Abstract
Thyroid cancer is the most common endocrine cancer in the world. Noting that the NOS3 gene polymorphism interferes with nitric oxide production, this study aims to identify and analyze the NOS3 gene polymorphism in the intron 4 region in patients with papillary thyroid cancer. A case-control study was conducted with 31 papillary thyroid cancer patients of both genders who underwent thyroidectomy and treatment with sodium iodide radiopharmaceutical (131I) compared with 81 control patients. Through papillary thyroid cancer, the results were observed, compiled, and analyzed using SPSS version 25.0. The significance level of 5% was adopted. Genotypic frequencies of healthy subjects were in the Hardy-Weinberg equilibrium (P = 0.503). There was a significant genotypic difference between papillary thyroid cancer and healthy individuals (P <0.001). The BB genotype conferred a protective factor for papillary thyroid cancer (P <0.001, odds ratio (OR) 0.16; 95% confidence interval (CI) 0.06, 0.42), while the presence of the A allele appears to be a risk factor for papillary thyroid cancer (P <0.001, OR 3.54; 95% CI 1.86, 6.73). The intron 4 polymorphism of the NOS3 gene was associated with susceptibility to papillary thyroid cancer. Thus, future research into the effects of this polymorphism is essential.
Introduction
Thyroid cancer is the most common endocrine cancer in the world. 1 In Brazil, 9610 new cases—in which 1570 were male and 8040 were female—are predicted for the 2018–2019 biennium. Therefore, the frequency of this carcinoma is observed three times more in women than men. 2
Thyroid neoplasms are classified histologically by their shape, which is subdivided into tumors derived from follicular cells (papillary, follicular, and anaplastic carcinoma) and parafollicular or C cells (medullary carcinoma). 3 Papillary thyroid carcinoma (PTC) is the most common (80%) and has the highest cure rate in patients. It is also quite common in women 30–50 years old. Derived from the follicular epithelium, this type of cancer invades the lymphatic system, generating multifocality and lymph node metastases. 4
Nitric oxide (NO) is a radical and acts as a biological mediator in various processes, including the blood flow regulation and neuronal activity modulation. Mutations in the nitric oxide synthase 3 (NOS3) gene directly interfere with NO production and the regulation of NOS3 gene expression.5 –8 Located on human chromosome 7q36.1, its variable number of tandem repeats (VNTR) polymorphism in intron 4 is closely associated with an altered concentration of plasma nitric oxide and increased reactive oxygen species (ROS) production. 9 This polymorphism has a 27-base-pairs sequence, with one larger and one smaller allele. The larger allele “b” has five tandem repeats of 27 base-pairs, while the smaller allele “a” has four repeats. 10
The excessive ROS presence may cause oxidative stress in cells and, consequently, damage deoxyribonucleic acid (DNA), ribonucleic acid (RNA), lipids, and proteins. Furthermore, ROS can also affect the DNA repair system, rendering the individual more susceptible to the development of diseases, such as cancer. 11 Noting that the NOS3 gene polymorphism interferes with NO production, this study endeavors to investigate whether the NOS3 intron region 4 gene polymorphism acts as a protective or a risk factor in PTC patients.
Material and methods
Subjects
As an observational descriptive case control study, the research participants were divided into two groups. The case group consisted of 31 patients of both genders (20 female and 11 male) over 18 years old (average age: 48 ± 13 years) diagnosed with PTC. These patients were residents of the Federal District (Brazil) and underwent thyroidectomy and radioiodine therapy at a private clinic in the Federal District. The control group consisted of 81 participants (43 female and 38 male) with a mean age of 52 ± 5 years, healthy volunteers, paired and unrelated to the case group.
Participants from both groups were excluded if they were (a) younger than 18 years old, (b) had a thyroid cancer diagnosis but were not eligible for radioiodine therapy, and (c) refused to participate in the research or for whom legal representatives did not consent.
The attending physician chose participating candidates for iodotherapy from their current clinical condition and treatment indication. 12 The following items were assessed: (a) the extent of surgery, complications, residual or metastatic presence; (b) the histopathological diagnosis; (c) the date of diagnosis; (d) the initial disease presentation; (e) the tumor extension; (f) the use of levothyroxine and others (radiotherapy and previous surgery); (g) evaluation of risk factors and medications that may interfere with treatment efficacy; and (h) recent exposure to iodinated contrast. A whole-body survey (WBS) with low iodine-131 doses was requested to estimate the volume of remaining tissue or metastatic disease. In summary, iodotherapy is indicated in patients with incomplete tumor resection or apparent metastases after thyroidectomy and who are not candidates for surgical reintervention, and in patients with presumably complete tumor resection but classified as a high or intermediate risk for persistent disease. 12 The potential adverse effects were also considered: transient changes in gonadal function; acute sialadenitis; advanced menopause; persistent xerostomia and xerophthalmia; increased risk of second cancer; hypersensitivity reactions; spinal depression; leukopenia; thrombocytopenia; acute leukemia; anemia; chromosomal abnormalities; radiation syndromes; thyroiditis; gastritis; sore throat; cough; and death.
The number of participants in the case group was determined according to the sample calculation: estimating a 1% thyroid cancer prevalence among the cancer types in the adult population, a sampling error of 5% and a 95% confidence interval (CI). In the number of patients (n = 8450) registered with this cancer in the studied geographical region, the sample size reached was 12 participants. With the compensation of losses, a sample of 31 PTC patients was considered.
The UNICEUB Ethics Committee approved this study under CAAE No. 57382416.6.0000.0023, and all participants had a detailed clinical history, and their resulting evolution followed.
Laboratory methods
After signing the Free and Informed Consent Terms and the Biological Material Guard Terms, and having answered a proposed questionnaire, each participant had 10 mL of their venous blood collected.
DNA was extracted from blood collected by Invitek's Invisorb Spin Blood Mini Kit (250) (catalog # CA10-0005, lot # 1031100300) and had an average concentration of 20 ng/μL. NOS3 polymorphism genotyping was performed using the polymerase chain reaction-restriction on fragment length polymorphism (PCR-RFLP)-based analysis. The primers used to evaluate NOS3 intron 4 polymorphism were: Sense 5′AGG CCC TAT GGT AGT GCC TT 3′ and Antisense 5′TCT CTT AGT GCT GTG GTC AC 3′.
The DNA amplification used the following thermocycling conditions: 94°C/4 min (initial denaturation), succeeded by 36 denaturation cycles at 94°C/1 min, followed by 60°C/30 s and 72°C/1 min for oligonucleotide annealing. The final extension occurred at 72ºC/10 min. The PCR products were the 393bp allele “a” (mutated) and the 420bp allele “b” (ancestral) fragments.
Statistical analysis
The Chi-square test evaluated departures from Hardy-Weinberg equilibrium and frequency differences between groups. The data comparison of quantitative measurements between genotypes groups underwent a normality test that resulted in an abnormal distribution and, hence, was analyzed by the Kruskal-Wallis statistic test. The statistical analyses were carried out with SPSS (version 26.0, SPSS, Chicago, IL, USA) for Windows, with a 5% significance level adopted a priori.
Results
Table 1 describes the genotypic and allelic frequencies of the NOS3 intron 4 polymorphism. Most participants in the case group had at least one mutated allele. In the genotypic analysis, a statistically significant difference was noted between the studied groups, and that the bb genotype appears to act as a protective factor against PTC (P <0.001; odds ratio (OR) 0.16; 95% CI 0.06, 0.42). A statistical difference was also found between the groups when analyzing the allele frequencies; however, the presence of the “a” allele seems to be a risk factor for PTC (P <0.001; OR 3.54; 95% CI 1.86, 6.73). Nevertheless, the control group’s genotype distribution abided by the Hardy-Weinberg principle (P >0.05).
Genotypic and allelic distribution of papillary thyroid carcinoma (PTC) patients and control subjects.
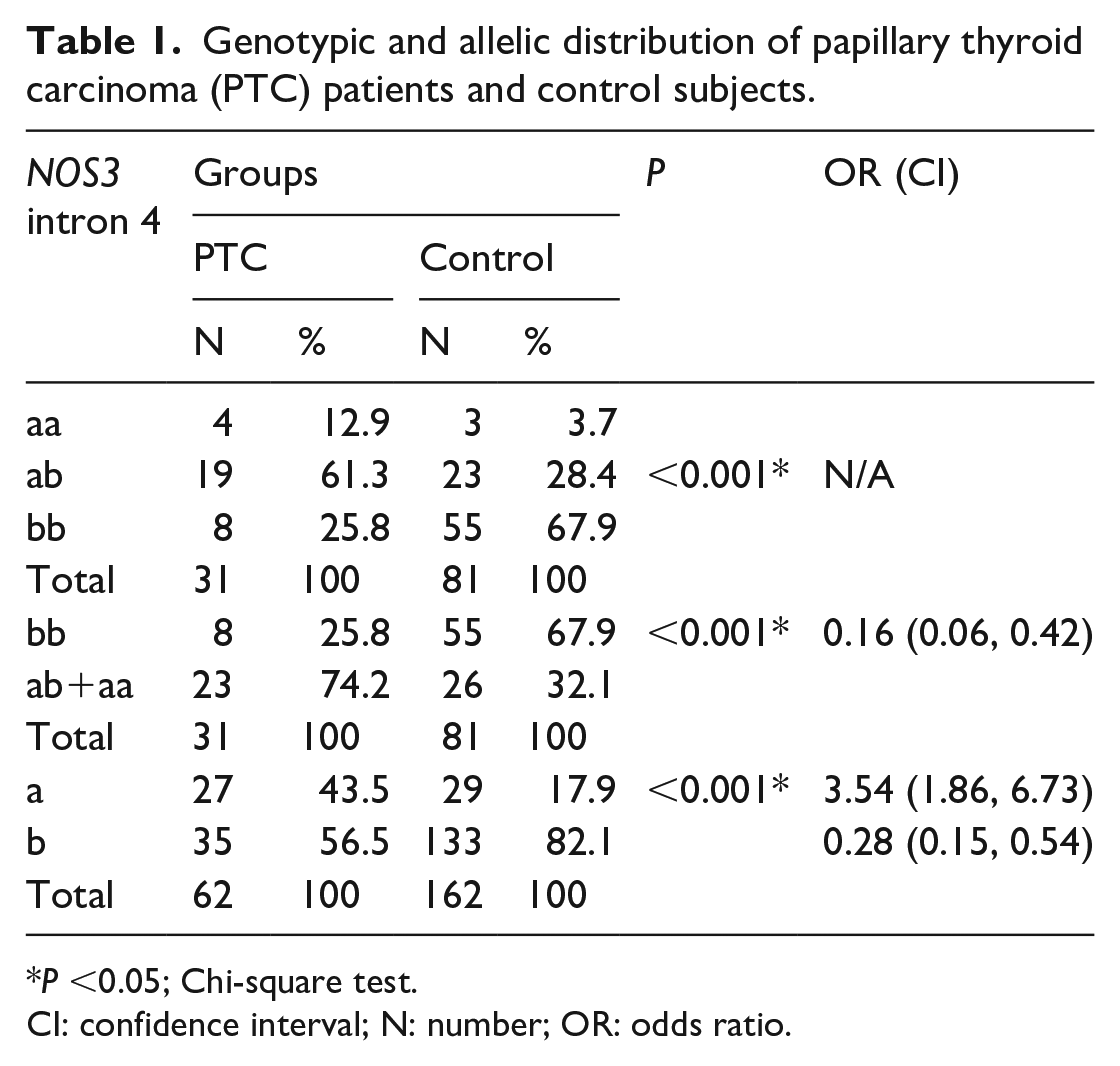
P <0.05; Chi-square test.
CI: confidence interval; N: number; OR: odds ratio.
Clinical characteristics of PTC patients analyzed in relation to polymorphism were: serum thyroglobulin concentration, thyroid-stimulating hormone (TSH), antithyroglobulin, gender, body mass index (BMI), and administered dose. No associations were found between the NOS3 intron 4 polymorphism and these variables (Tables 2 and 3)
Medians, median intervals, and P-value of thyroglobulin, TSH, and BMI measurements in PTC patients, according to genotype.
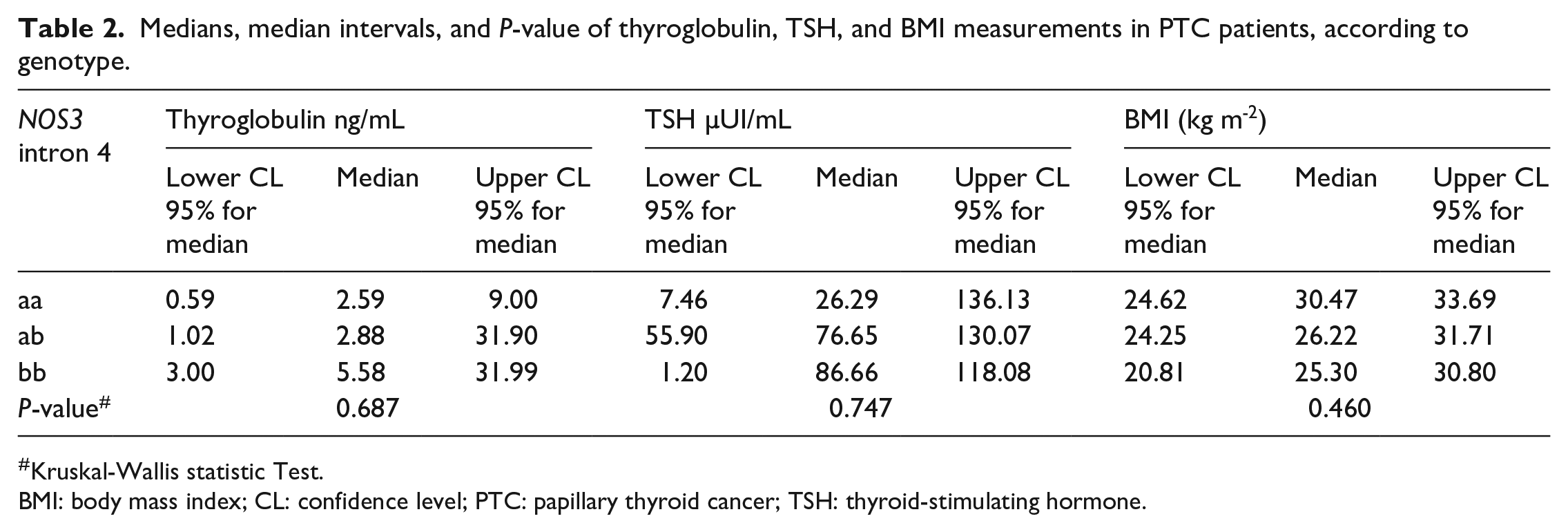
Kruskal-Wallis statistic Test.
BMI: body mass index; CL: confidence level; PTC: papillary thyroid cancer; TSH: thyroid-stimulating hormone.
Distribution of antithyroglobulin concentration, gender, and the radiopharmaceutical administered dose in PTC patients, according to genotype.
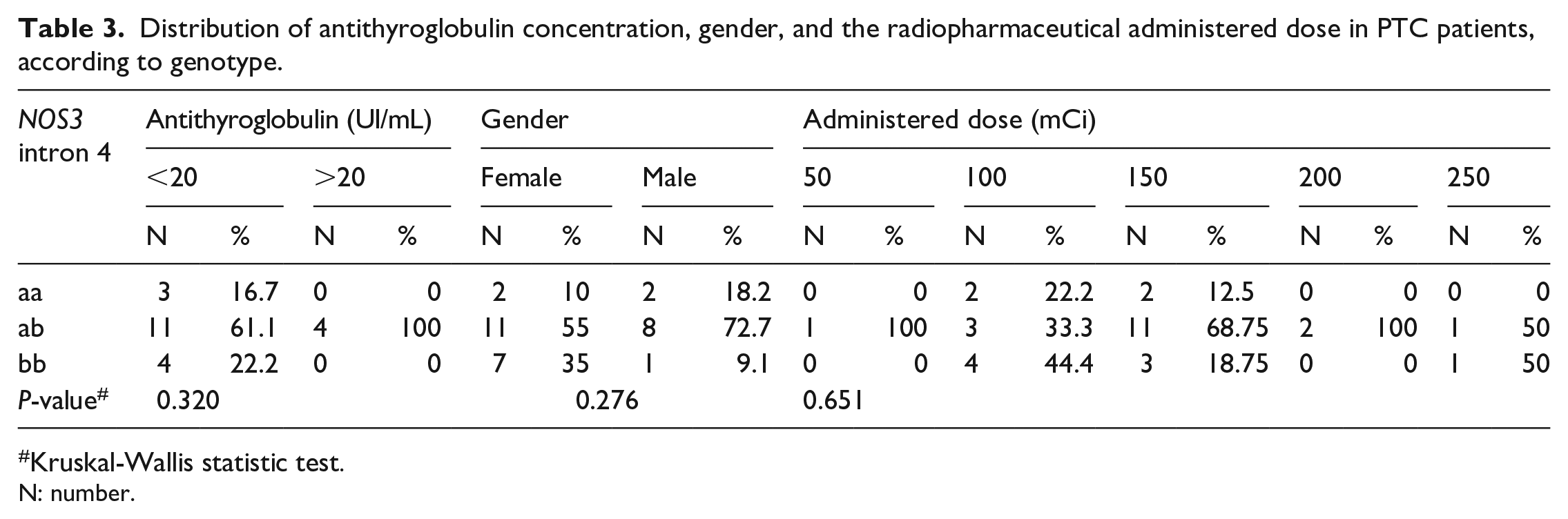
Kruskal-Wallis statistic test.
N: number.
Discussion
The present study revealed that the presence of a polymorphism in the NOS3 intron 4 gene region was associated with susceptibility to PTC (Table 1). Currently, there are no studies that relate PTC with the NOS3 gene intron 4 region, making this work a pioneer in its field. Nonetheless, some studies verified the association of this polymorphism with other cancers.13–18
A meta-analysis study observed that the many studies linking NOS3 G894T and NOS3 intron 4 polymorphisms with cancer risk were conflicting.18,20 The 14 case control studies examined suggested that the analyzed polymorphisms do not alter susceptibility to cancer 17. Nonetheless, Haque et al. 18 suggest that further studies should be done with more detailed information so the possible association could be more precisely evaluated. The authors also recommended that these studies consider gene–gene and gene–environment interactions.
Some studies considered whether there was an association of this polymorphism with prostate cancer. 13 ,14 However, the results are conflicting. A meta-analysis investigated polymorphisms in three NOS3 regions, including intron 4, for an association with prostate cancer. From 11 articles reviewed, Nikolić et al. 21 concluded that the a allele presence is a risk factor for prostate cancer (OR 2.29; 95% CI 1.51, 3.49; P <0.01). In contrast, another study analyzed paraffined prostate tissue and concluded that NOS3 intron 4 polymorphism was not associated with prostate cancer (P = 0.301). 22
A case-control study with Mexican women sought to associate the NOS3 intron 4 polymorphism with breast cancer. In this study, the association between the polymorphism and breast cancer was statistically significant, and the presence of the “a” allele seemed to be a risk factor for its development (P = 0.001; OR 1.9; 95% CI 1.29, 2.95). 16
In contrast, Fu et al.(19), analyzing a meta-analysis by Hao et al.(20), did not find an association between NOS3 G894T polymorphism and breast cancer risk.
In a case-control study involving Caucasian women with vulvar cancer, two NOS3 gene polymorphisms were analyzed for a possible association. The NOS3 intron 4 polymorphism appeared to reduce the duration of disease-free survival (P = 0.03) but was not related to the neoplasia phenotype. The other polymorphism studied in NOS3 exon 7, Glu298Asp, had no relation to disease development or disease-free survival time. 17
The NOS3 intron 4 polymorphism was associated with susceptibility to PTC in the Brazilian population studied. No associations were detected between this polymorphism and the patients' clinical characteristics analyzed: serum thyroglobulin concentration, TSH, antithyroglobulin, gender, BMI, and dose administered. Information on genetic analysis can be a valuable method to help treat and understand diseases that affect the population. Hence, further population studies should be conducted to increase our comprehension of PTC and thus contribute to the diagnosis, prognostic analysis, and gene therapies, which will promote a better quality of life for the carrier.
Footnotes
Acknowledgements
This work was carried out with the support of the Higher Education Personnel Improvement Coordination - Brazil (CAPES) - Financing Code 001. DPI/UnB.
CAPES - Financing Code 001 is a postgraduation scholarship and independent of the research subject.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received financial support from CAPES and DPI/UnB.
Ethical approval
All procedures performed in studies involving human participants were per the institutional ethical standards, national research committee, or both (UNICEUB Ethics Committee - CAAE No. 57382416.6.0000.0023) and 1964 Helsinki declaration and its later amendments or comparable ethical standards.
