Abstract
Objective
To conduct a systematic review and meta-analysis to compare the effects of statins and placebo on systolic blood pressure (SBP) and diastolic blood pressure (DBP) in patients with hypertension.
Methods
We used different databases such as MEDLINE and the Cochrane Library Central Register of Controlled Trials (CENTRAL) to search for randomized controlled trials published from 1 January 2000 to 1 January 2022 to retrieve relevant studies and full-text articles evaluating statins versus placebo in patients with hypertension. Data were taken from eligible studies. The included articles also performed Cochran’s Q statistics, forest plots, funnel plots, Egger’s test, and sensitivity analyses.
Results
This study included 8 studies with a total of 3086 patients. We sought to assess patients' SBP and DBP. The results were expressed as mean differences (MD) with 95% confidence intervals. There was no difference in DBP between the statin and placebo groups (MD = 2.48, 95% CI [−2.00, 6.96], p = .28, random effects model). In terms of SBP, the statin group was superior to the placebo group (MD = 4.37, 95% CI [0.72, 8.02], p = .02, random-effects model). Sensitivity analyses and funnel plots showed that our study was robust, with low publication bias.
Conclusion
The results show that statins could be clinically more effective than placebo in patients with hypertension.
Introduction
Essential hypertension is a common and frequent disease in cardiovascular medicine. 1 It is easily overlooked by patients because of its insidious onset and mild symptoms. However, hypertension causes dangerous complications in vital organs such as the heart, brain and kidneys, 2 and as such it has attracted increasing attention in the medical field.
Statins (HMG-CoA reductase inhibitors) are the most widely used and effective lipid-lowering drugs in clinical practice. With intensive clinical and basic research on statins, it has been found in recent years that statins have anti-hypertensive effects in addition to lipid-lowering effects.3,4 Studies have found that statins also significantly lower blood pressure in hypertensive patients without dyslipidemia compared with the placebo group and other lipid-lowering drugs.
Some studies have shown that statins lower blood pressure, while others do not.5,6 A recent meta-analysis of a number of trials relating to the effect of statins on blood pressure showed that patients using statins had significantly lower systolic (but not diastolic) blood pressure. 4 A large randomised sample study of individuals in the community confirmed that statins significantly lowered both systolic and diastolic blood pressure. Statins lowered diastolic blood pressure by similar amounts in patients taking and not taking anti-hypertensive drugs. In addition, the antihypertensive effect of statins increased with increasing diastolic blood pressure in non-antihypertensive drug users. 7
In this study, several newly published randomised controlled trials compared the clinical effects of statins and placebo in patients with hypertension. Relevant articles are included in this meta-analysis in order to fully understand the role of statins and placebo in patients with hypertension.
Methods
This research is based on PICO process: In patients with hypertension (Population), does the administration of statins (Intervention) compared to placebo result in SBP and DBP (Outcomes).
Literature search strategy
We searched MEDLINE and the Cochrane Library Central Register of Controlled Trials (CENTRAL) for randomised controlled trials published between 1 January 2000 and 1 January 2022, using the search terms. Our search strategy varied by database, but covered the search term ‘statins and hypertension’. We conducted a comprehensive search using multiple databases, trial registries, grey literature and conference proceedings, with no restrictions on language of publication or publication status. To achieve maximum sensitivity of the search strategy and to identify all studies, we manually screened the reference lists of all retrieved articles to further identify potentially relevant studies.
Study selection
To be included in the analysis, the original research articles had to meet the following criteria. (1) articles published in peer-reviewed journals and (2) journal articles with outcome measures related to hypertension. Exclusion criteria were. (1) A research paper not written in English. (2) No full-text studies were provided. (3) Not an original research report. (4) A summary or presentation of the conference. (5) Not a hypertensive patient. (6) The study did not measure blood pressure.
In addition, if there are multiple publications from the same queue, we extract the data from the largest or most recent dataset.
Data extraction and quality assessment
The following data were extracted: study characteristics, number of patients, clinical outcome results including SBP and DBP. Where available, the following details were extracted: first author, study title, country, year of publication, study design, study characteristics, sample size, mean age of patients and male/female ratio.
For the quality assessment, we considered the following domains: random sequence generation, allocation concealment, blinding (self-reporting), blinding (objective results), incomplete and selective reporting of results, and the presence of other biases. We assessed each domain separately and critically, and our score system is based on GRADE (Grading of Recommendations, Assessment, Development and Evaluations), which is a transparent framework for developing and presenting summaries of evidence and provides a systematic approach for making clinical practice recommendations. We classified them as low risk of bias, unclear risk or high risk of bias according to the criteria set out in the Cochrane Handbook. 8
Statistical analysis
Initially, we analysed all studies separately, replicating the originally reported findings using the methods described in the respective publications. Any inconsistencies were resolved with the authors of the original studies. To optimise the interpretability of our findings for continuous outcomes, we used network estimates of treatment effects to model the odds ratio (OR) of achieving a mean difference (MD), i.e. the smallest change in patient-reported outcomes that patients considered important. The broad Cochran’s Q statistic was used to assess homogeneity in multivariate meta-analyses. In detail, an I2value of 0% indicates no heterogeneity, 25% indicates low heterogeneity, 25–50% indicates moderate heterogeneity and 50% indicates high heterogeneity. In addition, a random effects model was used when heterogeneity was observed and a fixed effects model was used when no heterogeneity was modelled. Potential publication bias was assessed by visual inspection of funnel plots and Egger’s test for small study effects. Further sensitivity analyses were conducted to assess the robustness of the results via exponential skewing.
Results
Search process
A total of 780 titles and abstracts were identified through the screening electronic search strategy, of which 8 were eligible for assessment. Eight studies describing 16 study groups met the inclusion criteria and were included in the meta-analysis.
After title and abstract review, 372 studies were excluded due to duplication and 400 studies were excluded because they did not meet inclusion criteria based on article type, study design, study population or relevant outcomes. After exclusion, eight articles were included in our meta-analysis. In total, we retrieved 8 studies that met all inclusion criteria (Figure 1).9–16 Flowchart of study selection.
Characteristics of included studies
Baseline characteristics of included studies.

Results of quality assessment
A qualitative assessment was conducted by using the Cochrane Risk of Bias tool to assess various indicators for each study (Figure S1). Scores ranged from 7 to 8, with nine studies scoring above 7. Given the bias summary, selection bias, detection bias and other biases were not an issue. Two trials were at risk of bias and seven were not (Figure S2).
Results of heterogeneity test
In eight studies involving 3086 patients, a comparison of systolic blood pressure (SBP) between the statin and placebo groups was reported. A meta-analysis showed a difference between the two groups (MD = 4.37, 95% CI [0.72, 8.02], p = .02, random effects model) (Figure 2). Forest plot: comparison of SBP between the statin and placebo groups, experimental = statin, control = placebo.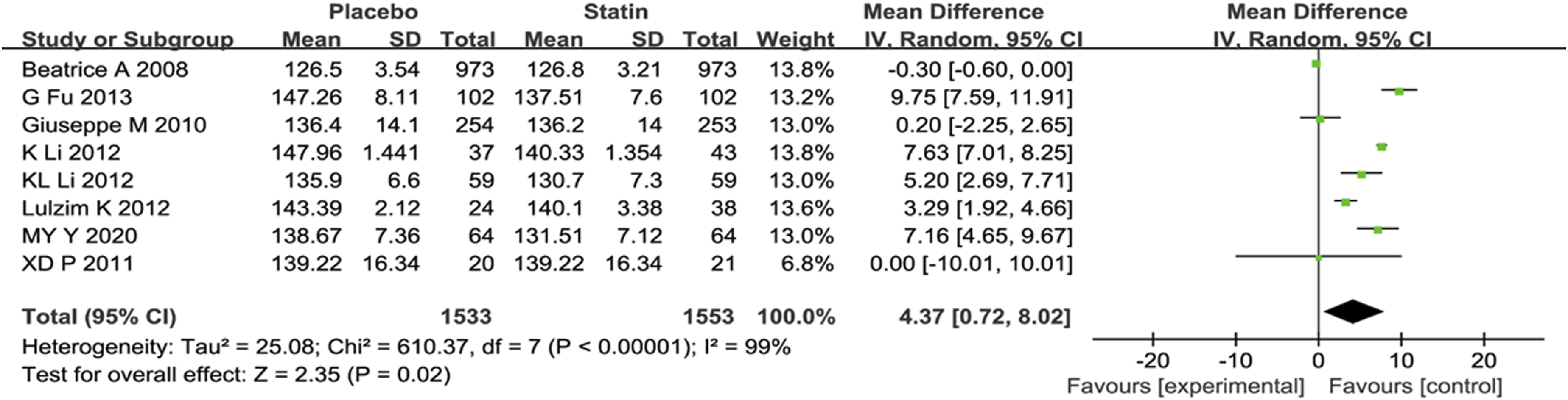
A meta-analysis was also performed on the diastolic blood pressure (DBP) of patients in the statin and placebo groups. The results showed no significant difference in DBP between the two groups (MD = 2.48, 95% CI [−2.00, 6.96], p = .28, random effects model) (Figure 3). Forest plot: comparison of DBP between the statin and placebo groups, experimental = statin, control = placebo.
Results of Sensitivity Analysis and Publication Bias
A total of eight studies reported SBP. The forest plots showed that the statin group was superior to the placebo group (MD = 4.37, 95% CI [0.72, 8.02], p = .02, p = .02, I2 = 99%). We performed a sensitivity analysis by removing Beatrice’s 2008 study and the results were little changed, with I2 = 92% from 99% (Figure 4), indicating that the results of the included articles were robust. Sensitivity analysis forest plots: comparison of SBP between the statin and placebo groups, experimental = statin, control = placebo.
We also drew a funnel plot to assess the publication bias of SBP between the statin and placebo groups; the plot shows its shape to be symmetrical. This result indicates that there was little publication bias in this meta-analysis (Figure S3).
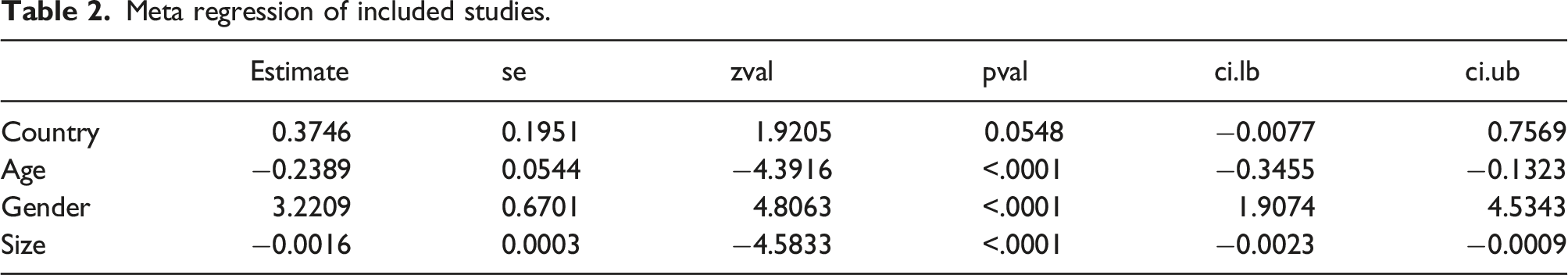
Results of meta regression
Meta regression of included studies.
Discussion
Eight studies met the inclusion criteria to assess the effect of statins on hypertension. Meta-analysis of these studies showed that statins were more effective than placebo for SBP. However, there was no difference in DBP between the statin and placebo groups, suggesting that the two treatments have similar clinical effects on DBP.
There are two main mechanisms by which statins provide protection against cardiovascular disease. Firstly, statins reduce the incidence of disease and fatal cardiovascular events by lowering total serum cholesterol and, more generally, lipid composition. Second, statins may have multiple effects (e.g. anti-inflammatory and anti-proliferative) in addition to causing changes in lipid composition, directly protecting tissues and organs from cardiovascular risk factors. 17 In the past few years, a third mechanism for the cardiovascular protective effects of statins has been proposed - that statins also lower blood pressure, thereby reducing blood pressure-related risks. 18 The effect of statins on cardiovascular disease has been described in the included studies.
One study reported that the addition of pravastatin to antihypertensive therapy resulted in a significant and sustained reduction in total serum cholesterol and LDL cholesterol compared with the placebo group. However, this increase did not result in further reductions in systolic or diastolic blood pressure values, either with regular measurements in the physician’s office or with repeated outpatient measurements over 24 h19,20 This provides evidence for a substantial antihypertensive effect of statins in patients with hypertension, suggesting that the protective effect of these drugs on the cardiovascular system is not dependent on reducing the risk of blood pressure-related cardiovascular disease.
In addition, one study reported significant improvement in systolic left ventricular hypertrophy and left ventricular systolic function in patients with essential hypertension after one year of treatment with simvastatin. 21 The trend of decreasing left ventricular diastolic function was the same as before treatment, but without significant changes. Simvastatin reversed left ventricular hypertrophy and improved left ventricular systolic function in patients with essential hypertension. 22 A large body of data shows that statins, in addition to lowering plasma cholesterol, reduce morbidity and mortality from coronary artery disease; this beneficial effect is independent of their cholesterol-lowering effect. 23
Limitation
This study also had some limitations. Firstly, dynamic changes in blood pressure were not analysed in this study. Secondly, due to the limited raw data, no further stratified comparisons of complications were made.
Conclusions
In a systematic meta-analysis, we found that statin might be clinically more effective than placebo in the treatment of hypertension. It might be more effective in controlling systolic blood pressure (SBP) and reducing the conversion to hypertension, which could help clinician making a good choice for hypertension. The limited number of included studies reduces the reliability of the conclusions. However, a large number of relevant studies should be included in the future to validate the conclusions.
Supplemental Material
Supplemental Material - Effects of statin on hypertension patients: A systematic review and meta-analysis
Supplemental Material for Effects of statin on hypertension patients: A systematic review and meta-analysis by Jianwei Wan and Min Chen in European Journal of Inflammation
Footnotes
Acknowledgements
Sincere gratitude is extended to the Copenhagen Nordic Cochran Center for granting us permission to utilize the Cochrane bias risk assessment tool.
Author contribution
JW and MC participated in the conception and design of the study, library searches and assembling relevant literature, critical review of the paper, supervising the writing of the paper, and database management and participated in data collection, library searches and assembling relevant literature, writing the paper, and critical review of the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Important Weak Discipline Construction Project of the Pudong New Area Health System (PWZbr2022-11).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
