Abstract
It seems that Porphyromanas gingivalis is carcinogenic, because it activates a number of inflammatory immune responses in the host and causes disorders in bacterial clearance mechanisms. There is little information on the prevalence of this bacterium in cancer patients. The aim of this study was to evaluate and compare the prevalence of P. gingivalis in cancer patients by meta-analysis methods.
Different databases including PubMed, EMBASE, the Cochrane Library, Scopus, and ISI web of Knowledge were investigated and eight relevant articles published in 2000–2013 were finally analyzed. Data were analyzed by meta-analysis method, fixed effect model. I² statistics were calculated to examine the heterogeneity of papers. The information was analyzed by R and STATA Ver 12.2.
A total of 711 people infected with P. gingivalis were included in this study. In total, the prevalence of P. gingivalis was 40.7% (95% CI, 19.3–62.1). The prevalence of P. gingivalis was evaluated in four case-control studies. The results of this study showed that P. gingivalis increased the chance of cancer development and periodontal disease as much as 1.36 times (OR, 1.36; 95%CI, 0.47–3.97).
Although there was no significant correlation between P. gingivalis and cancer, this bacterium increased the chance of cancer and periodontal disease and could be considered as a main potential risk factor.
Keywords
Introduction
Recent studies have reported the relationship between periodontal diseases and many systemic disorders such as cardiovascular diseases, diabetes, pulmonary diseases, cancers, and neonatal malformations prior to child birth. The rationale for these problems would probably be related to chronic and long-term aspects of inflammation in periodontal diseases.1,2
Porphyromonas gingivalis is an anaerobic gram negative bacterium which resides in the mouth and is highly associated with periodontal diseases. 3 Since this bacterium has a high genetic capability to develop resistance against any unfavorable interventional condition against its proliferation; it can also develop resistance against different currently used antibiotics. Most studies have shown that non-surgical attempts to remove P. gingivalis have failed due to the special niche of this bacterium (packets deeper than 5 mm).4,5 On the other hand, due to the painful outcome of surgical interventions and higher expenditures associated with these kinds of treatment, patients with periodontitis do not lean to select surgical treatment and therefore finding a non-surgical strategy for treatment of these patients is highly important. According to the studies from America and Europe, the most common antibiotics applied for the control of aggressive periodontitis in Iran, were combinations of amoxicillin and metronidazole. However, the prevalence of antibiotic resistance against these two antibiotics (particularly amoxicillin) is high among the Iranian population, due to the common prescriptions of these antibiotics by the physicians. 5 One of the most important points for antibiotic prescription in periodontal diseases is to choose narrow spectrum and not commonly used antibiotics. Many studies have reported the gastrointestinal adverse effects of antibiotic over- application. 6 It seems that P. gingivalis bacteria may be carcinogenic, since it activates some immunologic and inflammatory reactions in the host, creating some disturbances in the clearance of bacteria. 7 P. gingivalis has the capability to invade and penetrate different epithelial cells and possesses a complex mechanism that enables the bacterium to change the cellular defence, particularly, some special genes of the host. Some studies have shown the effects of P. gingivalis, in different levels, on some molecules associated with cellular cycles inside the cell. An important carcinogenic effect of P. gingivalis is the inhibition of epithelial cells’ apoptosis which is an intrinsic protective mechanism of cancerous cells.8,9 This process may be created via the prolongation of cell survival and increasing of the cell proliferation. 10 This bacterium has the ability to invade, proliferate, and survive inside the cytoplasm of infected cells and, thereby, spread to contiguous cells. 11 The prevalence rate of periodontal bacteria among cancer patients may be associated with patients’ health status, existence of any oral pathogenic condition, time of chemotherapy, surgery, disease stage, type of cancer, patients’ age, previous or current antibiotic therapy, et cetera. 12 Moreover, investigating the prevalence rate of periodontal agents such as P. gingivalis among cancer patients is important, since its findings can help physicians to control correlated periodontal complications and have a better view for the next treatment planning of cancer disease. There are little knowledge about the prevalence rate of P. gingivalis among cancer patients and this study aims to evaluate and compare the prevalence rate of P. gingivalis among cancer patients via a meta-analysis method.
Materials and methods
Search method
Motor searches of PubMed, EMBASE, the Cochrane Library, Scopus, and ISI web of Knowledge were interrogated to find the correlated studies. The key words of periodontal disease, Porphyromonas gingivalis bacteria, and cancer were used in this search and English language studies were identified by two independent researchers and the relevant studies were selected for evaluation. For a more comprehensive search, the references of identified papers were also searched.
Paper selection
First, a list of all the existing titles and paper abstracts were identified in the literature by the researchers and were evaluated for selection of the most correlated articles. Then, all the correlated studies were entered into the analysis. Out of the 57 evaluated studies, 17 were irrelevant or repeated. Among the 40 remained papers, 30 were inconsistent with the defined criteria of this study and the prevalence rate of the bacteria for two other studies were not reported. Finally eight consistent studies were selected and entered into the final analysis (Figure 1). The correlated information of these papers was entered into a prepared check list and was entered into the SPSS 16 for meta-analysis (Table 1). The criteria for the selection of appropriate papers in this meta-analysis were as follows: (1) randomized control trial (RCT) papers; (2) case-control studies; (3) the prevalence of P. gingivalis; and (4) different kinds of cancers. Repeated articles were removed.
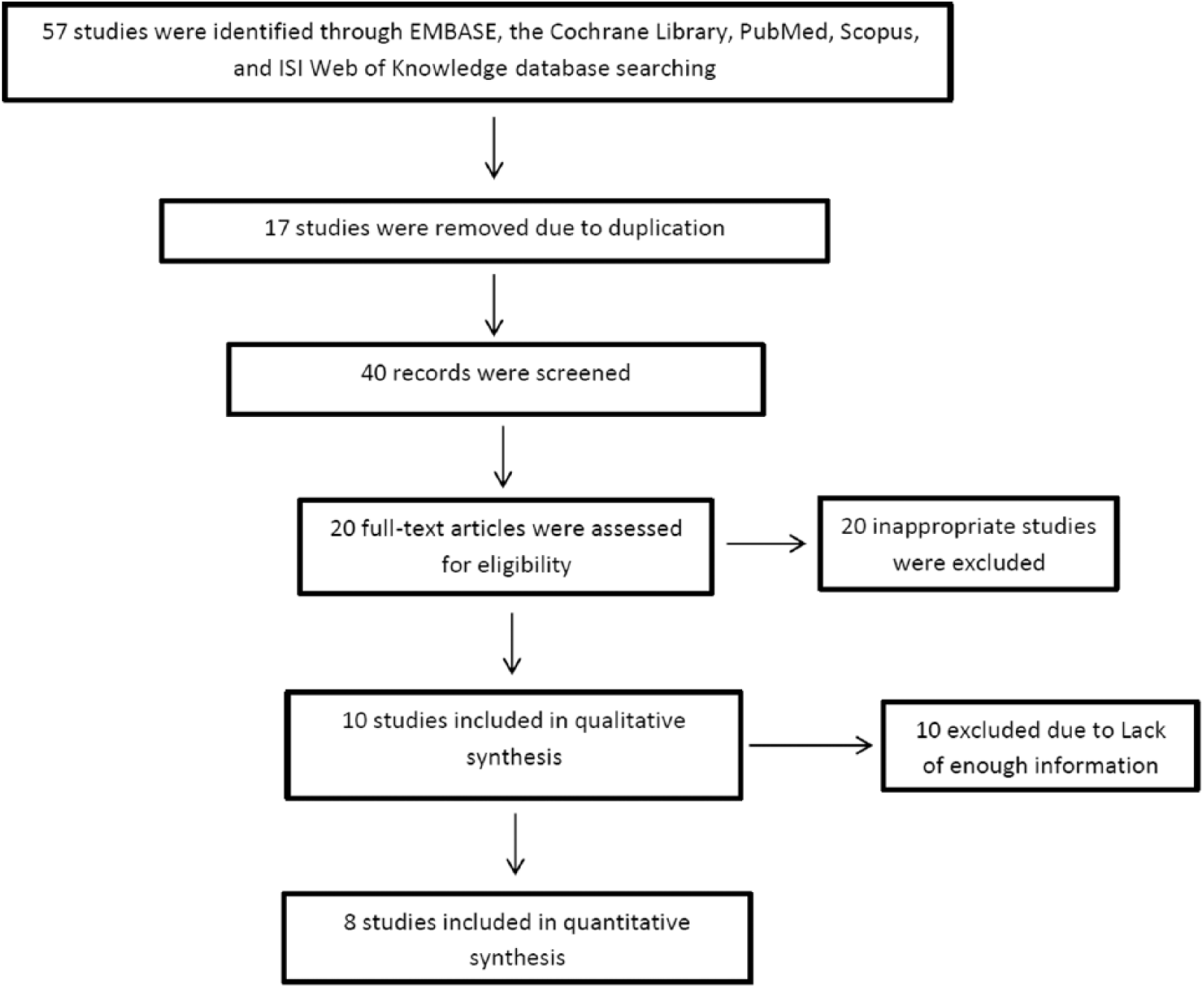
The flowchart of selected articles for final analysis.
General characteristics of selected studies for meta-analysis.

Data extraction
Data were extracted according to the following markers: (1) methodology of study; (2) type of cancer; (3) prevalence of P. gingivalis in the cases and control groups; (4) number of participants in each study and either in case or control groups; (5) type of sampling; (6) serotypes of P. gingivalis; and (7) name of author, year of publication, and the studied country. Abstracts and full text of all the studied papers were evaluated by two of our co-workers separately and in case of any inconsistency, both authors matched the results together and solved the discrepancy.
Statistical analysis
Since the main evaluated marker in this study was the prevalence rate of P. gingivalis, its variance was measured via binominal distribution and 95% confidence interval was considered for this analysis. To combine the different prevalence rates reported by papers, their mean weights were applied. Each study was dedicated a weight in accordance with inverted variance. For evaluation of no heterogeneity of data, Q test and I 2 marker with 10% error level were applied. In four case-control studies, odds ratio and 95% CI were calculated. In case of heterogeneity of results, data were analyzed using random effect model. Stata version 11/2 and R version 2/15/1 software were used for data analysis and all the P values were reported as two-tail figures and P <0.05 was considered as significant.
Results
In this meta-analysis, eight consistent studies published between 2000 and 2013 were identified and entered into the final analysis, to assess the prevalence of P. gingivalis among patients suffering from periodontal diseases (Figure 1). The total prevalence of P. gingivalis was 40.7% (95% CI, 19.3–62.1) (Figure 2). In four studies, the prevalence of P. gingivalis was reported among groups of cases and controls (Figure 3). The results of this study revealed that this relationship was not statistically significant; however, P. gingivalis increased the prevalence of cancer and periodontal diseases, most of which consequently resulted in gingival cancers; therefore, this bacterium could be considered as a potent risk factor. In other words, this study revealed that P. gingivalis increased the odds ratio of cancer and periodontal diseases as much as 1.36 (OR, 1.36; 95% CI, 0.47–3.97). According to the results of the Jamaican study, P. gingivalis increased the chance of periodontal diseases by 6.6-fold, which was statistically significant (OR, 6.6; 95% CI, 2.57–16.91). The publication bias figure showed that the bias effect among studies was not significant, since most of the studies were inside the funnel plot and it can be presumed that the results of most correlated studies have been entered into this meta-analysis (Figure 4).
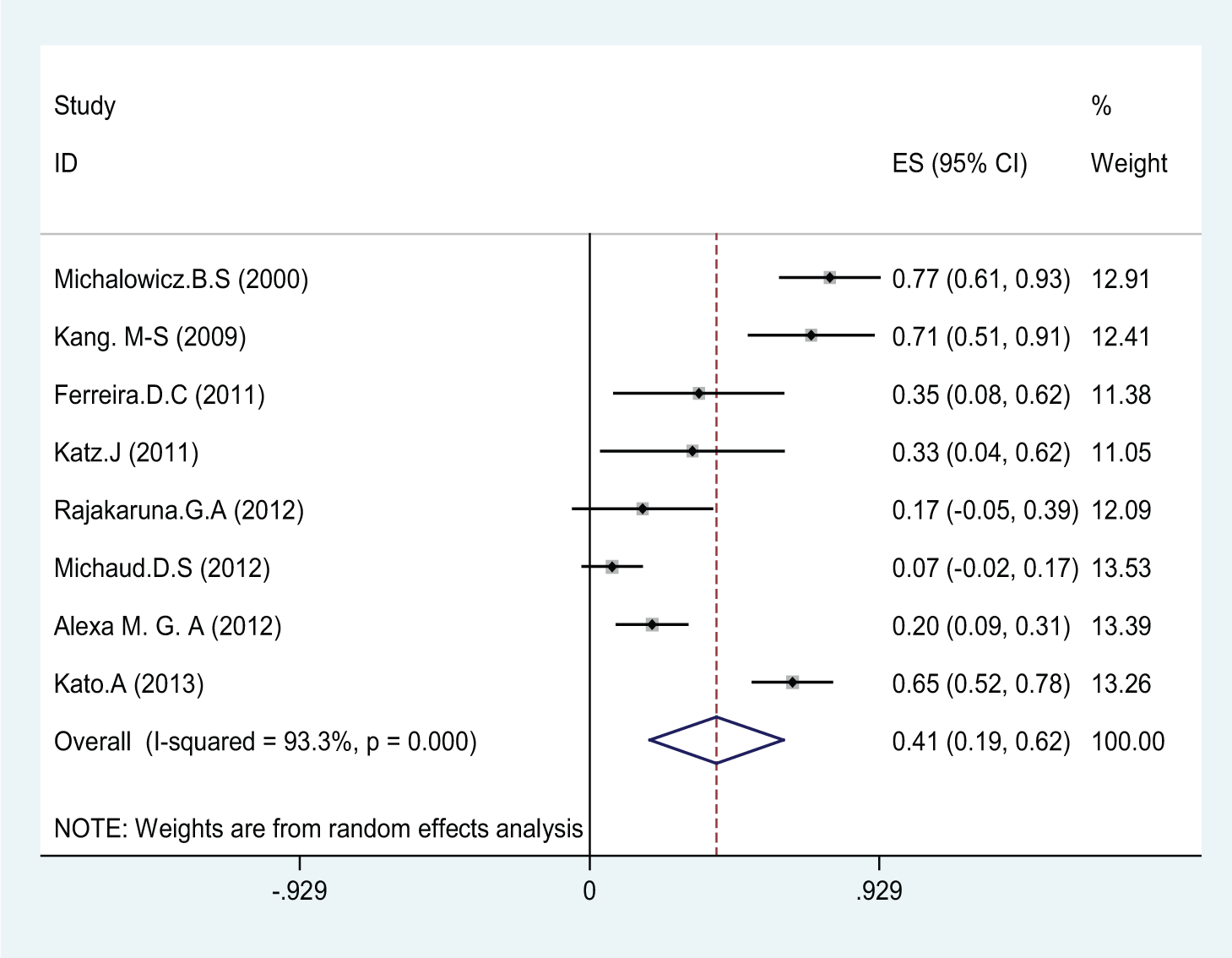
The prevalence rate of P. gingivalis among all selected studies upon random effects model. The middle point of each line indicates the prevalence rate and length of line indicates 95% confidence interval of each study. Rhombus shape indicates the prevalence rate for all studies.

Relationship between P. gingivalis and cancer and/or periodontal studies among groups of cases and controls and 95% CI upon random effects model. The middle point of each line indicates the prevalence rate and the length of line indicates 95% confidence interval of each study. Rhombus shape indicates 95% confidence interval for all studies.
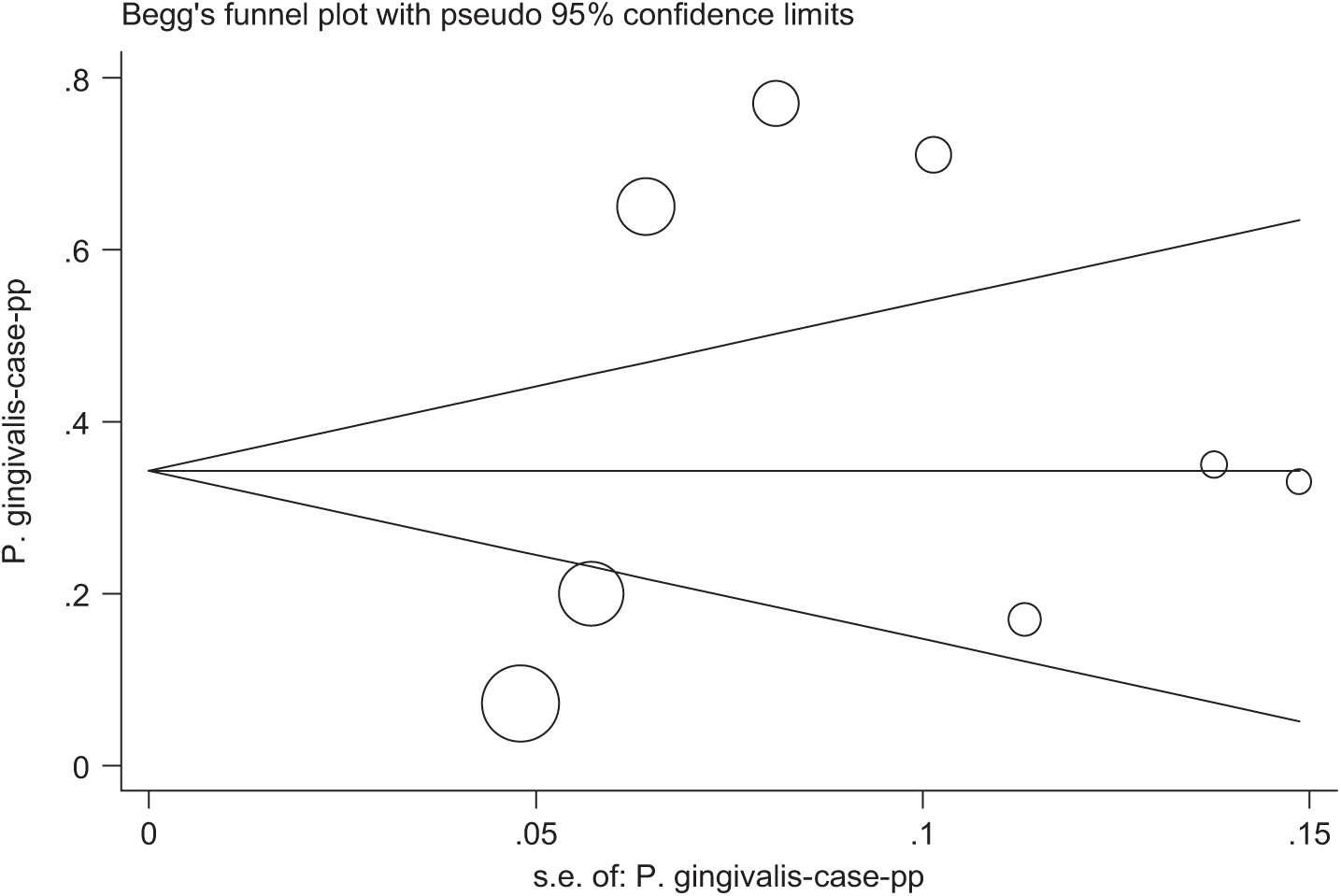
Begg’s funnel plot for publication bias in the risk difference (RD) analysis. Each circle represents the RDs for eradication success. The diameter of each circle represents the weight in the meta-analysis.
Discussion
Periodontal diseases are created by complex function of a group of anaerobic periodontal bacteria which are mostly gram negative.23,24 In the current study the relationship between P. gingivalis bacteria and cancer was investigated via meta-analysis of relevant papers. The results of this study revealed that the prevalence of P. gingivalis among cancer patients was significantly higher than healthy individuals. The results of our study were in accordance with those reported by Kutz and others in 2011. They reported that P. gingivalis may cause mouth cancer and showed that P. gingivalis induced the alteration of epithelial cells to neoplastic forms and its concentration rate was higher among cancer cells than normal tissues of mouth. 20 According to Kang et al.’s study, there was a significant difference between the group of head and neck cancer patients and group of healthy individuals for the frequency of P. gingivalis. Contrary to our results, Kang et al.’s study revealed a higher prevalence of P. gingivalis among healthy individuals than cancer patients (Figure 3). This discrepancy may be associated with different conditions such as patients’ health status and types of cancer treatment. 13 Also, the prevalence of P. gingivalis was compared between groups of solid and non-solid cancers by Kang et al. and it was revealed that its prevalence was higher among non-solid cancers. 12
Another study by Michaud and others showed that the prevalence rate of pancreatic cancer among patients with >200 ng antibody of P. gingivalis was two-fold higher than that among those with ⩿200 ng. This result may support the idea that periodontal diseases increase the risk of pancreatic cancer. 15 Their study also reported that higher serum levels of P. gingivalis (ATCC 53978) may be the best marker for the higher burden of the bacteria and invasive periodontal diseases which may justify why this bacterium is the only periodontal pathogen possibly correlated with higher risk of pancreatic cancer. Also, a recent study compared the saliva samples of healthy individuals and those with pancreatic cancer patients and reported that there was a significant relationship between higher prevalence of P. gingivalis and this type of cancer. 25 Different studies have investigated the possible relationship between mouth’s cancer and periodontal diseases26,27 and most of these studies have shown the positive relationship between these diseases. A study from Jamaica reported a positive relationship between the presence of P. gingivalis and periodontal diseases and also revealed that P. gingivalis was the most potent predictor for periodontal diseases among adolescents, 16 which was in accordance with our results. The importance of our findings about a higher prevalence rate of P. gingivalis in cancer tissue compared to healthy tissue is not certain; however, this bacterium invades both healthy and neoplastic tissues more than other bacteria such as S. gordonii. Meanwhile, P. gingivalis can intervene in the cancerogenesis process by inhibiting the host’s cellar death and induction of cell proliferation.
Some carcinogenic properties of P. gingivalis are as follows: it attaches to different lines of gingival cancer cells; 28 invades the epithelial cells of gingiva in experimental conditions; 29 it prevents and restrains apoptosis of epithelial cells; 30 it proliferates and survives in the epithelial cells and spreads to other cells; 31 it promotes cell survival and increases cellular proliferation; 32 it induces COX-2 gene expression; 33 and it increases the production of TNF-a, IL-6, IL-8, and IL-1b. 34 P. gingivalis possesses a lipoprotein and LPS on its external surface that can provoke proliferation of human fibroblast cells. 35
Evidences show that micro-RNA (miRNA) has an important role in the incidence of cancer. 36 During the inflammatory responses by macrophages, special types of miRNA are induced which have the ability to regulate the host’s cell responses against pathogenic agents. Furthermore, pathogenic agents themselves may regulate miRNA expression. For example, P. gingivalis can induce the expression of several miRNA among gingival epithelial cells. Totally, miRNA influences intrinsic and adaptive immunity control networks as well as apoptosis via regulation of signaling pathways such as TLR paths,37,38 signaling pathways of cytokine 37 and Stat 3. 39 Recent studies have reported that cells can secrete miRNA 40 and send it to the receiver cells, where it can influence the gene expression.41,42 Therefore, there is a probability that miRNA, associated with immunity, is transferred to neoplastic tissues, even if the pathogenic agents are located at another place.
There are differences between the prevalence rates reported by different studies for P. gingivalis that may be associated with the variations of P. gingivalis strains used in different studies. There are two completely different P. gingivalis strains which differ in several aspects. For example, the strain P. gingivalis ATCC53978 has a recognized capsule as the main antigen associated with pathogenesis 43 and the strain P. gingivalis ATCC33277does not have this antigen and, therefore, causes a slight inflammation.44,45
There were not enough data associated with age, gender, smoking, and alcohol consumption in this meta-analysis. These variables are important and may impress the ability of bacteria to invade the immune system during invasion to tissues and, therefore, potentially influence the malignancy process.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from The Clinical Microbiology Research Center, Ilam University of Medical Sciences, Ilam, Iran.
