Abstract
Objective
To compare primary test positivity in cytology and human papillomavirus-based screening between different Nordic cervical cancer screening programs using harmonized register data.
Methods
This study utilized individual-level data available in national databases in Finland, Iceland, Norway, and Sweden. Cervical test data from each country were converted to standard format and aggregated by calculating the number of test episodes for every test result for each calendar year and one-year age group and test method. Test positivity was estimated as the proportion of positive test results of all primary test episodes with a valid test result for “any positive” and “clearly positive” results.
Results
The age-adjusted rate ratio for any positive test results in primary human papillomavirus-based screening compared to cytology was 1.66 (95% CI 1.64–1.68). The age-adjusted rate ratio for clearly positive test results was 1.02 (95% CI 1.00–1.05). A decreasing rate ratio by age was seen in both any positive and clearly positive test results. Test positivity increased over time in Iceland, Norway, and Sweden but slightly decreased in Finland.
Conclusions
The probability of any positive test result was higher in human papillomavirus testing than in primary cytology, even though the cross-sectional detection of a clearly positive test result was the same. Human papillomavirus testing can still lead to an improved longitudinal sensitivity through a larger number of follow-up tests and the opportunity to identify women with a persistent human papillomavirus infection. Further research on histologically verified precancerous lesions is needed in primary as well as repeat testing.
Introduction
Screening for cervical cancer (CC) has been effective in all the Nordic countries leading to reductions of 60–85% in incidence and mortality due to CC compared to the situation without screening.1–3 Recently however, a growing trend in CC incidence has been reported both in Sweden 4 and Finland 5 and in the Nordic countries combined. 3 This increase in incidence emphasizes the need for improvement in screening performance and quality assurance of screening. Fortunately, prophylactic vaccines against human papillomavirus (HPV) infection will also likely reduce the burden of CC in vaccinated cohorts during the following decades. 6
Cervical cytology has for decades been used for screening of cervical abnormalities that can then be treated to avoid invasive cancer. Within the last two decades, tests that detect viral oncogenic HPV have become more prevalent. HPV-based screening has been shown to be more sensitive than cytology-based screening in detecting precancerous lesions. 7 However, rates of repeat testing and colposcopy referrals are also expected to increase during the first round of screening when switching to a more sensitive test method. 8 HPV-based screening also needs effective risk stratification to reduce unnecessary referrals since most HPV infections are benign and clear spontaneously. 9 Several triage options, such as HPV genotyping, immunocytochemistry, and methylation, exist with cervical cytology still being the most common method in practice. 10 This transition from cytology-based screening to HPV-based screening will evidently lead to changes in the performance characteristic of screening and requires new tools to monitor screening programs.
HPV-based screening is currently being implemented in most Nordic countries. Norway has introduced this gradually in a randomized setting, starting in 2015, and from 2021 onwards all women aged 34 or older will be screened with an HPV test. In Sweden, the National Board of Health and Welfare issued a recommendation in 2015 to use HPV tests in screening for women who are aged 30 or older, but all counties have not yet implemented the new recommendations. In Finland, around 1,00,000 women had been invited to primary HPV screening during 2003–2014 as part of a large-scale randomized evaluation study. 11 Thereafter, an increasing number of municipalities have switched to the HPV test in primary screening with approximately 18% of women screened in this way in 2018. 5 In Iceland, primary HPV tests are only used as an exit test for the oldest age group.
In several Nordic countries, screening programs publish reports on screening activity, but the comparison between different programs is challenging because of differences in how the test results are reported. The main differences are whether all tests or only primary tests are included, which age groups are included, and whether the results are stratified by test method.
This study is part of the NordScreen project, which is a pilot model for cross-country comparison of cancer screening programs. 12 Performance indicators based on aggregated test result data are published online on the NordScreen platform (nordscreen.org). Estonia is also involved in the project, but data from Estonia were excluded from this article due to a high proportion of missing test results.
Our aim in this study was to compare primary test results in the Nordic CC screening programs using harmonized register data and investigate whether there are differences in test positivity between countries, age groups, or test methods. We also report the proportion of cytological abnormalities in primary HPV screening by country and age group.
Material and methods
Setting
All Nordic countries have implemented population-based CC screening, but the target age range of screening, the interval between screening tests, and the primary screening method used vary. The current CC screening programs in the Nordic countries included in this study are summarized in Table 1. Further details on screening programs can be found from a recent article on Nordic CC screening programs, 13 from fact sheets on the NordScreen website (nordscreen.org), and from national documentation. 5 , 14 , 15
Summary of cervical cancer screening programs in January 2020 and cervical test data used in the analyses.
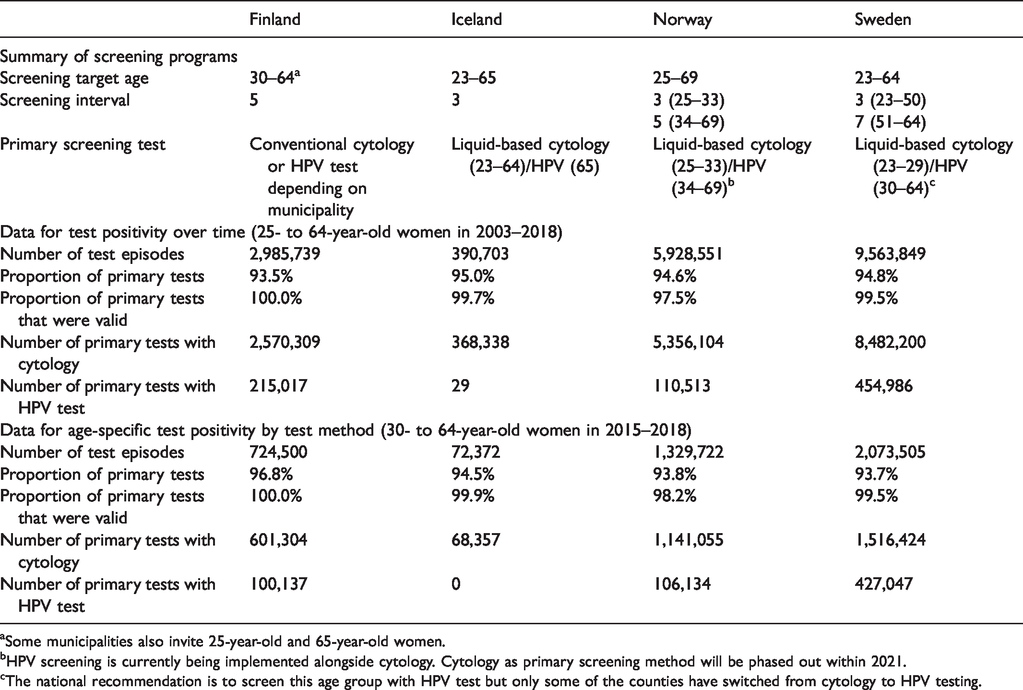
aSome municipalities also invite 25-year-old and 65-year-old women.
bHPV screening is currently being implemented alongside cytology. Cytology as primary screening method will be phased out within 2021.
cThe national recommendation is to screen this age group with HPV test but only some of the counties have switched from cytology to HPV testing.
Data sources
This study utilized individual-level data available in the national screening registers or databases that contain test result data in four Nordic countries. All data sources used unique personal identifiers, so within each country we were able to link together tests that belonged to the same individual. The Finnish Mass Screening Registry contains data on statutory cervical screening activities, whereas the Cancer Detection Clinic in Iceland, the Cancer Registry of Norway, and the Swedish National Cervical Screening Registry contain data on all cervical tests taken in their respective countries. Data from Denmark were not available for the present study but will be added to the NordScreen website when they become available.
All countries had data from year 2000 onwards until the end of year 2018.
Harmonization of data
Cervical test data from each country were converted to standard format in each collaborative center using the same R program script.
All test observations in the national collaborating databases (Finland, Iceland, Norway, and Sweden) were combined into test episodes. Any subsequent results occurring within 90 days of the first test in the episode were considered part of the same test episode. For episodes containing multiple results, only the most severe cytology and HPV test results were considered. This 90-day interval was chosen to combine the results of primary test and potential reflex cytology triage after a positive HPV test while excluding repeat tests, which should not be performed within three months of initial test. 16 The combining of tests into test episodes slightly increased the proportion of valid (i.e., not missing or unsatisfactory) test results, but this varied by country depending on whether the new test after an unsatisfactory test was taken within 90 days of the first test.
Cytological test results in the national databases were classified according to either Bethesda or Papanicolaou classification. HPV test results were classified as positive or negative for any oncogenic HPV. The combined test result values used in the involved countries are presented in Supplement 1. The test episodes were defined as primary tests if the latest test episode within the last 2.5 years was not positive, or there were no preceding tests within the last 2.5 years. A similar method for separating primary tests from all tests has been used in Norway. 15
Since our interest was to compare primary screening, we excluded repeat tests and tests of cure by including only primary test episodes for further analysis (difference between test positivity for all tests and primary tests is shown in Supplement 2). As 2.5 years of history was required to define primary tests, the primary test data were available from the year 2003 onwards.
The test method (whether cytology or HPV test) was nationally defined in various ways. In Finland, registry data include a variable for test method, whereas in other countries the test method had to be inferred using information on year, age, and geographic location of test. The test method for each test episode was defined as the test method of the first test within the episode.
Aggregation of data
The harmonized test episode data were aggregated by the same R program script in each collaborating center. The script calculated the number of test episodes for every test result for each calendar year and one-year age group, while also stratifying between primary tests (primary/other) and test methods (cytology/HPV test).
Statistical analysis
All statistical analyses have been performed with the aggregated data. Descriptive statistics of this screening data are presented in Table 1. In the analysis, all positive test results were simplified into two categories: “any positive,” and “clearly positive.” Any positive results include all abnormal test results (atypical squamous cells of undetermined significance (ASC-US)+ and/or HPV+) whereas clearly positive results (atypical squamous cells, cannot exclude high-grade squamous intraepithelial lesion (ASC-H)+/atypical glandular cells, not otherwise specified (AGC-NOS)+) warrant a colposcopy according to most screening algorithms. Thus, the clearly positive category in HPV testing includes only tests that were positive for oncogenic HPV and for which the cytology triage was ASC-H+/AGC-NOS+.
Test positivity over time
We estimated the prevalence (with 95% confidence intervals estimated by binomial exact method) of positive test results as a proportion of all primary test episodes with a valid test result. This was calculated for “any positive” and “clearly positive” test results separately. Only test episodes of women aged 25–64 were included to increase comparability, since that age range is shared by all involved countries. Primary HPV tests for women aged under 30 were also excluded since recommendations discourage HPV testing in women aged under 30, and the few existing tests (1% of HPV tests since 2003 in all involved countries together) might lead to erroneous conclusions.
Age-specific test positivity by test method
We estimated the prevalence (with 95% confidence intervals estimated by binomial exact method) of positive test results as a proportion of all primary test episodes with a valid test result. This was calculated for any positive and clearly positive test results separately. Data from tests taken in years 2015‒2018 were used, since 2015 was the first year when primary HPV test data were available from all countries except Iceland. For cytology, test episodes of women who were aged 25–64 were included. For HPV tests, test episodes of women who were aged 30–64 were included for Finland and Sweden, but the age range of HPV tests from Norway was 35–64 since HPV-based screening starts at age 34. HPV tests in Iceland were also excluded because it is only used as an exit test.
The comparison of test positivity by test method was performed by pooling data from Finland, Norway, and Sweden together. Rate ratios (with 95% confidence intervals estimated by normal approximation method) for comparison of test positivity between cytology and HPV tests were calculated using Poisson regression with cytology as the reference category. Rate ratios were calculated separately for each of the five-year age groups for any positive and clearly positive results. We also calculated an age group-adjusted rate ratio between HPV test and cytology for any positive and clearly positive results separately.
Age-specific prevalence of cervical abnormalities in primary HPV screening
We estimated the results of reflex cytology (with 95% confidence intervals estimated by binomial exact method) for positive primary HPV tests in Finland, Norway, and Sweden taken between 2015 and 2018. The results of reflex cytology were stratified into three groups: no abnormality, low-grade abnormality (ASC-US or low-grade squamous intraepithelial cells), and high-grade abnormality (ASC-H or AGC-NOS+).
Statistical software
All analyses were done with statistical software R (version 3.6.1).
Results
Test positivity over time in different countries
The proportion of positive primary tests increased over time in Iceland, Norway, and Sweden but slightly decreased in Finland (Figure 1(a)). Overall, the proportion of positive test results during 2003–2018 was between 2.2% and 8.6% in all countries except Iceland. The sharpest increase over time was in Iceland, where the test positivity rate increased from approximately 5% in 2014 to over 10% since 2016. The rates for clearly positive results during 2003–2018 were much lower and varied mostly between 0.5–1.5% (Figure 1(b)), increasing slightly over time.
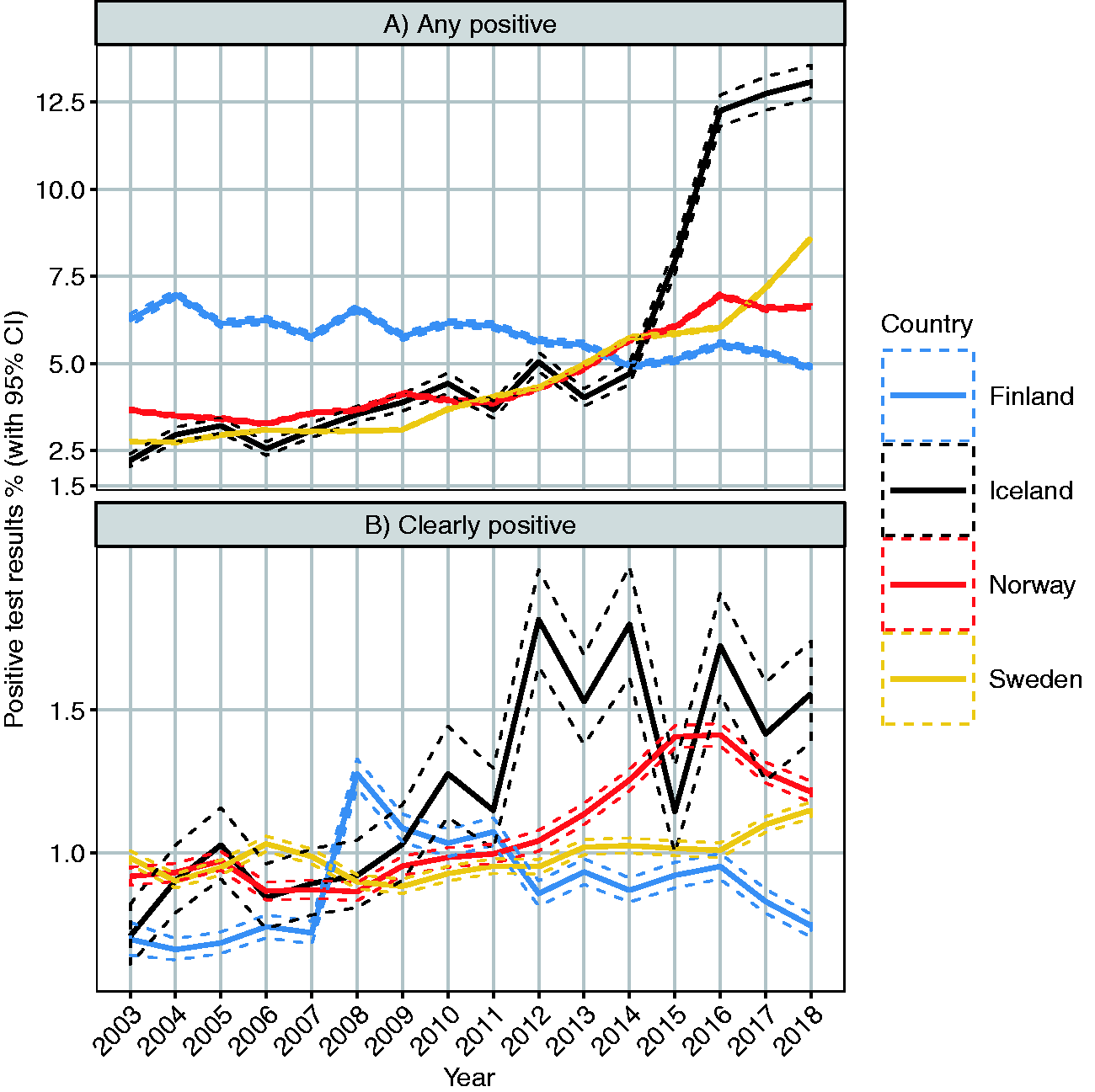
Proportion of positive primary test results in 2003–2018 among women aged 25–64 with 95% confidence intervals irrespective of test method. Proportion of any positive test results is presented in panel A and proportion of clearly positive results is shown in panel B. Any positive results include all abnormal results (ASC-US+/HPV+) whereas clearly positive results are defined as high-grade abnormal cytology (ASC-H+/AGC-NOS+).
We also looked at the positivity rate over time by test method in different countries, which revealed that the time trend varies between countries (Supplement 3). In Finland, the positivity rate of cytology has a decreasing trend, whereas in Norway and Sweden the rates have increased over time. The positivity rates of HPV tests have increased over time only in Finland and Sweden.
Age-specific test positivity by test method
Rates for any positive test results were higher with HPV tests than cytology in all age groups in all countries where both test methods are used (Figure 2). With both test methods, the positivity rate was highest among the youngest age groups. The positivity rate in HPV testing was similar between countries.
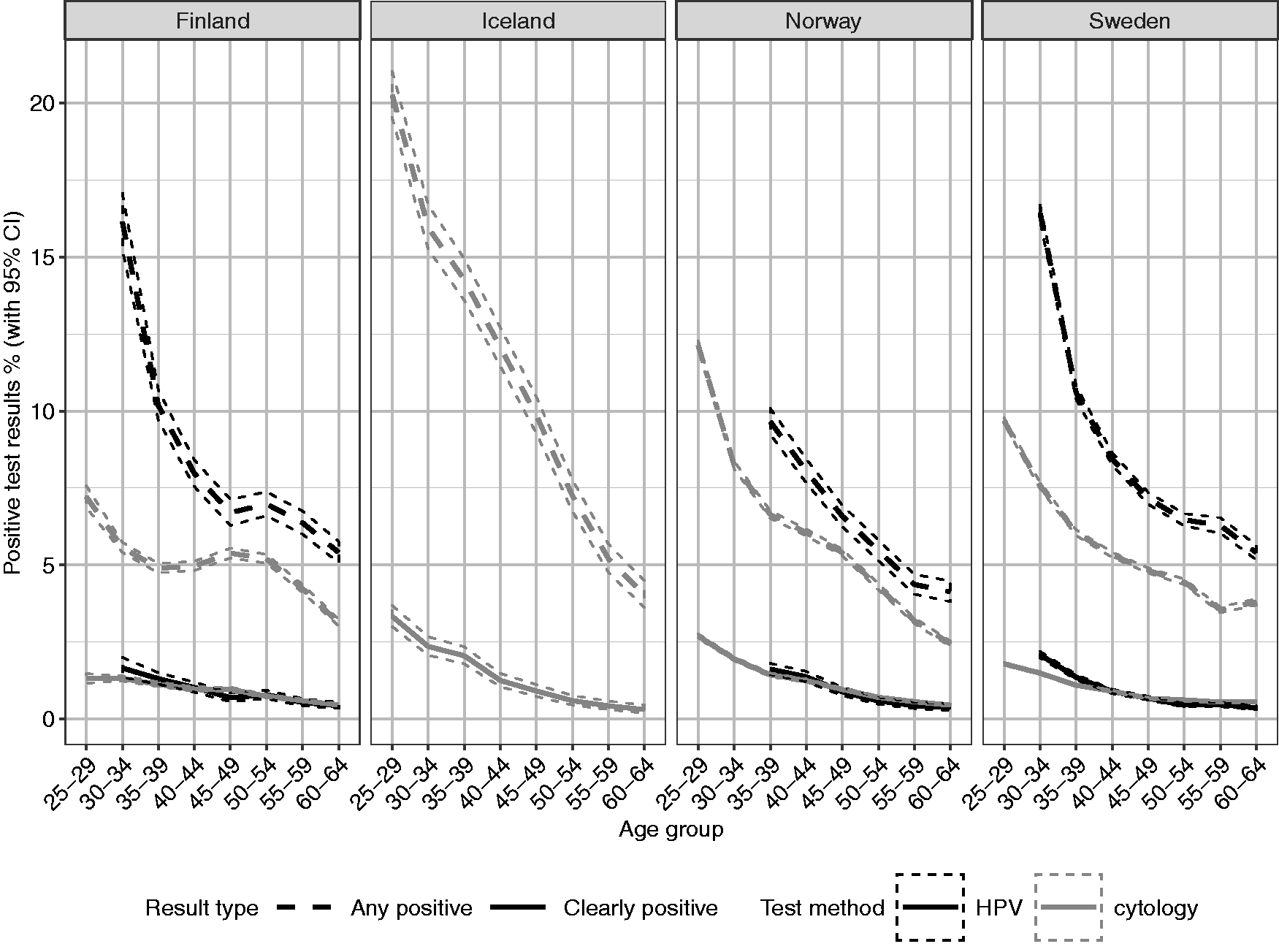
Age-specific distribution of any positive (dashed line) and clearly positive (solid line) test results with 95% confidence intervals in primary screening by type of screening method (cytology or HPV test) for women aged 25–64 in 2015–2018. For HPV tests, the youngest age group was 30–34 years in Finland and Sweden and 35–39 years in Norway. Any positive results include all abnormal results (ASC-US+/HPV+), whereas clearly positive results are defined as high-grade abnormal cytology (ASC-H+/AGC-NOS+).
For clearly positive results, the proportions are mostly similar between the test methods. With HPV-based testing, there were slightly more clearly positive results among women in the youngest age groups in all countries.
In the pooled analysis, when the test result data from Sweden, Norway, and Finland were combined, the rate ratio of any positive test result for HPV tests compared to cytology when adjusted for age group was 1.66 (95% CI 1.64–1.68) with point estimates ranging from 1.37 to 2.18 by age group as can be seen from Figure 3. The rate ratio of clearly positive test result for HPV tests compared to cytology when adjusted for age group was 1.02 (95% CI 1.00–1.05) with point estimates varying between 0.78 and 1.26 by age group. A decreasing rate ratio by age was seen in both any positive and clearly positive test results.
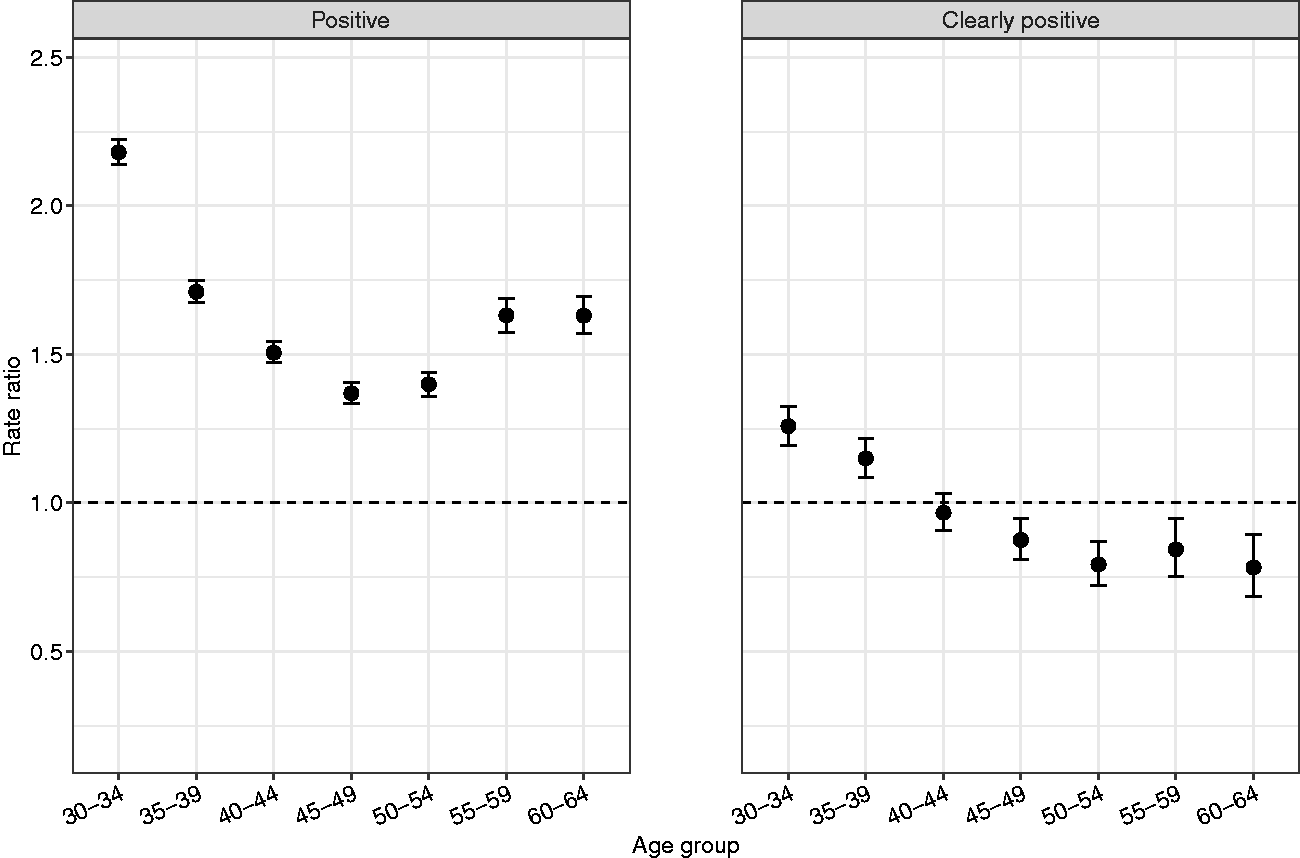
Pooled analysis of age-specific rate ratio of test positivity with 95% confidence intervals for HPV tests compared to cytology in Sweden, Norway and Finland during 2015–2018. Any positive results include all abnormal results (ASC-US+/HPV+) whereas clearly positive results are defined as high-grade abnormal cytology (ASC-H+/AGC-NOS+).
Age-specific prevalence of cytological abnormalities in primary HPV screening
The prevalence of cervical abnormalities in cytology triage of positive primary HPV tests varied by age group and country, but in most positive HPV tests, no abnormalities were found. Abnormalities were more common in younger age groups in all included countries and the prevalence of high-grade abnormalities was slightly higher in Norway compared to Finland and Sweden (Figure 4).
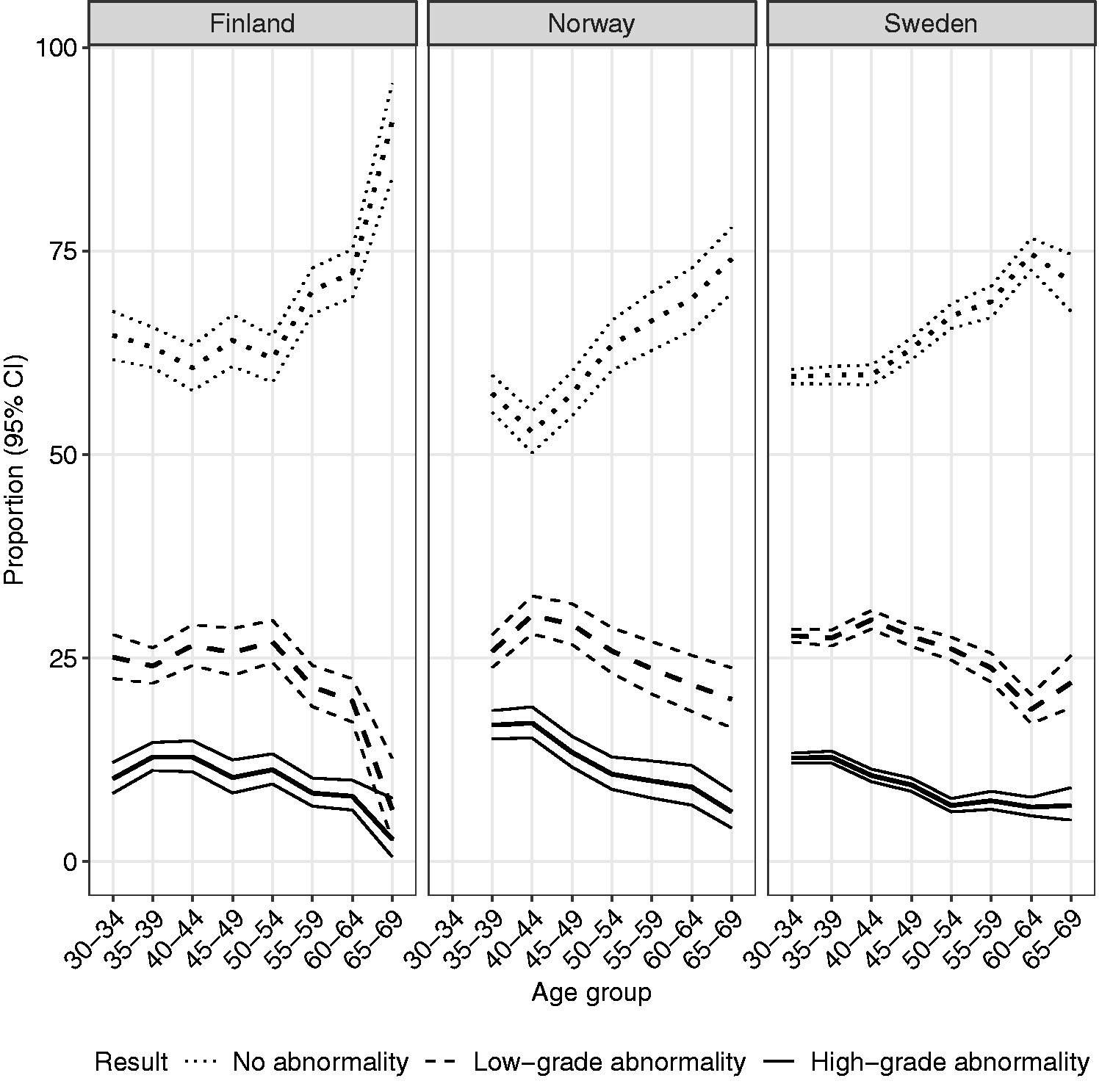
Age-specific proportion of no abnormality (dotted line), low-grade abnormality (dashed line), and high-grade abnormality (solid line) with 95% confidence intervals in cytology triage of positive primary HPV tests for women aged 30–64 (35–64 years for Norway) in 2015–2018. Low-grade cytological abnormality is defined as ASC-US/LSIL and high-grade abnormality as ASC-H+/AGC-NOS+.
Discussion
Key findings
The proportion of any positive primary test results in screening was higher in HPV-based screening compared to cytology with a rate ratio of 1.66. The difference between test methods was most pronounced in women aged 30–34 with a rate ratio of 2.18. Still, the probability of a clearly positive test result, leading to referral for colposcopy according to most recent guidelines, was the same in primary testing episodes between the two test methods.
Test positivity increased over time in Iceland, Norway, and Sweden but slightly decreased in Finland. The most striking increase in test positivity was seen in Iceland where the proportion of positive test results increased from 5% in 2014 to over 10% in later years.
Interpretation of results
It is important to keep in mind when interpreting these results that we have focused on primary screening by excluding the test episodes that were preceded by a positive test within the last 2.5 years. Thus, repeat tests following an earlier positive test or tests of cure are not included and the results reflect the situation in the population without recent abnormalities. Proportion of positive HPV tests can also be interpreted as HPV prevalence, which shows that there are only minor differences between these countries. The age gradient in prevalence of oncogenic HPV infection was also similar to previously reported figures from some Nordic countries. 17 Since the rate of “any positive” test results was higher in HPV-based screening compared to cytology, the longitudinal program sensitivity could be increased through increased volume of repeat testing. 11 , 18 Most HPV-positive women in our study had normal cytology in reflex triage. The negative age gradient of cervical abnormalities following a positive HPV test is in line with other studies reporting results of cytology triage for primary HPV-based screening.19–22 HPV-positive women have also been shown to have a similar age gradient in histological outcomes with more high-grade abnormalities in younger women. 23 This finding needs to be verified in the Nordic countries as well. The observed differences between the test methods when comparing proportions of “clearly positive” results in younger age groups were surprising. Clearly positive results are, by our definition, the same as high-grade cytological abnormalities. There should be no increase in high-grade abnormalities in HPV-based screening among the tested women since only a subset of women are tested with cytology. One possible explanation for this discrepancy is that HPV-based screening is not implemented in a randomized manner nationwide in the involved countries. In Norway, randomization has been done within some regions, but the potential for selection bias remains in nationwide analysis.
The threshold for a clearly positive result could also be defined differently and not only based on the result of cytology. Referring women who are both HPV positive and have a low-grade cervical abnormality in cytology, or women who are HPV positive for genotypes 16 or 18, are potential strategies for increasing detection of precancerous cervical lesions.19,20 The threshold for a clearly positive result in the present study was defined as high-grade cytology to increase comparability between countries, as HPV tests as triage are not used in every country, and data on HPV genotype was not available in all countries.
The coauthors from Iceland report that the sudden increase in test positivity in the latest years can be explained by a switch from conventional cytology to liquid-based cytology (ThinPrep) and the introduction of HPV tests as a triage test. In cases where there is uncertainty over the result of cytology, the staff will often label the result as ASC-US and then perform an HPV test as triage to define the proper management for screened women. The increase in test positivity rate in Sweden and Norway in the last years is partially caused by the increasing proportion of HPV tests. Since the interval in HPV screening is usually longer and countries started HPV-based screening only recently, the results are mostly from the first round of HPV screening and it is possible that the proportion of positive results in the second round will be lower. Nevertheless, as countries are increasingly switching from cytology to HPV-based screening, the proportion of women with positive test results will likely increase substantially.
It is also worth noting that we have only compared primary testing, and the true performance of screening cannot be comprehensively assessed without considering the results of diagnostic verifications. The number of these colposcopies will likely increase since in HPV-based screening more women are referred to repeat testing. The next study in the NordScreen project will focus on the detection rate and predictive values of both screening methods by linking histological data from colposcopies with the test data.
Strengths and limitations
The obvious strength of our study is that we used comprehensive population-based registers that include unique personal identifiers allowing us to combine tests from the same individual. Therefore, we could harmonize data between countries and focus on primary tests. This improves comparability between countries and gives a better indication of the screening activity by excluding tests in other settings.
The biggest limitation in comparing HPV- and cytology-based screening is that the implementation of these methods has not been randomized in all included countries and the comparison has been done on nationwide data. The background risk of HPV infection might differ between regions and be positively correlated with the patterns of implementation of HPV-based screening.
We also focused strictly on test results since we could not easily follow actual management of screened women. The management of women varies by country and over time, and is dependent on factors such as age, test technology, and prior history of test results, such as persisting ASC-US. The next phase of the NordScreen project, where histological data are combined with primary test data, will illuminate potential differences in management of women with similar test results.
Conclusion
The probability of any positive test result was higher in HPV testing than in primary cytology, even though the cross-sectional detection of a clearly positive test result was the same. HPV testing can still lead to an improved longitudinal sensitivity through a larger number of follow-up tests and the opportunity to identify women with a persistent HPV infection. The number of colposcopies will also likely increase and further research on histologically verified precancerous lesions is needed in primary as well as repeat testing.
Supplemental Material
sj-pdf-1-msc-10.1177_0969141321992404 - Supplemental material for Comparison of cytology and human papillomavirus-based primary testing in cervical screening programs in the Nordic countries
Supplemental material, sj-pdf-1-msc-10.1177_0969141321992404 for Comparison of cytology and human papillomavirus-based primary testing in cervical screening programs in the Nordic countries by Veli-Matti Partanen, Joakim Dillner, Ameli Tropé, Ágúst Ingi Ágústsson, Maiju Pankakoski, Sirpa Heinävaara, Tytti Sarkeala, Jiangrong Wang, Gry Baadstrand Skare, Ahti Anttila and Stefan Lönnberg in Journal of Medical Screening
Footnotes
Acknowledgements
We would like to thank all those involved with the NordScreen project and the data managers who provided the cervical test data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research grant from the Nordic Cancer Union.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
