Abstract
Countries are now challenged by the rapid development of vaccines aimined at the primary prevention of infections. In the years to come, several vaccines will need to be considered as potential candidates in routine immunization programs. Recently, two new vaccines against two/four types of human papillomavirus (HPV) have been commercialized. Bivalent HPV 16 and 18 (Cervarix™) and quadrivalent HPV 6, 11, 16 and 18 (Gardasil®) vaccines are now extensively used in some countries. These vaccines will prevent infection and long-running complications, such as cervical cancer, other HPV-related cancers and genital warts (for the quadrivalent vaccine). The beneficial effect of these vaccines will be largely observed in women. This article summarizes the burden of HPV preventable disease worldwide and briefly describes the impact of secondary prevention and the most relevant aspects of the current available vaccines, their efficacy and safety. Finally, some major aspects that are likely to impact the introduction of these vaccines around the world are outlined, with particular emphasis on developing countries.
Burden of the human papillomavirus-related cancer disease
One of the most frequent severe consequences of human papillomavirus (HPV) infection is cervical cancer, a disease that arises in the special cells that form the uterine cervix and that represent a transition between the vagina and the uterine body. Cervical cancer is the second most common malignant neoplasm among women worldwide after breast cancer (
Burden of breast and cervical cancer in women worldwide.
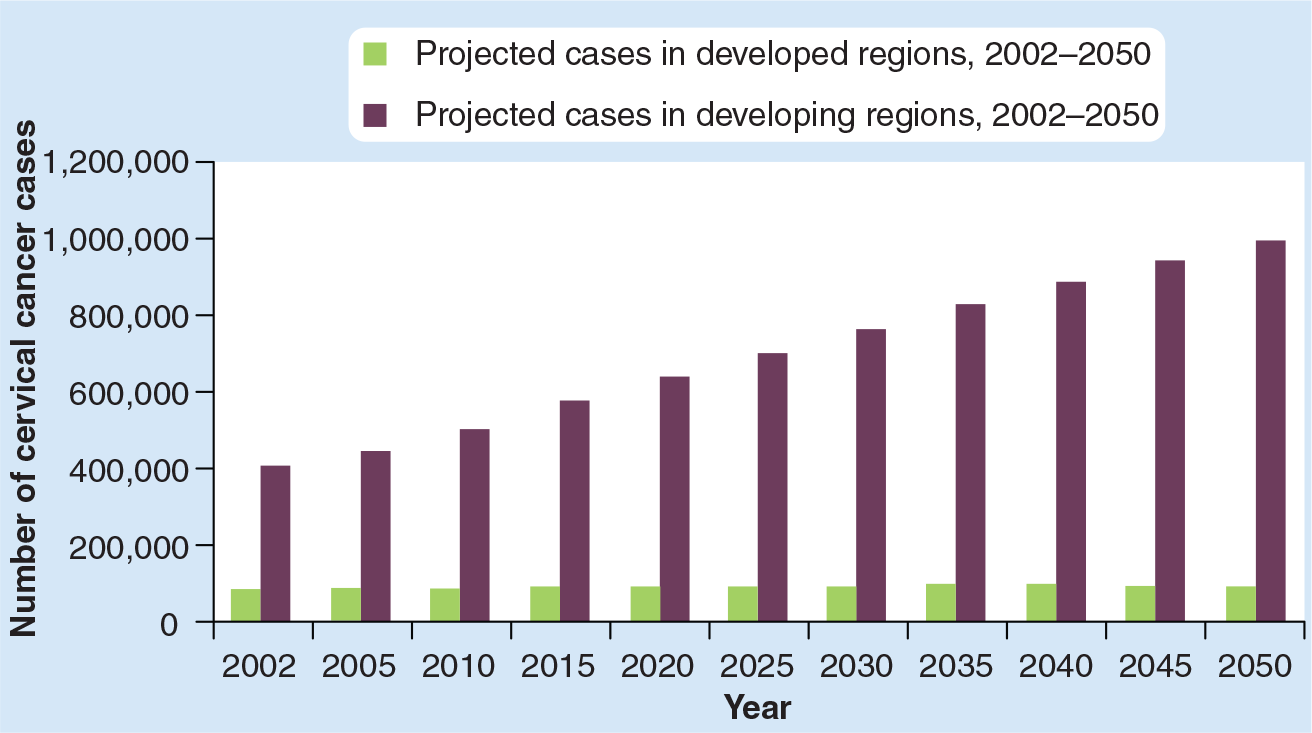
Cases of cervical cancer: projection for 2002–2050.
Human papillomavirus infection has been firmly established as a necessary cause of invasive cervical cancer [3–5]. However, the acquisition of an HPV infection is not sufficient to develop cervical cancer, suggesting that factors other than HPV infection are also involved in the progression to cancer. Among them, probably the host response and viral characteristics are the most crucial elements for persistence, progression and invasion [6]. Moreover, other factors such as cigarette smoking, long-term use of oral contraceptives, high parity and concomitant coinfections, such as HIV, increase the risk of cancer in the presence of certain HPV infections [7,8].
Today, over 100 different HPV types have been identified, and more than 30 of these are known to infect the cervical epithelium [9]. Genital HPV types have been grouped according to their association with cervical cancer as low-risk types, which are found in normal epithelium and also in genital warts (mainly caused by HPV 6 and 11); and high-risk types, which are associated with cervical cancer (HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 66) [10,11]. Other potential carcinogenic HPV types are 26, 30, 34, 53, 67, 68, 69, 70, 73, 82 and 85 [10,12]. The first and second most frequent types in invasive cervical cancer are HPV 16 and 18, which combined account for over 70% of the cases worldwide [13–15]. In an ongoing study on a large series of invasive cervical cancer samples, conducted at the Catalan Institute of Oncology (Spain), the next most common type identified is HPV 45 in all world regions, except for Europe where HPV 33 ranks as the third [15].
Approximately 10% of the women with normal cytology in the general population worldwide will harbor HPV cervical infection at a given point in time. The HPV types that are most frequently identified are from the α9 and 7 families: HPV 16, 18, 31, 58, 52 and 33 [16]. The prevalence of the infection varies considerably by world region and by age. The highest HPV prevalence estimates are observed in women younger than 34 years, after which prevalence decreases followed by a gentle increase in older age groups (
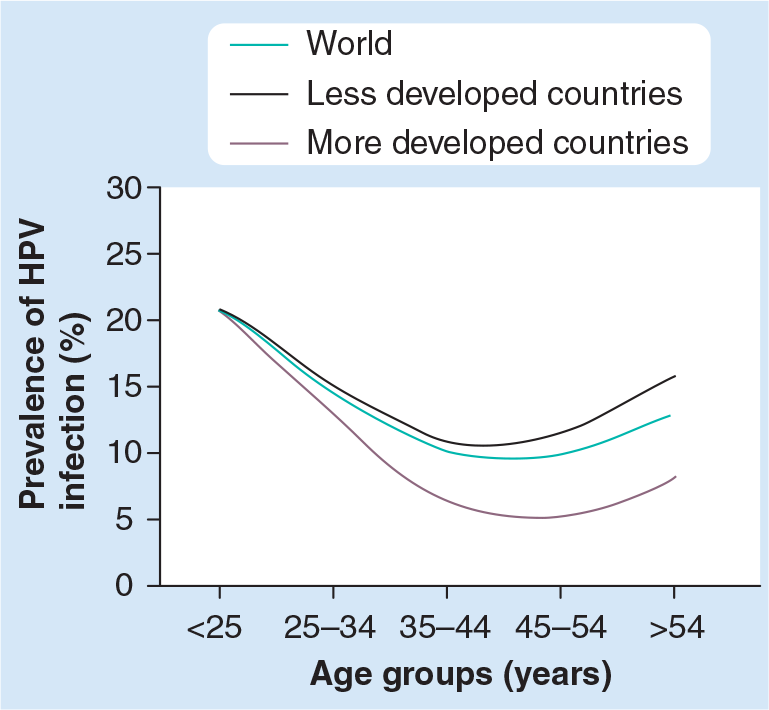
HPV prevalence in women with normal cytology by age groups and world regions.
In addition to cervical cancer, there is now enough evidence for the carcinogenicity of HPV 16 in cancer sites other than the cervix, such as the vulva, penis, vagina, anus, oral cavity and oropharynx [10]. The number of cancer cases attributable to any HPV type including all these sites and both sexes for the year 2002 was estimated to be approximately over 68,200 cases [17]. A large international effort is now underway to provide a more precise estimation of the HPV contribution in these cancer sites [18].
Secondary prevention: cervical cancer screening
The recognition of the necessary role of HPV infection in the etiology of virtually all the cervical cancers has dramatically changed the perspectives of the diagnosis and prevention of this neoplasia. The two axes in which this change is being articulated refer mainly to preventive measures: the introduction of reliable HPV detection tests in cervical cancer screening (secondary prevention) [19] and the HPV vaccination (primary prevention) [20,21].
It is important to recognize that repeated conventional cytology screening tests have been shown to reduce the incidence and mortality of cervical cancer by up to 70% in populations where they have been intensive and systematically applied [22]. However, current standard screening recommendations generally involve an average of 14 cytologies per lifetime and result in 2–9% of low-grade squamous intraepithelial lesions/high-grade squamous intra-epithelial lesions depending on the study [22]. The large majority of these lesions are a reflection of acute infections that, in a considerable proportion, will require special follow-up and medical intervention. In these circumstances, anxiety cannot be underestimated. Moreover, in many countries, screening occurs at a woman's demand, which inevitably results in over-screening those women who are more aware of the benefits of prevention, while other women remain underscreened. This results in an ineffective and expensive preventive intervention. A successful screening program needs to be well-structured: it must warrant a call–recall system to follow-up target women, it has to reach a coverage of over 80% of the target population and it has to offer adequate treatment regimens. Screening covers less than 5% of the women at risk in many African and Pacific countries, approximately 20% in the majority of Latin American countries and over 60% in Europe, North America, Australia and in some Asian countries [101]. Owing to the high cost of these programs, active research is under investigation to evaluate screening alternatives that can be in place when resources are limited while establishing adequate coverage and treatment.
In addition, new options for cervical screening are of great importance, such as the introduction of HPV DNA testing. A recent review has demonstrated that there are clear benefits for the use of HPV DNA testing in the triage of equivocal smears, low-grade lesions in older women and in the post-treatment surveillance of women after treatment of cervical intraepithelial neoplasia [23]. The use of these tests is now being recommended in some countries alone or as adjuvant to cytology [22]. HPV DNA testing used as primary screening has shown to be more sensitive than cytology, but has a lower specificity [23,24].
Cancer prevention stakeholders in many settings aim to improve screening coverage; however, the costs and benefits of secondary prevention need to be weighted with those of primary prevention through HPV vaccines. In some settings, HPV vaccines may be the most realistic option to impact cervical cancer burden. Even in the most desirable setting, the need to continue screening for cervical neoplasia will remain, even after vaccination, since current available HPV vaccines do not cover all HPV types that lead to cervical cancer. However, the frequency, spacing and tools for cervical cancer screening in vaccinated cohorts must be evaluated in order to allow more efficient protocols.
Main data on HPV vaccines
To date, two prophylactic HPV vaccines have been licensed by several regulatory agencies such us the European Agency for the Evaluation of Medicinal Products (EMEA) or the US FDA. HPV vaccines have been approved in over 100 countries and have been introduced in some national immunization programs, mainly in developed countries.
A summary of the main findings of each vaccine has recently been reviewed [25,26]. The two existing vaccines are Gardasil® (Merck & Co., Whitehouse Station, NJ, USA) and Cervarix™ (GlaxoSmithKline Biologicals, Rixensart, Belgium). Both vaccines are made from L1 viruslike particles, are noninfectious and are administered on a three intramuscular shot series over a 6-month period. Although similar, the two vaccines have some differences in terms of their composition and their manufacture system: the quadrivalent vaccine (Gardasil) targets four HPV types (6, 11, 16 and 18), includes an aluminium hydroxyphosphate sulfate adjuvant and is produced in a yeast system [21,27]; conversely, the bivalent vaccine (Cervarix) targets two HPV types (16 and 18), includes the ASO4 adjuvant and is manufactured in an insect-cell system [28].
The results from randomized, controlled trials have demonstrated high immunogenicity, efficacy and safety of these vaccines [28–31].
Current findings indicate that the available vaccines are immunogenic in a wide age range, with high seroconversion rates – essentially 100%. Antibody titers reach the highest levels 1 month after the third dose; this is followed by a decrease after which they remain stable at or above levels observed after natural infection. In addition, the adjuvant contained in the bivalent vaccine aims to ensure prolonged immune response by enhancing antibody titers and stimulating cellular immune response, although full evaluation has not yet been reported. In the quadrivalent vaccine, booster ability has been already reported as evidenced by the increase in vaccine type-specific antibodies in response to a booster vaccine dose [32].
Both vaccines have demonstrated prophylactic efficacy against a range of end points from HPV infection to cervical preneoplastic lesions (cervical intraepithelial neoplasia grade 3 [CIN 3]) related to the HPV types included [30,31]. Moreover, the quadrivalent vaccine has demonstrated high efficacy against preneoplastic lesions of the vulva (VIN 2/3) and vagina (VAIN 2/3) related to HPV 16 and 18 and genital warts related to HPV 6 and 11 [33]. Limited cross-protection in some closely related types has been demonstrated against incident HPV infection for the bivalent, persistent infection for both vaccines and high-grade disease for the quadrivalent vaccine [28,30].
Regarding available safety data within the limits of 6–7 years of follow-up trials, both vaccines appear to be safe and well tolerated, the most common vaccine-related adverse events being the injection-site reactions, such as pain, redness or, in some cases, syncope. It is important to note that the proportion of women participating in the vaccine trials experiencing serious adverse events of any type was similar in those receiving HPV vaccine as compared with controls. These vaccines are based on noninfectious proteins that do not carry the early viral proteins with oncogenic capacity. Side effects are likely to be similar to those of other vaccines of similar composition, such as the hepatitis B vaccine. HPV vaccines are not recommended for pregnant women owing to limited safety data on this issue. Despite the evidence of a good safety vaccine profile from clinical trials, an accurate monitoring on adverse events must continue after licensing vaccines by the industry and public health agencies. With this purpose, the FDA and the CDC established the Vaccine Adverse Event Reporting System (VAERS) in 1990. Since the quadrivalent HPV vaccine was licensed in June 2006, more than 16 million doses of the vaccine have been distributed in the USA and the number of adverse events reported after vaccination has not been higher than the number of adverse events that would otherwise be expected in an unvaccinated population [102]. To avoid misunderstandings and false alarms, any serious disease or death occurring in a vaccinated person has to be evaluated in the context of the probability that this disease affects the nonvaccinated population. It can be extremely alarming for the general population to attribute important side effects to the vaccine without serious statistical analysis and full medical evaluation [34].
Several questions about HPV vaccines remain unanswered, including long-term safety, long-term protection, full description of cross-protection with relevant HPV types and vaccine integration with other preventive measures. Additional follow-up of the participants from existing clinical trials, performing Phase IV studies and evaluating efficacy in selected populations, such as among immunocompromised patients, will be important in investigating these issues. Moreover, current vaccines have some weaknesses and second generation vaccines are under development. Research on these new vaccines is moving towards widening HPV-type coverage, inducing long protection, lowering the cost, being thermostable, delivered via noninjectable methods and providing therapeutic efficacy [35]. However, the commercialization timeline for these vaccines will be long since they should be fully developed and safety, immunogenicity and efficacy should be proven. Thus, for the coming years the only available option is the current available virus-like particle HPV vaccines.
Issues for a successful introduction of HPV vaccines
A selected group of women around the world has benefited from prevention of cervical cancer through cervical cytology. As mentioned previously, in countries where massive screening efforts have been systematically undertaken, reduction of cervical cancer can be in the order of 70–80%. This is observed in the Nordic European countries. Even though the scenario of secondary prevention delivery to those most at need is a global failure, many countries with limited resources still consider its introduction. Specifically, the impact of opportunistic screening is variable, but can be as low as 20% in many settings in Latin America and less than 1% in many African settings [22].
With the recent development and availability of HPV vaccines, we are now facing a unique opportunity to introduce them as a new tool for prevention, which by its nature, alone or in combination with secondary prevention, could clearly impact the target population in a more efficient manner than by secondary prevention alone. Globally, vaccination is one of the most successful and cost-effective public health interventions. Massive vaccine introduction by extensive immunization programs, such as the Expanded Program of Immunization (EPI), has been demonstrated to be effective in the control and even eradication of immunopreventable diseases, such as smallpox, measles or poliomyelitis (

Impact of massive vaccine introduction over the incidence of measles disease.

Impact of massive vaccine introduction on the incidence of poliomelytis disease.
Several reviews analyze all the recognized aspects for a successful introduction of a vaccine in the general population [36,37]. The critical steps could be summarized as follows:
Prevention of the disease by the vaccine must rank as a health priority;
There must be licensure for the safety and efficacy of the vaccine;
There must be an adequate delivery system and vaccine supply;
The vaccine must meet the criteria of a financially sustainable program;
The vaccine needs to be acceptable to health professionals and society;
There must be surveillance for vaccine coverage, effectiveness and safety.
Some of the major issues that could be considered as potential obstacles for a satisfactory vaccine introduction, taking into account the specificities of the HPV vaccine as compared with previous vaccines are listed in
Economical
Price is now considered the main barrier for the full introduction of HPV vaccines [38,39]. HPV vaccines have been marketed at over US$100 per dose with country variability. While the HPV vaccination of teenagers has started in the majority of the northern hemisphere, stakeholders in developing countries are confronted with an unaffordable price as it is stands today. A full evaluation of the price components and of strategies to purchase vaccines would be relevant for a tiered pricing. Moreover, the cross-licensing of the two vaccines has not helped to reduce the prices. Many international agencies are devoting efforts to identify strategies to ameliorate the acquisition of drugs or vaccines. Many hours and lots of manpower are being dedicated to reduce the gap of inequity and, on some occasions, these efforts are successful. The use of public resources requires careful evaluation, even if the final outcome results in improved health. Unfortunately, purchase facilities as well as human power need to be prioritized. Health-impact interventions should be evaluated in the context of competitive interventions. For this, sophisticated methods using mathematical models are becoming almost a necessary prerequisite to plan massive interventions. Cost–effectiveness models and affordability evaluation of screening and vaccine interventions facilitate discussion and understanding of the health impact of each intervention. In India, for example, a vaccination cost of $2 in preadolescence followed by screening three-times per lifetime using visual inspection or testing for HPV DNA would be considered a cost-effective intervention under regular economical standards [40]. A similar analysis to estimate affordability has now been carried out for most of the Latin American countries in which average financial costs are estimated in order to evaluate a vaccine coverage of 70% of five consecutive birth cohorts. The evaluation is carried out for different vaccine prices per dose. The analysis demonstrates that a very cost-effective intervention could cost over $100 million/year [41]. We should be pleased to see that groups such as the Global Alliance for Vaccines and Immunization (GAVI) Alliance or the Pan American Health Organization (PAHO)-Revolving Fund for the Latin American region, are being active in obtaining important logistic and budgetary reduction for different interventions in the region and they are now considering the introduction of HPV vaccination in most at-need and at-risk populations. While these evaluations are more valuable in those settings in which the scarce funds need to be carefully allocated, data necessary to build-up these models are often of poor quality and many times nonexistent.
Managerial
There are some issues related to the HPV vaccines that may contribute some difficulties to the wide use of them, including the fact that the vaccine has a target age before sexual initiation and the need for three doses. Regarding the latter point, the evaluation of the long-term duration of vaccine protection is crucial in order to determine the need for booster doses. On the contrary, data derived from ongoing studies investigating easier methods of vaccine administration and improved schedules may result in a potential reduction of vaccine doses and easier delivery programs [103]. Groups such as PATH are evaluating different strategies for vaccine implementation in different settings, such as use of call–recall system for second and third dose of HPV vaccines in school girls in Peru. Furthermore, countries that have poor adolescent health systems and/or absence of screening programs will encounter more difficulties in targeting young girls. However, comprehensive strategies can facilitate cancer prevention by introducing vaccination in pre-existing school-based programs or by combined actions, such as by vaccinating the girl and screening the mother during the same visit. Issues on the follow-up may also arise in these strategies and careful monitoring would therefore be needed.
Social acceptability
Several aspects could be addressed in relation to the acceptability of the HPV vaccine as a gender-specific vaccine that, in addition, relates to a sexually transmitted disease. In order to increase social acceptability, it is necessary to educate the public about the nature of HPV infection, the seriousness of the related diseases and the available opportunities for prevention. Furthermore, the information needs to be carefully evaluated in order that it is adapted and understood by different cultural environments. Health education campaigns should aim to reassure unjustified fears and anxiety [42].
To introduce the vaccine, cost permitting, it is important that the intervention is accepted by the medical and civil population. Education is therefore of great relevance, particularly when complex issues such as sex, adolescence and cancer are involved. Although vaccines have a historically recognized capacity to reduce disease and to increase life expectancy by reducing the number of deaths by thousands, they remain the target of opposition groups [34]. The introduction of interventions among a priori healthy subjects is regarded by opponents as an unreasonable intervention that is not justified by its benefits. Fortunately, the introduction of a new vaccine requires an important evaluation of potential side effects during its implementation and in the follow-up of vaccinated cohorts.
It is often heard, in some discussion forums, that women should be the ones most in favor of HPV vaccines. It is clearly easier for a woman to balance her own health and programmatic benefits of cancer prevention strategies (i.e., comparing HPV vaccination with a more spaced cytology screening versus routine cytology or versus nothing at all, as is the case in many countries). Women are probably the most suitable to lead the profound change needed to reduce cervical cancer burden. There are examples in which the voice of the most affected can be so powerful that they are the only ones able to modify established dynamics. Advocacy groups of those living with HIV/AIDS subjects have produced important pressure to obtain bioequivalent antiretroviral drugs at a low cost and to facilitate massive production [43]. Although the important role of women should not be undervalued, it is obvious that men, as part of the infection and disease dynamics, also need to be active in the prevention strategies. However, health education may be necessary in order to encourage them to become more clearly involved.
More worrisome is when women are subject to cultural boundaries with gender barriers. It is not unusual, for example, to hear in gynecological settings that some men would not allow their spouse to be visited by a male gynecologist. A long list of gender inequalities could be brought up here. Women should be able to express freely their wish to have a healthy sexual and reproductive life and to be adequately informed. To understand the benefits of HPV vaccines as well as those of secondary prevention, women and men need to know that cervical cancer can be prevented and that every woman can be at risk for the disease. However, expanding on this point is beyond the scope of this review and exploring cultural boundaries in depth would itself require a review.
Potential barriers to the introduction of human papillomavirus vaccines
Current high cost
Competitive diseases measured as years of life lost
Three-dose vaccine
Unusual age at vaccination
Need of cold chain
Poor screening programs
Poor adolescent health systems
Social acceptability
Low level of human papillomavirus knowledge
Absence of sexual health promotion programs
Lack of political will
The development of HPV vaccines has run in parallel with marked investments in medical education and public awareness. HPV marketing has been very important in some countries often seen as TV publicity, street propaganda, monument creation and more. As previously stated, sex, teenagers and cancer include three parameters of major social interest. The medical profession and scientists have developed many rules in order to avoid being influenced by marketing issues, and conflicts of interests are requested in the majority of scientific acts. It is in the interest of the public that drug-regulatory agencies, such as the FDA or EMEA, strongly control for the quality of the results while accepting that it is the industry that finally will make the necessary investments for massive vaccine production all over the world. Furthermore, the WHO, through its Global Advisory Committee on Vaccine Safety, provides independent scientific assessment of vaccine safety issues, particularly on adverse events attributed to vaccines and the creation of ad hoc task forces to commission, monitor and evaluate research on any reported association of specific vaccines/components with adverse events [104].
Executive summary
Cervical cancer is the second most common malignant neoplasm among women worldwide.
Approximately 493,200 new cases of cervical cancer and 273,500 deaths from this pathology were estimated to occur in 2002, with more than 80% of the cases clustering in developing countries.
In addition to cervical cancer, there is now enough evidence for the carcinogenicity of specific human papillomavirus (HPV) types in cancer sites other than the cervix, such as the vulva, penis, vagina, anus, oral cavity and oropharynx.
HPV infection has been firmly established as the necessary cause of invasive cervical cancer.
Today, over 100 different HPV types have been identified, and more than 30 of these are known to infect cervical epithelium.
The most frequent HPV types in invasive cancer are HPV 16 and 18 (high-risk types), which combined account for over 70% of the invasive cervical cancer cases worldwide.
Cervical cancer screening by cytology has been successful in populations where it has been intensive and systematically applied.
The high cost of these programs in terms of the resources required (i.e., infrastructure and treatment algorithms) makes them unachievable for many countries worldwide.
Alternative screening tools and algorithms are under evaluation, taking into account the existent options of screening techniques, their characteristics and country resources.
To date, two prophylactic HPV vaccines have been licensed by several regulatory agencies, such as the European Agency for the Evaluation of Medicinal Products (EMEA) or the US FDA.
The two existing vaccines are Gardasil® (Merck & Co., Whitehouse Station, NJ, USA), a quadrivalent vaccine (HPV 6, 11, 16 and 18) and Cervarix™ (GlaxoSmithKline Biologicals, Rixensart, Belgium), a bivalent vaccine (HPV 16 and 18).
Both are made from L1 virus-like particles, are noninfectious and are administered on a three intramuscular shot series over a 6-month period.
Results from randomized, controlled trials have demonstrated high immunogenicity, efficacy and safety of these vaccines.
Critical steps for a successful vaccine introduction could be summarized as follows:
– Prevention of the disease by the vaccine must rank as a health priority.
– The vaccine must have licensure for safety and efficacy.
– There must be an adequate delivery system and vaccine supply.
– The vaccine must meet the criteria of a financially sustainable program.
– The vaccine needs to be acceptable to health professionals and society.
Introduction of the vaccine needs to be followed by surveillance for vaccine coverage, effectiveness and safety.
Finally, it is necessary that politicians understand the existing tools that can impact the health of their population. This is a bidirectional path in which politicians need to be informed and scientific and medical professionals have to identify adequate channels to inform politicians. While in many countries HPV vaccination is a reality, many efforts are still needed to ensure the massive introduction of HPV vaccines in developing countries.
Future perspective
Ongoing research and programmatic activities in the file are clearly focusing on the evaluation of new screening tools and strategies, development of new polyvalent or more generic vaccines evaluating the actual vaccines in selected populations, such as HIV-infected patients and in ways to efficiently introduce vaccine to low- and medium-income countries.
Footnotes
Acknowledgements
The authors thank all the work of the HPV group at the Catalan Institute of Oncology.
Partial support has been obtained from the Spanish Ministry of Health (FIS PI030240 and FIS P1061246), Marató TV3, CIBERESP, GSK Biologicals and MSD Merck who had no role in the data collection, analysis or interpretation of the results. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
