Abstract
Purpose:
The present study aimed to evaluate the diagnostic roles of CD56 immunohistochemistry in differentiating various thyroid lesions.
Methods:
A meta-analysis was performed to evaluate the rate of loss of CD56 immunohistochemistry expression from 13 eligible studies regarding various thyroid lesions, including papillary thyroid carcinoma, follicular carcinoma, and follicular adenoma. To confirm the value of CD56 immunohistochemistry in differentiating various thyroid lesions, a diagnostic test accuracy review was conducted.
Results:
An 87.8%, 79.1%, 11.9%, 25.5%, and 19.6% loss of CD56 immunohistochemistry expression was identified in papillary thyroid carcinoma, follicular carcinoma, follicular adenoma, benign follicular nodule, and Hashimoto’s thyroiditis, respectively. In the normal thyroid tissue, the rate of loss of CD56 expression was 1.6%. Classical, follicular, diffuse sclerosing, tall cell, and encapsulated variants of papillary thyroid carcinoma showed an 88.4%, 75.3%, 97.2%, 91.7%, and 91.7% loss of CD56 expression, respectively. In the comparison between the follicular variant of papillary thyroid carcinoma and follicular adenoma, the pooled sensitivity and specificity of CD56 immunohistochemistry was 0.82 (95% confidence interval (CI) 0.70, 0.90) and 0.94 (95% CI 0.83, 0.99), respectively. The diagnostic odds ratio and the area under curve on summary receiver operating characteristic curve was 51.43 (95% CI 5.83, 453.88) and 0.9387, respectively.
Conclusion:
Collectively, these results indicate that the rate of loss of CD56 immunohistochemistry expression was significantly higher in malignant tumors, such as papillary thyroid carcinoma and follicular carcinoma, than in follicular adenoma, benign follicular nodule, and Hashimoto’s thyroiditis. As such, CD56 immunohistochemistry can be useful in differentiating follicular variant papillary thyroid carcinoma from follicular adenoma.
Introduction
Thyroid cancer is the most common malignant tumor in the endocrine system, and papillary thyroid carcinoma (PTC) accounts for at least 80% of thyroid cancer cases (1). Sometimes, differentiating malignant from benign thyroid lesions through histologic analysis of the primary tumor may be difficult. Ancillary tests, such as immunohistochemistry (IHC), can be helpful in distinguishing differentiating thyroid lesions. Recently, various immunohistochemical markers, such as HBME-1, galectin-3, CK19, and CD56, have been introduced in the diagnosis and differentiation of thyroid lesions (2). However, definitive markers or panels for thyroid lesions are yet to be determined. CD56, which is expressed in neurons, glial cells, and various epithelial cells, is a neural cell adhesion molecule involved in cell-to-cell adhesion (3, 4). CD56 is also expressed in the membrane of the normal thyroid follicular cells and is downregulated in PTC (4-12). The loss of CD56 expression is associated with lymphangiogenesis and metastasis, and eventually worse prognosis in colon and pancreatic cancers (6, 8-10). However, the diagnostic role of CD56 in malignant thyroid tumor is not elucidated.
The present study aimed to determine the diagnostic roles of CD56 IHC in differentiating various thyroid lesions. We investigated the rate of loss of CD56 IHC expression in thyroid lesions, including PTC, follicular carcinoma (FC), follicular adenoma (FA), benign follicular nodule (BFN), Hashimoto’s thyroiditis, and normal thyroid tissue, and performed a meta-analysis. In addition, a diagnostic test accuracy review was conducted to confirm the role of CD56 IHC as a single diagnostic marker in differentiating thyroid lesions.
Materials and methods
Published study search and selection criteria
Relevant articles were identified through a search of PubMed and MEDLINE databases through 30 April 2017 using the key words “thyroid,” “CD56,” and “immunohistochemistry.” The titles and abstracts of the articles were screened for exclusion. Review articles were further screened to find additional eligible studies. Searched results were then reviewed and included if (a) the study was performed in human thyroid tissue; and (b) information regarding CD56 IHC expression in various thyroid lesions is available. Studies were excluded if they were (a) case reports or non-original articles; or (b) non-English language publications.
Data extraction
Data from all eligible studies were extracted by two independent authors. The following data were extracted from each of the eligible studies (2-4, 13-22): the first author’s name, year of publication, study location, antibody clone and manufacturer, antibody dilution ratio and cut-off value, and number of patients analyzed. In addition, the included variants of PTC were investigated. For the meta-analysis, we extracted all data associated with IHC results.
Statistical analysis
To perform the meta-analysis, data were analyzed using the Comprehensive Meta-Analysis software package (Biostat, Englewood, NJ, USA). The rates of loss of CD56 IHC expression in various thyroid lesions were investigated for the meta-analysis. The subgroup analysis was performed according to the PTC variant and cut-off value for the loss of CD56 expression. Heterogeneity between studies was checked using the Q and I2 statistics and presented using P-values. Additionally, a sensitivity analysis was conducted to assess the heterogeneity of eligible studies and the impact of each study on the combined effect. To define the significant difference between two subgroups, a meta-regression test was performed, and the result was presented using P-values. Begg’s funnel plot and Egger’s test were performed to assess publication bias. When a significant publication bias was found, the fail-safe N and trim-fill tests were additionally conducted to confirm the degree of publication bias. The results were considered statistically significant when P < 0.05.
The diagnostic test accuracy review was analyzed using the Meta-Disc program (version 1.4, unit of Clinical Biostatics, the Ramon y Cajal Hospital, Madrid, Spain) (23). The summary receiver operating characteristic (SROC) curve was initially constructed by plotting “sensitivity” and “1-specificity” of each study, and the curve fitting was performed through linear regression using the Moses–Littenberg linear model (24). Because heterogeneity in the evaluation criteria was present, the accuracy data were pooled by fitting a SROC curve and measuring the value of the area under the curve (AUC) (23). An AUC close to 1 was regarded a perfect test, and an AUC close to 0.5 was considered a poor test. In addition, the diagnostic odds ratio (OR) was calculated via the Meta-Disc program.
Results
Study selection and characteristics
A total of 85 reports were identified in the database search, of which 72 were excluded because they focused on other diseases (n = 53), had insufficient information for CD56 IHC expression (n = 13), and were non-original articles (n = 4) and duplicated reports (n = 2). Finally, the meta-analysis included 13 eligible studies. The 13 studies comprised 1234 thyroid cases; namely, PTC (n = 1043), FC (n = 30), FA (n = 109), and BFN (n = 23) (Fig. 1 and Tab. I).
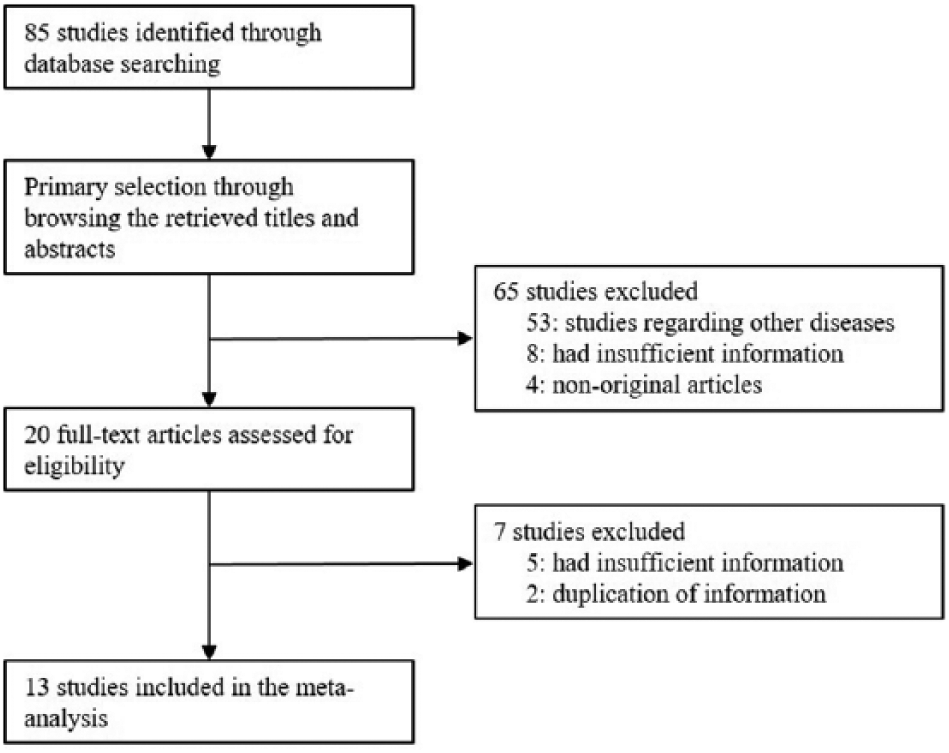
Flow chart of the study search and selection methods.
Main characteristics of the eligible studies
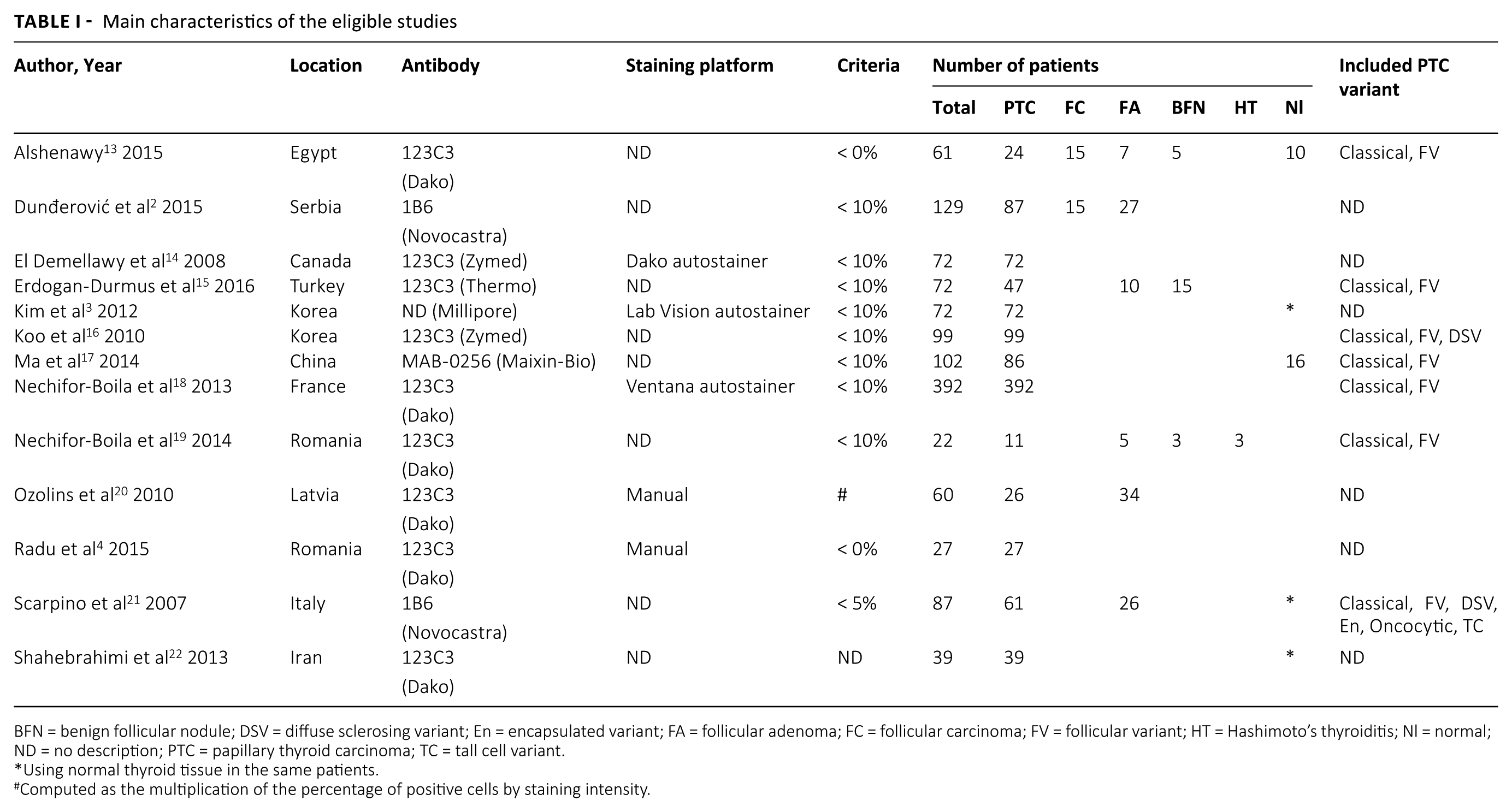
BFN = benign follicular nodule; DSV = diffuse sclerosing variant; En = encapsulated variant; FA = follicular adenoma; FC = follicular carcinoma; FV = follicular variant; HT = Hashimoto’s thyroiditis; Nl = normal; ND = no description; PTC = papillary thyroid carcinoma; TC = tall cell variant.
Using normal thyroid tissue in the same patients.
Computed as the multiplication of the percentage of positive cells by staining intensity.
Loss of CD56 expression in thyroid lesions
The rate of loss of CD56 expression in normal thyroid tissue is 0.016 (95% confidence interval (CI) 0.004, 0.063) (Tab. II). Meanwhile, the estimated rates of loss of CD56 expression were 0.878 (95% CI 0.815, 0.921) and 0.791 (95% CI 0.604, 0.904) in PTC and FC, respectively. In the subgroup analysis based on the PTC variant, the loss of CD56 expression in the follicular variant of PTC (FVPTC) (0.753; 95% CI 0.658, 0.829) was lower than that of other variants, such as classical (0.884; 95% CI 0.766, 0.946), diffuse sclerosing (0.972; 95% CI 0.799, 0.997), tall cell (0.917; 95% CI 0.378, 0.9950.917; 95% CI 0.378, 0.995), and oncocytic (0.929; 95% CI 0.423, 0.996). In FA, the rate of loss of CD56 expression was 0.119 (95% CI 0.050, 0.259). A significant publication bias was observed via primary tests, Begg’s funnel plot, and Egger’s test (P = 0.019) in PTC and normal thyroid tissue. However, in the secondary assessment of publication bias—including fail-safe N and trim-fill tests—no significant publication bias was noted.
Meta-analysis for the rate of loss of CD56 immunohistochemical expression in various thyroid lesions
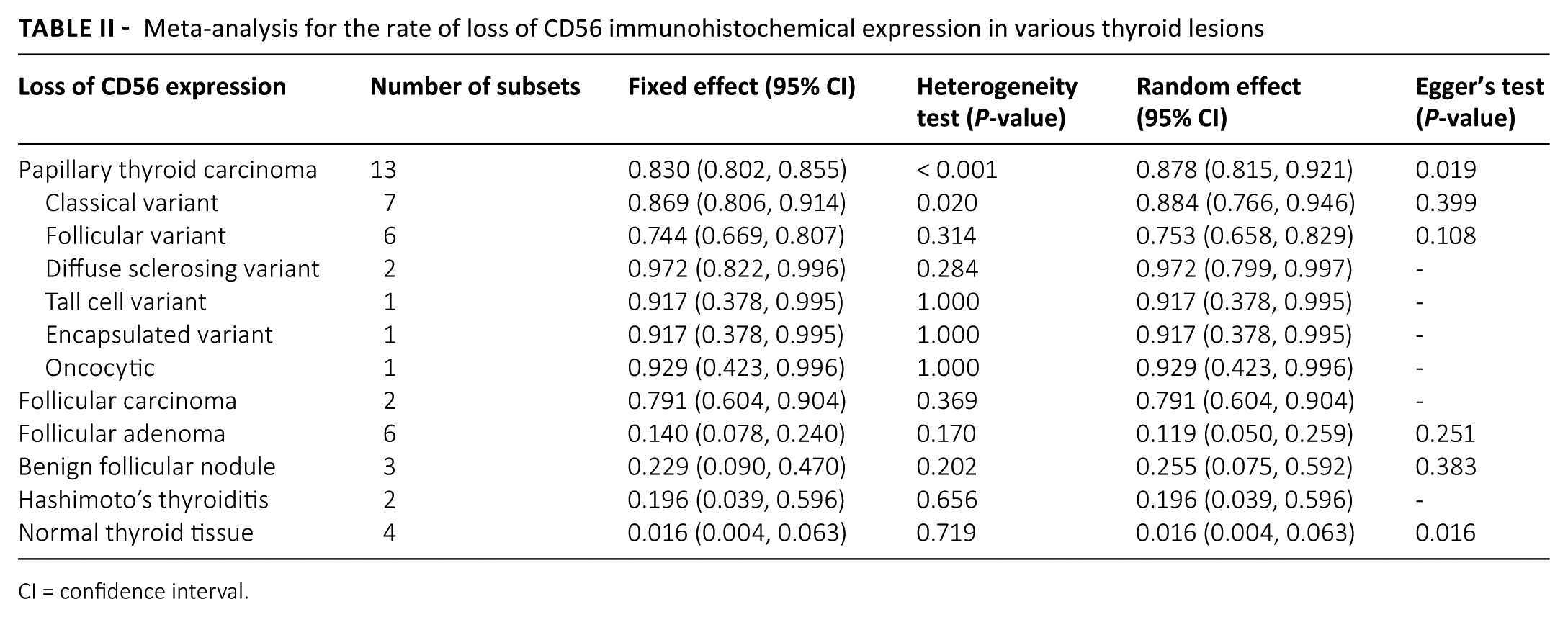
CI = confidence interval.
To confirm the optimal cut-off value of loss of CD56 expression, we performed a subgroup analysis dividing into 10% and 0% subgroups. The rate of loss of CD56 expression was lower in the low cut-off (0%) subgroup than that in the high cut-off (10%) subgroup in PTCs (Tab. III). However, in meta-regression tests, no significant difference was observed between high and low cut-off subgroups in various thyroid lesions and normal thyroid tissue.
Subgroup analysis and meta-regression test based on cut-off value for the rate of loss of CD56 immunohistochemical expression in various thyroid lesions
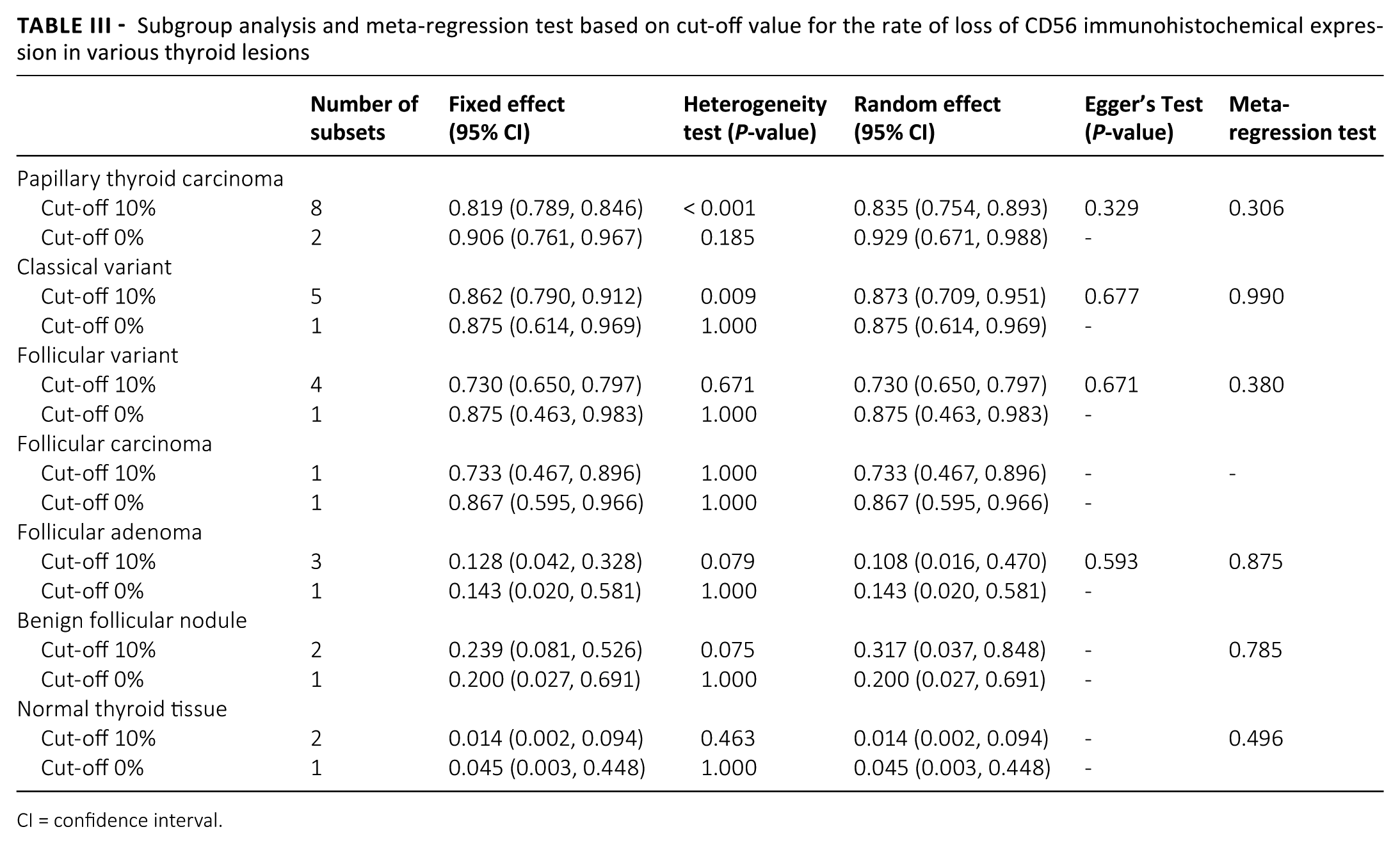
CI = confidence interval.
Diagnostic test accuracy review of CD56 immunohistochemistry
To determine the diagnostic role of CD56 expression in various thyroid lesions, a diagnostic test accuracy review was performed. In a comparison between PTC and FA, the pooled sensitivity and specificity were 0.32 (95% CI 0.28, 0.36) and 0.89 (95% CI 0.80, 0.95), respectively (Tab. IV). The diagnostic OR and the value of AUC on the SROC curve were 48.33 (95% CI 4.49, 520.62) and 0.9410. In differentiation of FVPTC from FA, the pooled sensitivity and specificity were 0.82 (95% CI 0.70, 0.90) and 0.94 (95% CI 0.83, 0.99), respectively. The diagnostic OR and the value of AUC on the SROC curve were 51.43 (95% CI 5.83, 453.88) and 0.9387. Compared with the normal thyroid tissue, the loss of CD56 expression had a significant diagnostic accuracy in PTC. In addition, in a comparison between FC and FA, the pooled sensitivity and specificity were 0.81 (95% CI 0.63, 0.93) and 0.79 (95% CI 0.66, 0.88), respectively. The diagnostic OR and the value of AUC on the SROC curve were 29.78 (95% CI 5.98, 148.25) and 0.8792, respectively.
Diagnostic test accuracy review of CD56 immunohistochemistry in thyroid tumors
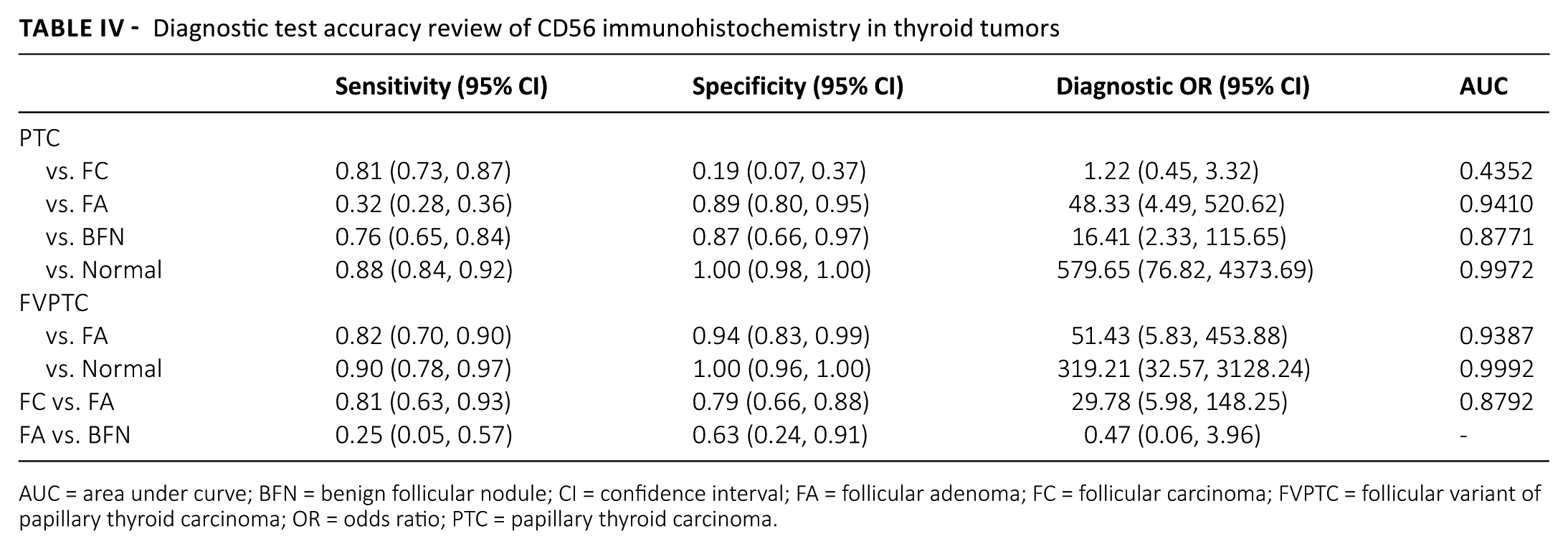
AUC = area under curve; BFN = benign follicular nodule; CI = confidence interval; FA = follicular adenoma; FC = follicular carcinoma; FVPTC = follicular variant of papillary thyroid carcinoma; OR = odds ratio; PTC = papillary thyroid carcinoma.
Discussion
Recently, the diagnostic value of various single markers or IHC panels in differentiating follicular cell-derived thyroid lesions has been introduced (2). However, definitive information for the diagnostic accuracy of these IHC markers is not fully elucidated. The present study is the first meta-analysis and diagnostic test accuracy review of previously published studies regarding the diagnostic accuracy of CD56 as a single IHC marker in various thyroid lesions.
Many studies have been reported for various panels for diagnosing and differentiating thyroid lesions using IHC markers, such as HBME-1, galectin-3, CK19, and CD56 (2). However, these IHC markers are not commonly used in daily practice. The immunoprofile patterns of various thyroid lesions may overlap (14). Even with the use of an IHC panel, accurately differentiating thyroid lesions is impossible (2-4, 13-22). Unlike other IHC markers, CD56 is a negative marker, and the loss of CD56 IHC expression is correlated with malignant thyroid lesions, including PTC and FC. CD56 expression is present in normal thyroid follicular cells and is lost in malignant thyroid lesions. However, clearly defining the diagnostic role of a single CD56 IHC marker clearly is difficult because eligible studies included only a small number of patients (2-4, 13-22). In the present study, we focused on the loss of CD56 expression in malignant thyroid lesions and investigated the diagnostic role of CD56 IHC through a meta-analysis. The malignant tumors, such as PTC and FC, showed a higher rate of loss of CD56 expression than other benign lesions, such as FA, BFN, and Hashimoto’s thyroiditis, and the normal thyroid tissue. A meta-regression test showed significant differences in loss of CD56 expression between malignant and these benign thyroid lesions. Evaluating the loss of CD56 expression as an ancillary test can be useful for differentiating malignant and benign thyroid lesions, such as FA, BFN, and Hashimoto’s thyroiditis.
In thyroid pathology, one of the most frequent challenges is to distinguish FVPTC and FC from FA (11, 14). Differentiating these thyroid lesions is important in deciding on further therapy and follow-up. However, differentiating such lesions may be difficult in limited specimens, such as those obtained through fine-needle aspiration cytology and core needle biopsy. In these limited specimens, the application of several markers can be ineffective as an ancillary test. Thus, defining the diagnostic roles of single markers in an ancillary test is important. In the present meta-analysis, because 11.9% of FAs showed a loss of CD56 IHC expression, CD56 IHC may be inadequate to distinguish FVPTC from FA. However, in the diagnostic test accuracy review, CD56 IHC had high sensitivity and specificity and high value of AUC on the SROC curve in a comparison between FVPTC and FA. CD56 was useful in the differentiation between PTC and FA, and between PTC and BFN, and this result is similar to that of a previous report (14). Differentiating between FC and FA via core needle biopsy is challenging because determining the presence of capsules and capsular and vascular invasions of the tumor is difficult. Although evaluating CD56 IHC expression is useful in differentiating between FC and FA according to the present diagnostic test accuracy review, confirmative diagnosis for FC and FA should be performed by evaluating the entire tumor. CD56, using the rapid IHC method, can be useful for differentiation between malignant and benign lesions, including FC from FA in frozen section. In the present study, the encapsulated variant of PTC showed a loss of CD56 expression in 91.9% of cases. Our data supported the results of a previous report that CD56 IHC can be used to differentiate PTC from other benign thyroid lesions with papillary structures (13). However, the information on CD56 expression in non-invasive follicular thyroid neoplasm with papillary-like nuclear features was not found.
Dunđerović et al. (2) also compared the value of a single IHC marker and panels in differentiating benign and malignant thyroid lesions. Their data supported that CD56 is the most specific marker among single IHC markers. However, in differentiating thyroid lesions, the sensitivity of combining CD56 with other IHC markers was lower than other marker combinations (2). According to a previous report, IHC panels, including CD56, have lower sensitivities (40.16%–52.46%) than that of CD56 as a single marker (58.20%) (2). In addition, interpreting the results of IHC panels may be difficult if the item on each panel shows overlapping results. Markers with high specificity can be suitable to confirm thyroid malignancies. Although the panel is more sensitive than the single CD56 marker, it has to be evaluated carefully, taking into account the cost and running time. Another study reported that IHC markers combined with CD56 showed higher sensitivity and specificity than panels combining other markers (7). When several IHC markers were combined, the specificity for malignant lesion did not reach 100% (2). In addition, the sensitivity and specificity can vary, depending on the compared thyroid lesions. In our results, a single CD56 marker showed high sensitivity and specificity. Therefore, including CD56 in the marker combination for differentiating malignant and benign lesions can increase the specificity of the IHC panel. A single marker, rather than the combination of several markers, can be a suitable substitute for frozen sections and limited specimens in evaluating thyroid malignancies. Cumulative studies for the comparison between the single marker and the panel are needed.
Because the loss of CD56 expression is evaluated via IHC, setting a cut-off value is important in determining the rate of loss of expression. Eligible studies used various cut-offs, ranging from 0% to 10%. When the cut-off value is low, the rate of loss of CD56 expression is also low. In our meta-analysis, the rate of loss of CD56 expression increased in the higher cut-off subgroup (Tab. III). However, no significant difference in the rate of loss of CD56 expression was noted between the 0% and 10% cut-off subgroups of various thyroid lesions in the meta-regression test. These results suggested that the 0%–10% range of cut-off value might have no effect on the rate of loss of CD56 expression. However, in the current meta-analysis, the optimal cut-off value could not be confirmed due to insufficient information. If CD56 expression is lost during malignant transformation, the possibility of heterogeneity inside the tumor should be considered.
The current study has a number of limitations. First, FVPTC and FC, including encapsulated FVPTC, could not be compared due to insufficient information in eligible studies. In addition, cases with non-neoplastic lesions or normal tissue contained less than thyroid carcinomas in the present meta-analysis. Further cumulative studies will be needed to obtain the confirmative information. Second, in each eligible study, if the diagnosis of debatable cases was not confirmed based on hematoxylin and eosin staining, the diagnostic role of the IHC marker should be re-evaluated. However, the present meta-analysis could not confirm the diagnosis of each case in the eligible studies. Third, the present meta-analysis was performed to evaluate the diagnostic usefulness of CD56 as a single marker in differentiating various thyroid lesions. However, the diagnostic role of CD56 for metastatic lesions in the lymph nodes and distant organs could not be evaluated. Fourth, eligible studies used various antibody clones, IHC methods, and cut-off from various populations. However, comparisons based on antibody clones and IHC methods could not be conducted due to insufficient information. Fifth, on pathologic examination, IHC was performed and evaluated for only a portion of the tumor. Confirming complete loss of CD56 expression before examination of the entire lesion may be challenging. Conclusive information regarding the heterogeneity of loss of CD56 expression was unavailable. Additional cumulative studies regarding the heterogeneity of loss of CD56 expression are needed.
In conclusion, the rates of loss of CD56 IHC expression were significantly higher in malignant thyroid tumors, such as PTC and FC, than in FA and other benign lesions, such as FA, BFN, and Hashimoto’s thyroiditis. In thyroid lesions, CD56, as single marker, can be useful for differentiating PTC and FC from FA in daily practice.
Footnotes
Disclosures
Conflict of interest: The authors declare that they have no conflict of interest.
