Abstract
Purpose:
The aim of this study was to investigate the rate of ROS1 rearrangement and concordance between ROS1 immunohistochemistry (IHC) and molecular tests in non-small cell lung cancer (NSCLC).
Methods:
The study included 10,898 NSCLC cases from 21 eligible studies. ROS1 rearrangement rates were evaluated in NSCLC by a meta-analysis, including subgroup analyses. In addition, we performed a concordance analysis and a diagnostic test accuracy review of ROS1 IHC in NSCLC.
Results:
The estimated overall rate of ROS1 rearrangement and IHC positivity was 2.4% (95% confidence interval (CI) 1.5, 3.7). In the subgroup analysis, which was based on tumor subtype, the rate of ROS1 rearrangement and IHC positivity was 2.9% (95% CI 1.9, 4.5) and 0.6% (95% CI 0.3, 1.2) in adenocarcinoma and non-adenocarcinoma, respectively. The overall concordance rate between ROS1 IHC and molecular tests was 93.4% (95% CI 78.3, 98.2). In ROS1 IHC positive and negative cases, the concordance rates were 79.0% (95% CI 43.3, 94.9) and 97.0% (95% CI 83.3, 99.5), respectively. The pooled sensitivity and the specificity of ROS1 IHC were 0.90 (95% CI 0.70, 0.99) and 0.82 (95% CI 0.79, 0.84), respectively. The diagnostic odds ratio and the area under the curve of the summary receiver operating characteristic curve were 118.01 (95% CI 11.81, 1179.67) and 0.9417, respectively.
Conclusion:
The rates of ROS1 rearrangement differed by tumor histologic subtype in NSCLC. ROS1 IHC may be useful for the detection of ROS1 rearrangement in NSCLC. Detailed criteria for evaluating ROS1 IHC are needed before it can be applied in daily practice.
Keywords
Introduction
Although lung cancer has long been a disease characterized by late-stage diagnosis and no progress in treatment options, the last decade has yielded substantial progress with systemic therapies for molecular subgroups of patients with non-small cell lung cancer (NSCLC). 1 NSCLCs are subdivided by histologic type, including adenocarcinoma, squamous cell carcinoma, and adenosquamous carcinoma. Known oncogenic drivers of NSCLC include EGFR, ALK, MET, KRAS, HER2, RET, and ROS1.2–4 These genetic alterations may be important for the application and prediction of various targeted therapies in NSCLC patients. 5
ROS1, a tyrosine kinase receptor, belongs to the insulin receptor family.6,7 Constitutively activated ROS1 fusion proteins induce the activation of oncogenic pathways, such as the PI3K/AKT/mTOR and RAS/MAPK/ERK pathways, and are involved in cell proliferation, survival, and cell cycling.8,9 ROS1 genetic alterations are found less frequently in cancer, including NSCLC, and the clinicopathological significance of ROS1 rearrangement is controversial.10-12 For the detection of ROS1 rearrangement, molecular tests, such as in situ hybridization (ISH) or DNA sequencing, are required. Although molecular tests are confirmative, it is difficult to test all NSCLC patients in daily practice. In addition, the disadvantages of these molecular tests include high costs, multiple steps, requirement of advanced technical skills, and high turnaround time compared to immunohistochemistry (IHC). Recently, ROS1 IHC has been introduced and studied; however, its diagnostic accuracy has not been fully elucidated in NSCLC.
In order to evaluate its role, we performed a concordance analysis between ROS1 IHC and molecular tests, and a diagnostic test accuracy review of ROS1 IHC in NSCLC. In addition, ROS1 rearrangement was investigated by subdividing histologic types and detection methods.
Materials and methods
Published study search and selection criteria
Relevant articles were obtained by searching the PubMed and MEDLINE databases until 30 November 2017.These databases were searched using the following key words: “non-small cell lung cancer or NSCLC” and “ROS1.” The titles and the abstracts of all searched articles were screened for exclusion. Review articles were also screened to find additional eligible studies. Search results were then reviewed and included if (a) the study was performed on human NSCLCs; and (b) there was information about ROS1 genetic alteration and IHC expression. Articles were excluded if they were (a) case reports or non-original articles; or (b) non-English language publications.
Data extraction
Data from all eligible studies were extracted by two independent authors. The included data were extracted from each of the eligible studies:2,13–32 the first author’s name, the year of publication, the study location, the detection method for ROS1 genetic alteration, and the number of patients analyzed. For the meta-analysis, we extracted all data associated with the results of ROS1 genetic alteration and IHC.
Statistical analyses
For the meta-analysis, all data were analyzed using the Comprehensive Meta-Analysis software package (Biostat, Englewood, NJ, USA). We investigated the rates of ROS1 genetic alteration and IHC expression in NSCLC. The concordance rates were measured by agreement rates between ROS1 IHC and molecular tests. Because the eligible studies used various methods for the detection of ROS1 genetic alteration in various populations, a random-effects model was more suitable than a fixed-effects model. Heterogeneity between the studies was checked using the Q and I 2 statistics, and presented using P-values. Additionally, a sensitivity analysis was conducted to assess the heterogeneity of eligible studies and the impact of each study on the combined effect. To assess publication bias, Begg’s funnel plot and Egger’s test were used. If a significant publication bias was found, the fail-safe N and trim-fill tests were performed to confirm the degree of publication bias. The results were considered statistically significant at P< 0.05.
The review of diagnostic test accuracy was performed using the Meta-Disc program (version 1.4, Unit of Clinical Biostatics, the Ramon y Cajal Hospital, Madrid, Spain). 33 In order to calculate the pooled sensitivity and specificity, data were collected from each eligible study and forest plots were obtained. The summary receiver operating characteristic (SROC) curve was initially constructed by plotting “sensitivity” and “1-specificity” of each study, and the curve fitting was performed through linear regression using the Littenberg and Moses linear model. 34 Because the data were heterogeneous owing to differences in the evaluation criteria, the accuracy data were pooled by fitting a SROC curve and measuring the value of the area under the curve (AUC). 33 An AUC close to 1 indicated a strong test and an AUC close to 0.5 was considered a poor test. In addition, the diagnostic odds ratio (OR) was calculated by the Meta-Disc program.
Results
Selection and characteristics
A total of 354 reports were identified in the database search for the meta-analysis. Among them, 156 were excluded because of insufficient information on ROS1 gene rearrangement as determined by molecular tests and/or ROS1 IHC expression. Also, 150 were excluded because they were non-English or non-original articles, and 23 were excluded because they reported the results of other diseases or used animal models or cell lines. Four duplicate articles were also excluded. In total, 21 studies were included in the meta-analysis (Figure 1 and Table 1), and the number of patients was 10,898. Table 1 shows the ROS1 rearrangement and IHC positivity data for the eligible studies. In all immunohistochemistry, the antibody clone was D4D6, which was manufactured by Cell Signaling Technology.
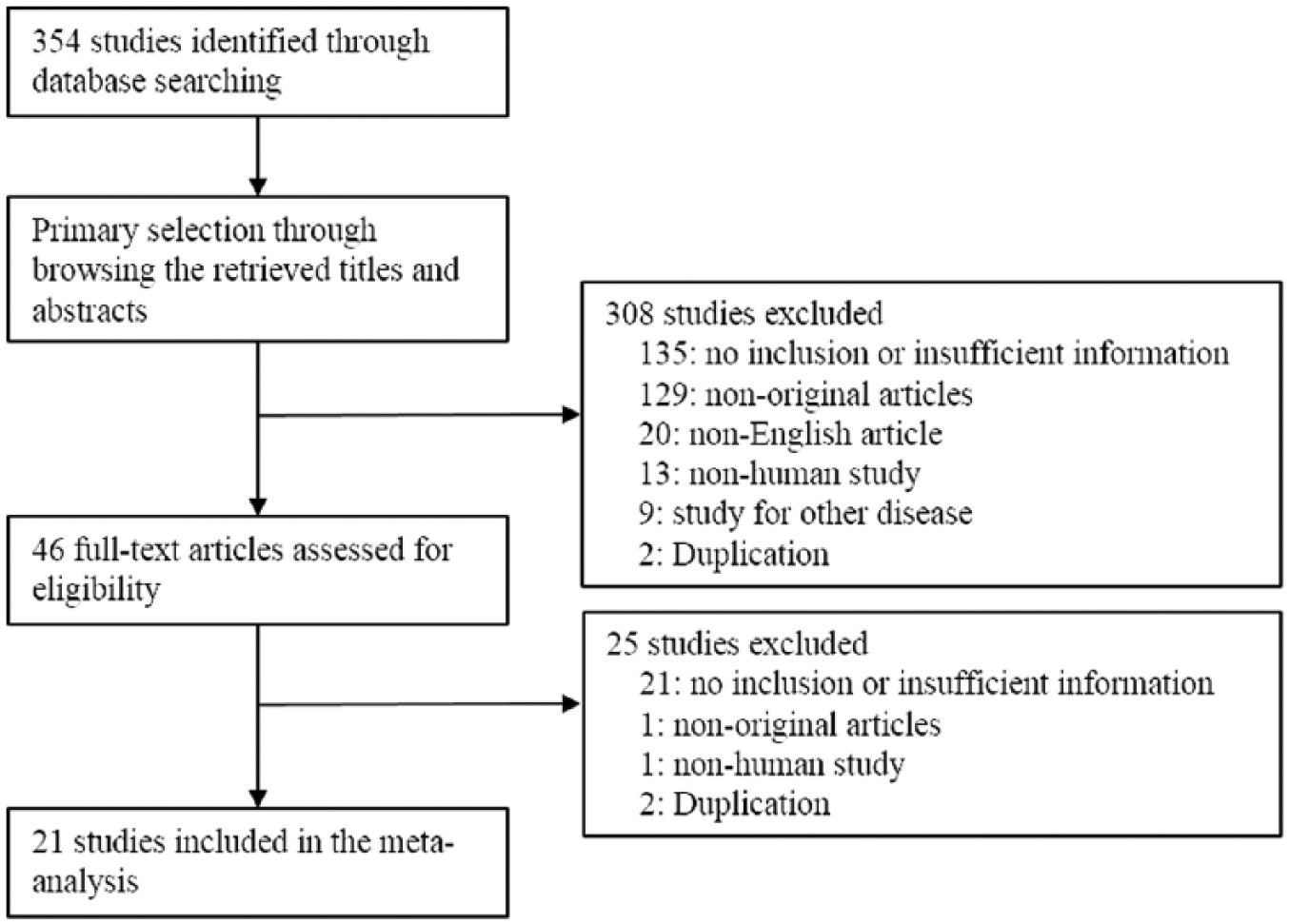
Flow chart of study search and selection methods.
Main characteristics of the eligible studies.
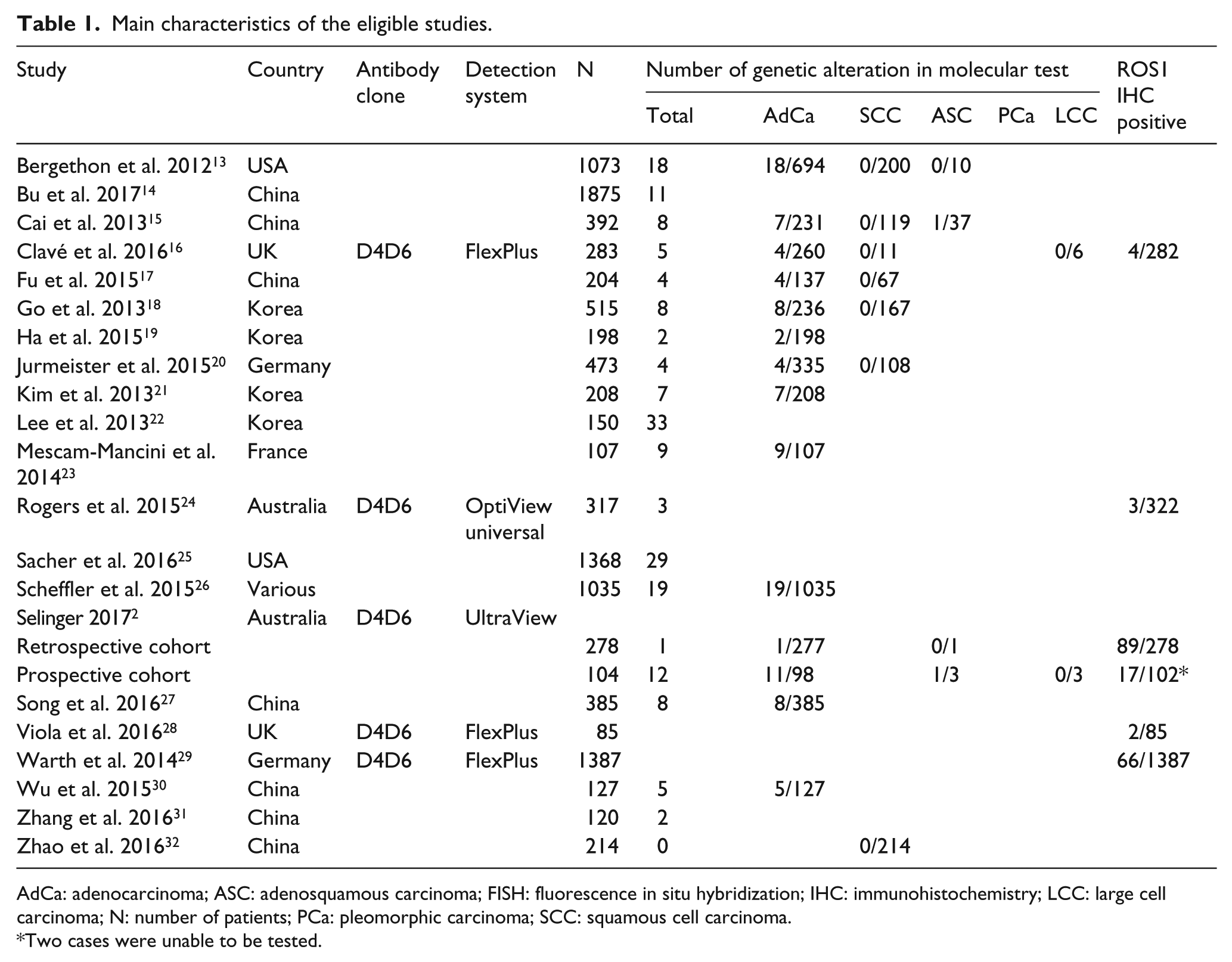
AdCa: adenocarcinoma; ASC: adenosquamous carcinoma; FISH: fluorescence in situ hybridization; IHC: immunohistochemistry; LCC: large cell carcinoma; N: number of patients; PCa: pleomorphic carcinoma; SCC: squamous cell carcinoma.
Two cases were unable to be tested.
Meta-analysis for ROS1 genetic alteration in non-small cell lung cancer
The estimated overall rate of ROS1 rearrangement and IHC positivity was 2.4% (95% CI 1.5, 3.7; Table 2). There was no significant publication bias identified by Egger’s test (P = 0.072) or Begg’s funnel plot. Subgroup analysis based on histologic type was performed. The rate of ROS1 rearrangement and IHC positivity in adenocarcinoma and non-adenocarcinoma was 2.9% (95% CI 1.9, 4.5) and 0.6% (95% CI 0.3, 1.2), respectively. Among non-adenocarcinomas, the rate of ROS1 rearrangement and IHC positivity in adenosquamous carcinoma and pleomorphic carcinoma was 3.5% (95% CI 1.1, 10.3) and 3.8% (95% CI 0.6, 19.1), respectively. However, ROS1 rearrangement was not found in squamous cell carcinoma. The ROS1 rearrangement rates varied by the detection method. The range of ROS1 rearrangement rates was 0.6, 22.0% and 2.0, 2.1% by fluorescence in situ hybridization (FISH) and DNA sequencing, respectively. In a subgroup analysis based on the detection method, the estimated rate of ROS1 rearrangement was 2.5% (95% CI 1.4, 4.5), 2.1% (95% CI 1.3, 3.3), and 0.8% (95% CI 0.2, 2.6) by FISH, DNA sequencing, and the NanoString technique, respectively. The range of ROS1 IHC positivity was 0.9, 32.0% in four eligible studies. All studies used the clone D4D6 (Cell Signaling Technology, Danvers, MA, USA) at a dilution of 1:50 or 1:300. The positive rate of ROS1 IHC was 2.0% (95% CI 0.8, 5.3). In addition, for the subgroup analysis based on the evaluation criteria, the eligible studies were classified into two subgroups: (a) staining intensity alone; and (b) a combination of intensity and percentage of positive tumor cells. The ROS1 IHC positivity of the intensity-only subgroup was significantly higher than that of the combination subgroup (4.8%, 95% CI 3.8, 6.0 vs. 1.3%, 95% CI 0.7, 2.6).
Meta-analysis for ROS1 genetic alteration and IHC expression in non-small cell lung cancer.
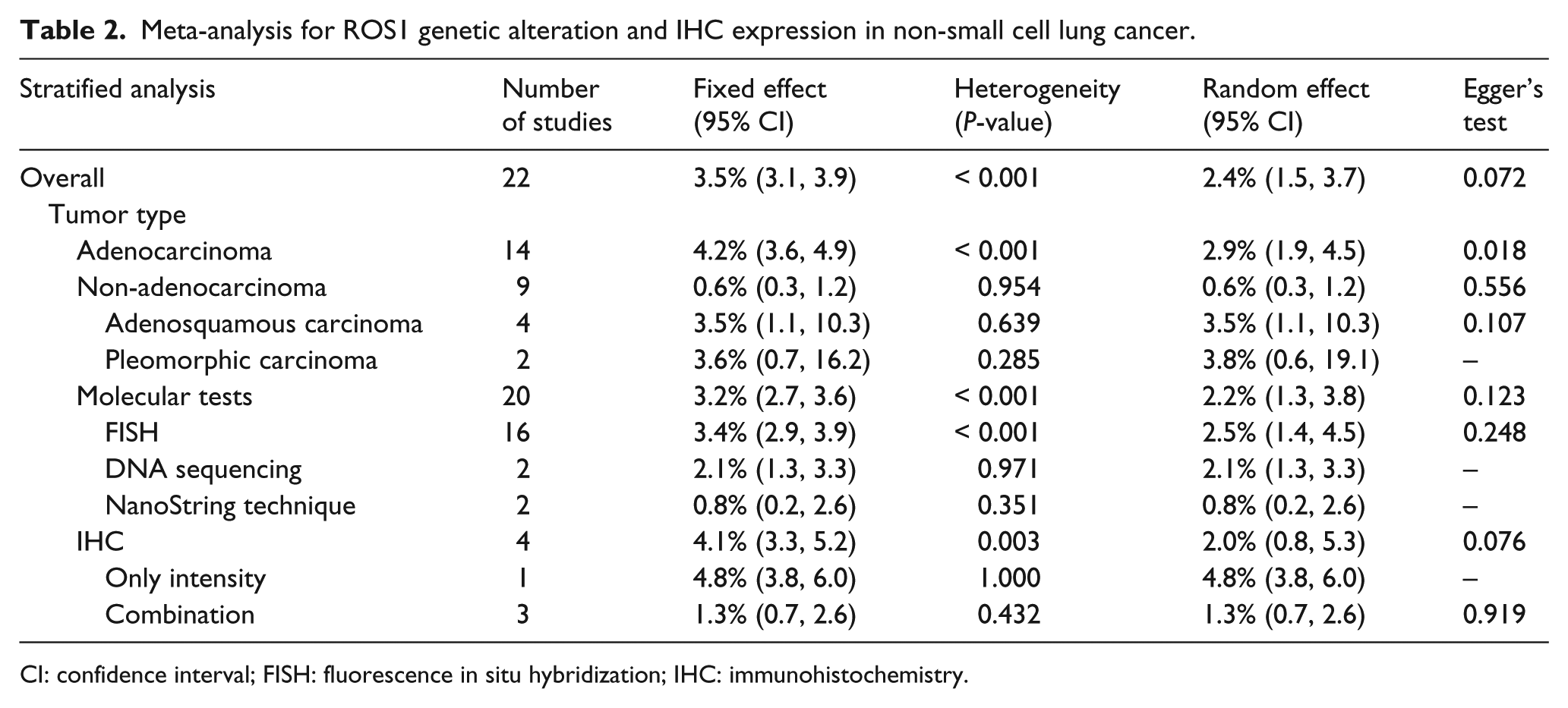
CI: confidence interval; FISH: fluorescence in situ hybridization; IHC: immunohistochemistry.
Next, to assess the usefulness of ROS1 IHC, a concordance analysis between ROS1 IHC and molecular tests was performed in NSCLC cases (Table 3). The overall concordance rate was 93.4% (95% CI 78.3, 89.2). In the subgroup analysis based on ROS1 immunoreactivity, the estimated concordance rate was 79.0% (95% CI 43.3, 94.9) and 97.0% (95% CI 83.3, 99.5) in the ROS1 IHC positive and negative subgroups, respectively. In the subgroup analysis based on evaluation criteria, the concordance rate of the intensity-only subgroup was significantly higher than that of the combination subgroup (98.6%, 95% CI 56.1, 100.0 vs. 90.7%, 95% CI 69.7, 97.6). In ROS1 IHC positive cases, the concordance rate was 90.0% (95% CI 32.6, 99.4) and 74.2% (95% CI 26.3, 95.9) in the intensity-only and the combination subgroup, respectively. In ROS1 IHC negative cases, there was no difference in the concordance rate between the intensity-only and the combination subgroup (99.8%, 95% CI 97.2, 100.0 vs. 94.9%, 95% CI 73.7, 99.2).
The concordance analysis of ROS1 immunohistochemistry and genetic alteration in non-small cell lung cancers.
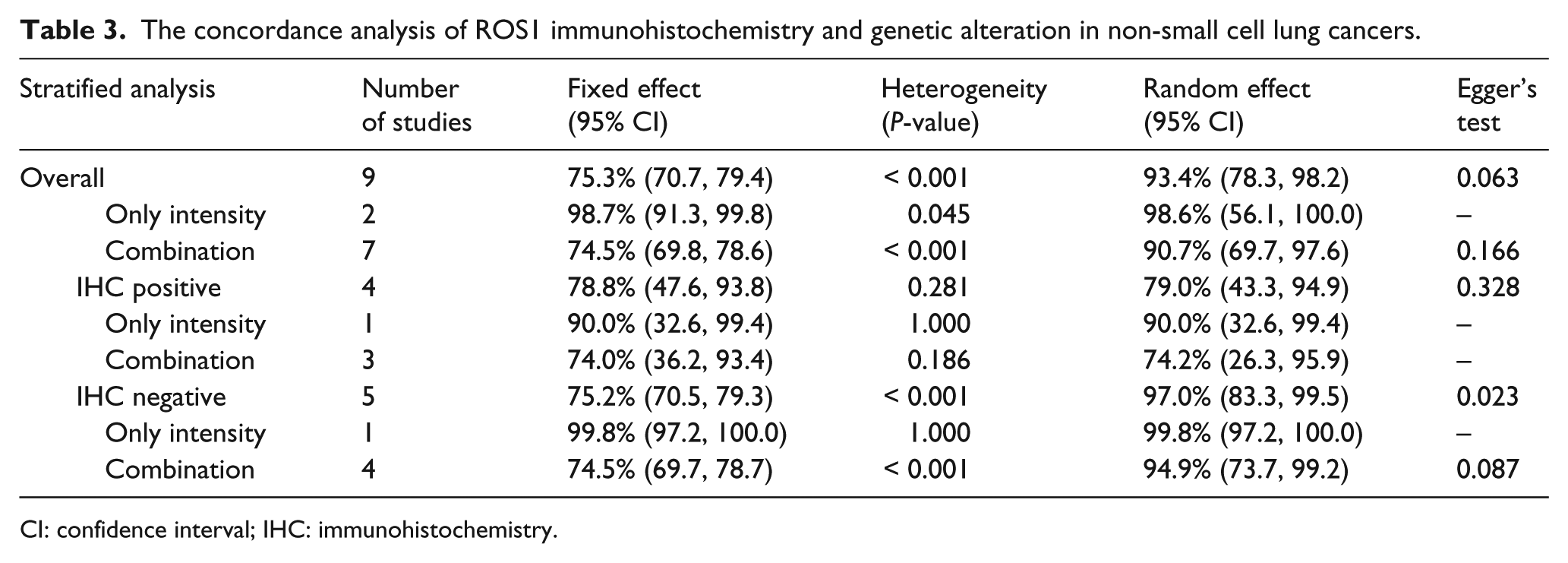
CI: confidence interval; IHC: immunohistochemistry.
Diagnostic test accuracy review
To confirm the diagnostic accuracy of ROS1 IHC for predicting ROS1 rearrangement, we performed a diagnostic test accuracy review. The pooled sensitivity and specificity were 0.90 (95% CI 0.70, 0.99) and 0.82 (95% CI 0.79, 0.84), respectively (Figure 2). The sensitivity of the eligible studies ranged from 0.33 to 1.00, and the specificity ranged from 0.50 to 1.00. The AUC of the SROC curve was 0.9417. The diagnostic OR was 118.01(95% CI 11.81, 1179.67).
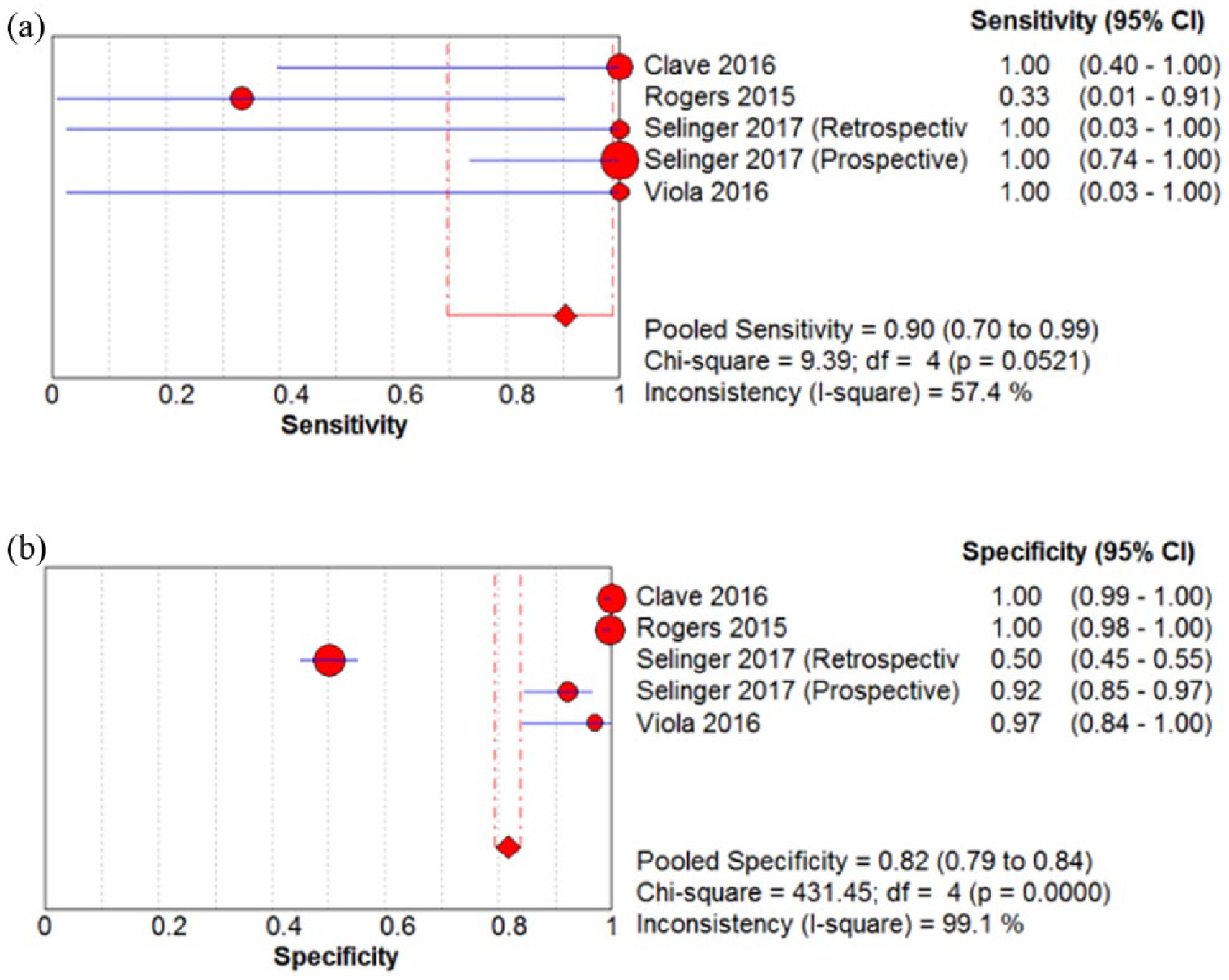
The sensitivity (a) and specificity (b) of ROS1 immunohistochemistry in non-small cell lung cancer (NSCLC).
Discussion
Various biomarkers, such as BRAF, ERBB2, MET, ROS1, and RET, have been evaluated for targeted therapies in daily practice. In addition, their genetic status can correlate with prognosis. Accurate tests for various biomarkers are important for predicting therapeutic effects in NSCLC. In previous studies, we confirmed the diagnostic role and accuracy of ALK and c-MET IHC in NSCLC.35,36 Taken together, an IHC panel including ALK, c-MET, and ROS1 can be useful for screening genetic alterations in NSCLC.35–40 However, the clinical usefulness and diagnostic accuracy of ROS1 IHC is not fully understood. The present study is the first meta-analysis to assess the ROS1 rearrangement rate and the diagnostic accuracy of ROS1 IHC in NSCLC.
Although treatments using tyrosine kinase inhibitors (TKIs) have been developed and applied in NSCLC, therapeutic effects can differ according to tumor nature, such as genetic alteration and histologic type. 5 Patients with ROS1 rearrangement had poorer outcomes after treatment with EGFR-TKIs. 21 Crizotinib, an ALK inhibitor, shows therapeutic efficacy in NSCLC patients with ROS1 rearrangement.41,42 Of 50 patients, 72% had objective response to crizotinib with a median progression-free survival of 19.2 months. Tumor responses were observed regardless of the specific ROS1 fusion partner. 37 A European retrospective study of 32 patients reported showed similar results. 42 Other ROS1 inhibitors are currently being evaluated, including ceritinib, cabozantinib and entrectinib. 1 To obtain optimal therapeutic effect, accurate molecular tests are warranted in daily practice. Studies for ROS1 genetic alteration in NSCLC have been reported since 2007. ROS1 rearrangement status correlates with patient status, including female gender, history of non-smoking, and adenocarcinoma histology. 43 However, in some studies, the correlation between ROS1 rearrangement and tumor stage is controversial. 29 In addition, the correlation between ROS1 rearrangement and patient survival is unclear. Some studies have reported that ROS1 rearrangement significantly correlated with poor survival in patients with NSCLC.15,44,45 In other studies, there was no significant correlation between ROS1 rearrangement and survival.13,29,46-48 To obtain the conclusive information regarding ROS1 genetic alteration, large-scale prospective studies and/or meta-analyses are required.
Adenocarcinoma and squamous cell carcinoma are the two most common histological types of NSCLC, accounting for 50% and 30% cases, respectively. 4 The incidence of ROS1 rearrangement was 2.4% of patients with adenocarcinoma. 43 Concordant with our results, the rate of ROS1 rearrangement in adenocarcinoma was higher than that in non-adenocarcinoma in a previous meta-analysis. 43 However, there was no ROS1 rearrangement in squamous cell carcinoma found in the eligible studies. The rates of ROS1 rearrangement in other histologic types, such as adenosquamous carcinoma and pleomorphic carcinoma, were higher than that in adenocarcinoma (Table 2). Therefore, molecular tests for ROS1 can be required for all NSCLCs, except squamous cell carcinomas. 43 In addition, ROS1 rearrangement was frequently found in female patients, non-smokers, and patients with advanced stage cancer. In selected patients with triple wild type EGFR/KRAS/ALK genotype and non-smoking history, ROS1 rearrangement accounted for 7.4–8.3%.23,30,44 Patient selection according to clinicopathological characteristics would not be effective for detecting ROS1 genetic alterations. Since the incidence rate of ROS1 genetic alterations is very low in NSCLCs, molecular tests cannot be applied to all patients, and an effective screening method is needed for daily practice.
Although ROS1 IHC in NSCLC has been introduced and studied, its diagnostic accuracy has not been fully elucidated. Whether ROS1 IHC in NSCLC can replace molecular tests, such as FISH or direct sequencing, is not yet known. In many laboratories, IHC is used more commonly than molecular tests. In addition, molecular tests remain expensive and labor-intensive. In the present meta-analysis, the overall concordance rate analysis between ROS1 IHC and molecular tests was 93.4% (95% CI 78.3, 98.2). In ROS1 IHC negative cases, the concordance rate was very close to 1 (97.0%, 95% CI 83.3, 99.5). According to our results, the ROS1 IHC negative cases could be considered as negative for rearrangement without additional molecular testing, such as for c-MET and ALK IHC in NSCLC.35,36 Therefore, ROS1 IHC can be useful for screening ROS1 rearrangement in NSCLC. However, the concordance rate between ROS1 IHC and molecular tests in ROS1 IHC positive cases was lower than that in ROS1 IHC negative cases. The ROS1 IHC positive rates were variable, ranging from 0.6% to 4.8%.16,24,28,29 The variability among the reports may be caused by different evaluation criteria and patient populations. In addition, ROS1 immunostaining can be heterogeneous, such as in cases of breast and gastric cancers with 2+ HER2 scores, and the possibility of false positivity of ROS1 IHC should be considered. In the current meta-analysis, subgroup analysis based on the evaluation criteria of ROS1 IHC was also performed. The concordance rate in the intensity-only subgroup was significantly higher than that in the combination subgroup (4.8%, 95% CI 3.8, 6.0 vs. 1.3%, 95% CI 0.7, 2.6, P < 0.001). Therefore, cumulative studies are needed to obtain the definitive evaluation criteria of ROS1 IHC.
There are some limitations in the current meta-analysis. First, the ranges of the sensitivities and the specificities of the eligible studies were 0.33–1.00 and 0.50–1.00, respectively. Only four eligible studies showed the correlation between ROS1 IHC and molecular tests. Therefore, to obtain more conclusive information for the sensitivity and the specificity of ROS1 IHC, further cumulative studies are required. Second, concordance analysis based on subgroups by molecular tests could not be performed due to insufficient information from eligible studies. Third, in ROS1-positive cases, the concordance rate was as low as 79.0% (95% CI 43.3, 94.9), compared to ROS1 negative cases. However, in the current meta-analysis, the cause of the low concordance rate in ROS1-positive cases could not be found because there were too few eligible studies analyzed.
In conclusion, the rates of ROS1 rearrangement differed according to histologic types of NSCLC and detection methods. In addition, ROS1 IHC showed good concordance with ROS1 molecular tests in NSCLC. Further studies of detailed evaluation criteria and diagnostic flow for the detection of ROS1 rearrangement are required before application in daily practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2017 Inje University research grant.
