Abstract
Background
Few studies have addressed the clinicopathological features of colorectal cancer (CRC) that express programed death-ligand 1 (PD-L1). Various assays and scoring methodologies were used and thus inconsistent results were obtained. In this study, we aimed to investigate the relationship of PD-L1 expression in CRC with various clinicopathological variables using a standardized assay and scoring algorithm.
Design
Tissue microarrays were constructed from 91 random cases of CRC diagnosed at King Hussein Cancer Center (KHCC). Immunohistochemical (IHC) staining using the monoclonal antibody 22C3 was performed. Scoring using the standard “Combined Positive Score” (CPS) method was done. CPS of ≥1 was considered positive. Various clinicopathological features were collected from the medical records of the patients.
Results
Of the 91 cases, 49 (53.8%) were PD-L1 positive (CPS ≥1). PD-L1 expression was more frequent among moderately differentiated carcinomas (43 of 72 (59.7%) were positive compared to 6 of 19 (31.5%) poorly differentiated cases (P = 0.029)); among node negative cases (21 of 24 (87.5%) N0 cases were PD-L1 positive in contrast to 28 of 67 (41.8%) N1/N2 cases (P = <0.001)); among mucinous subtype (12 of 15 (80%) of cases (P = 0.02)); and among mismatch repair deficient (dMMR) (16 of 16 (100%) versus 11 of 30 (36.6%) MMR proficient (P = <0.001)). Age, gender, localization, and T or M stages were not significantly associated with PD-L1 expression.
Conclusion
PD-L1 expression in CRC is associated with favorable prognostic features; namely, lower grade, N0, mucinous variant, and dMMR tumors.
Introduction
Colorectal cancer (CRC) is the third most common cancer and the fourth leading cause of cancer death worldwide, also its incidence and mortality are increasing each year. 1 Despite the improvement in treatment modalities of CRC, cancer recurrence and metastasis remain major causes of treatment failure. Recent advances in cancer therapy, including targeted therapies as well as immunotherapy, are starting to produce an impact on the outcome of many types of cancer.2-4
Cancer immunotherapy has now been established as the fourth cornerstone of cancer therapy, with surgery, radiation, and traditional chemotherapy being the remaining cornerstones. 5 From adoptive T-cell therapy to immune checkpoint blockers, immunotherapy has continued to achieve significant success and has revolutionized cancer treatment. Amongst these therapies are the anti-programmed death-ligand 1 (PD-L1) and anti-programmed death-1 (PD-1).4,5
PD-L1 is an immune-regulatory molecule that, upon interacting with its receptor, PD-1, suppresses the CD8 cytotoxic immune response in both physiologic and pathologic pathways.4-6 It is mainly expressed on the surface of activated T cells and regulates their proliferation and activation. The overexpression of PD-L1 by cancer cells has been co-opted as an immunoediting approach to avoid immunologic surveillance and thereby facilitate cancer growth. 7
Accumulating data are in favor of a relationship between the expression of PD-L1 in tumors and response to treatment. Preliminary studies that are based on immunohistochemical (IHC) detection of PD-1, PD-L1, and PD-L2 expression in melanoma, lung cancer, kidney cancer, and CRC have suggested that PD-L1 expression by tumor cells (TC) might be most closely correlated with a response to anti-PD-1 blockade. 8 As for the expression of PD-L1 in CRC and its prognostic significance, studies are still limited and conclusions are controversial. Some studies have revealed a negative association between PD-L1 expression and the overall survival rate of CRC patients9-11; on the other hand, others have demonstrated a contradicting effect.12,13
Molecular mechanisms of CRC tumorigenesis mainly include three pathways: chromosomal instability, microsatellite instability (MSI), and epigenetic instability. MSI is mainly caused by dysfunction of mismatch repair (MMR) proteins (MLH1, MSH2, PMS2, and MSH6). Most CRCs develop via a chromosomal instability pathway, and approximately 12–15% have MMR dysfunction, which is responsible for MSI-high/MMR-deficient (dMMR). MSI-high CRC displays high infiltration with CD8 + cytotoxic T lymphocytes, which is associated with the upregulation of T-cell checkpoints such as PD-L1.14,15
Clinically, initial trials suggested a minimal role for immunotherapy in CRC yet showed a significantly higher response rate and survival in patients with MSI-high/dMMR CRC treated with pembrolizumab (anti-PD-1) when compared to patients with CRC-mismatch repair proficient (pMMR).15-17
The present study aimed to evaluate the expression of PD-L1 in Jordanian patients with CRC, investigate its prognostic significance, and explore its association with clinicopathological variables. Additionally, we also aim to clarify the relationship between the microsatellite status and PD-L1 expression in CRC and its effect on CRC prognosis.
Materials and methods
Formalin-fixed, paraffin-embedded tissue blocks were collected from patients who underwent colectomy at King Hussein Cancer Center (KHCC), Amman, Jordan. Through an archival search at the Department of Pathology and Laboratory Medicine at KHCC, 91 CRC cases of stages II, III, and IV were retrieved. Tumor blocks, their respective hematoxylin and eosin (H&E) sections, and pathology reports were reviewed for each of the selected cases. Furthermore, data regarding each patient's clinical stage, survival, and MMR status were obtained from their medical and pathology records at KHCC.
Upon H&E slide evaluation, the block with the highest TC density was selected, and the precise tissue area of interest was marked for tissue microarray construction.
Tissue microarray blocks were constructed containing 25–30 tissue cylinders, and one extra control cylinder of normal tissue was introduced into the recipient block.
Two 4-μm sections were prepared for each block; the first was stained with H&E as a control slide and was used to evaluate the appropriateness of the tumor cores. The second slide was used for IHC. Immunostaining was performed on the slides using the anti-PD-L1 22C3 antibody clone (Agilent Technologies, Santa Clara, CA, USA) run on the Ventana Benchmark-Ultra (Ventana Medical Systems Inc., Tucson, AZ, USA). The choice of the anti PD-L1 clone (22C3) was based on the fact that this clone was readily available at our institution with a vast experience in its use and interpretation; also, it has the largest US Food and Drug Administration (FDA) approvals of all anti PD-L1 clones for clinical indications including esophageal and gastric cancers. IHC staining interpretation was based on a combined positive score (CPS) ≥1 cutoff, which is defined as the ratio of PD-L1 membrane-stained, TC, and cytoplasmic and membranous staining mononuclear inflammatory cells (TCs, macrophages, lymphocytes) in the tumor microenvironment to the total TCs present, multiplied by 100. Immunoreactivity was considered negative if the CPS was <1.
Immunohistochemical stains for MMR proteins were also performed using an automated immunostaining method on Ventana BenchMark Ultra. Antibody clones used were as follows: MLH1 clone M1, MSH2 clone G219-1129, PMS2 clone EPR3947, and MSH6 clone 44 (all from Ventana Medical Systems Inc.). Intact or lost staining in the TC nuclei was reported. Appropriate external and internal controls were evaluated. The external control consisted of a section of normal human appendix placed on each test slide for every case while the internal control in each case consisted of known reactive and non-reactive tissue elements. Normal lymphocytes in lymphoid follicles as well as crypt base epithelial cells in the appendix and the test cases were evaluated as positive internal and external controls while surface epithelial cells and non-proliferating cells served as negative controls. Confirmed lack of staining was considered indicative of dMMR according to their well-known associations.
Statistical analysis
Patient characteristics and their demographic and disease information were analyzed using descriptive statistics. Comparisons between PD-L1 + and PD-L1 − cases according to certain features were performed using the chi-square test. Where appropriate, the odds ratio was also calculated. Data were analyzed with IBM SPSS version 25 and MedCalc version 19.
Results
The total number of cases studied were 91. Of these, 53 (58%) were males, 38 (41.7%) were females, and 50 cases (55%) belonged to patients over 60 years. The number of PD-L1 positive cases (CPS≥1) was 49 (53.8%) while 42 (46.1%) of cases were negative. Examples of PD-L1 positive cases are displayed in Figure 1.
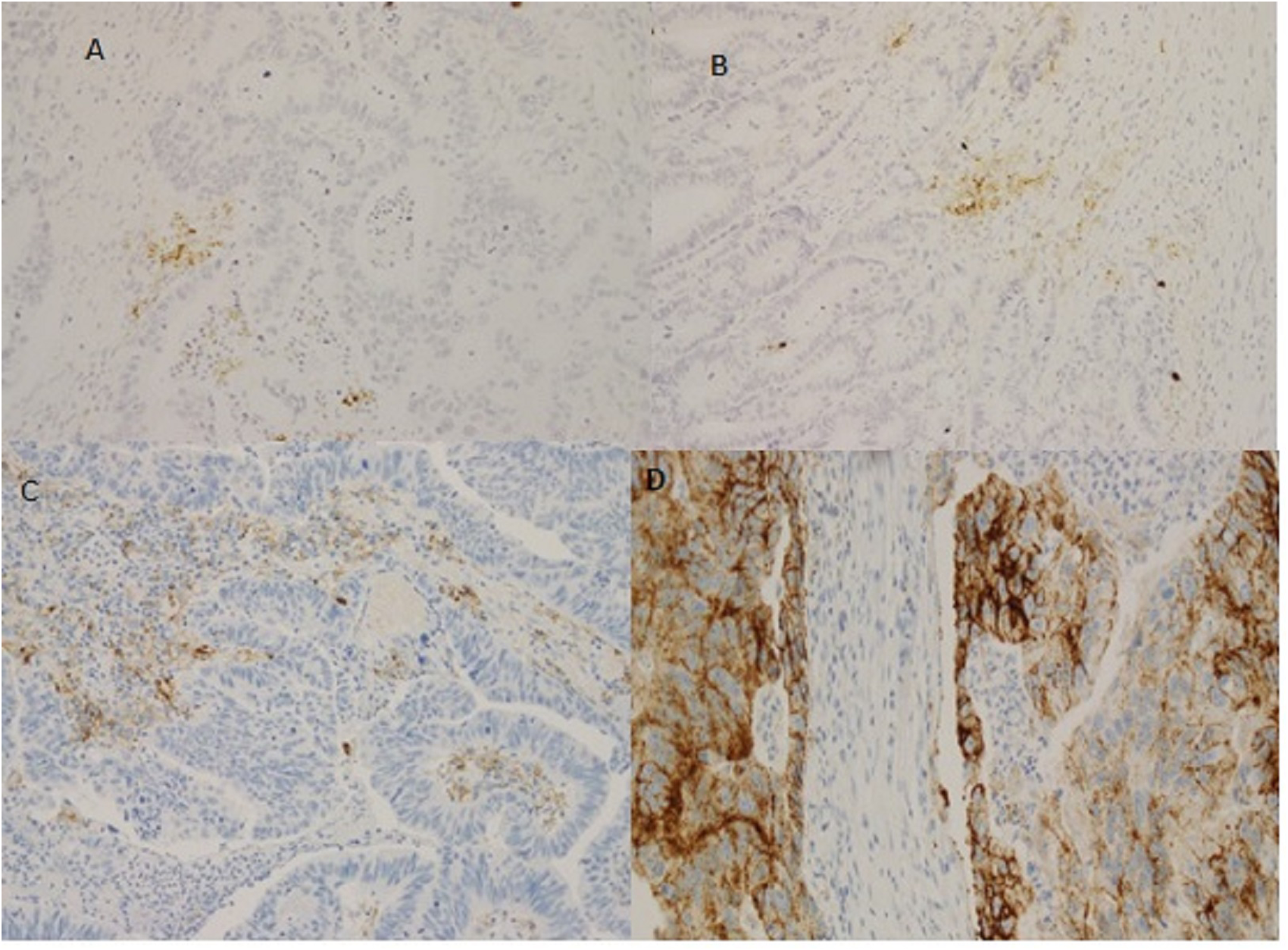
PD-L1 in colorectal cancer. (a) CPS = 3; (b) CPS = 8; (c) CPS = 20; (d) CPS = 90.
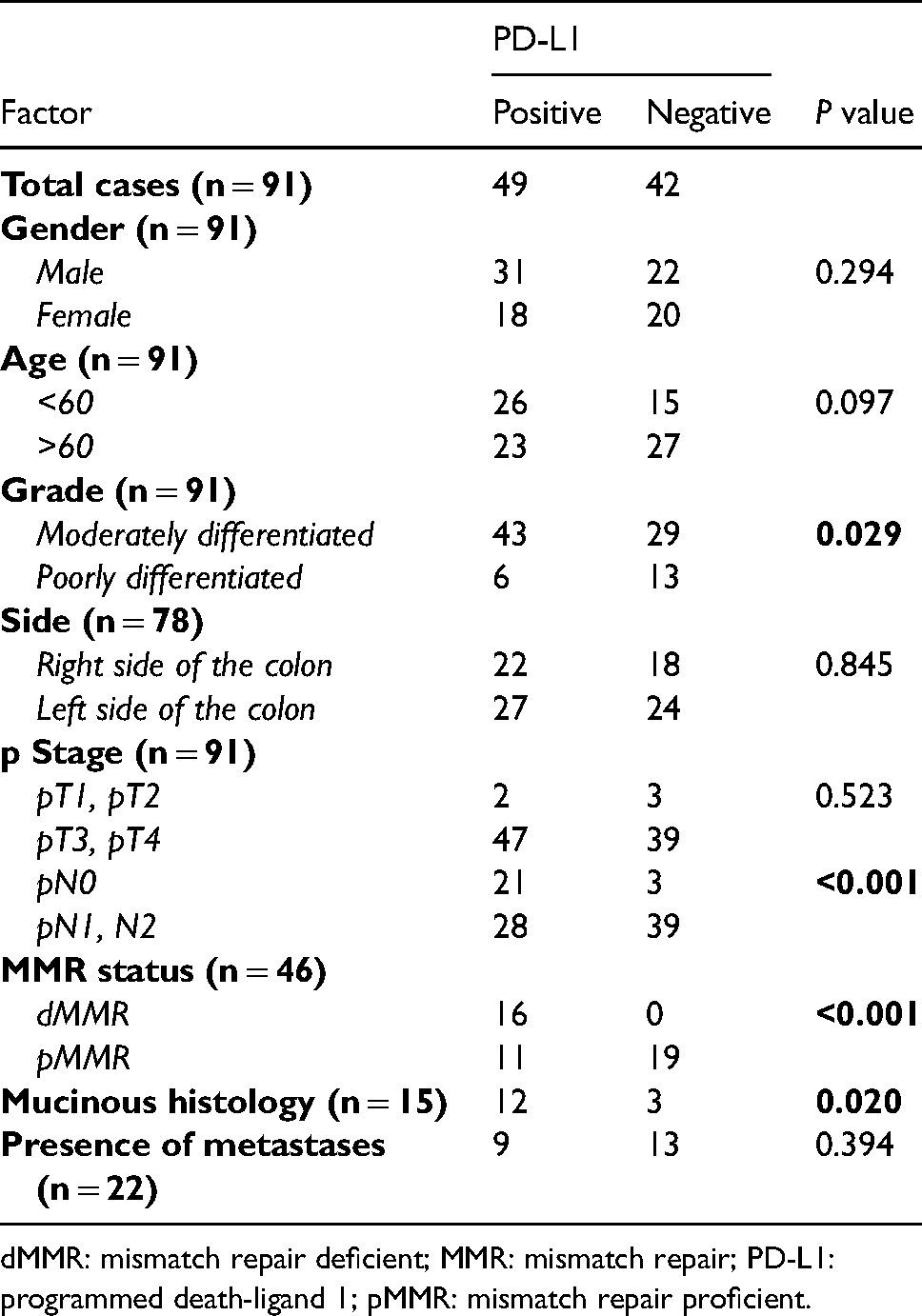
The association between PD-L1 expression (CPS≥1) and clinicopathological factors are shown in Table 1.
Correlations of PD-L1 with clinicopathological factors.
dMMR: mismatch repair deficient; MMR: mismatch repair; PD-L1: programmed death-ligand 1; pMMR: mismatch repair proficient.
Tumor grade, pathological N stage, MMR status, and mucinous histology were significantly associated with PD-L1 expression (Table 1).
Odds ratios were calculated, where appropriate, and the results are shown in Table 2. The odds of having a positive PD-L1 at the CPS cutoff of CPS≥1 when tumors are moderately differentiated were 3.2 times the odds of having positive PD-L1 when the tumors were poorly differentiated (P = 0.033). In addition, cases with pN0 stage had 9.8 times the odds of having a positive PD-L1 result compared to cases with pN1–2 (P<0.001). In addition, cases with dMMR had 56 times the odds of having a positive PD-L1 compared to pMMR cases (P = 0.007)
Odds ratio for PD-L1 positive cases with selected features.
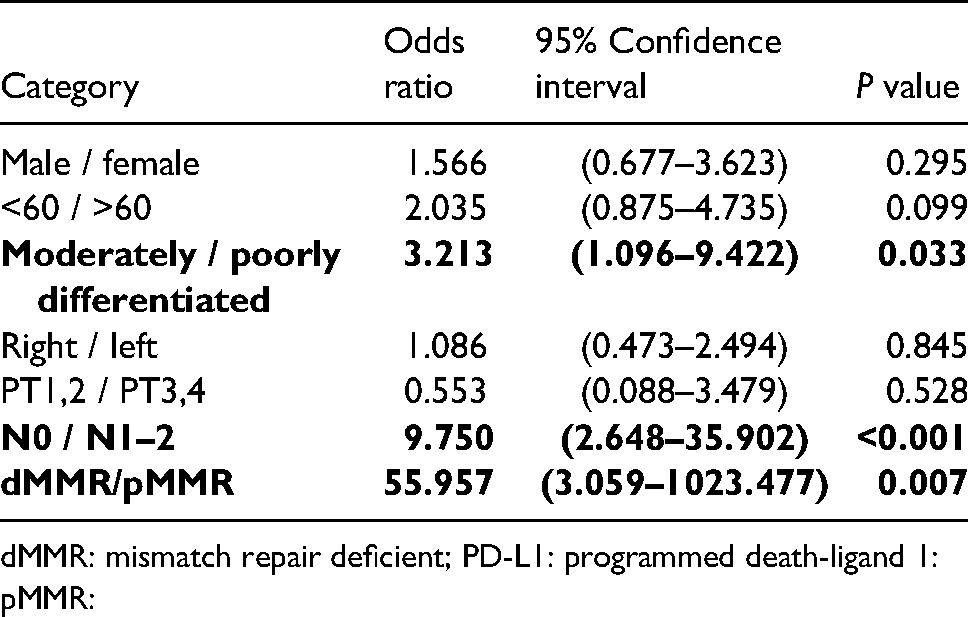
dMMR: mismatch repair deficient; PD-L1: programmed death-ligand 1: pMMR:
MMR status (MSH2, MSH6, PMS2, and MLH1) was available for only 50% of the cases (46 cases). Of these, 30 (65.2%) were pMMR tumors and 16 (34.7%) were dMMR CRC. PD-L1 expression was positive in all dMMR cases, while only 11 cases out of 30 pMMR cases (23.9%) displayed positive PD-L1. The association between PD-L1 expression and MMR status was significant (P<0.001).
Discussion
In the current decade, the world of oncology has dramatically changed with the introduction of checkpoint inhibitors and immunotherapy. Anti PD-1/anti PD-L1 are considered amongst the most important immunotherapies currently available and approved by the American FDA. 16 This made the identification and validation of prognostic and predictive biomarkers in the treatment of tumors of vital importance, especially since this can help in selecting patients who are most likely to benefit from a given therapy while sparing probable toxicity in those unlikely to benefit. 17
In CRC, there are many risk factors and genetic alterations that are associated with its tumorigenesis including viral and bacterial infections, alcohol, tobacco use, aging, ulcerative colitis, sedentary lifestyle and—most importantly—genetic instabilities, including chromosomal and MSI.18-20
PD-L1 is an important molecule in the tumor microenvironment, and its up-regulation is one of the key mechanisms of tumor immune evasion in CRC and many other malignancies. It plays a crucial role in the immune response and a key role in the body's antitumor immune responses. Using drugs to block the PD-1/PD-L1 pathway enhances the immune system and leads to TC lysis, thus opening a new avenue for cancer treatment. However, the role and prognostic value of PD-1/PD-L1 overexpression in regulating CRC has not been entirely clarified, and research discussing the relationship is still limited compared to other widely studied tumor types.9-13,21-30
In a meta-analysis, Wu et al. 21 found that PD-L1 overexpression was correlated with poor 5-year overall survival in CRC. In another meta-analysis, Pyo et al. 22 found a worse disease-specific survival association with PD-L1 expression in CRC. Similar results of worse overall survival was found in a third meta-analysis by Shen et al. 23 They also found the overexpression of PD-L1 to be associated with lymphatic invasion and tumor stage, but not gender, MSI, or tumor differentiation. Enkhbart et al. 9 found that overall survival rates were significantly poorer in PD-1 and PD-L1-positive CRC. Lee et al. 10 found PD-L1 that positivity in TC and negativity in tumor-infiltrating lymphocytes were independent predictors of poorer overall and disease-free survival in patients with CRC.
In contrast, in another meta-analysis, Xiang et al. 28 found no relationship between PD-L1 expression and prognosis in CRC; Kim et al. 29 found the same in their study on the subject. However, a study by Droeser et al. 13 found better prognosis and improved survival in CRC expressing PD-L1. Additionally, Li et al. 12 found a better overall and disease-free survival in CRC with over expression of PD-L1 in both TC and immune cells (IC). However, it should be noted that in all previously cited studies there was no unified or standardized methodology to assess the PD-L1 expression where different antibody clones, testing platforms, and scoring algorithms were used. For example, in one of the discordant studies mentioned above 9 the authors used clone ab174838 (1:100; Abcam, Cambridge, UK) employing manual methods rather than an automated platform, and scored cytoplasmic staining of TC in a scoring algorithm combining both the intensity and proportion of TC. This approach has not been used in published clinical trials, and TC scoring is based on membranous staining while cytoplasmic staining is usually disregarded. In another contradictory study by Lee et al. 10 three different assays (MIH1 clone antibody on a Ventana Benchmark platform, E1L3N clone antibody also on Ventana Benchmark platform, and a 22C3 clone antibody on a Dako Autostainer Link 48 platform) were used, which showed discrepancies between the different antibody clones and assays and, as previously mentioned, if scoring included TC only PD-L1 was associated with poor prognosis. However, in contrast, if only IC were scored PD-L1 was associated with better prognosis, which is similar to our results in that our scoring was largely based on IC. Therefore, one should pay attention when comparing studies as scoring in some of the studies was based on TC staining and in others on IC or both. Current guidelines for PD-L1 testing take into account IC staining in most adopted algorithms. The National Comprehensive Cancer Network guidelines in the United States as a rule recommend using the FDA-approved companion diagnostic tests for PD-L1 for all FDA-approved immunotherapy drugs. The majority of the companion diagnostic tests include IC as part of the scoring algorithms except for some of the non-small cell lung cancer (NSCLC) assays. The most common medication that has companion diagnostic testing is pembrolizumab, which requires CPS scoring for all indicated cancer types except for NSCLC that requires only tumor proportion scoring.
In the current study, we focused on evaluating PD-L1 expression in CRC as a biomarker for anti-PD-1 or anti-PD-L1 immunotherapy; we also investigated its prognostic significance and its possible correlation with clinicopathological factors.
PD-L1 was expressed (CPS ≥1) in 49 (53.8%) of the cases. This expression was significantly associated with lower nodal stage (P<0.001) and better differentiation (P = 0.03), despite the fact that the presence of metastasis showed no significant relationship with PD-L1 expression. This may be explained by the low number of patients (22) in whom metastasis information was available. Rosenbaum et al. 24 also found an association between lower tumor stage and PD-L1 expression.
Our results also showed that dMMR cases were significantly correlated with a positive expression of PD-L1 (P = < 0.001). We also found that the expression of PD-L1 was significantly associated with the mucinous histology subtype (P = .020).
Regarding the MMR status and PD-L1, Rosenbaum et al. 24 also found PD-L1 expression to be associated with microsatellite instability-high (MSI-H) CRC, which was similar to our results. Additionally, Siraj et al. 27 and Kim et al. 29 found an association between PD-L1 expression and dMMR. The association of PD-L1 expression with dMMR/MSI-H status was documented in a review by Zhao et al. 31
In addition, Droeser et al., 13 who studied PD-L1 expression in the two subtypes of CRCs (including 1197 pMMR and 223 dMMR-deficient CRCs) identified strong PD-L1 expression in 37% of pMMR and in 29% of dMMR CRCs. In their hands, strong PD-L1 expression in pMMR CRC was significantly associated with early T stage, absence of lymph node metastases, lower tumor grade, absence of vascular invasion, and significantly improved survival. These results are similar to ours in most of the mentioned variables except for the T stage, which was not significant in our hands. This may be due to the small number of T1/T2 cases in our cohort (only 5 cases).
It is clear from the presented literature and the studies reviewed so far that the prognostic role of positive PD-L1 expression in CRC is controversial. The lack of standardization and unified scoring algorithms that account for the IC in the microenvironment may be the reason for this discrepancy.
Considering specific populations similar to ours, a recently published study in patients from the Middle East found high PD-L1 expression in 37.3% of cases and was correlated with aggressive clinicopathological features such as high grade, larger tumor size, “mucinous histology,” and worst survival. They also found PD-L1 expression to be significantly higher in patients exhibiting dMMR and BRAF mutations. Their findings in terms of association with dMMR are similar to ours; however, their other findings are not, but this may be explained by the fact that they only scored PD-L1 in TC and didn't include the IC in the tumor microenvironment as we did. 27 Apart from NSCLC, all other solid tumors including gastrointestinal malignancies are currently scored by algorithms that account for the IC.
Ample evidence supports PD-L1, tumor mutational burden, number of tumor-infiltrating lymphocytes (TILs), peripheral blood lymphocyte count, dMMR, and MSI-H as predictive biomarkers that guide the clinical application of immune checkpoint blockade (ICB) therapies.30,31
Tumors with dMMR or MSI-H are more sensitive to ICB, particularly to PD-1 and PD-L1 inhibitors. This has led the FDA to approve the indication of ICB for all dMMR/MSI-H solid tumors regardless of the histology, type, or location (i.e., tumor agnostic).16,30,31
dMMR/MSI-H represents a good prognosis in early CRC settings without adjuvant treatment and a poor prognosis in patients with metastasis. However, several clinical trials have demonstrated that dMMR or MSI-H are significantly associated with long-term immunotherapy-related responses and better prognosis in CRC and non-colorectal malignancies treated with immune checkpoint inhibitors. 30
Our study suggests that high PD-L1 expression in tumor tissue is associated with factors of good prognosis in patients with CRC as it is significantly associated with better differentiation, dMMR, mucinous histology, and negative nodal status. Our data therefore suggest that PD-L1 expression could be utilized as a prognostic factor of CRC, with PD-L1 being correlated with a favorable prognosis.
However, one of the important limitations in our study is the small number of cases included. We recommend future studies with a much larger cohort of patients so that our results can be confirmed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
