Abstract
Purpose
The aim of this study was to elucidate the rates and prognostic roles of programmed cell death ligand 1 (PD-L1) immunohistochemical (IHC) expression in various malignant tumors through a systematic review and meta-analysis.
Method
The current study included 16,176 patients from 97 eligible studies. We investigated PD-L1 expression and its correlation with survival rate in various malignant tumors.
Results
The estimated rate of PD-L1 IHC expression was 0.449 (95% confidence interval [CI] 0.404-0.495). The highest and lowest PD-L1 expression levels were found in thyroid cancer (0.829, 95% CI 0.781-0.868) and small-cell neuroendocrine carcinoma (0.005, 95% CI 0.000-0.080), respectively. PD-L1 expression was significantly correlated with poorer overall survival and disease-free survival rates (hazard ratios 1.276, 95% CI 1.097-1.486 and 1.304, 95% CI 1.034-1.644, respectively). However, PD-L1 IHC expression was significantly correlated with worse overall survival rates in patients with esophageal cancer and renal cell carcinoma and with worse disease-free survival rates in patients with colorectal cancer, hepatocellular carcinoma, and renal cell carcinoma.
Conclusions
Our results show that PD-L1 expression rates and the correlations with survival varied between tumor types. Detailed evaluation criteria for PD-L1 will have to be standardized before application to specific tumor types.
Introduction
Programmed cell death ligand 1 (PD-L1), also known as CD274 or B7-H1, is a member of the B7 superfamily (1). PD-L1 is expressed by macrophages and dendritic cells and binds to PD-1 receptors. PD-L1 regulates excessive adoptive immune responses against antigens and autoimmunity. That is, PD-L1 is a negative regulator of the immune response during T-cell receptor activation. Several studies have reported that targeted therapies using a monoclonal antibody against PD-1/PD-L1 result in survival gains in patients with various malignant tumors (2). However, the correlation between the expression of PD-L1 measured by immunohistochemistry (IHC) and the therapeutic effects and prognostic role of PD-L1 expression have not been fully elucidated. PD-L1 IHC expression is significantly correlated with a worse survival rate in renal cell carcinoma (3–7). However, the prognostic role of PD-L1 IHC expression is controversial in some malignant tumors (8). Because studies used different antibody clones, IHC staining methods, and evaluation criteria, it may be difficult to obtain conclusive information from previous studies using PD-L1 IHC.
The current meta-analysis was performed to investigate the PD-L1 IHC expression rates in various malignant tumors. In addition, the correlation between PD-L1 expression and survival was investigated to confirm the prognostic roles of PD-L1 in malignant tumors.
Materials and methods
Published study search and selection criteria
Relevant articles were obtained by searching the PubMed and MEDLINE databases through March 31, 2016. The search was performed using the following keywords: “programmed death ligand 1 or PD-L1” and “immunohistochemistry.” The titles and abstracts of all returned articles were screened for exclusion. Review articles were also screened to find additional eligible studies. The search results were scanned according to the following inclusion and exclusion criteria: 1) PD-L1 IHC expression investigated in human malignant tumors; 2) case reports were excluded; and 3) all were English-language publications.
Data extraction
The following information was collected from the full texts of eligible studies and was verified (1, 3–46): first author's name, publication date, study location, number of patients analyzed, antibody manufacturer, dilution ratio, cutoffs for assessing PD-L1 IHC expression, tumor types, and data allowing estimates of the effects of PD-L1 IHC expression on overall survival (OS) and/or disease-free survival (DFS). We did not define a minimal number of patients to be included in a study. Any disagreements were resolved by consensus.
Statistical analysis
All data were analyzed using the Comprehensive Meta-Analysis software package (Biostat). The PD-L1 IHC expression rates of various malignant tumors were investigated, and meta-analysis was performed. The correlations between PD-L1 IHC expression and survival were measured by hazard ratios (HRs) obtained from data in the eligible studies. We aggregated the estimated HR and its standard error using given parameters, which were the HR point estimate, the log-rank statistic or its p value, the O–E statistic (difference between numbers of observed and expected events), or its variance (47). If the extractable data only included a survival curve, 2 researchers independently extracted the survival rates to reduce reading variability according to Parmar's recommendation (47). Although meta-analyses were performed using fixed-effects and random-effects models in the current study, the values were pooled using a random-effects model for interpretation. Because the eligible studies used various antibodies and evaluation criteria, a random-effects model was more suitable than a fixed-effects model. Subsequently, a study showing results of an estimated HR >1 without an overlapping 95% confidence interval (CI) suggested worse survival with PD-L1 IHC expression. Subgroup analyses based on specific tumors were performed. In addition, heterogeneous and sensitivity analyses were conducted to assess the heterogeneity of eligible studies and the impact of each study on the combined effect, respectively. Heterogeneity between studies was checked by the Q and I2 statistics and p values. Begg's funnel plot and Egger's test were performed to assess publication bias. If significant publication bias was found, the fail-safe N and trim-fill tests were additionally conducted to confirm the degree of publication bias. A p value <0.05 was considered significant.
Results
Selection and characteristics of the studies
A total of 376 reports were identified in the database search. Of these, 84 were excluded due to a lack of sufficient information. In addition, 195 reports were excluded for the following reasons: focus on other diseases (119 reports), non-human studies (33 reports), non-original articles (18 reports), duplicates (2 reports), and articles in a language other than English (23 reports) (Fig. 1). After applying the inclusion and exclusion criteria, 97 studies were selected for the meta-analysis (1, 3–46). The results regarding the most relevant studies are presented in the main section of this article, while the remaining results are shown in the supplementary materials. Eligible studies used antibodies with various manufacturers and dilution ratios, as shown in Table I and supplementary Table S1 (Tab. S1 - Main characteristics of the eligible studies. Available online at www.biological-markers.com).
Main characteristics of the eligible studies
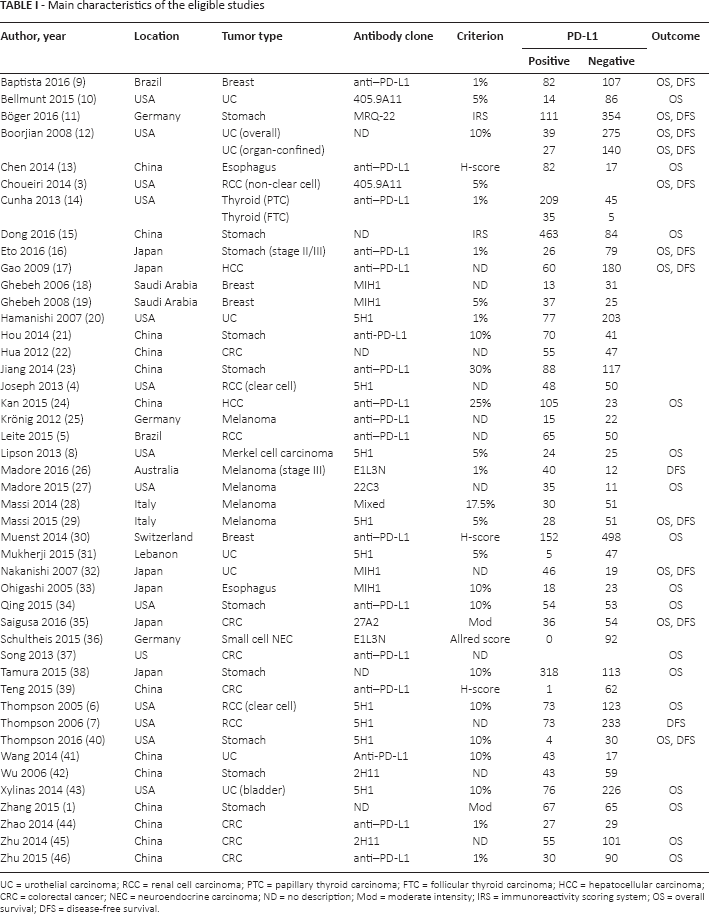
UC = urothelial carcinoma; RCC = renal cell carcinoma; PTC = papillary thyroid carcinoma; FTC = follicular thyroid carcinoma; HCC = hepatocellular carcinoma; CRC = colorectal cancer; NEC = neuroendocrine carcinoma; ND = no description; Mod = moderate intensity; IRS = immunoreactivity scoring system; OS = overall survival; DFS = disease-free survival.

Flowchart for study search and selection methods.
PD-L1 IHC expression rates in various malignant tumors
The estimated rate of PD-L1 IHC expression was 0.448 (95% CI 0.402-0.494; range 0.107-0.864). The PD-L1 IHC expression rates of hepatocellular carcinoma, colorectal, esophageal, and stomach cancers were 0.551 (95% CI 0.086-0.941), 0.354 (95% CI 0.247-0.478), 0.565 (95% CI 0.256-0.831), and 0.453 (95% CI 0.323-0.589), respectively (Tab. II), and those of renal cell carcinoma and urothelial carcinoma were 0.407 (95% CI 0.2 68-0.563) and 0.274 (95% CI 0.164-0.420), respectively. The rates of PD-L1 IHC expression by breast and thyroid cancers were 0.419 (95% CI 0.267-0.587) and 0.829 (95% CI 0.781-0.868), respectively. The PD-L1 IHC expression rates of non-small cell lung cancer (NSCLC), ovarian and testicular tumors were 0.422 (95% CI 0.327-0.523), 0.555 (95% CI 0.197-0.865) and 0.583 (95% CI 0.346-0.787), respectively (supplementary Tab. S2 - PD-L1 expression rates of various malignant tumors. Available online at www.biological-markers.com).
PD-L1 expression rates of various malignant tumors
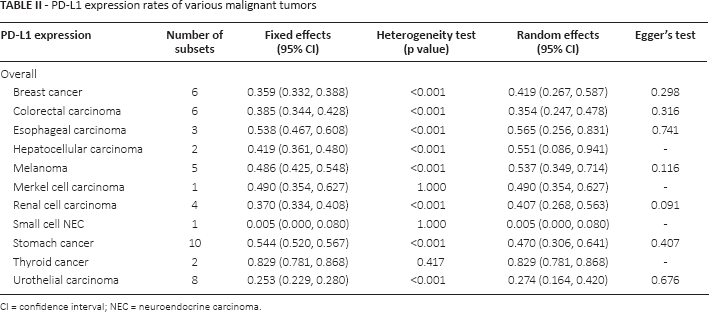
CI = confidence interval; NEC = neuroendocrine carcinoma.
Correlation between PD-L1 expression and survival
PD-L1 expression was significantly correlated with a worse OS rate (HR 1.276, 95% CI 1.097-1.486). The HRs were 2.351 (95% CI 1.553-3.558), 1.353 (95% CI 1.005-1.822), and 2.716 (95% CI 1.583-4.660) for esophageal cancer, mesothelioma, and renal cell carcinoma, respectively (Tab. III). However, PD-L1 expression and worse OS were not correlated in patients with some malignant tumors, such as adrenocortical carcinoma, breast cancer, colorectal cancer, glioblastoma, hepatocellular carcinoma, head and neck squamous cell carcinoma, melanoma, stomach cancer, thymic tumor, testicular tumor, urothelial carcinoma, or uterine cervical cancer (Tab. III and supplementary Tab. S3 - Overall survival rates according to the PD-L1 expression. Available online at www.biological-markers.com). However, PD-L1 expression was significantly correlated with a better OS in patients with Merkel cell carcinoma, (HR 0.448, 95% CI 0.204-0.982).
Overall survival rates according to PD-L1 expression
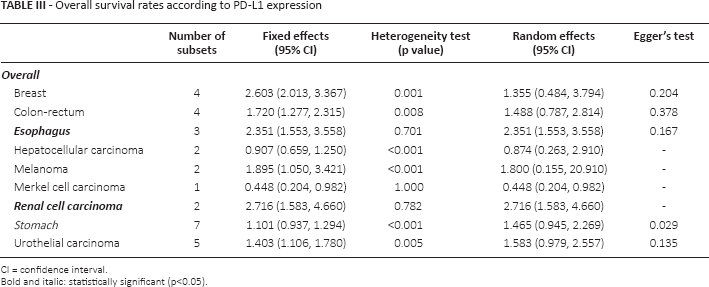
CI = confidence interval.
Bold and italic: statistically significant (p<0.05).
Next, we evaluated the correlation between PD-L1 expression and DFS in various malignant tumors. PD-L1 IHC expression was significantly correlated with a worse DFS rate (HR 1.304, 95% CI 1.034-1.644). Significant correlations were observed between PD-L1 IHC expression and worse DFS rate in patients with colorectal cancer (HR 2.856, 95% CI 1.141-7.149), hepatocellular carcinoma (HR 1.790, 95% CI 1.183-2.708), renal cell carcinoma (HR 2.360, 95% CI 1.280-4.354), testicular tumor (HR 2.251, 95% CI 1.113-4.551), and thymic carcinoma (HR 3.694, 95% CI 1.436-9.500) (Tab. IV and supplementary Tab. S4 - Disease-free survival rates according to the PD-L1 expression. Available online at www.biological-markers.com). Although some malignant tumors, such as head and neck squamous cell carcinoma, melanoma, nasopharyngeal carcinoma, stomach cancer, and urothelial carcinoma, showed HRs >1, no statistical significance was found. However, PD-L1 expression was significantly correlated with better DFS in patients with ovarian cancer (HR 0.410, 95% CI 0.233-0.720; supplementary Tab. S4).
Disease-free survival rates according to PD-L1 expression
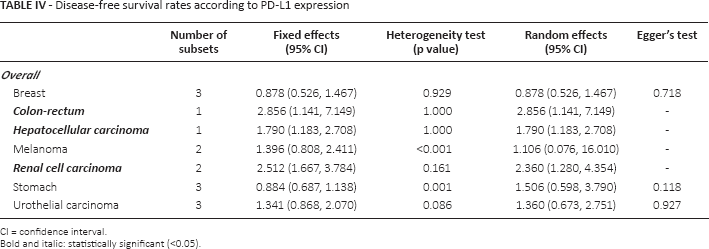
CI = confidence interval.
Bold and italic: statistically significant (<0.05).
Discussion
Targeted therapies using immune checkpoint inhibitors have been studied intensively in various malignant tumors. Although studies on the expression and prognostic role of PD-L1 IHC have been reported, no conclusive information is available. The current work is the first meta-analysis of published studies on the expression and prognostic roles of PD-L1 IHC in various malignant tumors.
PD-L1 is expressed on macrophages and dendritic cells as well as tumor cells. PD-1 expression activates T cells and B cells, and PD-L1 binding to the PD-1 receptor suppresses T-cell migration, proliferation, and secretion of cytotoxic mediators. PD-L1 expression can be induced in malignant tumors by interferon gamma secreted from surrounding CD8+ T cells and oncogenic signaling within tumor cells (40). Several clinical studies have reported that PD-L1-overexpressing NSCLC shows higher overall response rates to anti-PD-1/PD-L1 therapies (48). PD-L1 IHC expression is useful to estimate overall response rates using a 1% cutoff point, and is associated with improved survival (49). However, the correlations between PD-L1 IHC expression and survival vary depending on the tumor type (1, 3–46). To obtain conclusive information for this correlation, further studies and a meta-analysis will be needed.
This meta-analysis included studies that used IHC to evaluate PD-L1 expression. The rates of PD-L1 IHC expression were 10.7%-86.4% and varied according to tumor type, but the reported results of previous studies are discrepant. This discrepancy between studies may be caused by different PD-L1 antibody clones, various IHC methods, or population differences. In addition, various cutoff values (1%-50%) to interpret the IHC results were used in the eligible studies, and these results can be largely affected by the cutoff values. Because PD-L1 may be expressed in tumor cells as well as intratumoral inflammatory cells, the discrepancy between studies may be due to the rates of PD-L1 IHC expression. Consequently, this discrepancy might affect the rate of PD-L1 expression by specific tumors and the correlation between PD-L1 expression and survival. A subgroup analysis based on cutoff values and tumor type would help confirm the causes of the discrepancy. However, it is difficult to perform a subgroup analysis due to insufficient information on specific tumors.
In the current meta-analysis, PD-L1 expression was significantly correlated with worse OS and DFS in human malignant tumors (HR 1.276, 95% CI 1.097-1.486 and HR 1.304, 95% CI 1.034-1.644, respectively). Unlike the overall results of eligible studies, inverse correlations or no correlations were found for some malignant tumors. Among the eligible studies, NSCLC was the most frequently studied tumor. The range of PD-L1 IHC expression rates by NSCLC was 0.074-0.902. A subgroup analysis based on NSCLC subtype could not be performed due to insufficient information. In the current analysis, PD-L1 IHC expression was not correlated with worse OS and DFS rates in patients with NSCLC (HR 1.035, 95% CI 0.811-1.321 and HR 0.969, 95% CI 0.688-1.366, respectively; supplementary Tabs. S3 and S4). In some reports, PD-L1 IHC expression correlated significantly with better OS. Patients with NSCLC who expressed PD-L1 showed a higher overall response rate against anti-PD-1/PD-L1 monoclonal antibody therapy than those with no PD-L1 IHC expression (48). However, cases that did not express PD-L1 showed an overall response rate of 8%-20% against anti-PD-1/PD-L1 monoclonal antibody therapy. Therefore, PD-L1 IHC expression may not be the only, exclusive parameter to predict the response against anti-PD-1/PD-L1 monoclonal antibody therapy. Indeed, further cumulative studies are needed to confirm the prognostic role of PD-L1 IHC expression in patients with NSCLC. No association was found between PD-L1 IHC expression and OS in patients with colorectal carcinoma, hepatocellular carcinoma, melanoma, testicular tumors, or thymic tumors. The estimated rate of PD-L1 IHC expression in renal cell carcinoma was 0.407 (95% CI 0.268-0.563). PD-L1 IHC expression was significantly correlated with worse OS and DFS in patients with renal cell carcinoma.
A number of limitations of the current study should be mentioned. First, some tumor types had only 1 report, thus limiting the data for adrenocortical carcinoma, glioblastoma, testicular, and uterine cervical cancer (supplementary Tabs. S3 and S4). Second, the eligible studies used various evaluation criteria for PD-L1 IHC expression. However, we could not investigate the discrepancy according to the evaluation criteria due to insufficient information. Third, PD-L1 can be detected in tumor cells, tumor-infiltrating lymphocytes, and stromal inflammatory cells. However, further evaluation, such as a subgroup analysis based on expression pattern, could not performed due to insufficient information. Fourth, we extracted survival data at the 60-month follow-up point in eligible studies with only survival curves to avoid bias due to the follow-up period. However, the correlation between PD-L1 IHC expression and survival may have differed from those in the eligible studies.
In conclusion, PD-L1 IHC expression varied according to tumor type. Although PD-L1 IHC expression was significantly correlated with worse OS and DFS rates, some tumors showed an inverse correlation. This heterogeneity in the results is likely related to the different methodological approaches to evaluate PD-L1 in each tumor type. Standardized criteria of PD-L1 IHC expression in various malignant tumors are needed to apply PD-L1 IHC expression to predicting prognosis.
Footnotes
Financial support: This study was supported by a 2013 Inje University research grant.
Conflict of interest: The authors declare that they have no conflict of interest.
