Abstract
Background:
In France, approximately 40% of women, including menopausal women, do not participate in cervical cancer screening. Many studies and meta-analyses have shown that self-sampling devices for high-risk human papillomavirus (HR-HPV) testing are valuable tools to increase participation. The success of self-sampling screening strategies depends on several factors, including the manner and circumstances in which the women are invited to participate. The acceptability and effectiveness of these strategies should be evaluated before further implementation.
Objectives:
The primary objective is to evaluate the uptake of cervical cancer screening in under-screened women, based on a direct offer of a vaginal self-sampling device by a midwife.
Design:
RIDECA is an interventional research project located in the South of France.
Methods and Analysis:
Six hundred women aged 50–65 with no cervical smear or HR-HPV test for 3 years or more will be recruited at two sites. The device will be offered to enrolled women to utilize at home and return to the Montpellier Hospital laboratory for HR-HPV testing. Completion of self-sampling by women who have accepted the device and follow-up of those with positive HR-HPV DNA tests will be monitored. During recruitment, participants will complete questionnaires on their socio-economic environment and motivational factors based on the psychosocial I-Change model. Semi-structured interviews will be conducted with a sub-group to identify barriers and levers to participation. Statistical analyses will be conducted for the full research sample and for each recruitment site.
Ethics:
The project was approved by the Ethical Research Committee Ile de France VI and by the French Data Protection Authority.
Discussion:
The results will provide useful information on the effectiveness (acceptability, efficiency) of this outreach strategy and identify barriers and levers that facilitate its implementation.
Registration:
ClinicalTrials.gov (NCT04716127), January 20, 2021.
Plain language summary
In France, 40% of women, especially postmenopausal women, do not participate in cervical cancer screening. We already know that vaginal self-collection with HPV testing is highly accepted by women and increases their participation in screening. Different strategies have been proposed as the most appropriate approach to utilize when offering the vaginal self-sampling option to women.
The study, set up in the Occitania region of France, is still ongoing and is targeting women aged 50–65 who have not been screened for cervical cancer. The aim of the study is to assess whether reaching women in their everyday environment and offering them a vaginal self-collection device during an educational session with a midwife improves their adherence to and participation in cervical cancer screening.
To answer these questions, we will recruit six hundred women aged 50–65 who have not been screened for cervical cancer for three years or more at two sites:
- In the department of Hérault, where midwives will approach women visiting a mobile breast cancer screening unit,
- in the department of Aude, where a midwife can easily arrange appointments for interested women, following an information campaign.
Participants who agree to take part will receive a self-sampling kit to use at home and return to the laboratory for HR-HPV testing. During recruitment, we will also use questionnaires and interviews to assess women’s socio-economic situation and motivations for participating (or not) in cervical cancer screening.
This study will allow us to assess women’s adherence to this “direct” “face-to-face” cervical screening strategy and the potential influence of socio-economic and motivational factors on adherence. Through gaining these vital insights, we hope to identify the barriers and levers that will facilitate its implementation.
Keywords
Introduction
Epidemiology of cervical cancer and cervical cancer screening in France
In most industrialized countries, implementation of cervical cancer screening based on cervical cytology (Pap smears) has dramatically decreased the incidence and mortality of cervical cancer. In France, cervical cancer ranked 12th among all forms of women’s cancer, with incidence and mortality rates estimated in 2023 at 3159 and 1100 women, respectively.1,2 Nevertheless, the non-attendance rate among French women remains significant, amounting to about 40% of those selected for screening from 2018 to 2020.3 –5 To improve the screening coverage rates, the French Health Authorities have launched an organized population-based screening program (Dépistage Organisé du Cancer du Col de l’Utérus—DO-CCU) starting in 2019. 6 The DO-CCU program is based on mailing non-attendee women a reminder to perform cervical cancer screening. The screening methods adopted were also updated in 2021. Cervical cancer screening, performed as part of a gynecological examination by a healthcare professional, is now based on a cervical smear for women aged 25–30 years (every 3 years if normal) and high-risk human papillomavirus (HR-HPV) testing for those aged 30–65 years (every 5 years if normal).7,8 Regarding self-sampling, the French Health Authorities recently recommended its use for HR-HPV detection in two situations: first, when women do not respond to the invitation letter after 1 year, and second, when women participate in specific interventions or interventional research programs targeting non-attending and hard-to-reach women. 9
Self-sampling for cervical screening
A number of recent publications and meta-analyses have investigated the value of proposing self-sampling along with HR-HPV testing for cervical cancer screening. Their findings suggest that this approach could be an effective strategy for increasing participation in cervical screening, particularly among non-attendee women.10,11 In light of these results, an increasing number of countries, especially after the COVID-19 pandemic, have considered self-sampling as a method to reach under-screened women or as an option in an organized screening program, as in the Netherlands. 12 Technically, numerous studies and meta-analyses have shown that vaginal or urine self-sampling with PCR-based HR-HPV DNA detection has similar performance to clinician-collected cervical smears for the detection of CIN2+ lesions.10,13,14 In addition to the clinical accuracy of the self-sampling device and the HR-HPV test used, there are other important issues to consider before implementing self-sampling as a cervical cancer screening option. These include understanding the profiles of women who might choose to use a self-sampling device and their motivation to do so. For example, self-sampling is more effective than invitation letters in increasing screening participation among under-screened women. 10 It is also important to determine the most effective method of delivering the device to women. This may entail postal delivery, opt-in, community campaigns, or direct distribution through door-to-door visits. Of the aforementioned approaches, door-to-door delivery, which has been evaluated mainly in the context of middle- and low-income areas, has been found to be the most effective in terms of increasing women’s participation.10,11 However, the latter approach involves extensive logistical considerations. These include identifying the best locations and times to reach these women, as well as the human and physical resources needed to facilitate the delivery of self-collection devices. It also requires the allocation of resources to contact and follow up women with a positive HR-HPV test.
Context of the study and hypotheses
In France, women who are not screened for cervical cancer are often older (>50 years old) or present unfavorable socioeconomic conditions. 3 Recent data have also confirmed the existence of French territorial inequalities in terms of screening participation. 4 For instance, the estimated screening participation rates in the departments of Hérault and Aude, located in the south of France, during the 2018–2020 period were 61% and 56%, respectively. This rate declines to 47% and 40%, respectively, among women over the age of 60. In both of these departments, a preliminary study based on a self-administered questionnaire has revealed a high level of acceptability of vaginal self-sampling regardless of age, gynecological follow-up, and educational level. 15 Notably, in this and other studies, acceptability was significantly higher in the group of women over 50 years of age.15,16
The aforementioned data lead us to hypothesize that an outreach strategy for non-attendee women, which has rarely been evaluated in developed countries and never in France, will be a valid complementary approach to those previously organized and evaluated in this latter country (including postal invitations to cervical smears, sending a self-sampling device for use at home, opt-in options, and promotion by general practitioners). Our intervention will focus on under-screened women 50–65 years of age, in the low-participation departments of Hérault and Aude.
Methods
Objectives and endpoints
The primary objective of this study is to evaluate the uptake of self-sampling HR-HPV amongst under-screened older women when a device is proposed to them directly by a health professional (more details in Supplemental Material 1). The primary endpoint is the uptake rate, defined as the ratio between women who accept the self-sampling device and those to whom it has been proposed.
The secondary objectives are to evaluate:
– The psychosocial determinants and the role of the socioeconomic context in uptake of cervical cancer screening in this population.
– The adherence to the proposed screening strategy assessed in terms of the proportion of women who performed the self-sampling and returned the device to the laboratory, out of those who took the device home (at the latest, 1 month after the last reminder, mailed 6 months after recruitment).
– The efficacy of vaginal self-sampling with HPV testing for cervical cancer screening, assessed in terms of the proportion of women who test positive for HR-HPV and follow recommendations by having a cervical smear within the following 12 months.
Study design
RIDECA (Interventional Research for Cervical Cancer Screening) is a prospective interventional study. This protocol has been designed following the SPIRIT 17 guidelines (Supplemental Material 2). Schedule of enrolment, intervention, and assessments is summarized in Figure 1.

SPIRIT schedule of enrolment, intervention, and assessments.
Participants
Inclusion and exclusion criteria
The project targets women aged 50–65 years, with no cervical smears or HR-HPV tests for at least 3 years (⩾3 years) and no exclusion criteria (Table 1). The criterion “no cervical smears or HR-HPV tests for at least 3 years” is based on participants’ declarations at the time of recruitment, but this criterion will be verified for each woman at the end of the study by requesting confirmation from the Health Insurance Department database.
Inclusion and exclusion criteria of the RIDECA study.
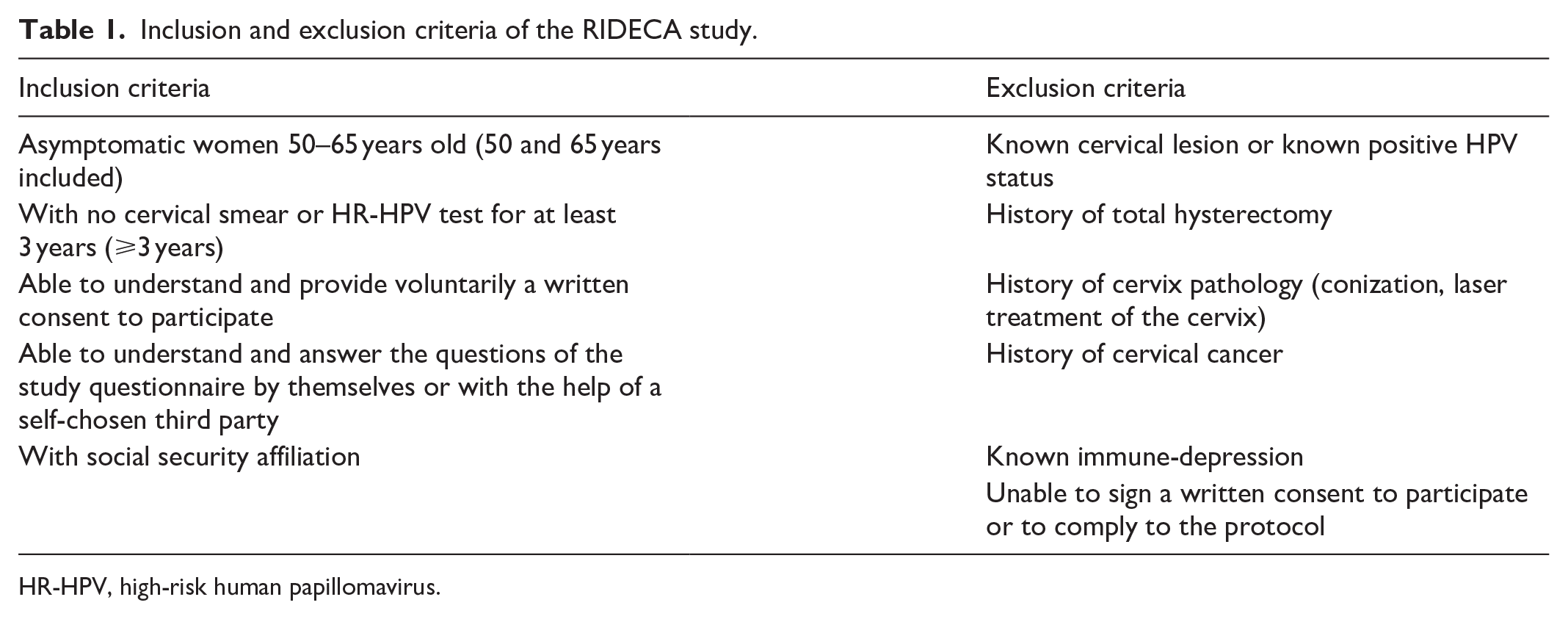
HR-HPV, high-risk human papillomavirus.
Setting
Women are recruited at two sites, one in each department covered by the study, through two different procedures:
– In the department of Hérault, women are recruited by the midwife while attending the mobile unit for breast cancer screening (the “mammobile”). This mobile unit consists of a truck giving easy access to mammography and clinical examination by a physician, for eligible women aged 50–74 years in the department of Hérault. Since 1991, the mobile unit has been traveling around nearly every working day, covering 250 communities in the department. The midwives are present at the mammobile site 6 days per month, from 8.30 am to 5 pm, in daily rotating shifts. In order to specifically target the most under-screened population, women are recruited in communities with an overall participation rate in cervical cancer screening of less than 52% (⩽52%), according to the Health Insurance Department database.
– In the department of Aude, recruitment takes place around the city of Limoux. The department of Aude, especially this area, is one of the French departments with the lowest social and economic resources and the lowest human health resources, showing considerable geographical differences. The midwife works for the project 1.5 days a week, giving women easy appointments, either at the Social and Medical Care Center in Limoux or by proposing the self-sampling device in medical centers, City Halls, during regional health promoting events or in local drugstores. These actions take place after promoting the RIDECA project in the community, through articles in local newspapers and information on social networks, and through meetings and telephone calls with women who have previously visited the Social and Medical Care Centre in Limoux.
Sample size calculation
The exact proportion of the target population, composed of non-attendee women 50–65 years of age who will predictably accept the proposed vaginal self-sampling device, is not yet known. In the preliminary study, 80% of the women interviewed declared that they would accept the vaginal self-sampling device for cervical cancer screening. 15 Since this acceptance rate was based only on declarative evidence and because of the bias identified in this preliminary study, we can expect to obtain a lower uptake rate in the present study, of approximately 50% among the women to whom the self-sampling device is proposed directly. With a sample size of 600, a two-sided 95% confidence interval (CI) for an expected uptake rate equal to 0.5, will therefore range from 0.46 to 0.54 (CI width of 0.08).
Duration of the recruitment
The duration of the recruitment will depend on the percentage of under-screened women aged 50–65 years and on the number of women attending the mobile unit or the various sites around Limoux identified by the midwives. Accordingly, the time required to recruit 600 women is estimated to be approximately 24 months.
Intervention
The proposed intervention consists of three stages (Figure 2).

Flowchart of the RIDECA study protocol.
Meetings with the midwives (stage 1)
At each of the two sites, the midwife recruited and trained for the project invites women to participate in the study, based on specific inclusion and exclusion criteria, and to declare in writing their non-opposition to the data collection process. In case of refusal, the mid-wife asks for the woman’s non-opposition to collecting selected data (age, city of residence, and the main reasons for not participating). Women who agree to participate are asked by the midwife to sign the informed consent form and give their address and name their referring health professional. They are then helped to complete a questionnaire (see “Data collection and management” section) and invited to participate 4 months later in a telephone interview conducted by a researcher in the field of Health Psychology. The midwives next dispense some educational information about cervical cancer and cervical cancer screening, using various supports (illustrated leaflets in various languages and a short video) and present the vaginal self-sampling device with complete instructions, including how to use it at home and return it to the laboratory using a prepaid envelope, and explaining the meaning of HR-HPV test results and the follow-up process if relevant. At this stage, women are free to accept or refuse the self-sampling device.
Returning the self-sample (stage 2)
Women are asked to perform the self-sampling at home within 1 month of the meeting with the midwife, and to return it by prepaid mail to the laboratory within 2 weeks after performing the self-sampling procedure. Those who have accepted the vaginal self-sampling device at enrolment but have not returned it are sent letters of reminder 3 and 6 months after their recruitment. If the vaginal self-sample has not been returned in response to the two reminders, the case is classified as lost to follow-up.
Follow-up (stage 3)
Whatever the result of the HR-HPV DNA test, each woman and the health professional she named at enrolment (a general practitioner, a gynecologist, or a midwife) receive the results of the HR-HPV test along with a letter giving recommendations about cervical cancer screening. Women whose test is unsuccessful are sent another device along with a letter of explanation. The follow-up of those whose HR-HPV DNA test is positive is based on recommendations published by the French Health Authorities.18,19 In addition to the mailing, the midwife phones these women, the midwife phones these women to inform them that the HR-HPV test results are positive and recommend having a cervical smear performed within the next month. Completion of follow-up consists of mailing questionnaires to these women and their referring health professional 3, 6 and 12 months later, and calling any non-respondents by phone 6 months later. Those with no reported cervical smear at 12 months are considered lost to follow-up.
Laboratory procedures
Vaginal self-sampling device and HR-HPV testing
During their recruitment, women are asked to perform self-sampling at home using the vaginal self-sampling device (Evalyn® brush; Rovers Medical Devices B.V., Oss, Netherlands) as explained by the midwife, to label the device with their anonymous number and the date of sampling and to return it to the laboratory in a pre-stamped envelope within the next 2 weeks. Upon receipt in the laboratory, the vaginal self-sample is processed for the detection of HR-HPV using the Cobas 4800 HPV DNA test (Roche Diagnostics, Meylan, France). More details are available in Supplemental Material 3.
Detention of sample for future research
With women’s informed consent, DNA extracted from the vaginal self-sampling device is conserved at −80°C in a declared sample collection for future molecular research purposes.
Socioeconomic measures and qualitative study
In order to achieve the secondary objectives, a self-administered questionnaire is proposed to all the women included, containing multi-choice questions and open and semi-open questions about:
– Their socioeconomic situation (e.g. about their occupational status, lifestyle (living with a partner or single), level of education, level of medical education and literacy, medical follow-up, follow-up for chronic illness).
– The circumstances which prompted them to come and their prior knowledge about this screening program.
– Their motivational determinants as regards health, screening in general and cervical cancer screening in particular. These closed questions, using Likert scales (from 1 to 10) and semi-open questions (“key words”), were drawn up by the Epsylon laboratory team on the basis of the I-Change Model, 20 an integrated model of behavior change derived from ASE model (Attitude Social Influence Self-Efficacy Model), which includes the perception of autonomy support and autonomous motivations, self-efficacy, intention, and behavior states adapted to the topic of cervical cancer screening.
In addition, social deprivation will be quantified using the European Deprivation Index (EDI) score, 21 based on a territory unit (IRIS) defined by the French National Institute of Statistics and Economic studies (INSEE). EDI is generally categorized into quintiles from Q1 (the lowest deprivation quintile) to Q5 (the highest deprivation quintile).
Semi-structured interviews will be conducted 4 months after original recruitment cohorts with a view to exploring possible barriers and facilitators to the uptake of self-sampling as a means of screening. These interviews will be with 45 women who originally consented to the study, but will utilize purposive sampling to incorporate women from three different categories:
– 15 women who have refused the self-sampling device
– 15 who have accepted and performed the procedure
– 15 who have accepted it but have not performed the procedure 4 months after inclusion
These interviews will include both semi-open (key words) and open-ended questions exploring the following key thematic areas framed by the I-Change Model:
– Representations related to cervical cancer and its screening (referring to the “Awareness factors” in the I-Change Model)
– Perceived influence of information on decision-making (i.e., “Information factors”)
– Motives for performing/not performing vaginal self-sampling (i.e. “Motivation factors”)
– Perceptions of vaginal self-sampling and related perceived control (i.e., “Motivation factors” and “Ability factors”)
– Intentions and behavior states related to various cancer screenings, especially cervical cancer screening (i.e., “Intention state” and “Behavior state”)
Data collection and management
An electronic case-report form (e-CRF) has been developed using the ENNOV CLINICAL® software to check the quality of the data. This e-CRF also contains a specific section to monitor potential adverse effects. In order to ensure anonymity, participants are identified on the e-CRF by an identification number alone. The e-CRF is completed in each case by the mid-wife and the Clinical Research Assistant. An online input mask with controlled and limited access, contains the information necessary for sending the HR-HPV test results to the participants and the healthcare professional they have named and for the follow-up if appropriate.
During the meetings with the women, the acceptance of the self-sampling device by those to whom it has been proposed is recorded directly by the midwife, and the self-administered questionnaire is completed by the women themselves, with the help of the midwife if necessary. The Social Health Inequalities Platform, hosted by the INSERM UMR1027 in Toulouse, will calculate the EDI scores. It will be provided with a file containing an anonymized identifier and a postal address, in order to assign an EDI quantile to each woman, based on her place of residence. Lastly, follow-up data (use and return of the self-sampling device, HR-HPV test results, letters of reminder, participation in the semi-structured interview, follow-up data in cases with a positive HR-HPV test, lost to follow-up) are assessed directly by the Clinical Research Assistant.
Semi-structured interviews will be conducted by pre-arranged phone calls by a single researcher in the field of Health Psychology. The interview guide designed to facilitate discussions is quite flexible, allowing for the possible inclusion of new themes emerging during the exchanges. After obtaining permission to record, interviews will be audio-recorded before being transcribed.
Data analysis
Quantitative analysis
RIDECA is an interventional survey with a large descriptive content. The statistical analyses will be detailed in a statistical analysis plan finalized before the last patient is included in the study. Analyses will be performed both on combined study population and on the data obtained at each of the two recruitment sites separately. Quantitative variables will be reported using means, standard deviations, medians, and quartiles. Qualitative variables will be described using numbers and percentages with a CI of 95%. The 95% CIs will be calculated using the simple asymptotic method (as regards the primary endpoint, in particular). Qualitative variables will be compared using Chi-square or Fisher’s exact tests, and quantitative variables using Mann–Whitney or Student’s t-tests, depending on the normality of the distributions.
The study of the factors involved in uptake of cervical cancer screening will be performed using a logistic regression model. Clinically relevant factors or variables with a (univariate) p-value <0.10 will be selected for multivariate analysis and a backward covariate selection procedure will be as performed. OR will be reported with its 95% CI.
All statistical tests will be two-sided, and p-values <0.05 will be taken to be statistically significant. Statistical analyses will be performed using SAS software 9.3 (SAS Institute, Cary, NC, USA).
Qualitative analyses
Verbatim transcripts will be encoded and analyzed thematically applying open, axial then selective coding to textual data according the procedure addressed by Williams & Moser. 22 Thematic content analysis will be carried out with Nvivo software (Version 11, Lumivero, Denver, CO, USA). The respondents’ ideas will first be translated into inductive nodes (i.e. codes) gathered in a coding tree, then organized into themes and sub-themes in an axial coding stage. The coding tree will be presented graphically as a synoptic (thematic) tree, followed by a selective coding step to logically organize the determinants according to the conceptual models of compliance with health recommendations. In addition, Iramuteq software will be used to perform a lexicographic analysis based on discourse form, which can be used to analyze representations (of cancer, screening, etc.). Several validation procedures will be applied to check the robustness of our findings, 23 including the COREQ standards for presenting the results. 24
Intended time schedule
The intended time schedule is presented in Figure 3. The total estimated duration of the project is based on 18 months for drawing up the project (preparation of the questionnaires, study subject approval package, staff recruitment, and training), 24 months for women’s recruitment, and 12 months for the follow-up of women with positive HR-HPV DNA tests, data analysis, and drawing up the final report. Follow-up of women, including reminder letters for women who did not send back the vaginal self-sampling device and women with positive HR-HPV test, will end 12 months after recruitment completion. Recruitment of women started on September 21, 2021. Data collection is expected to end at the end of July 2024.

Time schedule of the RIDECA study.
Trial governance
The RIDECA project involves both French academic and field partners. The academic partners are the University Hospital of Montpellier (Laboratory of Solid Tumors, PCCEI, INSERM, Department of Clinical Research and Department of Methodology), which are in charge of the project management, HR-HPV testing and statistical analysis, and the University Paul Valery in Montpellier (EA 4556, Epsylon Laboratory), which will be involved in the analysis of the psychosocial factors associated with cervical cancer screening (drawing up the psycho-social model, preparing and analyzing the questionnaire, and semi-structured interviews). The field partners are the association AMHDCS (Association for the Mobile Unit for Breast Cancer Screening in the Department of Hérault), the Regional Cancer Screening Coordination Center in Occitania (CRCDC-OC), and the Aude Departmental Council. These partners are involved in the training of the midwives, the management and the schedules of their presence in the mobile unit or in various places in the department of Aude, the follow-up of women’s recruitment with respect to the organized screening program in Occitania (DO-CCU), and the identification of local social and health partners.
A project committee composed of the midwives, representatives of each partner, the Clinical Research Manager, the Clinical Research Assistant, and the methodologist, meets on a regular basis (every 2–3 months) in order to monitor the project and address any local and general problems.
Discussion
The European and international guidelines recommend a minimum screening coverage of 70% of women targeted in order to obtain a significant impact on cancer mortality.25,26 During the 2018–2020 period, approximately 60% of women in France participated in cervical cancer screening. 5 This number needs to be increased, especially among older women.
RIDECA is an interventional study evaluating vaginal self-sampling procedures and an outreach strategy as a means of increasing the participation in screening of unattended women. A number of French studies have been published or are in progress based on self-sampling in order to reach non-attendee women.27 –31 However, the outreach approach has not yet been evaluated in France for cervical cancer screening. This approach, which is similar to the door-to-door strategy used in low- and middle-income countries, therefore requires careful evaluation of its effectiveness in the French context. Another factor that makes the RIDECA project unique is the population targeted: women aged 50–65 years. It seems necessary to address this population, as participation in cervical screening is lower in this age group, decreasing progressively in France after the age of 45 years. 4 During the RIDECA project, women are approached in two ways: either at a mobile screening unit where women come for breast cancer screening, or by giving easy appointments with a midwife following extensive publicity campaigns. The opportunity of using the mobile unit for breast cancer screening in the department of Hérault provides us with an original approach to cervical cancer screening. These mobile units have been shown in France and other countries to be an effective strategy for increasing participation in breast cancer screening programs, while reducing social and geographical health inequalities.32 –34
Some limitations of the RIDECA intervention need to be mentioned. First, the design of the study may limit access to screening by severely deprived populations such as nomadic and homeless populations and those with poor French-language skills. Indeed, despite adopting an outreach strategy and offering the device free of charge, recruitment takes place on a voluntary, non-systematic basis. This could lead to recruitment bias, with under-representation of these populations. In addition, the study does not include people without social security affiliation, or those whose language level is too low to understand the questionnaire, even with the help of a third party. Second, the results will be obtained from a limited number of women (N = 600) living in a specific part of France, which may limit their generalizability to other regions. Third, the criteria used to identify women who have not been screened are only declarative. This may lead to errors in recruitment, particularly in recruiting of women who were screened less than 3 years ago or who have had a total hysterectomy years ago. Therefore, as mentioned above, the criterion “no cervical smear or HR-HPV test for at least 3 years” will be checked at the end of recruitment using the Health Insurance Authority database. Finally, follow-up of women with positive HR-HPV DNA test is planned by regular mailings and phone calls to women and their referring health professional. However, despite these efforts, it is possible that some women may not be reachable or may not respond, restricting follow-up data.
If the RIDECA study shows significant improvements in women’s uptake, screening rates and screening efficacy, the results and conclusions of this project will be presented to Health Authorities, starting at the local level. A population-based organized screening program (DO-CCU) is already operational in the Occitania region. The outreach strategy proposed by RIDECA can then be discussed as a possible complementary alternative. This study will also have another positive impact on educating women about cervical cancer screening through their discussions with midwives during recruitment, the use of information tools developed for the project, and the information letter sent with the HR-HPV results. Asking women about their gynecological follow-up during the recruitment process will be another way of informing them about cervical cancer screening and recommendations on gynecological issues in general. In addition, the project is expected to improve the health of the local population by disseminating information about cervical cancer screening and the role of HR-HPV testing and self-sampling through health professionals, social centers, and community-based associations. This project will also help to identify the psycho-social factors involved in the decision-making process and the potential barriers to compliance with cervical cancer screening. These determinants will be studied at the two recruitment sites, which correspond to two groups of under-screened women with different attitudes towards screening: a group of women already involved in breast cancer screening (at the mobile unit) and another group not involved in screening processes (in the Limoux area in the department of Aude). In addition to providing some valuable insight into women’s behavior, this information will facilitate the development of useful tools and strategies for increasing cervical cancer screening participation among women. Finally, this intervention will also serve as a means to identify some organizational and practical issues that must be addressed in order to improve preventive measures for deprived populations. These latter points will be particularly important as they will enable public health decision-makers to consider the transferability of our findings to other local and regional levels.
Supplemental Material
sj-docx-1-whe-10.1177_17455057241292693 – Supplemental material for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol
Supplemental material, sj-docx-1-whe-10.1177_17455057241292693 for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol by Olivier Lareyre, Caroline Mollevi, Guillaume Broc, Marie-Nina Caspar, Gergana Pehlivanska, Hélène Boyer, Soraya Groc, Giulia Corradi, Silvia Loy-Morel, Marine Guy, Nathalie Tricheux, Florence Cousson-Gélie and Nathalie Boulle in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057241292693 – Supplemental material for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol
Supplemental material, sj-docx-2-whe-10.1177_17455057241292693 for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol by Olivier Lareyre, Caroline Mollevi, Guillaume Broc, Marie-Nina Caspar, Gergana Pehlivanska, Hélène Boyer, Soraya Groc, Giulia Corradi, Silvia Loy-Morel, Marine Guy, Nathalie Tricheux, Florence Cousson-Gélie and Nathalie Boulle in Women's Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057241292693 – Supplemental material for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol
Supplemental material, sj-docx-3-whe-10.1177_17455057241292693 for An outreach strategy to increase uptake of vaginal self-sampling for cervical cancer screening in older French women: The RIDECA interventional research protocol by Olivier Lareyre, Caroline Mollevi, Guillaume Broc, Marie-Nina Caspar, Gergana Pehlivanska, Hélène Boyer, Soraya Groc, Giulia Corradi, Silvia Loy-Morel, Marine Guy, Nathalie Tricheux, Florence Cousson-Gélie and Nathalie Boulle in Women's Health
Footnotes
Acknowledgements
The authors wish to warmly thank all the women who participated to the study; Joëliane Battery, Sylvine Brochot, Aude Chalier, and Charlotte Kaan in the Department of Clinical Research; Credo Dossou in the Department of Public Health at the University Hospital of Montpellier; all the staff of the mobile unit (the mammobile); Sylvie Joubert, Marie Christine Lecq, and Jean Chapelle at the Laboratory of Solid Tumors; and Jean Loup Chappert, Antoine Khreiche, and Jean Pierre Daures, who contributed most helpfully at the very beginning of the project.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
