Abstract
Purpose:
This study aimed to elucidate the clinicopathological significance and prognostic role of tumor-infiltrating lymphocytes in colorectal cancer.
Methods:
The immunohistochemistry of CD3 and CD8 was performed on 265 human colorectal cancer tissues to investigate the tumor-infiltrating lymphocytes using Immunoscore. The correlation between Immunoscore and clinicopathological characteristics, including survival rates, was elucidated. In addition, the impact of tumor-infiltrating lymphocytes on programmed death-ligand 1 (PD-L1) protein expression was evaluated through immunohistochemistry.
Results:
Of the 265 colorectal cancer tissues, 40.8% had high Immunoscore, while 59.2% had low Immunoscore. A high Immunoscore was significantly correlated with favorable tumor behaviors, including lower rates of vascular, lymphatic, and perineural invasion; lymph node metastasis; and distant metastasis. PD-L1 expressions of tumor and immune cells were significantly higher in patients with high Immunoscore than in those with low Immunoscore. In addition, colorectal cancer tissues with high CD8-positive lymphocytes showed higher PD-L1 expressions of tumor and immune cells than colorectal cancer tissues with low CD8-positive lymphocytes. There was a significant correlation between high Immunoscore and better overall survival. However, there was no significant difference in survival rate according to PD-L1 expressions of tumor and immune cells in high and low Immunoscore subgroups.
Conclusions:
Taken together, our results showed that high tumor-infiltrating lymphocytes were significantly correlated with favorable tumor behaviors and better survival. In addition, there was a significant correlation between PD-L1 expression and tumor-infiltrating lymphocytes.
Introduction
The anti-tumor effect of immunotherapy has been demonstrated in various cancer types. The evaluation of programmed death-ligand 1 (PD-L1 (CD274)) expression might be important in predicting the effects of immunotherapy and prognosis of patients. However, the prognostic implication of PD-L1 expression in patients with colorectal cancer (CRC) remains unclear. 1 Although PD-L1 can be expressed in tumor and immune cells, the prognostic implication of cell-specific PD-L1 expression in CRC has not been fully elucidated. In addition, the PD-L1 expression pattern has not been fully elucidated in various tumor microenvironments. In the understanding of tumor behaviors, the tumor microenvironment is important in various tumors, including CRC. 2 A tumor microenvironment includes blood and lymphatic vessels, stromal cells, and infiltrating immune cells. 3 The correlations between tumor-infiltrating lymphocytes (TILs) and prognosis have been reported in various cancers, including CRC.4-6 The TILs were evaluated using Immunoscore, which is defined as a summation of scores of two regions (the core of the tumor and the invasive margin) for the immunohistochemistry of CD3- and CD8-positive T lymphocytes.7,8 In patients with kidney cancer and glioma, a significant correlation was observed between TILs and worse prognosis. 9 However, some studies found that CD8-positive T lymphocytes are associated with a better prognosis in patients with CRC.2,10 Recent studies reported that the prognostic implications differed according to the types of immune cells. The prognostic implication of immune cells differed according to the type of infiltrating immune cells. For example, T-helper 1 cells, M1 macrophages, and natural killer cells, but not M2 macrophages and B cells, were significantly correlated with better prognosis. In addition, the localization of intratumoral immune cells can influence the patient’s survival.11-19 In addition, tumor-infiltrating lymphocytes may be differed by microsatellite instability and may be frequently found in colorectal cancers with MSI-H. 20
This study aimed to elucidate the clinicopathological significance and prognostic role of TILs using Immunoscores in CRC. In addition, the correlation between TILs and PD-L1 expression of tumor (T-PD-L1) and immune cells (I-PD-L1) was investigated in CRC.
Materials and methods
Patients and tissue array methods
The files of 265 patients who had undergone surgical resection of CRC at the Eulji University Medical Center, between 1 January 2001 and 31 December 2010, were analyzed. We reviewed medical charts, pathological records, and glass slides in order to assess clinicopathological characteristics such as age; sex; tumor size; tumor location; tumor differentiation; vascular, lymphatic, and perineural invasion; depth of tumor; lymph node metastasis; metastatic lymph node ratio; distant metastasis; and pathologic tumor node metastasis (pTNM) stages. These cases were evaluated according to the 8th edition of the American Joint Cancer Committee TNM classifications. 21 This protocol was reviewed and approved by the Institutional Review Board of Eulji University Hospital (Approval No. EMC 2017-04-018). Five array blocks containing a total of 265 resected CRC tissue cores obtained from patients were prepared. Briefly, tissue cores (2 mm in diameter) were taken from individual paraffin-embedded CRC tissues (donor blocks) and arranged in recipient paraffin blocks using a trephine apparatus, as previously described. 22 The staining results of the different intratumoral areas in these tissue-array blocks showed excellent agreement. A core was chosen from each case for analysis. An adequate case was defined as a tumor occupying more than 10% of the core area. Each block contained internal controls consisting of non-neoplastic colon tissue. Clinical outcomes were followed from the date of surgery to the date of death, resulting in a follow-up period ranging from 0 to 60 months.
Immunohistochemical staining and evaluation
Sections 4 μm in thickness for immunohistochemistry were cut from each tissue-array block, deparaffinized, and dehydrated. For antigen retrieval, sections were treated with 0.01 M citrate buffer (pH 6.0) for 5 min in a microwave oven followed by treatment with 3% H2O2 to quench endogenous peroxidase. Sections were treated with normal serum of the host animal of the secondary antibody to block non-specific binding. Sections were then incubated with anti-PD-L1 antibody (clone SP263, Ventana Medical Systems, Inc., Tucson, AZ, USA), anti-CD3 (Leica Biosystems, Newcastle Upon Tyne, UK), and anti-CD8 (Leica Biosystems). Immunohistochemical staining was conducted following a compact polymer method using a VENTANA benchmark XT autostainer (Ventana Medical Systems, Inc.). Visualization was performed by treatment with OPTIVIEW universal 3,3′-diaminobenzidine kit (Ventana Medical Systems, Inc.). To confirm the reaction specificity of the antibody, a negative control stain without primary antibody was performed. All immunostained sections were lightly counterstained with Mayer’s hematoxylin. The immunohistochemical result for PD-L1 were evaluated by two independent researchers. PD-L1 showed immunoreactivity in the cell membrane. The positivity for PD-L1 was defined as a membranous expression of any intensity in ⩾10% of tumor or immune cells, regardless of the intensity of PD-L1 expression.
Determination of Immunoscore
All immunohistochemical stained slides for CD3 and CD8 were scanned using Pannoramic MIDI II (3DHISTECH, Budapest, Hungary). Images were captured from two regions, core of tumor and invasive margin using CaseViewer 2.0 (3DHISTECH). From captured images, CD3- and CD8-immunoreactive lymphocytes were qualified using NIH Image Analysis software (version 1.6.0, National Institute of Health, Bethesda, MD, USA) after setting one consistent intensity threshold. CD3- and CD8-immunoreactive lymphocytes were expressed as pixels at each region. In the present study, the cut-off used median value of pixels in each region. By cut-off, patients were classified into two groups as high (score 1) or low (score 0). Immunoscore is defined as a summation of scores of two regions and is divided into high (IS 3-4) and low score (IS 0-2). 8
Statistical analysis
Statistical analyses were performed using SPSS version 22.0 software (IBM Co., Chicago, IL, USA). The significance of the correlation between Immunoscore and the clinicopathological characteristics was determined by χ2 test. The comparisons between Immunoscore and age, tumor size, or metastatic lymph node ratio were analyzed using the two-tailed Student’s t test. Survival curves were estimated using the Kaplan–Meier product-limit method, and differences between the survival curves were determined to be significant based on the log-rank test. Results were considered statistically significant for P < 0.05.
Results
Correlation between TILs and clinicopathological characteristics
We analyzed the correlations between TILs and clinicopathological characteristics in CRC. Figure 1 shows representative images of CD3 and CD8 immunostaining in CRC. The proportion of patients with high Immunoscore (score, 3–4) was 40.8% (108 out of 265). In contrast, the proportion of patients with low Immunoscore (score, 0–2) was 59.2% (157 out of 265). High Immunoscore was significantly associated with favorable tumor behaviors, such as lower rates of vascular invasion, lymphatic invasion, perineural invasion, lymph node metastasis, and distant metastasis. There were significant correlations between high Immunoscore and lower pT and pTNM stage (Table 1). In addition, patients with high CD3-positive lymphocytes showed high CD8-positive lymphocytes (P < 0.001).
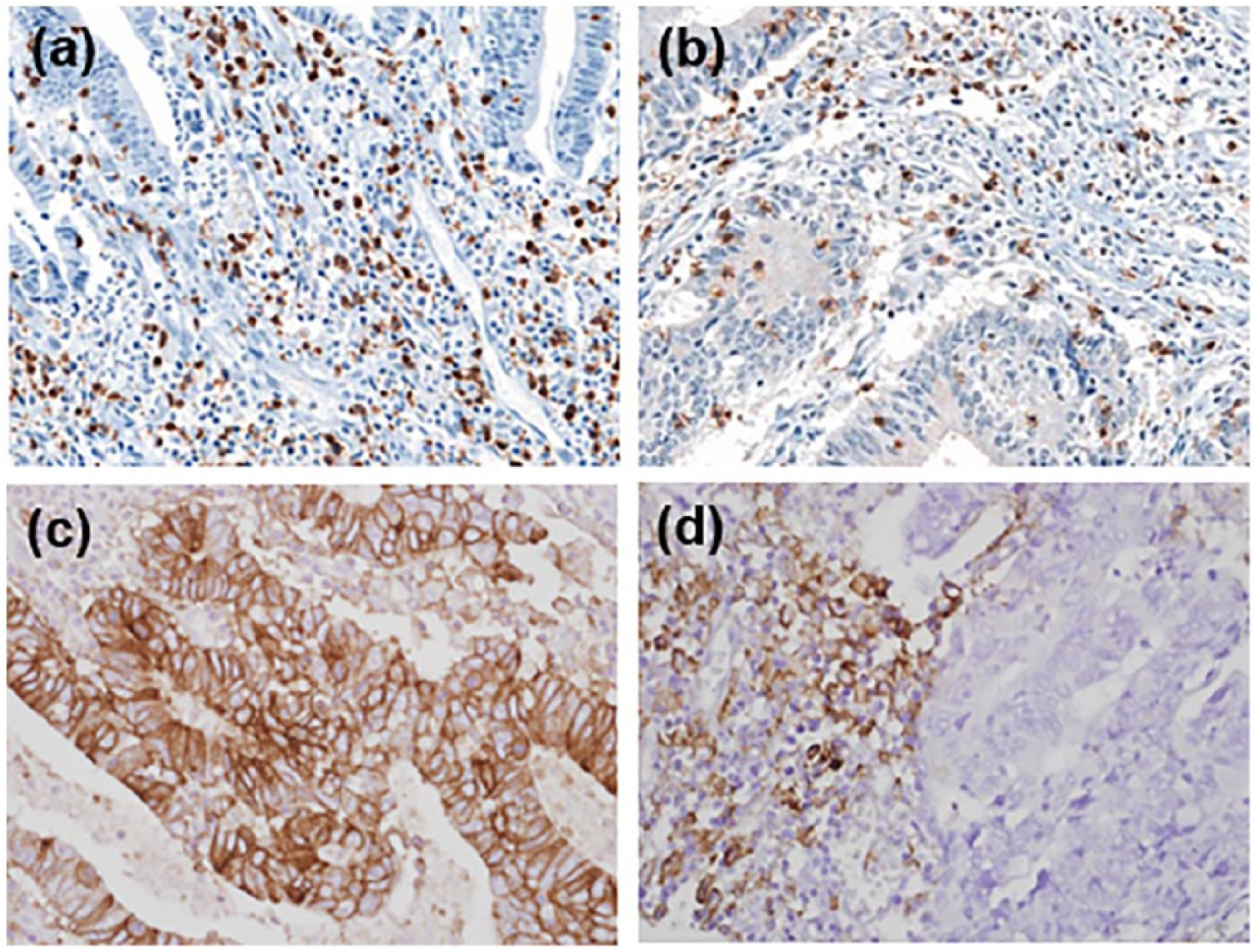
Representative images showing CD3 (a), CD8 (b), and PD-L1 expressions of tumor (c) and immune cells (d) in colorectal cancer (×400).
The correlation between Immunoscore and clinicopathological parameters in colorectal cancers.
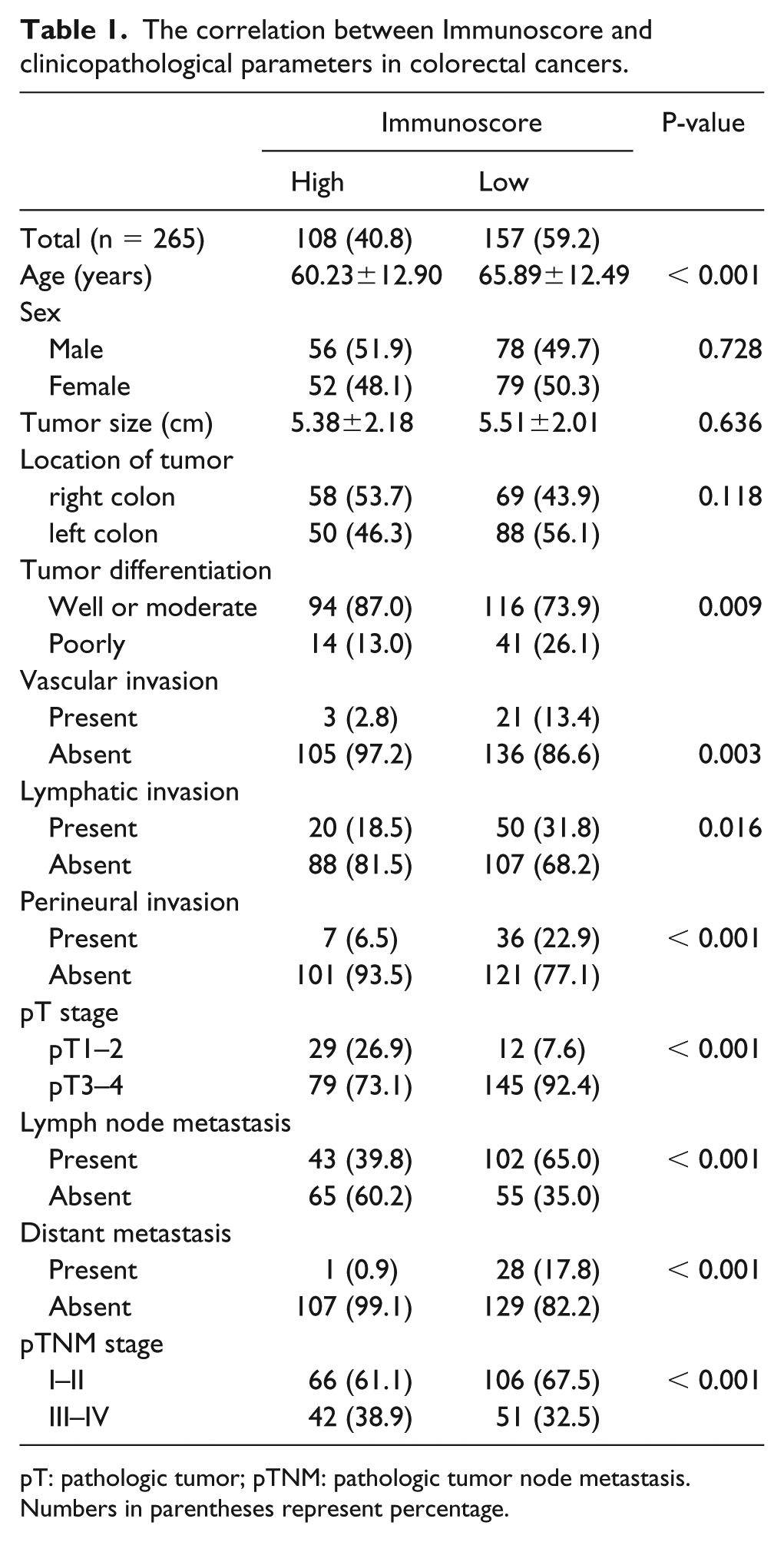
pT: pathologic tumor; pTNM: pathologic tumor node metastasis.
Numbers in parentheses represent percentage.
Correlation between Immunoscore and PD-L1 expression
The correlations between Immunoscore and T- and I-PD-L1 expressions were investigated. High Immunoscore was significantly correlated with high T- and I-PD-L1 expressions (P = 0.040 and P < 0.001, respectively;Table 2). In addition, there were significant correlations between high CD8-positive lymphocytes and high T- and I-PD-L1 expressions (P = 0.024 and P < 0.001, respectively). However, in comparison with CD3-positive lymphocytes, the correlation with I-PD-L1 expression, but not T-PD-L1 expression, was identified (P < 0.001 and P = 0.063, respectively).
The correlation between tumor-infiltrating lymphocytes and PD-L1 expression in colorectal cancers.
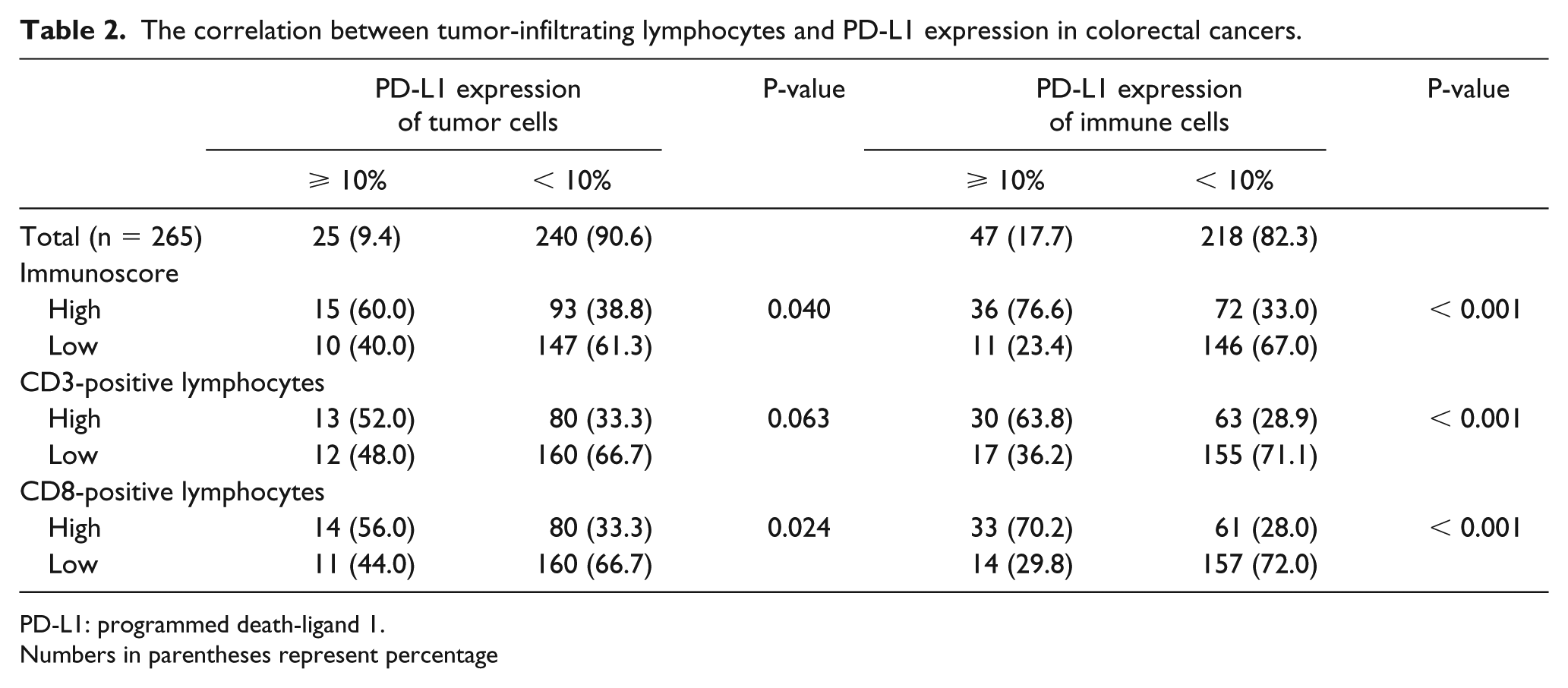
PD-L1: programmed death-ligand 1.
Numbers in parentheses represent percentage
Correlation between Immunoscore and survival
The correlation between Immunoscore and survival was investigated in CRCs. Patients with low Immunoscore showed poor overall survival compared with patients with high Immunoscore (P < 0.001; Figure 2). The prognostic implication of Immunoscore was investigated in detail according to the T- and I-PD-L1 expressions. However, the effects of T- and I-PD-L1 expressions were not significant on the survival of patients with high and low Immunoscores (Figure 3). There was a significant correlation between I-PD-L1 expression and poor overall survival (P = 0.014); however, there was no significant correlation between T-PD-L1 expression and overall survival (P = 0.295).
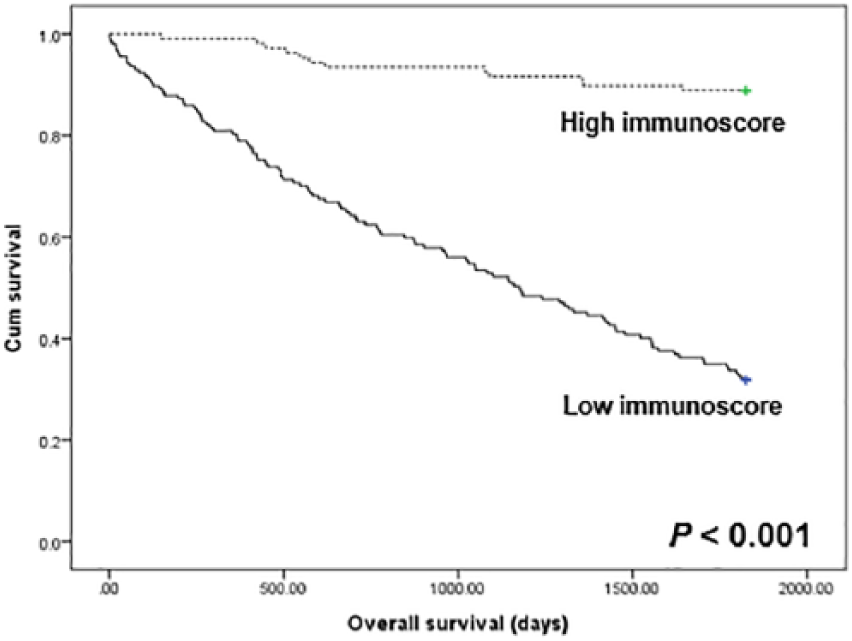
Kaplan–Meier curves for patient survival according to Immunoscore. Patients with high (dotted line) and low Immunoscore (solid line) showed overall survival.
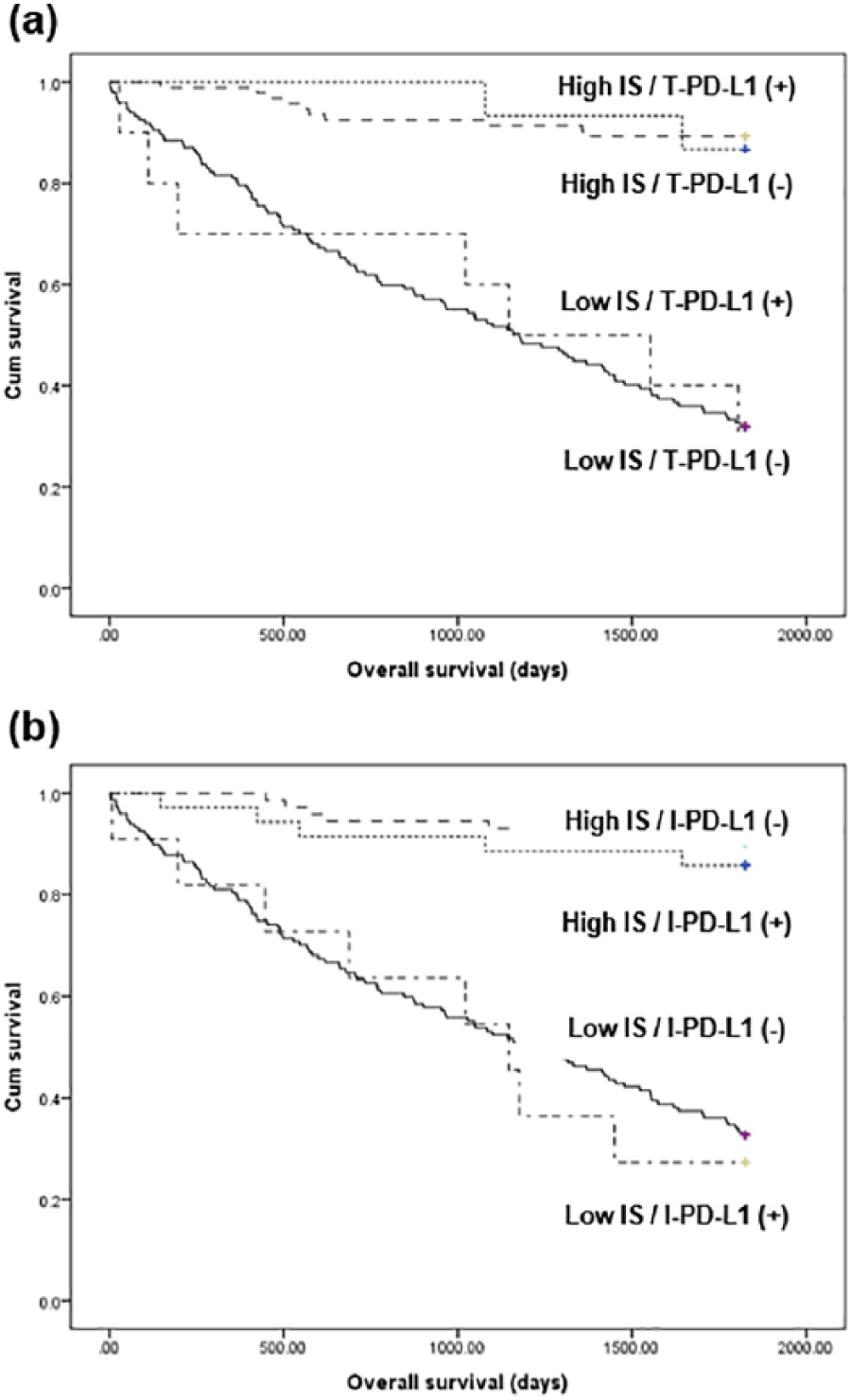
Kaplan–Meier curves for patient survival according to IS and PD-L1 expressions. (a) Survival according to Immunoscore and PD-L1 expression of tumor cells (T-PD-L1). (b) Survival according to Immunoscore and PD-L1 expression of immune cells (I-PD-L1). IS: Immunoscore; PD-L1: programmed death-ligand 1.
Discussion
Immunotherapy using tumor immunity against cancer cells is a treatment that has recently been in the limelight. The therapeutic effect of immunotherapy can be predicted through PD-L1 expression of tumor and/or immune cells.1,2 Although previous studies have shown that TILs may be useful for predicting patients’ prognoses, 23 Immunoscore is not commonly used in daily practice. The evaluation of TILs can be useful for the prediction of the therapeutic effect of chemotherapy and immunotherapy. 24 The correlation between TILs and PD-L1 expression is not fully elucidated in CRC. The present study aimed to show the prognostic role of Immunoscore and the correlation between Immunoscore and PD-L1 expression through immunohistochemistry using human CRC tissues.
In the tumor microenvironment, CD3- and CD8-positive lymphocytes may be important in tumor progression. 23 Although previous studies reported the correlation between high Immunoscore and survival in CRC,17,18,23 the impact on other clinicopathological characteristics has not been fully elucidated. In the present study, high Immunoscore was significantly correlated with a better survival. Additionally, patients with high Immunoscore had significantly favorable tumor behaviors, such as lower rates of lymphovascular invasion, lymph node metastasis, and distant metastasis, than those with low Immunoscore. Our results showed that there was a significant correlation between CD3- and CD8-positive lymphocytes. Similar to a previous study, patients with high CD3- or CD8-positive TILs had better survival rates than those with low CD3- or CD8-positive TILs (P < 0.001 and P < 0.001, respectively; data not shown). 2 In the subgroup analysis, the survival rate was low in patients with high CD3 and CD8 expression, in patients with high CD3 or CD8 expression, and in patients with low CD3 and CD8 expression. However, there was no significant difference in the survival rate between patients with high CD3 expression and high CD8 expression (data not shown). However, the relationship between TIL-associated pathway and tumor progression remains unclear.
Previous studies showed that PD-L1 expression was associated with TILs.2,3,24,25 Similar to our study, Lee et al. 2 reported the correlation between CD3- and CD8-positive lymphocytes and PD-L1 expression in CRC. However, there was no finding showing the correlation between Immunoscore and PD-L1. In our results, the subgroups were divided based on the level of T- and I-PD-L1 expressions. High Immunoscore was significantly correlated with higher T- and I-PD-L1 expressions. The subgroup with high CD8-positive lymphocytes showed higher PD-L1 expressions of both tumor and immune cells. However, no correlation was found between CD3-positive lymphocytes and T-PD-L1 expression. Therefore, the TILs can be useful for the prediction of PD-L1 expression in CRC. As described above, the prognosis of CRC patients differed by Immunoscore. In the present study, no significant difference was found in the survival rate between subgroups with high Immunoscore and those with low Immunoscore based on the level of PD-L1 expression. There was a significant correlation between poor overall survival and I-PD-L1 expression, but not T-PD-L1 expression. Taken together, the Immunoscore of immune cells and the PD-L1 expression pattern will be important in the prediction of a patient’s prognosis. However, in the present study, high Immunoscore was significantly correlated with T- and I-PD-L1 expressions. These findings indicated that CD3- and CD8-positive T lymphocytes and PD-L1-expressing immune cells are important components of the adaptive immune system in CRC. In patients with malignant melanoma, the density of CD8-positive TILs was the best predictive marker of the therapeutic effect of PD-1 blockade rather than PD-1 or PD-L1 expression. 26 However, there was no significant correlation between CD4-positive T lymphocytes and PD-L1 expression of tumor and immune cells. 27 PD-L1 expression was significantly higher in cases with CD20-positive B lymphocytes than in cases without CD20-positive B cells. 27 Since the possibility of TILs as the predictive marker of immunotherapeutic effect is not clear in CRC, further cumulative studies are required.
The present study showed the prognostic role of Immunoscore. However, after the introduction by Galon et al., 28 the Immunoscore in CRC was evaluated through various criteria. High Immunoscore was evaluated at the center of the tumor and the invasive front. Basically, the intratumoral heterogeneity of TILs can affect the Immunoscore. Because our study used tissue microarray with 2 mm core at two sites, intratumoral heterogeneity can be considered. However, there is the potential to show heterogeneity, even if it is not a tissue microarray, since it does not essentially CD3 and CD8 immunohistochemistry on all tissue samples. In the present study, tissue core was obtained from the hot spot of TILs on histologic examination. As described above, the selection of the status of TILs on hot spot may be important in predicting the prognosis of CRC patients. Even though the CD3- and CD8-negative lymphocytes are counted using image analysis, a hot spot for analysis should be selected. Thus, the process of selecting hot spots can be similar in either the daily practice or in the tissue microarray used in this study. If an immunohistochemistry is implemented in the 4-μm thick section, many immune cells can be present in the thickness. Therefore, a counting method using an image analyzer cannot be guaranteed owing to the overlapping and gathering of immune cells. In addition, a counting method is not comparable to the usual method because the commercial image analyzer may not be present in all pathological laboratories. The present study used the median value of pixel of CD3/CD8 immunohistochemistry as a cut-off using an image analyzer with an open source. Therefore, further studies are needed to validate the Immunoscore evaluating methods.
We previously reported the prognostic role of PD-L1 expression in solid malignant tumors. 1 In the previous meta-analysis, the rate of T-PD-L1 expressions in CRC patients was 35.4%. 1 T-PD-L1 expression was significantly correlated with worse disease-free survival, but not overall survival. 1 However, in our data, although there was no significant correlation between T-PD-L1 expression and survival rate, patients with I-PD-L1 expressions had significantly better overall and disease-free survival than those without PD-L1 expression (P = 0.014 and P = 0.031, respectively; data not shown). Our results are inconsistent with those of previous reports. 1 This could be caused by various PD-L1 clones, evaluation criteria, and population. In daily practice, the evaluation criteria for PD-L1 expression is introduced based on the cancer type. Reliable cut-offs for PD-L1 expression have been suggested based on the predicted immunotherapeutic effect and survival.29-31 However, the detailed criteria for PD-L1 expressions is not clear in CRC. The present study used 10% as the cut-off for PD-L1 expression. CRC with PD-L1 expression, regardless of the rate, was significantly correlated with Immunoscore (data not shown). Therefore, the Immunoscore may be useful for predicting PD-L1 expression.
In conclusion, our results showed that high Immunoscore was significantly correlated with favorable tumor behaviors and better survival. In addition, PD-L1 expression was significantly correlated with high Immunoscore and CD8-positive lymphocytes. In daily practice, Immunoscore may be a useful prognostic marker in CRC patients and a predictive marker of patients’ response to anti-PD-1 and PD-L1 therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Research Foundation of Korea (NRF) grant funded by the Korea government (Ministry of Science, ICT & Future Planning) (NRF-2017R1C1B5018063).
