Abstract
Keywords
Introduction
Heart failure (HF) is caused not only by coronary heart disease (CHD) (or ischemic heart disease) but also by cardiomyopathies. HF refers to the pathophysiological process in which the heart cannot pump the blood supply commensurate with venous reflux and body tissue metabolism; meanwhile, the cardiac output cannot meet the need for body metabolism. In patients with HF, abnormal water and salt retention usually occur due to cardiac dysfunction and cardiomyopathy. This is the pathophysiological process of most cardiovascular systems. HF has become a public health problem seriously threatening human health.1–3 According to the basic situation of heart disease, some factors can lead to HF. Dilated cardiomyopathy could be a major cause of HF.4–6 The most typical symptom of heart failure is dyspnea in varying degrees, which worsens during activity. In severe cases, sitting breathing, coughing, accompanied by a large amount of white velvet pink foam sputum, decreased appetite, and edema of the lower extremities.2,7–9 HF’s end-stage progresses rapidly, limiting the time-consuming diagnosis and treatment. Therefore, finding a reliable diagnostic marker is still important to seek effective early intervention in HF.
CORIN is a type II transmembrane serine protease mainly expressed in cardiomyocytes. It usually converts atrial natriuretic peptide precursor (ANP precursor) and brains natriuretic peptide precursor (BNP precursor) into active ANP and BNP, respectively.10–12 These active ANPs and BNPs play an essential role in reducing blood volume and blood pressure, regulating the balance of water and sodium in the body.13–15 ANP and BNP regulate the level of metabolites (such as cGMP) to improve systolic dysfunction and water retention associated with HF.16–18 Clinical and translational studies conducted over the past decade strongly support the pro-ANP convertase CORIN as a biomarker and effector of HF and myocardial infarction.16–29
CORIN is proteolytically cleaved from the cell membrane and released into circulation as soluble fragments of different sizes. Heart failure and acute myocardial infarction affect cardiac corin expression and shedding by different mechanisms.24,30,31 Plasma and cardiac CORIN levels decreased in patients with HF and animal models of HF.14,16,21,24,29,32
So far, several newly published randomized controlled trials have compared the evaluation of CORIN between heart failure group and control group. In order to fully evaluate the role of CORIN, relevant articles are included to evaluate CORIN levels from a different direction.
Methods
Literature search strategy
We systematically searched articles on the relationship between CORIN and HF published from 1 January 2000, to 1 January 2020. The search databases include PubMed, Cochrane database, EMBASE, and CNKI. The search keywords are as follows: (1) heart failure or HF; (2) CORIN; (3) Correlation or relationship. Boolean operators are used to combine grid terms and text words. Combine the three filters by applying Boolean “and” in turn. In order to maximize the scope of research, the author also studied the searching link list to find other relevant studies, not defined in the search policy. Access to information in the literature is not limited by publication date, language, and status.
Study selection
Included in the study if: (a) They are considered randomized trials or case–control studies. (b) They compared heart failure with the control group. (c) They analysed CORIN.
The study was excluded if: (a) They are case studies/meta-analyses/letters to editors. (b) There was no comparison between heart failure and the control group. (c) The patient did not receive CORIN analysis. (e) The data in the study are limited or insufficient. (f) They are replicas.
Data extraction and quality assessment
The two experts independently scanned the full text of the manuscript and extracted the following data from each qualified study: the first author’s name, the patient’s age and gender, country, year of publication, number of samples, and researching cycle of each article. The Cochrane bias risk assessment tool is used to assess the quality of methodological information. 33
Statistical analysis
Meta-analysis and related evaluations were performed using the statistical package Review Manager Version 5.2 (Copenhagen, Nordic Cochran center, Cochran collaboration, 2011). To ensure the continuity of the results, we calculated the effect size by dividing the standard deviation by the average difference between the groups. Studies with I2 statistics of 25% - 50%, 50% - 75% and >75% were regarded as low heterogeneity, medium heterogeneity and high heterogeneity, respectively. If I2 > 50%, sensitivity analysis was used to test the potential source of heterogeneity. Sensitivity analysis excluded one study in each cycle and assessed the effect of that study on the overall outcome. Furthermore, in the absence of heterogeneity, a fixed-effects model was used, whereas a random-effects model was used. Finally, a funnel was used to examine possible release bias.
Results
Data sources, retrieval and selection
The initial search found 905 relevant publications, of which 63 were excluded from duplicate publications. After filtering by title and summary, there were 49 articles left. Forty-two studies were excluded because of unsatisfactory article types and insufficient data. Finally, five articles18,29,34–36 were selected for meta-analysis. The research selection process was shown in Figure 1. Flow chart of study selection.
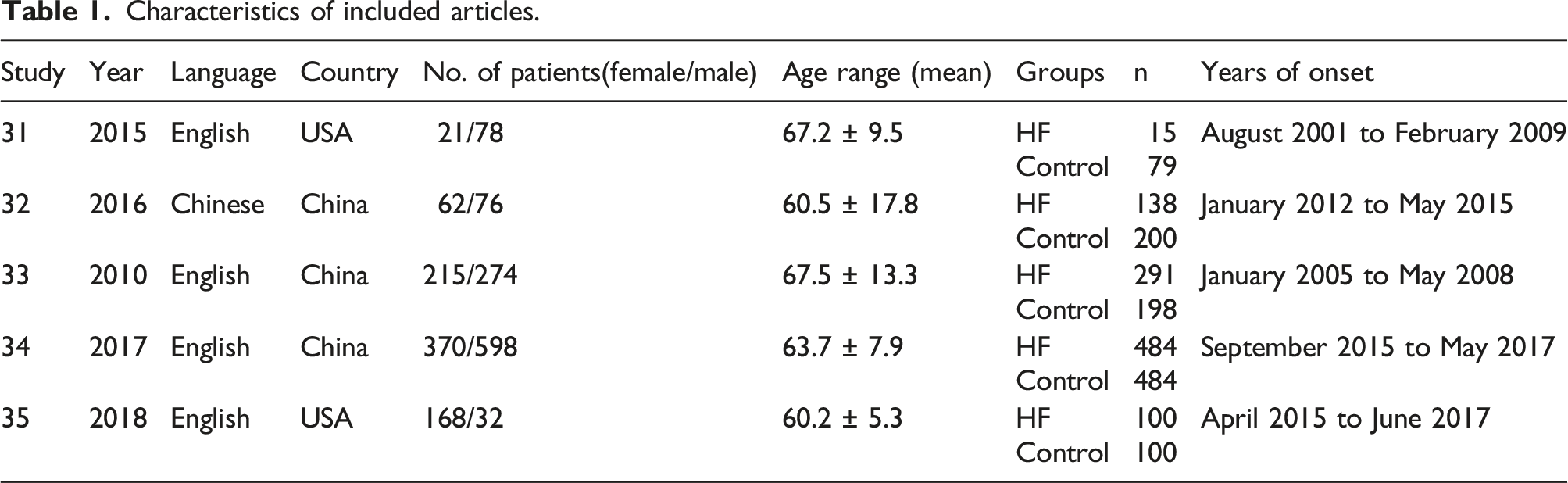
Characteristics of included studies
Characteristics of included articles.
The retrieval data quality ensured and found reliable
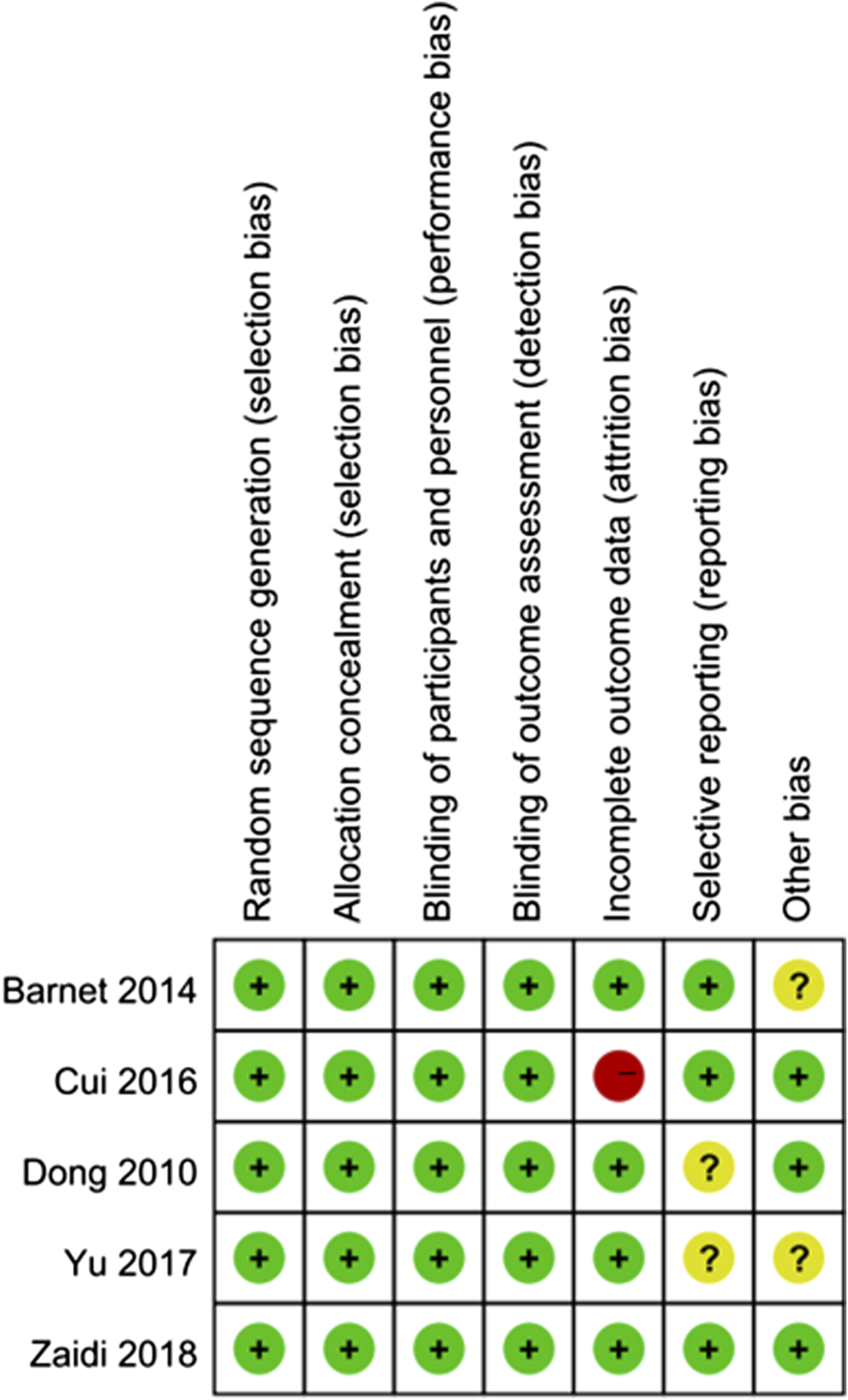
Review Manager 5.2 software was used for meta-analysis. The risk bias of this study was assessed using the bias assessment tool of Cochrane Collaboration. Figures 2 and 3 showed the evaluation in this study. As we can see, there were limited biases in the included articles. In general, only one trial had a risk of bias, and six trials had no risk. Only one study showed the problem of wear bias. The inherent deviation was inevitable by the backscattering method used in this study. Quality assessment summary, low deviation risk (green hexagon), unclear deviation risk (yellow hexagon) and high deviation risk (red hexagon). Quality assessment details.
The meta-analysis found CORIN level is statistically different in a different group
Figure 4 showed a forest diagram of CORIN levels between the HF group and the control group. This comparison involved seven studies. The Meta results of CORIN level were different (MD = −293.88, 95% confidence interval [-380.26, −207.49], p < 0.00001; heterogeneity p < 0.0001, I2= 97%). Forest diagram of Corin level between HF group and control group.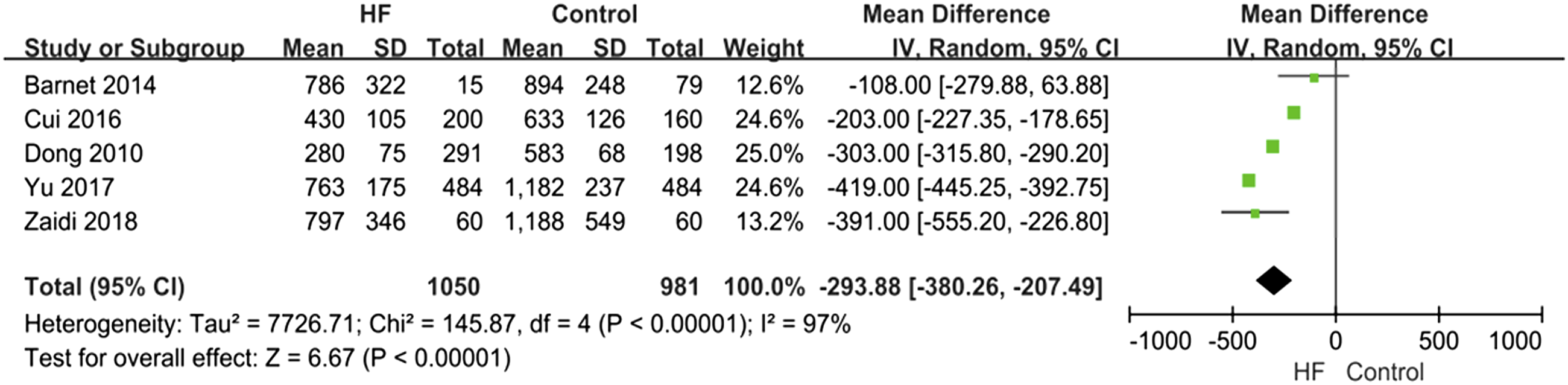
The CORIN forest between ischemic heart failure and non-ischemic heart failure was shown in Figure 5. The analysis showed that the CORIN of ischemic group was higher than that of non-ischemic group (MD = 88.79, 95% CI [70.46, 107.12], heterogeneity p < 0.0001, p = 0.94, I2= 0%). Corin forest diagram between ischemic heart failure and non ischemic heart failure.
Three articles were included in the CORIN subgroup analysis at different HF levels. The heterogeneity test results showed that there were significant differences in CORIN levels under three different HF levels. At HF level II, the total MD was −234 and the 95% CI was [- 406.49, - 61.50] (p = 0.0008, I2= 98%) and HF class III, the total MD was −337.11, and the 95% CI was [- 501.27, - 172.96] (p < 0.0001, I2= 98%), at HF level IV, the total MD was −439.37, and the 95% CI was [- 665.51, - 213.23] (p < 0.0001, I2= 99%) (Figure 6). Subgroup forest diagram of Corin analysis under different HF levels.
The result found with high sensitivity and low bias
In order to check the stability of the results, a sensitivity analysis was required. When Yu’s article was deleted, the results showed that in the heterogeneous part, I2 changed from 98% to 95%. This indicated that heterogeneity was stable (Figure 7). Sensitivity analysis of Corin level between HF group and control group.
To assess publication bias in this study, funnel plots were performed. The funnel plot results showed that there was no significance in publication bias (Figure 8). Publication bias funnel.
Discussion
Pro-natriuretic peptides are activated by the cardiac membrane protease known as CORIN. 35 The function of CORIN in pathological diseases like HF is yet unknown. In our study, we speculated on the presence of CORIN in human plasma, demonstrating the CORIN’s release from cells and subsequent circulation. The results indicated that plasma CORIN might be used as a biomarker to assess the status of HF or other cardiovascular diseases.
We compared plasma CORIN in the HF and control groups to test this hypothesis. According to our findings, CORIN levels in HF patients were considerably lower than in controls groups. However, there was a significant correlation between the reduction of CORIN levels and the severity of the decrease. In our study, we observed the control group’s mean age was lower than the HF group’s. Interestingly, plasma CORIN levels in healthy participants seemed to be greater in males than females, while the cause of this apparent gender difference is unknown. However, this difference between males and females did not achieve statistical significance in HF patients.
The heart is where CORIN is manifested most voluminously. 37 Recently reported that CORIN mRNA was only identified in the human heart. 37 Moreover, low quantities of mouse or rat CORIN mRNA have also been observed in various other tissues, including lung cancer cells, chondrocytes, developing kidneys, scar myofibroblasts, and certain brain regions.37–40 In the mouse, skin hair follicles had identified CORIN mRNA and protein. 41 It is yet unknown how CORIN works in these extra-cardiac tissues. The prolonged loss of cardiomyocytes, decreased CORIN synthesis in the heart or other tissues, accelerated clearance of plasma CORIN, and down-regulation of CORIN shedding that was linked to failing hearts are likely the causes of the low plasma CORIN levels observed in HF patients.
Our results showed that soluble CORIN was different in patients with ischemic and non-ischemic heart failure (Figure 5), although both groups were lower than those in the control group. This finding suggested that plasma-CORIN levels could be used to track acute coronary syndrome. The results included previous findings described by Zhou et al. 2016. 15 Soluble CORIN level was related to cardiac prognosis for a long time. 15 The results showed that low soluble CORIN levels were associated with a poor and slow prognosis of heart disease.
Our results show that soluble plasma CORIN can be used as a potential biomarker in clinical practice. In addition, soluble cortisone may also be considered a drug for treating heart failure. However, there are some challenges in using CORIN as a biomarker or therapeutic agent. First, most studies are case studies, lacking the details of CORIN and heart failure mechanism. Second, how do we separate the normal function of CORIN from the role of heart failure? Third, the study used cardiac samples from patients who were not prospectively enrolled but were simply available for evaluation; cardiac samples and HF were not correctly described. Thus, cardiac samples were defined as from the patients with dilated cardiomyopathy and at the same time described as “hypertrophic.” Therefore, this study is not suitable for analysis. 42 Therefore, in order to better understand the relationship between HF and CORIN level, CORIN activity measurement and CORIN level are needed to be considered. Fourth, cardiovascular disease is widely related to demographic parameters, such as age, gender, race, income, occupation, marital status, social status, stress, etc. Before accepting CORIN as a biomarker, more researches in field in detail are necessary.
Limitations
There are several limitations of this current study. First, the included studies are heterogeneous, including different types of diseases, various donor sources, different types of drugs used by patients, and different doses. Secondly, more studies from other countries can be analyzed in the following research. Thirdly, the study countries were limited, and we could include more articles in various countries to guarantee the representativeness. Fourthly, the heterogeneity was obvious in this research, which we need include much more eligible articles to reduce it in the future.
Conclusion
In our current meta-analysis study showed that, HF and CORIN have a substantial association in the progression and diagnosis of HF. The results are consistent with previously published articles. There was little evidence of publication bias, and the study was robust. Consequently, it may be concluded that CORIN may aid in the diagnosis of HF and that it is closely associated to HF. Given the limitations of this investigation, more RCTs with high standards, big sample sizes, multi-centres’, and assessment criteria that were mostly consistent were still required to confirm the findings and offer solid support for the superiority of CORIN in the management of HF.
Footnotes
Acknowledgements
We sincerely thank to the Copenhagen Nordic Cochran center for providing permission to use the Cochrane bias risk assessment tool.
Author contribution
ZH and JN participated in the conception and design of the study, library searches and assembling relevant literature, critical review of the paper, supervising the writing of the paper, and database management. JB, ZX and WY are participated in data collection, library searches and assembling relevant literature, writing the paper, and critical review of the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Talents Training Program of the Seventh People’s Hospital, Shanghai University of Traditional Chinese Medicine BDX2020-02 and The Outstanding Clinical Discipline Project of Shanghai Pudong PWYgy2018-05.
Ethics approval and consent to participate
This study is a Meta analysis. We have obtained a statement from the ethics committee of Seventh People’s Hospital of Shanghai University of Traditional Chinese Medicine that ruled that no formal ethics approval was required in this particular Meta analysis.
