft Excel were used to aid the analysis. The results are presented below, with illustrative quotations. Quotations were translated from Swedish to English by KNW, edited by a professional proof reader and compared to the original quotations for accuracy.
Results
The analysis resulted in three themes with sub-themes:
Themes and subthemes identified in the analysis.
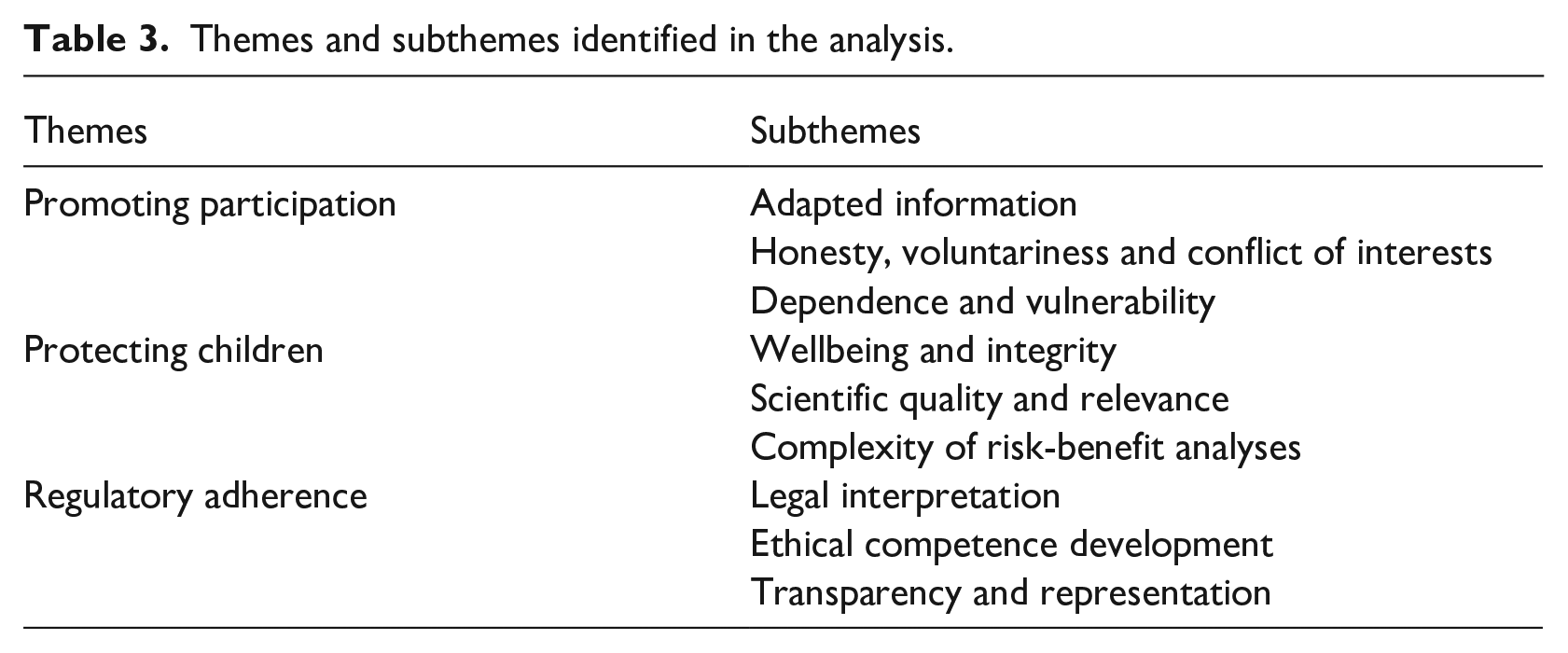
Promoting participation
Of fundamental value to the interviewed REC members was the promotion of children’s and parents’ participation in recruitment and informed consent. This was however seen as a challenge. Concerns were raised in relation to information for children, honesty, the voluntary nature of participation, conflict of interests (COI), dependency, and vulnerability.
Adapted information
The REC members highlighted children’s rights to receive age-adapted, concrete, and clear study information, as this was regarded as essential for promoting children’s participation.
When children are included, it’s really important to use age-adapted information. . .and that you have information for the youngest children . . .and adapted to the child’s reading ability, and include a lot of pictures and focus on the difficult parts. (P7)
The most common reason for requesting amendments to study applications was lack of, or insufficiently adapted information for children, which frustrated the REC members.
Often, the study information is useless and not adapted for children at all, and we’re really strict with that. Do it again, and do it right. . . (P10)
This perceived negligence was seen as a barrier to children’s self-determination and participation. As the quality of information for children was reported to vary substantially, the REC members called for methodological and paedagogical development alongside increased efforts from researchers.
To be honest, methodological development is needed. There are researchers who have given it a lot of thought. . .But there are many applications where I have to say. . .there’s remarkably little effort and reflection. (P11)
Some of the REC members described a lack of praxis for assessing the appropriateness of adaptations for different age groups. Others considered it problematic that reviews were made from an ‘adult perspective’ which did not represent children’s perspectives. Involving children of different ages to improve information and assent procedures was raised as a solution.
After all, we review through the eyes of an adult, when really, we should have some seven-, eight-year-olds. . . who could read [information] just like we have representatives from the general public. But we don’t have that. So we have to guess. (P8)
Honesty, voluntariness and conflict of interests
Honest, transparent, and neutral study information was reported as essential for promoting informed consent, especially in situations where parents may feel desperate because their children have serious health conditions.
If it’s a serious or rare disease, people can get desperate and agree to anything without really reflecting on it. (P2)
REC members underscored the importance of researchers being aware of, and respecting, children’s legal rights to dissent, regardless of their parents’ consent. They emphasized that a child should never be forced to participate.
Children do have the right to dissent even if the parents want them to participate. (P9)
Violations of honesty were sometimes identified in study information. For example, researchers exaggerated the individual benefits of participating, used overly optimistic or appealing wording, or were not fully transparent about the risks.
Often researchers sugarcoat information, for example by unconsciously or consciously understating the effects of taking the [study] medication. It says that the side effects aren’t too serious, but in reality, it makes you puke every day for a week. (P9)
In addition to looking at the information provision, the REC members considered the proposed recruitment process and context to identify risks to the voluntary nature of participation.
It’s not only about the written information but also how you invite people. . . that there’s some sort of realistic voluntary choice. . .that you’re not putting the child in a pressured situation. (P13)
To ensure proper informed consent, the REC members acknowledged the importance of identifying potential risks associated with COI among researchers and funding bodies. This included financial and non-financial interests, such as strong motivation to recruit participants.
The researcher. . . the doctor, can be very committed to the study and really eager to recruit. I know how it is from my own studies; you want to be able to include the patient. (P4)
Dependency and vulnerability
The REC members noted that research with children required special consideration, especially when it came to recruitment, due to children’s dependency on parents and healthcare. Overall, they took two main positions concerning children’s vulnerability and capacity for self-determination. Some REC members positioned children as vulnerable, relatively incompetent in terms of decision-making, and in need of special protection. These members emphasized the parent’s role in informed consent.
They’re so dependent on adults. They’re in a vulnerable situation. . .and have difficulty making themselves heard. Reason and argue, they can’t do that. They can’t ask the right questions. (P6)
Other REC members stressed children’s competency and capacity, and explicitly advocated for promoting children’s self-determination through actively and appropriately engaging children of different ages in informed consent procedures.
You should try to treat the child as the competent individual they can be. . . And always adapt information to the maturity of the child and take the child’s questions seriously. . .to take assent seriously. (P13)
Other concerns REC members raised were parents’ vulnerability and burdens related to being invited to participate in research during an emotionally difficult life situation.
It’s an extreme situation for parents, and to be asked about research at that time, I mean that could be the last drop making the cup overflow; it’s just too much. Or they put too much hope in it. (P5) They [parents] may not be capable of reading long texts, and they might misunderstand. It’s a tricky situation, and you have to think carefully about how you inform them. (P9)
To address vulnerability, the REC members encouraged researchers to provide clear differentiation between research and regular care, and to allow sufficient time for informed consent procedures.
Conflicting views and preferences between parents, or between parents and the child, were raised as a possible source of children’s vulnerability. The REC members challenged the assumption that parents always act in their children’s best interests.
Of course, parents should represent their children’s best interests, but how can you be certain that that’s always the case? (P13)
Independent decision-making support was suggested in cases of, for example, intrafamilial conflict, brain injury in the child, or highly complex research.
Protecting children
Protecting children from harm was viewed as a primary ethical obligation for the REC members. They also described a duty to protect children with severe health conditions from poorly designed and irrelevant research. Concerns were described regarding neglect of psychological and integrity risks in applications, and challenges were encountered in risk-benefit analyses due to complexity, subjectivity, and ambiguity.
Wellbeing and integrity
The REC members evaluated applications with regard to medical and psychological risks as well as the burden of participation. They tried to assess the implications of participation from a child’s perspective.
Imagine yourself in the child’s place, and try to see it from the child’s perspective. . . I put myself in the shoes of a child and think, if I’d been exposed to this, what would I have thought of it? (P1)
Their perception was that ethical applications tended to report medical and physical risks to a higher degree than psychological and integrity related risks.
The applicant often considers the physical risks. . .they aren’t focusing so much on the other [risks]. . . (P8)
However, the REC members were concerned with ensuring that children’s self-esteem, sense of normality, and wellbeing would not be negatively impacted. This was seen as particularly ethically problematic when it came to children with terminal illness in drug studies.
A study can require children to spend more time at the hospital. . . and here’s a conflict. . .you take the child’s valuable and, in some cases, short time remaining, time that would perhaps be better spent at home with their parents. But because of this drug study that has quite a small chance of being effective, the child has to come to the hospital. (P4)
Integrity-related risks were also assessed, and the REC members sometimes identified shortcomings related to data management.
That they [researchers] don’t know how they’re going to handle sensitive personal data. (P14)
Integrity issues were also raised in relation to retrospective studies using medical journal data. The study invitation might violate children’s privacy, evoke painful memories of being ill, and affect the children’s future partner relationships.
These retrospective journal review studies involving children are also difficult. . .How much fun is it to get this [research invitation] and be reminded? It’s also an ethical problem if a future partner might begin to wonder and ask questions like, you haven’t told me about this [disease], is it hereditary? (P10)
Further, genetic screening studies involving families were seen as problematic from an integrity perspective, as they may impose information and decision burdens on children.
Genetic screening. . .for a hereditary disease, the difficulties of children being involved. . .there’s this horrible disease in our family. . .do I want to know [if I have it] or not, just knowing that this information exists must be a really heavy burden for the child. . .genetic information, it’s hard. (P10)
Scientific quality and relevance
While the REC members stressed their duty to protect children, they also expressed that they had a duty to promote – and not to prevent – important and well-designed research that could benefit and improve the health of children.
If you think your job is to stop all research, then you’ll behave unethically. . . if you base things on an excessively precautionary principle and have extremely strict or unreasonable demands for medical research involving children, and you think you’re doing children a favour. . . you aren’t. (P11)
Scientific quality and relevance were therefore important aspects to consider for the majority of these REC members.
Does the proposed design have sufficient scientific quality to answer the research questions? If the design is poor, the scientific benefits will be minimal. So, even if the risks for the child are negligible, we should reject [the proposal]. (P10)
Nevertheless, the interviews made it clear that there had been some indecision within the authority regarding praxis and directives for evaluation of scientific quality in ethical review. As a result, the emphasis on scientific quality aspects varied between the REC members.
The REC members felt that burdening children with severe health conditions with research and associated risks had to be ethically outweighed and motivated by evident prospects of benefit to children.
If we’re going to involve young children in research that will take up their time, severely ill children, it has to be outweighed by good things, either for these children or for other children later on. It doesn’t make sense that they should make sacrifices for something that won’t benefit anyone. (P13)
Besides quality, the REC members considered the relevance of the study to be ethically important. They thought that children with severe health conditions should not be subjected to irrelevant or already answered research questions.
If your research question has already been answered, there’s no point in exposing a group again, and especially not severely ill children. I mean, the benefits should really be high. . . Otherwise, I think we should leave the children and the family alone. . . If we already know, we shouldn’t be asking the same questions again. (P5)
Complexity of risk-benefit analyses
Alongside assessment of information for children, risk-benefit analyses were at the core of ethics review. The REC members argued that risks should be motivated and weighted more heavily in research with children, and demands for benefit should be higher than in research with adults. In reality, it was not always easy to determine benefit requirements and acceptable risk levels for children with severe health conditions. The REC members had to balance risks against benefits that involved ambiguity and subjective judgement.
Risks and benefits are not well defined, something you can assess through a couple of checkboxes. . .It’s quite a difficult process. . .and you may end up with different results. (P8)
The REC members also pointed out that perceptions of risks and benefits are not objective but rather affected by personal factors, situations, and life history. This highlighted the importance of transparent risk information, to allow adequately informed consent.
The participant reading the study information might have different experiences. Researchers cannot really evaluate information until it is presented to the participant. (P12)
Uncertainty around benefits and risks, for example in early exploratory drug studies with critically ill children, complicated risk-benefit analyses further.
In phase I and II trials, you don’t know the effect. . .and [the drug] probably won’t work, because most drugs don’t make it past phases I and II. (P3)
Regulatory adherence
The REC members emphasized their obligation to ensure adherence to legal and formal research requirements, such as study information, informed consent, and protection of privacy. They stated that they relied primarily on Swedish law for ethical review as the formal basis for ethical decision-making. Although deficiencies in applications were seldom considered significant enough for rejection, the RECs frequently requested amendments based on non-adherence to regulatory requirements.
Legal interpretation
Although the law was generally perceived as a solid basis for decision-making, with the relevant ethical principles incorporated, some challenges were experienced in relation to interpreting and applying the law. The REC members explained that interpreting the law required a broad set of specific competencies connected to various fields of research, which might not always be represented in the REC.
Legal interpretation is demanding. . . and specialist expertise in various research fields is needed to figure out how to comply with the law. . .an extreme amount of different research methods and research logics. . . you won’t cover that. (P11)
Moreover, REC members from different professional backgrounds might interpret the law differently, and also might assess risks differently. However, different perspectives and disagreements were seen as crucial in seeking consensus and in ensuring the quality of ethical review.
A medically-oriented person might find a procedure completely OK, but from a psychosocial viewpoint it might be too much to expose people to. Your own perspective will affect which ethical values you find important in research. . .There can be conflicting perspectives among committee members, but the important thing is reaching consensus. (P1)
When consensus was not achieved, the REC members valued the possibility of seeking advice from a higher central body, The Ethics Review Appeals Board.
Ethical competence development
Besides the law, and clinical and scientific competence, the REC members perceived personal characteristics as important for ethical deliberation. Specific personal virtues that were mentioned included being flexible, perspective-taking, problem-solving oriented, conscientious, judicious, analytical, reflexive, impartial, liable, and aware of one’s power position. Teamwork skills such as communication and paedagogical skills were also seen as crucial for REC decision-making.
You have to be good at communicating and listening to others. Some people lack the ability to listen, and just run their own race, which prevents good discussions leading up to a joint decision. After all, it has to be a joint decision. (P7)
Some RECs described relying on a personal moral compass and personal experiences. This moral compass was described as integral to being a clinician, or as part of being a human who has relationships with significant others. The moral compass was enhanced through perspective-taking.
I’m also here reviewing as a human being. Many of us will say things like, I have a son, or a mother, who’s in exactly this situation, and I would never agree to this study. . . ethics, we carry within us at all times, and our values and legal pathos, and we must bring it with us into ethical review. (P5)
At the same time, the REC members acknowledged that there was a risk of their personal experiences and engagements biasing their decision-making and causing COI. They felt it was important to abstain from participating in the discussion or decision-making when they were too personally engaged.
Some of the REC members were concerned about the predominant focus on regulatory aspects in ethical review. Concerns were raised that a narrow focus on regulatory adherence might preclude other important ethical aspects from being raised in relation to paediatric research.
There seems to be a lot of emphasis on legal compliance, which of course is very important. But it isn’t the only thing that’s important. . .there’s a risk that the primary focus will be on the legal aspects. (P13)
The REC members also raised concerns that formal ethics competence and training within RECs might be insufficient. Educational efforts within the authority were encouraged, to ensure basic formal ethics competence and ethical deliberation based on established research ethics principles.
Some basic research ethics education probably wouldn’t hurt. It would help, because you wouldn’t have to repeat the basic arguments each time. . .the education that’s offered and the discussions are of a very legal nature. . .as a result, the development of the committee is not going in the moral philosophical or ethical direction, and I think that’s unfortunate. (P11)
Ethics competence development within the authority, especially in complex research areas such as genetic or paediatric research, was seen as important.
Transparency and representation
The REC members said that while individual competencies were important, decisions were made through a group effort, and the REC’s collective competence was based on representation of different backgrounds and perspectives. Representation from the public is required by Swedish law, and the REC members from the scientific community felt that this played an important role, especially in reviewing information and in increasing the REC’s transparency towards the public.
We shouldn’t be a secluded society that reviews and approves research. (P14)
Concerns were raised that there seemed to be barriers to the active participation of the public representatives in REC meetings.
Some of them are just quiet. But some are a bit more active and comment, especially on things like understanding and study information. The engagement varies a lot. (P4)
Power imbalances and a predominant focus on the formal and legal aspects of research were raised as possible explanations for the public representatives not being active or raising potential concerns.
It’s very rare for a [representative from the public] to object, so I’m not sure this works. What if they sit there and think this is so stupid, but don’t dare to say anything because there are ten of us with PhDs who all think this idea is great? (P3)
This limited engagement was perceived as a problem as it meant there was a risk that ethical review would consist solely of researchers reviewing other researchers, without real representation from public society.
Discussion
Ethical challenges in paediatric research have been described by previous researchers and pertain, for example, to inclusion, recruitment and obtaining parental consent/child assent (See, e.g. Dupont et al., 2016). This is the first study to present Swedish scientific REC members’ experiences and perspectives on research applications involving children with severe health conditions. In accord with previous research, REC members in this study highlighted aspects related to information for children, protection and integrity, scientific quality and regulatory adherence (Angell et al., 2010; Bergstraesser et al., 2020; Taplin et al., 2022). The REC members saw it as their responsibility to promote research with children while ensuring that children’s and parents’ rights to participation should always be promoted through appropriately adapted information. They strove to protect children from medical, psychological, and integrity-related risks, and burdens of research. The REC members also perceived that they had a duty to ensure that children were not subjected to research that would not produce beneficial results or advance knowledge. Shortcomings in age-adapted information as well as the limited regulatory specificity concerning child assent were perceived as challenging, in line with previous research (Bergstraesser et al., 2020; Harcourt and Quennerstedt, 2014; Whittle et al., 2004). Results also align with established principles in research ethics and provide illustrative examples of
REC members described how promoting children’s participation can raise challenges. Children may have limited decision-making capacity or legal authority to make decisions, and difficulties protecting their rights and interests (Council for International Organizations of Medical Sciences, 2016). Therefore, children’s assent is recognized as morally important by international research ethics guidelines (Swedish Ministry of Education and Research, 2004; World Medical Association, 2013). To promote children’s participation, REC members focused extensively on the study information presented to children and parents in their review, and the language used in recruitment materials. They advocated for children’s rights to adapted information, but often identified problems with insufficiently adapted or lacking information for children. Similar deficiencies have been reported by RECs in Switzerland (Bergstraesser et al., 2020). Researchers are required by Swedish law to inform children as far as possible about research. This includes aspects such as the plan and purpose of the research, methods, consequences and risks, reference to the research principal, the voluntary nature of participation and the right to withdraw (Swedish Ministry of Education and Research, 2004). Children’s preferences have moral weight, without appeal to autonomous decision-making (Navin and Wasserman, 2019). Assent processes fulfil an important role in supporting children’s development of autonomy, promoting children’s self-esteem and wellbeing, recognizing children’s moral worth and dignity, and safeguarding their right to express themselves (Archard and Skivenes, 2009; Diekema, 2003; Sibley et al., 2016). Therefore, researchers should promote children’s active participation (Council for International Organizations of Medical Sciences, 2016). However, best practices for designing information and assent processes to fit children’s needs are largely lacking (Cayouette et al., 2022). This also applies to Sweden, where there is currently little formal practical guidance for child researchers (Harcourt and Quennerstedt, 2014). Our study suggests that increased efforts are needed to improve the ethical aspects of information and assent in research involving children. This is important both from a children’s rights perspective and from a research ethics perspective. Some of the REC members felt that the absence of children’s perspectives on information and assent was problematic.
REC members highlighted that the vulnerable situation of many children who are diagnosed with severe health conditions, and their parents, calls for specific considerations in designing information and consent; the burden a severe health condition often has on the lives and wellbeing of children and parents, is described by many others (Boles and Daniels, 2019; Cohn et al., 2020; Emerson and Bögels, 2017; Law et al., 2019). Further, REC members recognized that decisions about research participation may be highly distressing for parents, who might have difficulties comprehending information. In the literature, early-phase trial recruitment has been found especially distressing due to desperation and uncertainty about the benefits and side-effects (Alahmad, 2018; Robertson et al., 2019).
The REC members were concerned that, for different reasons, parents may not always protect or consider their children’s best interests when making decisions. Consent regulation places children in legal dependency towards parents, who are regarded as the primary protectors for children. Therefore, any circumstances that affect the parents’ ability and resources to protect the child’s interests in the context of recruitment also constitutes a source of vulnerability for the child. Parents’ abilities to protect may be affected by psychological distress or social disadvantages, for example (Hurst, 2015). Despite the wide range of vulnerabilities that may arise in conjunction with children’s participation, the results suggest that while medical risks and harms might be emphasized by researchers, less attention might be paid to psychological or integrity risks. Researchers are legally obliged to protect children from psychological harm and intrusions of privacy and confidentiality. By neglecting such risks, researchers may contribute to children’s vulnerability in research. Research suggests that children may experience burdens that often go unnoticed, such as feelings of obligation to partake (Barned et al., 2018). The interviewed REC members largely approached vulnerability with consideration of the unique medical, psychological, and social situation of both children and parents. This bears resemblance to the contextual approach to vulnerability increasingly supported by various scholars (Hurst, 2015; Luna, 2009). Viewing risks and vulnerability from a holistic and long-term perspective might be especially important in research involving children with severe health conditions; these children may be adversely impacted by their illness for a long time, and in many areas of life. Systematic analysis of contextual vulnerability may be a helpful tool for RECs, in the identification of adequate protections to mitigate children’s vulnerability in research (Bracken-Roche et al., 2017). Sources of vulnerability can, for example, be identified based on the taxonomy proposed by Gordon (2020), which includes cognitive, communicative, institutional, deferential, economic, social or medical aspects (Bracken-Roche et al., 2017; Gordon, 2020).
One interesting and important point emerging from our results was that the REC members varied substantially in how they perceived children in terms of vulnerability, decision-making capacity, and need for protection. Opposing views within a REC can cause tensions between the desire to protect children versus including children, as Friesen et al. (2022) have also described (Friesen et al., 2022). It is important for REC members to critically reflect on the views they hold around children, and how these views can influence their reviewing (Carter, 2009; Lambert and Glacken, 2011). Little is currently known about how RECs understand and apply the concept of vulnerability, and more guidance may be needed for the concept of vulnerability to be practically useful. As Bracken-Roche et al. (2017) propose, such guidance should be informed by children’s voices and experiences, which are missing from current guidelines (Bracken-Roche et al., 2017).
The REC members in this study made it clear that the dependent and vulnerable situation of families brings a particular duty to protect the voluntary nature of participation. Without a voluntary decision-making process, informed consent is invalid (Appelbaum et al., 2009). The REC members especially highlighted honesty and the voluntary nature of participation as central values, but noted that full transparency concerning side-effects, for example in drug trials, was sometimes lacking in applications. Deciding on the depth and width of information required to make an informed and voluntary decision, without overwhelming children and parents, can be a difficult task. Health benefits and side-effects related to trial participation have been reported to be the most valued and relevant information for families considering participation in drug trials (Koonrungsesomboon et al., 2022).
The REC members considered COI to have ethical relevance in research with children with severe health conditions. They were concerned that the financial interests of funding bodies might impact moral research conduct, or that the presence of non-financial academic interests among researchers, such as the desire to recruit to target, or attain academic achievements, might lead to undue pressure on families to enrol. Regulation has primarily focused on financial COI, which may be easier to identify than non-financial COI. Further, non-financial COI can often be unavoidable (Romain, 2015; Saver, 2012). As an example, due to the relative rarity of childhood cancers, paediatric oncology research in Sweden is performed in a relatively narrow context. COI situations might be difficult to avoid since the same people can be involved in two possibly competing interests: research and care (Schröder Håkansson et al., 2020). The REC members in this study brought up the management of COI as an important ethical question. If not dealt with properly, these secondary interests may negatively impact scientific and moral integrity, the protection of participants, and public trust (Saver, 2012). In terms of researchers’ interest in recruiting patients, the REC members felt it was important to clearly differentiate between research and care, to emphasize the voluntary nature of research, and to offer impartial decision-making support to the families when needed.
It was evident from the interviews that risk-benefit assessments and legal interpretation could be complex and demanding for RECs, especially given the rapid scientific and technological development of healthcare research today. As acknowledged by some of the REC members, ethical deliberation goes beyond mere regulatory adherence. Ethical competence, personal moral virtues and teamwork skills are important for RECs, as regulations and ethical guidelines alone cannot ensure well-founded ethical decision-making (Eriksson et al., 2007; Johnsson et al., 2014). Moral virtues that signify personal character traits which manifest themselves in habitual action are beneficial for a person to have (Rachels and Rachels, 2015). Ethics competence has been conceptualized in terms of ethical sensitivity, ethical knowledge, ethical reflection, and ethical decision-making (Lechasseur et al., 2018). This corresponds with the type of ‘ethical preparedness’ that Lyle et al. (2023) advocate should be fostered by researchers and RECs (Lyle et al., 2023).
This study identified barriers to the active participation of REC members from the public in ethical review. Ethics review is a group effort in which diversity is critical for quality, fairness, trust, and equality, and where public representatives can promote openness and counterbalance scientific interests (Staley, 2013). However, RECs tend to be homogenous and some research challenges the assumption that public representatives adequately represent the patient perspective (Masterton et al., 2014). Absence of a public perspective could compromise the system and quality of ethics review (de Veras Santos and Guerriero, 2022). Maintaining the public’s trust in biomedical research, and interpersonal trust between clinical researchers and patients and their families, is crucial for the scientific enterprise and to recruit and retain participants (Norbäck et al., 2023; Resnik, 2011). Research on how to appropriately include the perspectives of children with severe health conditions about relevant aspects of ethics review and approval is warranted. Further, the perspectives of REC representatives from the public on this topic need to be investigated, as they are not included in this study.
Strengths and limitations
This study has methodological strengths as well as limitations. One strength is that all interviews were conducted by the same author. In reflexive thematic analysis, knowledge is actively produced through the researcher’s active, subjective, and reflexive analytic input, which is considered a resource (Braun and Clarke, 2021a, 2021b). Collaborative coding was used to enrich the quality of analysis, and expertise in psychology, paediatrics, nursing, nursing ethics, clinical ethics, and research ethics was represented among the study co-authors. However, none of the co-authors had prior experience of being a REC member. To maintain authenticity, we adopted an inductive approach, and revisited the transcripts and codes while refining the themes. The REC members were specifically asked about applications involving children with severe health conditions and cancer was used to exemplify to the REC members what a ‘severe health condition’ could entail. However, the term ‘severe health condition’ can be ambiguous and it is possible that the interviewed REC members assigned it different meanings. Moreover, the large quantity of studies that REC members review might make it difficult for them to recall or differentiate ethical issues specific to research with children with severe health conditions from research with children more generally.
We included scientific REC members from all six operating regions to capture meaningful variation and account for potential local differences in culture and perceptions within the different RECs. We found the sample specificity satisfactory, as the REC members were experienced in ethics review and represented a wide range of professional, scientific, and medical specialities (Malterud et al., 2016). However, there might be a self-selection bias among scientific REC members who agreed to participate. Moreover, the perspectives of REC members from the public are, for practical reasons, not represented in this study. Members of the public fulfil a vital function in RECs, for example in ensuring that research participants are protected and in checking the quality and comprehensibility of written materials (Staley, 2013). Further, their representation is required by Swedish law. The absence of their perspectives in this study should be considered a shortcoming and further studies exploring their perspectives are warranted. To facilitate transferability, we have thoroughly described the Swedish REC context and regulations. Nonetheless, to ensure confidentiality, we did not report professional background, which may restrict evaluation of the sample composition and thereby transferability. The REC members’ perspectives and experiences, as well as the analytic work of this study, should be considered with regard to the societal and regulatory context in which they were produced (e.g. the Swedish context). There are similarities as well as differences between countries in terms of how research with children is regulated, including requirements for parental consent and child assent (European Union Agency for Fundamental Rights, 2019). This study reports issues which are widely recognized as relevant in paediatric research, regardless of regulatory context (e.g. rights to information and choice, protection and regulatory adherence). Nonetheless, there may be certain limits to applicability in other settings or countries where RECs and paediatric healthcare and research are differently organized and regulated (Braun and Clarke, 2021b).
Conclusions
Ethical review of paediatric research is demanding and requires ethical competence to reach well-grounded decisions. The interviewed scientific REC members’ perspectives on paediatric research applications revolved around participation, protection, and regulatory adherence. Adapted information and assent processes are crucial to support children’s active participation and decreasing vulnerability. Therefore, efforts might be needed to develop best practices for information and assent. REC members’ narratives largely support a contextual and holistic approach to vulnerability and risk, acknowledging the unique medical, psychological and social situation of paediatric patients and their parents. However, results also indicate that individual REC members may have different perspectives on children’s capacities, vulnerability, and opportunities for participation. Children’s perspectives on assent are essential but have largely been left out in development of research ethics guidelines.
Footnotes
Acknowledgements
The authors would like to thank the REC members who contributed to this study by sharing their experiences and perspectives.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() . This work was funded by the Swedish Childhood Cancer Fund [PR2019-0107]. The funder had no role in the design, data collection, analysis, interpretation or writing of the manuscript. Open access funding provided by Uppsala University.
. This work was funded by the Swedish Childhood Cancer Fund [PR2019-0107]. The funder had no role in the design, data collection, analysis, interpretation or writing of the manuscript. Open access funding provided by Uppsala University.
Ethical declarations
According to Swedish legislation, this study did not require formal ethical approval as it did not fall under the Swedish Ethical Review Act. However, an advisory statement was granted by the Swedish Ethical Review Authority (ref: 2021-00570) and no objections were raised. The study followed the ethical requirements for research with human subjects as outlined by the Declaration of Helsinki (World Medical Association, 2013). Data were managed following the General Data Protection Regulation (2018).
