Abstract
Arthritis is a chronic condition worsened by systemic inflammation, inactivity, poor nutrition, and disrupted sleep, yet it is often managed reactively with symptom focused pharmacologic care. Emerging evidence suggests lifestyle interventions may target root causes of disease, offering a proactive approach to reduce pain and preserve joint function. This narrative review examined the impact of lifestyle interventions on arthritis-related pain and joint health, focusing on osteoarthritis and rheumatoid arthritis. A search of PubMed, EMBASE, Cochrane Library, and Google Scholar (2000 to 2025) identified randomized trials, meta-analyses, and cohort studies on nutrition, supplementation, physical activity, and sleep. Outcomes included pain and function scores, inflammatory biomarkers, and imaging markers of joint integrity. Lifestyle interventions reduced pain, improved function, and supported joint health. Anti-inflammatory diets, such as the Mediterranean diet, were linked to lower inflammation and symptom severity. Supplements like curcumin, glucosamine, and Boswellia showed modest benefits with favorable safety. Resistance training and Tai Chi improved strength, mobility, and pain, with added benefits for balance. Poor sleep was associated with greater pain and inflammation, while behavioral sleep interventions improved outcomes. Lifestyle medicine is safe, effective, and feasible to integrate into arthritis care, with strong support for diet and movement therapies.
“Addressing diet, exercise, sleep, and supplementation as discrete therapeutic targets may improve patient-reported outcomes, reduce medication burden, and mitigate comorbid disease risk.”
Introduction
Arthritis is a progressive, disabling condition that affects hundreds of millions worldwide and contributes substantially to pain, mobility loss, and diminished quality of life. 1 Traditionally framed as an inevitable consequence of aging or mechanical “wear and tear,” arthritis is now increasingly understood to be a chronic, multifactorial disease driven by systemic inflammation, metabolic dysregulation, oxidative stress, and lifestyle-related factors. 2 Despite this evolving understanding, current clinical approaches remain predominantly reactive, centered on pharmacologic symptom control, procedural interventions, or joint replacement, rather than disease prevention.3,4 A growing body of evidence highlights the powerful role of lifestyle behaviors, such as diet quality, physical activity, sleep patterns, and supplements in shaping the trajectory of arthritis. 5 As the burden of arthritis rises in parallel with aging populations and lifestyle-related chronic disease, there is an urgent need to promote integrative treatment models alongside traditional care. This review explores the 4 most common, accessible, and robustly researched lifestyle interventions as an alternative therapeutic strategy for arthritis and joint pain.
Objectives
This narrative review aims to summarize and synthesize the current evidence supporting lifestyle-based interventions in the management of arthritis, with a focus on dietary patterns, physical activity, sleep optimization, stress reduction, and targeted supplementation. We explored the biological mechanisms through which these lifestyle factors modulate systemic inflammation, joint integrity, and disease progression. We assess the clinical relevance and feasibility of integrating these interventions into a patient-centered, non-pharmacologic model of arthritis care that prioritizes long-term function, quality of life, and whole-person health.
Methods
A narrative review methodology was selected to allow for integrative, cross-disciplinary analysis of heterogeneous evidence spanning clinical trials, biological mechanisms, and behavioral interventions. This approach supports a multidimensional synthesis relevant to both research and real-world arthritis care.
A comprehensive search of PubMed, EMBASE, Cochrane Library, and Google Scholar was conducted to identify relevant studies published between January 2000 and February 2025. Search terms included combinations of: “osteoarthritis,” “rheumatoid arthritis,” “arthritis” “joint pain,” “inflammation,” “Mediterranean diet,” “curcumin,” “omega-3,” “glucosamine,” “collagen,” “Boswellia,” “resistance training,” “aerobic exercise,” “walking,” “running,” “Tai Chi,” “sleep,” and “lifestyle medicine.” Reference lists of included articles were screened manually to identify additional sources. The search was restricted to English-language publications, and gray literature was excluded.
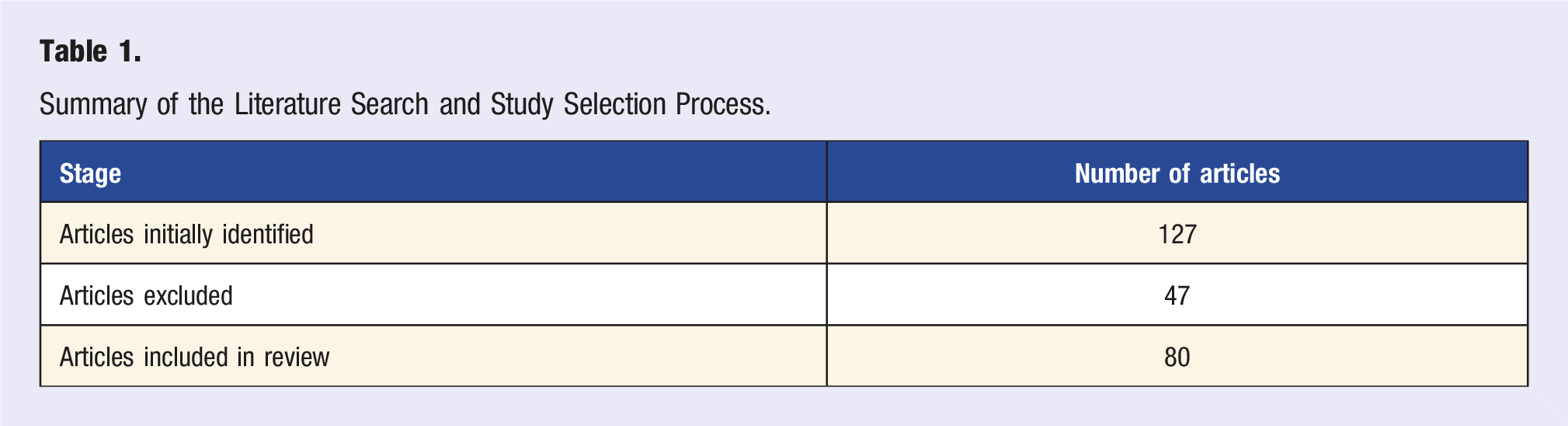
Studies were eligible for inclusion if they evaluated non-pharmacologic interventions targeting arthritis-related outcomes across 4 domains: (1) nutrition, (2) supplementation, (3) physical activity, and (4) sleep. Eligible study designs included randomized controlled trials, meta-analyses, systematic reviews, and prospective cohort studies. Preclinical and non-systematic literature was included selectively when contributing material insight into pathophysiological mechanisms or informing clinical context. Studies focused exclusively on surgical or pharmaceutical interventions were excluded. We included 80 studies altogether.
Primary outcomes of interest were changes in validated arthritis pain and function scales, including the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Knee injury and Osteoarthritis Outcome Score (KOOS), Visual Analog Scale (VAS), and Health Assessment Questionnaire (HAQ), as well as objective biomarkers of inflammation (e.g., C-reactive protein [CRP], interleukin-6 [IL-6], tumor necrosis factor-alpha [TNF-α]) and imaging-based assessments of joint structure. Studies were appraised for methodological rigor, reproducibility, effect magnitude, and translational feasibility.
We grouped findings by type of intervention and also looked at how different interventions interact and share biological effects. We focused on treatments that affect multiple body systems and could reduce the need for medications or surgery. While this review does not cover all existing studies, we selected papers based on relevance, consistency of themes, and contribution to the overall understanding. Throughout the process, we followed established guidelines for conducting a high-quality narrative synthesis.6,7
Summary of the Literature Search and Study Selection Process.
Results
Diet
Growing evidence supports the role of diet as a modifiable factor in the prevention and management of osteoarthritis (OA) and inflammatory arthritis (IA), including rheumatoid arthritis (RA). Recent randomized controlled trials, observational cohorts, and Mendelian randomization studies have highlighted the impact of specific dietary patterns, food groups, and nutrient profiles on inflammatory processes, symptom burden, and long-term outcomes. While the magnitude of effect varies, dietary interventions generally demonstrate consistent benefits across inflammatory markers, pain scores, and disease progression. We included 11 studies related to diet.
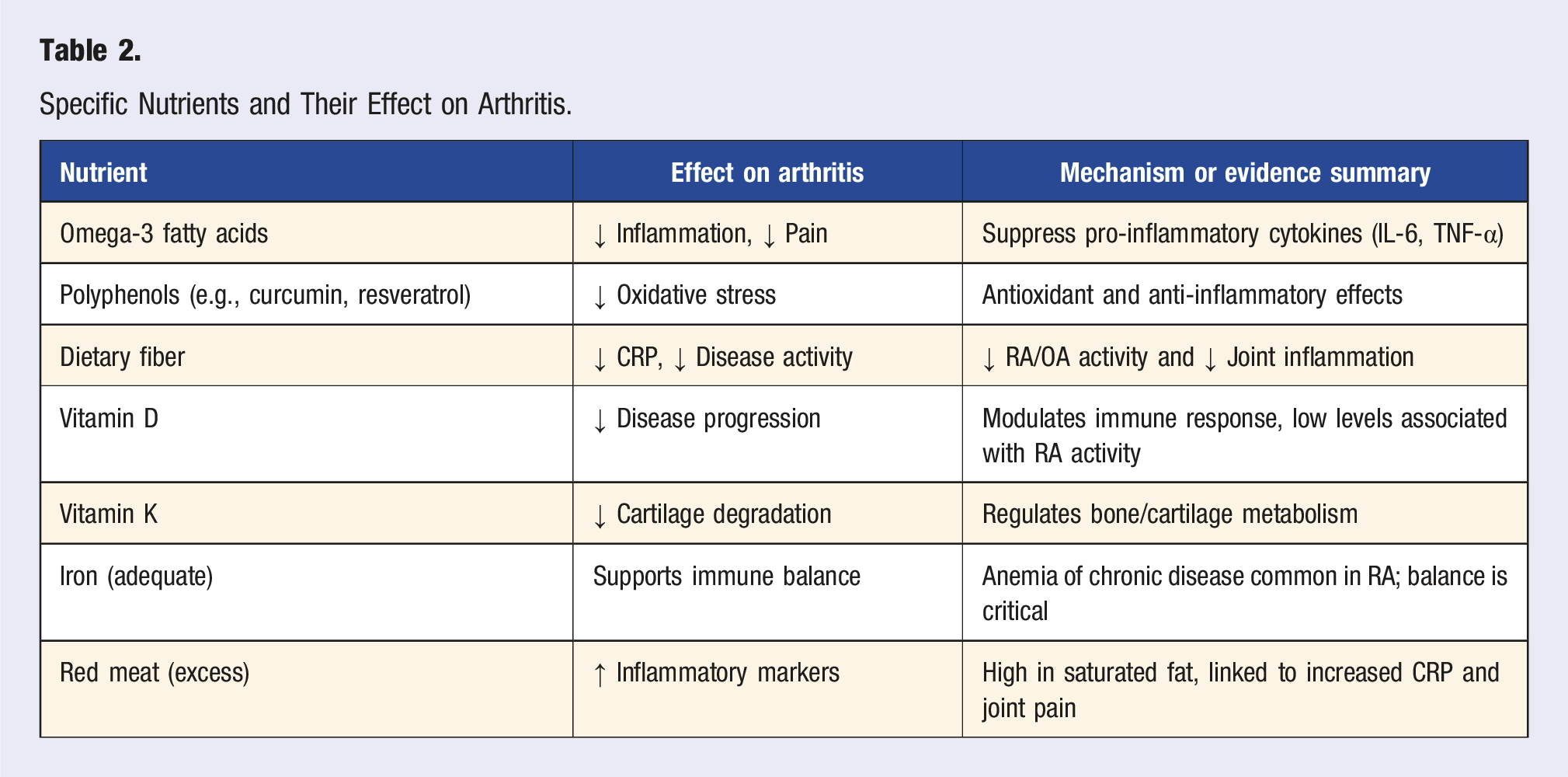
Several studies support the idea that adherence to anti-inflammatory dietary patterns is associated with lower systemic inflammation and improved arthritis symptoms. In a randomized controlled trial (n = 144), Law et al 8 (2025) found that patients with chronic musculoskeletal pain and metabolic syndrome who had higher Dietary Inflammatory Index (DII) scores, reflecting more pro-inflammatory diets, had significantly higher pain scores. The Dietary Inflammatory Index (DII) is a validated scoring system that quantifies the inflammatory potential of an individual’s diet based on the intake of specific nutrients, foods, and bioactive compounds. It was developed through a comprehensive review of nearly 2000 peer-reviewed articles linking dietary components to 6 inflammatory biomarkers (IL-1β, IL-4, IL-6, IL-10, TNF-α, and CRP). Higher DII scores indicate a more pro-inflammatory diet, whereas lower (more negative) scores reflect anti-inflammatory dietary patterns. In a comprehensive umbrella review of 14 meta-analyses, Cheng et al 9 (2025) found that polyunsaturated fatty acids (PUFAs), probiotics, and traditional anti-inflammatory diets (AIDs) significantly reduced RA disease activity scores, though most included studies were of low-to-very low quality, underscoring the need for higher-quality trials. Similarly, Sharma et al 10 (2024) reviewed 9 systematic reviews and found modest benefits of Mediterranean, vegetarian, and fasting diets in improving pain, stiffness, and inflammation, particularly through modulation of the gut microbiota and oxidative stress pathways.
Dietary fiber, particularly from cereal sources, appears to have a protective effect against RA, largely mediated by reductions in dietary inflammation. In a cross-sectional analysis of 15 114 NHANES participants (2011-2020), Wan et al 11 (2024) found that each 5 g/day increase in cereal fiber intake was associated with lower RA prevalence, and this association was significantly mediated by the DII. This highlights cereal fiber’s potential as a modulator of inflammatory load, commonly derived from whole grains such as oats, barley, and wheat bran. In a cross-sectional analysis of 37 275 NHANES participants (2001-2018), Zhu et al 12 (2025) found a higher intake of ultra-processed foods (UPFs) was significantly associated with increased odds of self-reported arthritis, particularly rheumatoid arthritis (RA). Each 10% increase in UPF consumption was linked to 4% higher odds of arthritis and 6% higher odds of RA. No significant association was found for osteoarthritis. The association was strongest among individuals who were overweight or obese.
At the nutrient level, PUFAs, fiber, phenols, and certain micronutrients show consistent anti-inflammatory effects. In an RCT involving 44 women with RA, Tavakoli et al 13 (2025) found that 16:8 intermittent fasting significantly reduced markers of oxidative stress (MDA), inflammation (NLR), and liver enzymes (AST/ALT), while increasing catalase levels, suggesting improved redox balance. In a meta-analysis of 8 RCTs (n = 778), Stanfar et al 14 (2024) reported that diets high in omega-3 fatty acids and phenols significantly reduced pain and improved physical function, with the most effective interventions being those combining multiple anti-inflammatory nutrients. In preclinical work, Búr et al 15 (2025) demonstrated that time-restricted feeding (TRF) reduced joint inflammation and IL-1β expression in a mouse model of arthritis exacerbated by a high-fat diet.
Large-scale observational studies consistently associate whole-food diets, especially those rich in fruits, vegetables, whole grains, and healthy fats, with reduced arthritis prevalence and severity. Bonaccio et al 16 (2014) analyzed data from 14 586 Italians and found that adherence to a Mediterranean diet was associated with lower white blood cell and platelet counts, indicating lower systemic inflammation. The protective effects were largely attributed to fiber, polyphenols, and antioxidants. Dong et al 17 (2024) conducted a meta-analysis of 2.99 million participants across 30 cohort studies and reported that higher consumption of fruits, cereals, and oily fish was significantly associated with lower RA risk, while tea and processed meats were associated with increased risk. Moderate alcohol consumption showed a modest protective effect (RR range: 0.96-0.97) that may be due to unmeasured lifestyle or socioeconomic factors. In an analysis of 109 591 women in the Women’s Health Initiative, Malani et al 18 (2024) found that higher Healthy Eating Index (HEI-2015) scores were associated with up to 19% lower risk of developing RA, particularly among postmenopausal women. Protective components included fruits, vegetables, and whole grains, while saturated fats and added sugars were linked to higher risk.
Specific Nutrients and Their Effect on Arthritis.
Dietary-Related Factors Effect on Arthritis.

Conclusion
The collective evidence suggests that dietary interventions, especially those emphasizing anti-inflammatory and whole-food patterns, can reduce arthritis risk, modulate systemic inflammation, and potentially improve survival in patients with inflammatory arthritis. While heterogeneity in dietary protocols and study designs persists, the consistency of findings across observational, interventional, and genetic studies strengthens the case for integrating dietary strategies into arthritis prevention and management. Future trials should focus on dose-response effects, patient-specific dietary responsiveness, and the synergy between diet and other lifestyle interventions.
Omega-3 Fatty Acids
Omega-3 polyunsaturated fatty acids (PUFAs), particularly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), have been extensively studied for their anti-inflammatory, metabolic, and chondroprotective properties in osteoarthritis (OA) and inflammatory arthritis (IA). While some evidence suggests omega-3s may modulate systemic inflammation and improve symptoms, findings remain mixed, with inconsistent results in OA but more favorable effects in RA. We included 8 studies related to omega-3s.
Observational studies suggest an inverse association between omega-3 intake and OA prevalence, particularly in middle-aged populations. A cohort study of 3467 NHANES participants by Huang et al 19 (2024) found that higher dietary omega-3 intake was associated with lower all-cause mortality (HR: 0.59-0.93, P = .011) in OA patients, though no significant effect was found for cardiovascular-specific mortality. Similarly, a meta-analysis of 22 834 NHANES participants by Liu et al 20 (2024) found that omega-3 intake was inversely associated with OA prevalence in adults aged 40-59 (OR: 0.85, P = .027). However, a Mendelian randomization (MR) analysis failed to establish a causal relationship, suggesting that omega-3 intake may be a marker of overall dietary quality rather than a direct protective factor. Conversely, a serum biomarker study by Felson et al 21 (2024) (n = 658) found no significant association between circulating omega-3 levels and OA risk, reinforcing the possibility that dietary patterns, rather than omega-3s alone, may influence OA development. Further supporting this, a genome-wide association study (GWAS) by Shi et al 22 (2025) analyzing 9 OA subtypes found no causal effect of dietary PUFA intake on OA risk, challenging the notion of omega-3s as a primary modulator of OA pathogenesis.
Despite the lack of strong causal evidence for OA prevention, omega-3s consistently show benefits in reducing inflammatory biomarkers and improving lipid profiles. A meta-analysis of 18 RCTs (n = 1018 RA patients) by Wang et al 23 (2024) found that omega-3 supplementation significantly increased serum EPA/DHA levels, lowered the omega-6: omega-3 ratio, and reduced triglycerides, but had only modest effects on RA disease activity scores. A broader review by Gammone et al 24 (2019) examined omega-3’s role in exercise-induced inflammation and found that EPA/DHA supplementation reduced muscle soreness, improved immune function, and modulated inflammatory responses, suggesting potential adjunct benefits in arthritis patients engaging in regular physical activity.
Evidence regarding omega-3’s effects on joint pain and function in OA remains inconsistent. A meta-analysis of 52 RCTs by Mathieu et al 25 (2022) concluded that omega-3 supplementation did not significantly improve OA-related pain or function compared to placebo, contradicting prior small-scale studies suggesting mild benefits. However, some systematic reviews suggest a potential role for omega-3s in RA management when combined with dietary interventions. A systematic review protocol by Raad et al 26 (2020) is currently assessing the effects of dietary interventions with or without omega-3 supplementation in RA management.
Figure 1 illustrates omega-3 effects on arthritis Omega-3 effects on arthritis.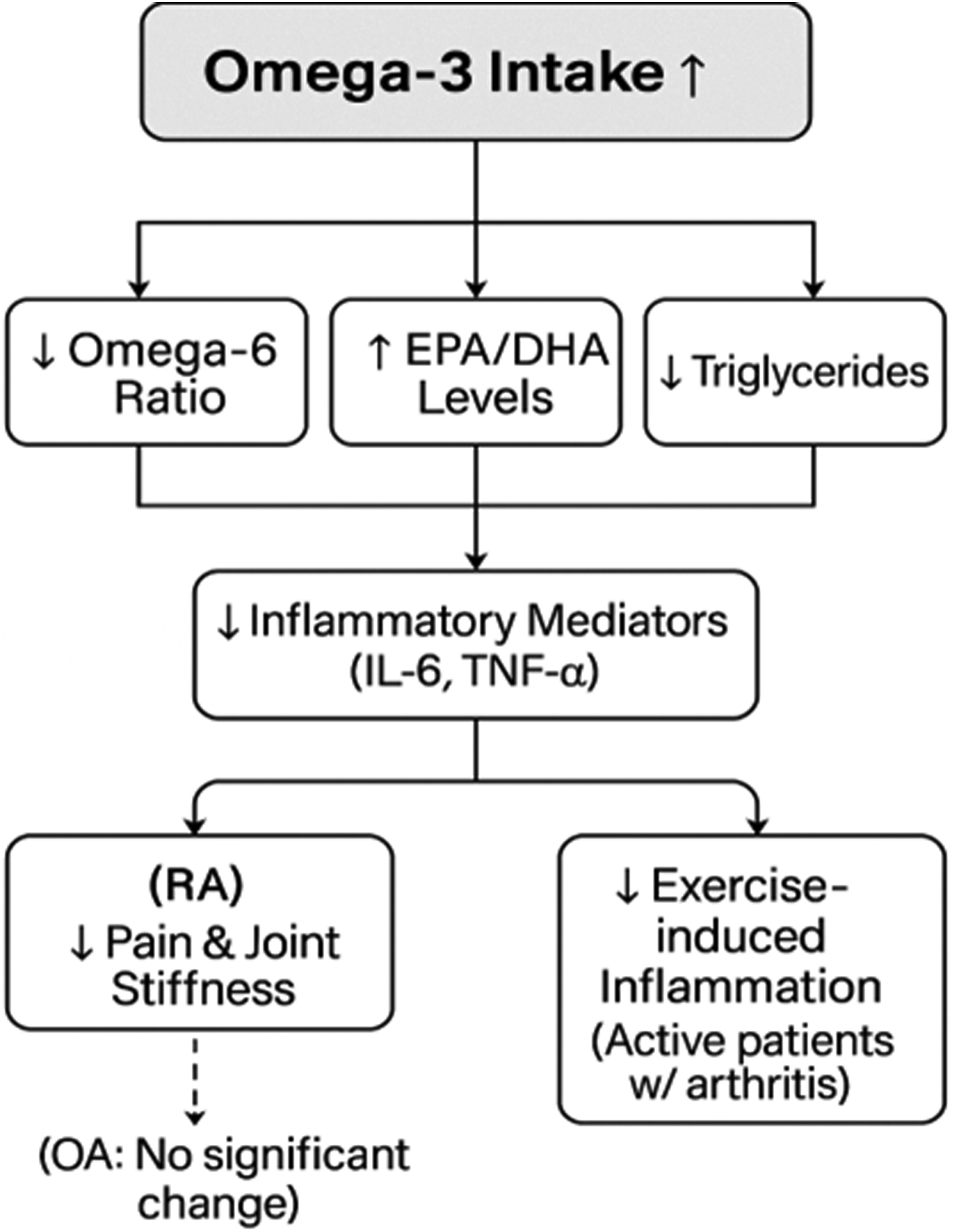
Conclusion
While omega-3 fatty acids exhibit strong anti-inflammatory and metabolic effects, their role in OA prevention and treatment remains uncertain, with limited evidence for direct symptom improvement. In contrast, omega-3 supplementation appears to offer modest benefits in RA, particularly in reducing lipid-associated inflammation. Future research should focus on targeted omega-3 dosing, synergistic dietary interventions, and long-term clinical outcomes to better elucidate its role in arthritis management.
Glucosamine and Chondroitin Sulfate
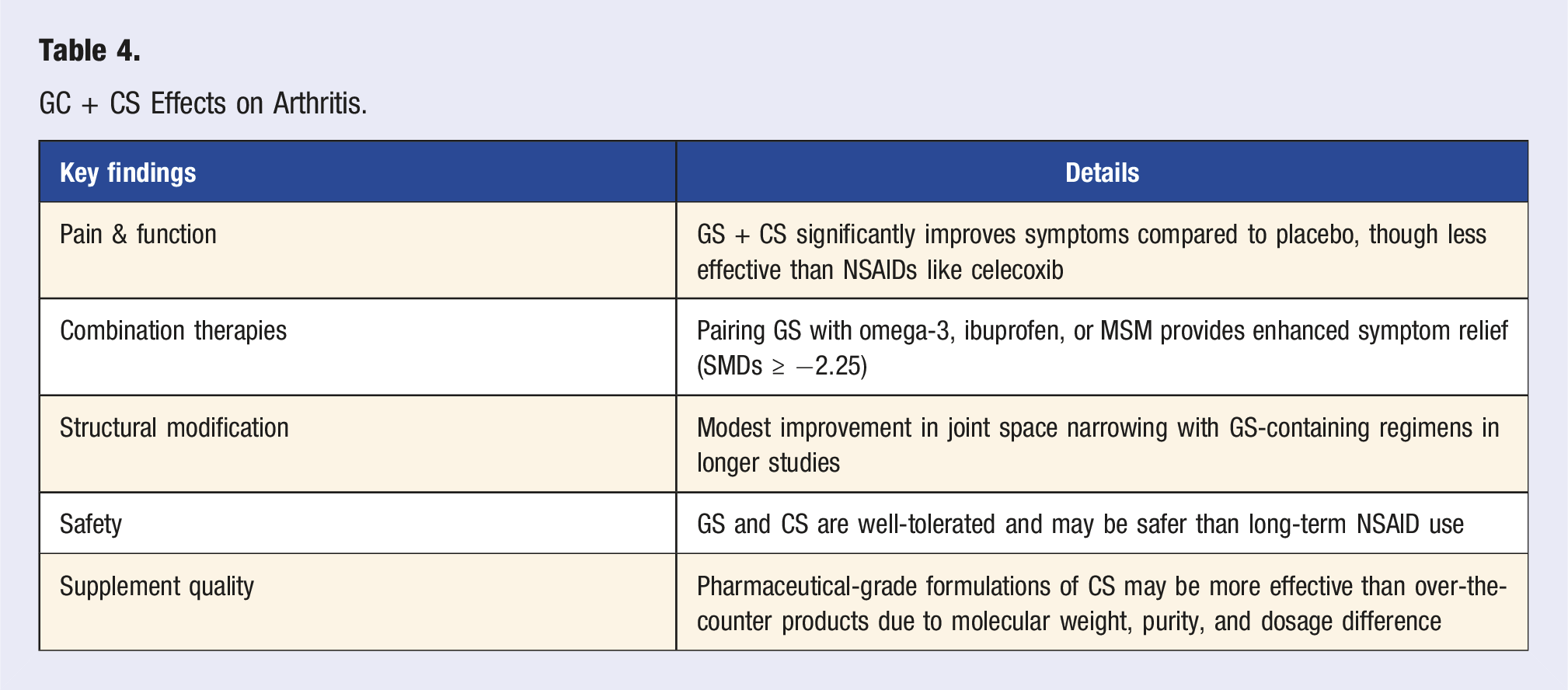
Glucosamine (GS) and chondroitin sulfate (CS) are widely used symptomatic slow-acting drugs for osteoarthritis (SYSADOAs). Despite continued use globally and inclusion in various guidelines, evidence on their clinical efficacy remains mixed. Recent large-scale analyses and network meta-analyses provide a more nuanced view of their role in OA management, suggesting selective benefits, particularly in combination therapies and long-term use. We included 10 studies related to GS and CS.
Several meta-analyses and RCTs indicate that glucosamine and chondroitin, especially in combination, can improve pain and joint function in patients with knee and/or hip OA. A network meta-analysis of 61 RCTs by Zhu et al 27 (2018) found that celecoxib was most effective for pain relief (SMD: −0.32; 95% CI: −0.38 to −0.25), followed by the combination of GS + CS. For stiffness, both glucosamine (SMD: −0.36; 95% CI: −0.67 to −0.06) and celecoxib showed statistically significant improvement over placebo. A similar conclusion was drawn in the systematic review of 54 studies (n = 16 427) by Zeng et al 28 (2015), which showed that GS + CS improved both pain and physical function scores compared to placebo, though celecoxib remained more effective overall for symptom control.
A recent network meta-analysis of 30 RCTs (n = 5265) by Sumsuzzman et al 29 (2024) evaluated different glucosamine-based combination therapies. The most effective pain-relieving interventions were Glucosamine + omega-3: SMD: −2.59 (95% CI: −4.42 to −0.75), Glucosamine + ibuprofen: SMD: −2.27 (95% CI: −3.73 to −0.82), Glucosamine + chondroitin + methylsulfonylmethane (MSM): SMD: −2.25 (95% CI: −3.84 to −0.67. These combinations exceeded the minimum clinically important difference (MCID) threshold of SMD ≥0.40 and were rated as moderate to low quality evidence, indicating a high potential for symptom relief without relying solely on NSAIDs.
Some evidence suggests that glucosamine may help slow joint space narrowing (JSN), though results are less consistent. In an analysis of 8 RCTs (n = 3793), Meng et al 30 (2023) found that GS + CS significantly reduced WOMAC total and stiffness scores, with modest improvements in joint space narrowing, especially in long-duration studies exceeding 6 months. Similarly, Rabade et al 31 (2024) found that glucosamine monotherapy was associated with a significant reduction in joint space narrowing, while chondroitin was more effective in improving pain and physical function. However, the GS + CS combination did not consistently outperform individual therapies.
One of the most consistent findings across studies is the favorable safety profile of GS and CS, especially compared to NSAIDs. In a systematic review of 15 RCTs, Vo et al 32 (2023) reported that glucosamine showed superior tolerability, with an SMD of −7.41 vs placebo for pain, though heterogeneity was high. Similarly, in a pilot RCT of 51 patients, Minoretti et al 33 (2024) found that GS + CS significantly improved WOMAC scores, with oral hyaluronic acid (HA) outperforming GS + CS in pain relief, but not in safety. Compliance was also high in a small observational study by Ayhan et al 34 (2024), which used an 8-week regimen of GS + CS + MSM + collagen, leading to significant reductions in VAS, WOMAC, and HAQ scores with minimal side effects.
A preclinical study by Malaguez et al 35 (2024) using a rabbit model of temporomandibular joint (TMJ) OA showed that GS + CS reduced TNF-α levels and preserved collagen, supporting a potential disease-modifying effect at the cartilage level. However, a narrative review by Brito et al 36 (2023) emphasized that supplement quality, particularly pharmaceutical-grade CS, may significantly influence outcomes. Variability in molecular weight, purity, and dosage complicates the interpretation of clinical trial data.
GC + CS Effects on Arthritis.
Conclusion
Glucosamine and chondroitin, particularly in combination with other anti-inflammatory agents, demonstrate moderate efficacy for symptom relief in knee and hip OA, with a favorable safety profile and high patient compliance. Although not as potent as NSAIDs, they remain a viable non-pharmacologic option, particularly for patients with contraindications to traditional pain medications. Further research should emphasize standardized supplement formulations, long-term outcomes, and patient-specific response profiles to optimize their role in OA management.
Collagen
Collagen, a structural protein composed of triple-helical polypeptide chains, has emerged as a promising non-pharmacologic intervention in osteoarthritis (OA) management. Collagen is enzymatically broken down during digestion into bioactive peptides: types I, type II, and hydrolyzed forms. These peptides serve as building blocks for connective tissue repair and may also exert immunomodulatory effects that reduce inflammation and support joint health. Clinical and preclinical studies suggest potential benefits for pain relief, functional improvement, and cartilage protection, with a favorable safety profile. However, findings vary depending on the collagen subtype, dosage, duration, and formulation used. We included 7 studies related to collagen.
Recent meta-analyses demonstrate that collagen supplementation leads to modest but statistically significant improvements in OA-related pain and physical function. A comprehensive meta-analysis of 35 RCTs involving 3165 OA patients by Liang et al 37 (2024) found that collagen reduced pain with a standardized mean difference (SMD) of −0.35 (95% CI: −0.51 to −0.18) and improved physical function (SMD: −0.31; 95% CI: −0.47 to −0.14). No increase in adverse events was reported, supporting its safety profile. This aligns with findings from Lin et al 38 (2023), whose meta-analysis of 4 RCTs (n = 507) reported that collagen peptide supplementation significantly reduced knee pain (SMD: −0.58; 95% CI: −0.98 to −0.18, P = .004) compared to placebo, with no significant difference in adverse events (OR: 1.66; 95% CI: 0.99 to 2.78, P = .05). Similarly, Mendia et al 39 (2025) reviewed 11 RCTs (n = 870) and observed significant improvements in both pain (mean difference [MD]: −13.63) and function (MD: −6.46) in patients receiving oral collagen-based supplements vs placebo.
Different collagen formulations and dosages appear to have comparable efficacy. A 5-arm RCT by Devasia et al 40 (2024) (n = 100) compared various collagen peptide regimens and found that 2.5 g/day of Type J collagen peptides produced equivalent improvements in WOMAC scores and cartilage degradation marker CTX-II as 10 g/day of conventional collagen. Improvements were also observed in quality of life (QoL) metrics, supporting the clinical equivalence of lower-dose formulations. In a systematic review of 36 RCTs, Brueckheimer et al 41 (2025) concluded that type I collagen hydrolysate significantly improved joint pain and mobility, with effects most pronounced in trials targeting joint-specific outcomes. These benefits were independent of the underlying OA subtype, suggesting a broad therapeutic role.
When used in combination with other joint-supportive agents, collagen appears to improve strength and quality of life. In a small 8-week study of 32 patients with meniscopathy, Serhat Genç et al 42 (2025) reported that a formulation combining collagen types I, II, III with glucosamine, chondroitin, and MSM significantly reduced pain and improved quality of life, though no change in proprioception was observed. Improvements in leg strength were also noted.
Emerging preclinical data support the immunomodulatory and chondroprotective properties of collagen. A mouse model of collagen-induced arthritis (CIA) by Pan et al 43 (2024) found that oral administration of undenatured type II collagen reduced arthritis incidence by ∼50%, normalized IL-17/IL-22 levels, and preserved gut barrier integrity via induction of oral tolerance mechanisms. These findings support the hypothesis that collagen may exert systemic anti-inflammatory effects beyond the joint itself.
Collagen Effects on Arthritis.
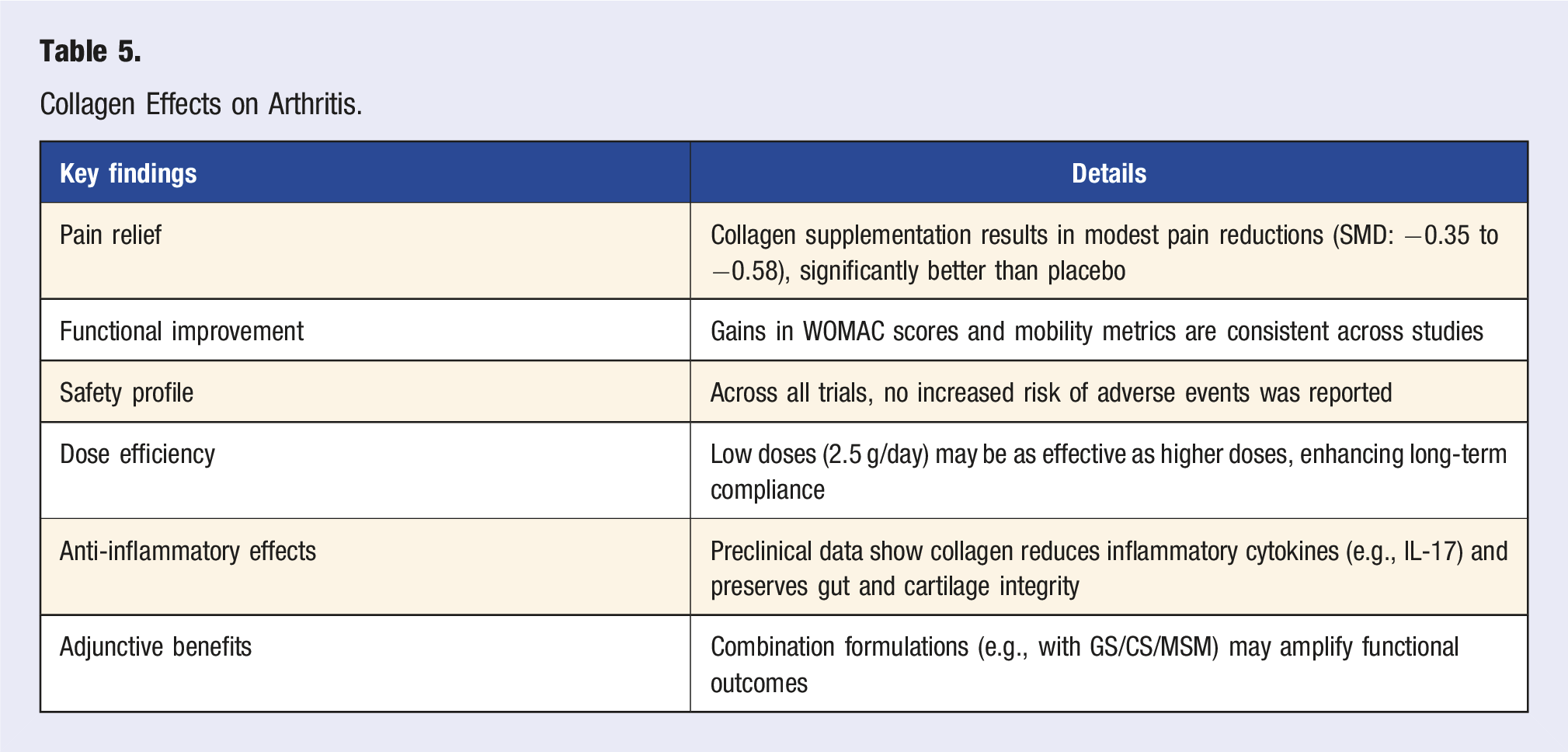
Conclusion
Collagen supplementation, especially hydrolyzed and type II formulations, demonstrates clinically meaningful improvements in pain and function for patients with knee OA, with minimal side effects and good tolerability. Preclinical evidence supports its immunomodulatory and chondroprotective effects, offering a biologically plausible mechanism of action. While not a replacement for first-line therapies, collagen represents a viable adjunct or alternative for patients seeking low-risk, long-term symptom relief. Future studies should prioritize head-to-head comparisons, biomarker analysis, and long-term joint structural outcomes.
Curcumin
Curcumin, the primary bioactive compound in Curcuma longa, has been extensively studied for its anti-inflammatory, analgesic, and chondroprotective properties in osteoarthritis (OA). Recent systematic reviews and meta-analyses suggest that curcumin supplementation significantly reduces pain, improves function, and decreases inflammatory biomarkers with a safety profile superior to NSAIDs. However, variability in study design, dosing regimens, and bioavailability formulations necessitates cautious interpretation of findings. We included 8 studies related to curcumin.
Curcumin supplementation has been shown to significantly reduce pain intensity in individuals with OA. A meta-analysis of 10 RCTs (n = 786) by Hidayat et al 44 (2025) found that Curcuma longa supplementation was significantly more effective than placebo in reducing pain (MD: 18.25, 95% CI: 7.79 to 28.72, P = .0006). Additionally, curcumin was comparable to NSAIDs for improvements in WOMAC total scores (MD: −11.99, 95% CI: −39.21 to 15.23, P = .39). Similarly, a Bayesian network meta-analysis of 23 RCTs (n = 2175) by Zhao et al 45 (2024). A clinical trial by Kuptniratsaikul et al (2014) demonstrated that 2000 mg/day curcumin is comparable in efficacy to 800 mg/day ibuprofen for managing knee osteoarthritis. 46 He reported that curcumin significantly reduced VAS pain scores (MD: −1.63, 95% CI: −2.91 to −0.45) and total WOMAC scores (MD: −18.85, 95% CI: −29.53 to −8.76) compared to placebo. Moreover, curcumin supplementation resulted in a significantly lower use of rescue medication (OR = 0.17, 95% CI: 0.08 to 0.36), particularly when combined with NSAIDs (OR = 0.01, 95% CI: 0.00 to 0.13). An umbrella meta-analysis of 11 meta-analyses by Bideshki et al 47 (2024) reinforced these findings, demonstrating significant reductions in VAS, WOMAC pain, stiffness, and function scores (P < .001 for all). These results support curcumin’s role as an effective pain-relieving supplement for OA, particularly in individuals who cannot tolerate NSAIDs.
Multiple studies confirm improvements in functional mobility and stiffness with curcumin supplementation. The systematic review by Zeng et al 48 (2022), analyzing 29 RCTs with 2396 participants, found that curcumin supplementation consistently improved joint function across OA, rheumatoid arthritis, and ankylosing spondylitis. Hidayat et al 44 (2025) further reported that both low-dose (<1000 mg/day) and high-dose (≥1000 mg/day) curcumin formulations led to significant improvements in VAS pain and functional scores (P < .05 for both). Additionally, a systematic review of aging-related conditions by Nunes et al 49 (2024) identified positive effects of curcumin on muscle health and joint mobility in older adults, suggesting potential benefits in maintaining functional independence in OA patients.
Curcumin exhibits strong anti-inflammatory and chondroprotective properties, largely mediated by its ability to inhibit NF-κB, COX-2, and inflammatory cytokines (TNF-α, IL-1β, IL-6). A scoping review of 50 studies by Ma et al 50 (2025) concluded that curcumin inhibits matrix metalloproteinase (MMP) activity, enhances collagen synthesis, and protects chondrocytes from apoptosis, which may help slow OA progression. A randomized controlled trial (RCT) of nano-micelle curcumin by Khamar et al 51 (2024) found that curcumin supplementation significantly reduced inflammatory biomarkers in rheumatoid arthritis patients, including erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) (P < .0001 for both).
Curcumin’s safety profile appears superior to NSAIDs, with fewer adverse events and comparable efficacy. Zhao et al 45 (2024) found that curcumin supplementation had fewer side effects than NSAIDs (OR = 0.51, 95% CI: 0.25 to 0.94) and that combining curcumin with NSAIDs yielded less adverse reactions (OR = 0.23, 95% CI: 0.06 to 0.9). Hidayat et al 44 (2025) concluded curcumin was non-inferior to NSAIDs for improving WOMAC scores (P = .39), suggesting it may serve as a viable alternative or adjunct to NSAID therapy.
Curcumin’s efficacy may be dose-dependent, with studies reporting optimal effects at doses of 500-1500 mg/day. Hidayat et al 44 (2025) found similar pain reductions at both <1000 mg/day and ≥1000 mg/day doses, suggesting a therapeutic window exists where higher doses do not necessarily confer additional benefits. Ma et al 50 (2025) emphasized that curcumin’s bioavailability is a major limitation, with standard formulations having poor absorption and rapid metabolism. Khamar et al (2024) Khamar et al 51 (2024) reported that nano-formulated curcumin significantly improved absorption and clinical outcomes in RA patients. These findings highlight the importance of optimized delivery systems (e.g., nano-micelles, liposomal formulations) for enhancing curcumin’s therapeutic potential.
Curcumin Effects on Arthritis.
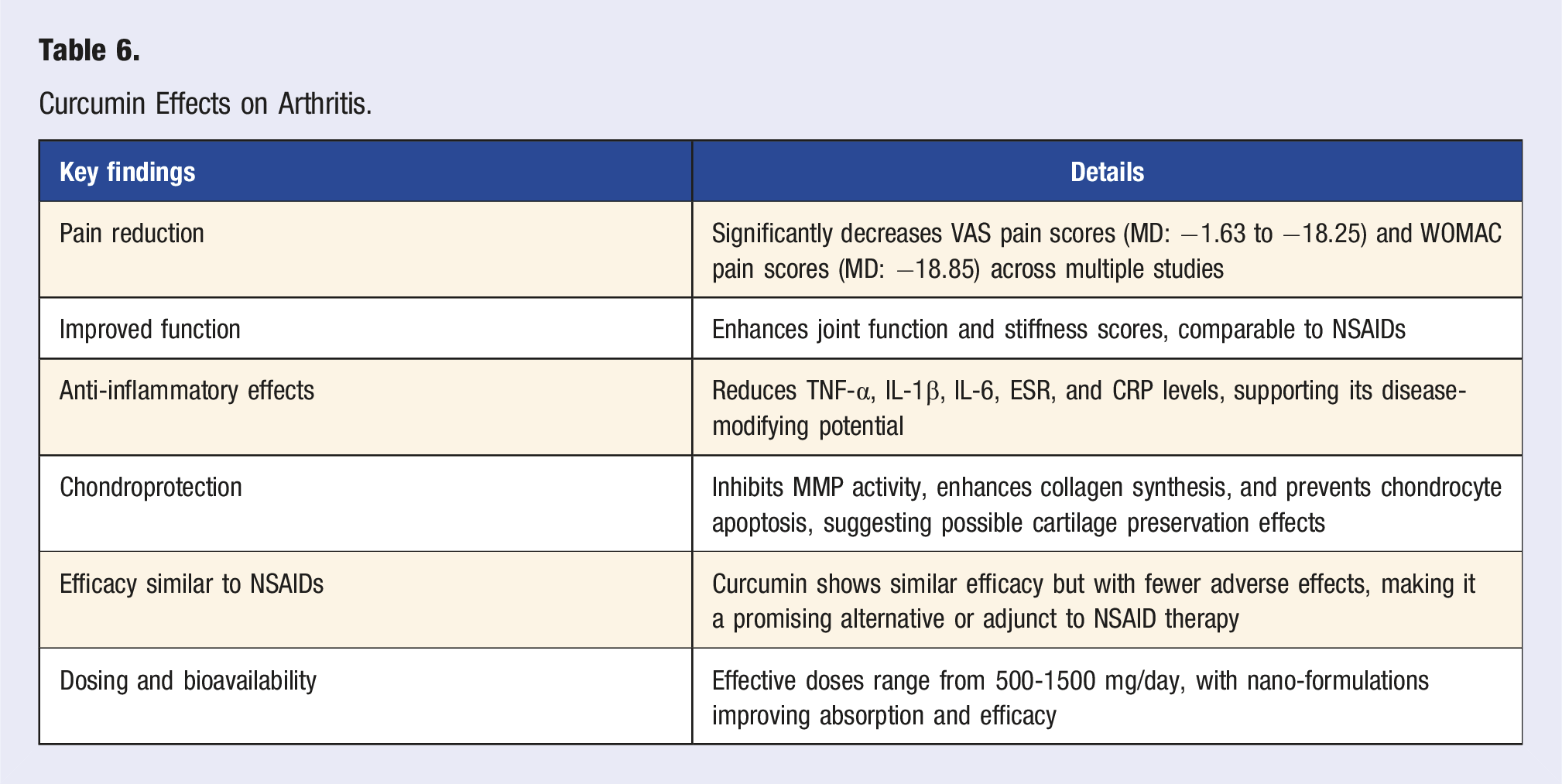
Conclusion
These findings support curcumin as an effective, well-tolerated intervention for OA pain, function, and inflammation. While not superior to NSAIDs in all aspects, curcumin’s chondroprotective and safety profile make it a valuable alternative or adjunct therapy. Future research should focus on long-term outcomes, optimal dosing, and bioavailability-enhancing formulations to maximize curcumin’s therapeutic potential in OA management.
Boswellia Serrata Extract
Boswellia serrata extract (BSE), particularly standardized forms such as Aflapin® and Boswellin®, has been investigated for its anti-inflammatory, analgesic, and cartilage-protective properties in osteoarthritis (OA). Recent human trials and preclinical models indicate that Boswellia supplementation may reduce pain, improve physical function, and modulate inflammatory markers, with minimal side effects. However, efficacy appears to depend on standardization, dosage, and formulation. We included 4 studies related to BSE.
Boswellia has demonstrated consistent efficacy in reducing OA-related pain and improving function. A systematic review of 9 randomized controlled trials (n = 712) by Dubey et al (2024) reported that Aflapin® (a highly bioavailable BSE) significantly outperformed other Boswellia formulations in reducing multiple OA symptom domains: VAS pain score: Mean difference (MD) = −16.09 mm, WOMAC-pain: MD = −18.68, WOMAC-function: MD = −14.99, WOMAC-stiffness: MD = −14.25. 52 All changes were statistically and clinically significant, with a better tolerability profile compared to conventional NSAIDs. These findings were reinforced by a 90-day RCT of 105 patients by Majeed et al 53 (2024), in which 300 mg/day of Boswellin® Super led to VAS pain reduction by 61.9%, WOMAC total score improvement by 73.6%, significant reductions in pro-inflammatory biomarkers: TNF-α, IL-6, and hs-CRP. No serious adverse events were observed, highlighting the safety of long-term BSE use.
Mechanistic evidence from preclinical studies supports Boswellia’s role as a chondroprotective and anti-inflammatory agent. In a rat model of monoiodoacetate-induced OA, Choi et al 54 (2024) found that Boswellia serrata resin extract (BSRE) at 50-100 mg/kg/day: reduced joint swelling and histological cartilage erosion, upregulated COL2A1 and aggrecan expression, downregulated IL-6, TNF-α, and matrix metalloproteinases (MMPs). These results suggest that BSE may inhibit cartilage degradation and promote matrix preservation, offering potential structural benefits in addition to symptomatic relief. Boswellia’s anti-inflammatory properties may also extend to musculoskeletal conditions beyond OA. In a double-blind RCT of 50 healthy men with delayed-onset muscle soreness (DOMS), Salter et al (2025) found that 60 mg/day of a standardized Boswellia serrata supplement (SBS) for 10 days: reduced post-exercise VAS pain by 8.0 mm, increased leg strength by +3.4 kg (1RM leg press), lowered serum IL-6 and CRP levels compared to placebo. 55 Although not OA-specific, these findings suggest a broader role for Boswellia in muscle recovery, systemic inflammation control, and physical performance enhancement.
Boswellia Serrata Extract Effects on Arthritis.

Conclusion
Boswellia serrata supplementation, particularly standardized extracts like Aflapin® and Boswellin®, offers clinically meaningful symptom relief in OA, including reductions in pain, stiffness, and inflammatory biomarkers. Preclinical findings support its cartilage-preserving and immunomodulatory properties, and its favorable safety profile makes it a compelling option for long-term management. While further large-scale, head-to-head trials are warranted, current evidence supports Boswellia as a viable adjunctive therapy in osteoarthritis care.
Exercise and Physical Activity
A growing body of evidence highlights the efficacy of movement-based and resistance training interventions in reducing osteoarthritis (OA) symptoms, improving physical function, and lowering inflammation levels. While variations exist based on intervention type, duration, and patient characteristics, the cumulative evidence supports exercise as a key therapeutic strategy American Geriatric Society guideline recommends progressive resistance training, aerobic exercise, and functional movement therapy as first-line interventions for OA. 56 We included 13 studies related to exercise and physical activity.
Resistance training is associated with moderate but clinically meaningful pain relief across multiple trials. A meta-analysis of 27 RCTs (n = 1712) by Lim et al 57 (2024) found that resistance training significantly improved pain scores (SMD: −0.48, 95% CI: −0.58 to −0.37, P < .001), strength (SMD: 0.40), and function (SMD: −0.56) compared to controls. This meta-analysis found that the benefits of resistance training were observed primarily in individuals with knee and hip osteoarthritis. A larger meta-analysis of 257 trials by Marriott et al 58 (2024) reinforced these findings, but with slightly larger effect sizes, particularly in pain reduction (SMD: −0.92) and function (SMD: −0.79). While both studies confirm resistance training’s effectiveness, the stronger effects in Marriott’s analysis may stem from differences in study inclusion criteria or a higher proportion of long-term interventions. Notably, Turner et al 59 (2020) identified 2-3 sets of 8-12 repetitions, performed 3 times per week for at least 8-12 weeks, as the most effective protocol for improving WOMAC pain scores. However, Messier et al 60 (2021) found no significant difference between high- and low-intensity resistance training over 18 months, suggesting that even moderate-intensity training can yield meaningful benefits. Taken together, these findings suggest that resistance training has potential to reduce pain in OA, with longer interventions and structured protocols yielding the best outcomes. However, intensity level may be less important than consistency and total training volume.
Beyond pain relief, resistance training improves physical function and mobility, particularly in lower extremity strength and gait performance. A cohort study of 2589 individuals Dunlop et al 61 (2011) found a graded relationship between activity level and gait speed. Participants in the lowest quartile of physical activity averaged 4.0 ft/s, while those in the highest quartile reached 4.5 ft/s, a statistically significant improvement (P < .001). Similarly, Chmelo et al 62 (2013) found that older, overweight adults with OA who engaged in higher spontaneous physical activity exhibited better lower extremity function and reduced knee pain. These findings align with the resistance training data, suggesting that both structured exercise and general movement contribute to functional improvements.
Resistance training appears to exert meaningful anti-inflammatory effects, though benefits vary based on protocol. A network meta-analysis of 52 RCTs (n = 4255) by Lin et al 63 (2024) found that adjunctive strategies such as blood-flow restriction and isokinetic training produced the largest reductions in inflammatory markers (SMD: −1.89 for CRP, TNF-α) and the greatest increases in muscle hypertrophy (SMD: 1.28). These findings suggest that exercise may contribute to joint preservation through anti-inflammatory and regenerative pathways.
Structured guideline-based exercise programs, such as GLA:D, can delay or reduce the likelihood of joint replacement in osteoarthritis
Across 13 RCTs involving 967 patients with RA, a meta-analysis by Ye et al 65 (2022) found that aerobic exercise, including jogging, walking, cycling significantly improved functional ability (MD = −0.25, P = .0002), pain (SMD = −0.46, P = .04), and aerobic capacity (MD = +2.41, P < .00001) in patients with RA, with moderate to low GRADE certainty. No serious adverse events were reported. In a network meta-analysis of 67 RCTs involving 4944 KOA patients, Luo et al 66 (2025) found that Pilates was the most effective aerobic intervention for improving overall function (WOMAC Function SUCRA = 0%) and total symptom reduction, while Tai Chi was superior in alleviating pain (VAS SUCRA = 17.4%). Weight-loss walking ranked highest for mobility improvements (TUG SUCRA = 27.1%). Other beneficial modalities included Baduanjin and yoga, which showed moderate effectiveness across domains. Cycling, conventional walking, and aquatic training ranked lower.
While higher long-term physical activity levels are associated with lower chronic pain, acute increases in activity may temporarily exacerbate symptoms, though the direction of the effect is unclear. A meta-analysis of 13 studies (n = 9363) by Burrows et al 67 (2020) found that participants with higher daily activity levels reported less pain overall, but those who suddenly increased activity experienced short-term pain flares. This aligns with the concept of load-dependent pain exacerbation, where excessive mechanical stress on compromised joints can temporarily heighten symptoms before adaptation occurs. These findings reinforce the need for gradual exercise progression in OA management.
A systematic review by Mauri et al 68 (2025) compared aerobics, resistance, and neuromuscular training, finding that neuromuscular training was the most effective for pain reduction (effect size: −2.26, 95% CI: −4.37 to −0.14). Neuromuscular training is a type of training that improves automatic motor control and joint stability by enhancing the body’s sensory and neural responses. 69 However, no single modality was superior across all outcomes, suggesting that multimodal approaches (combining resistance, aerobic, and neuromuscular training) may offer the greatest overall benefits.
Exercise and Physical Activity Effects on Arthritis.
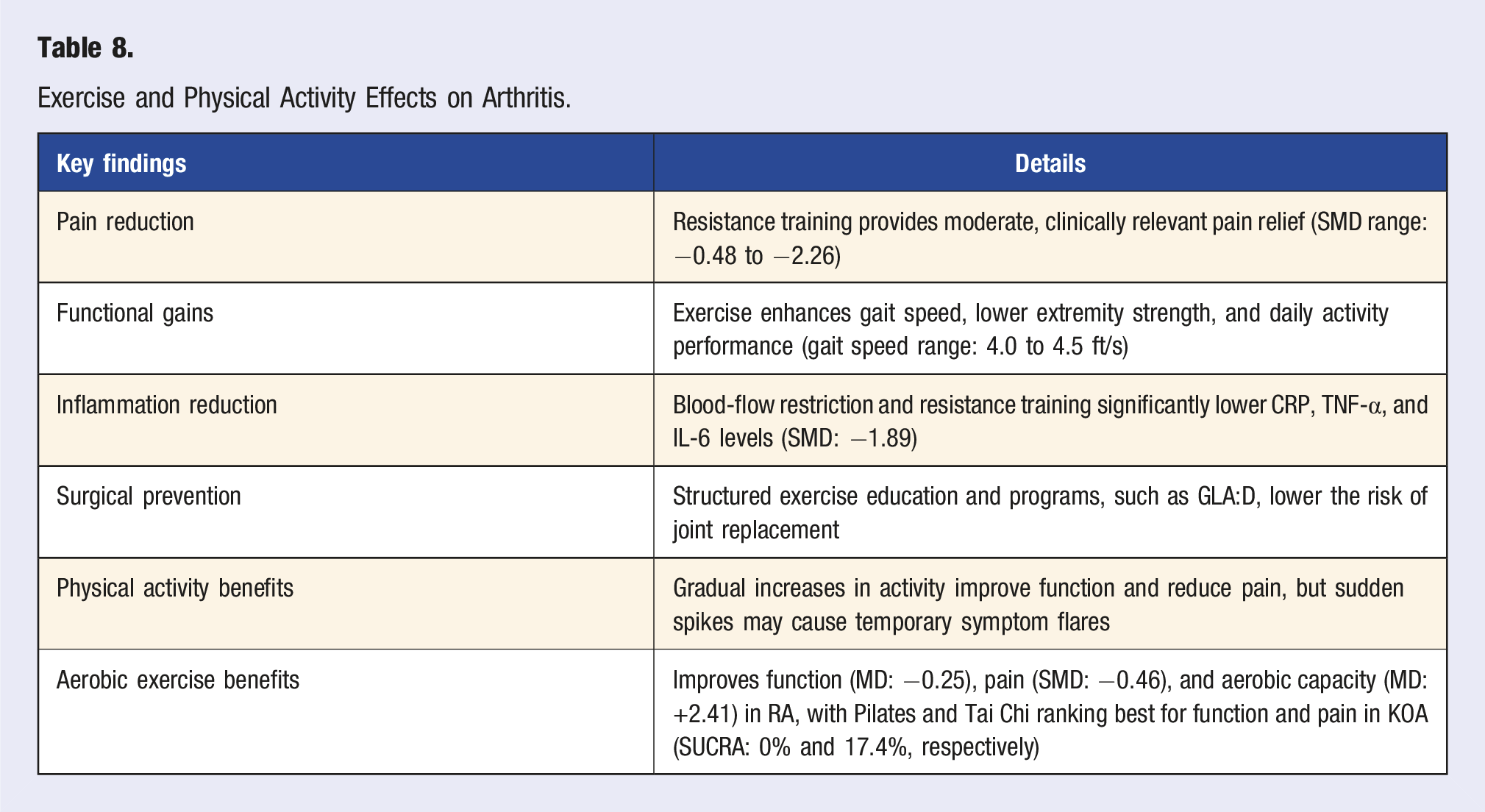
Conclusion
These findings underscore that structured movement-based interventions, particularly resistance training, are essential for OA management. A multimodal approach incorporating resistance, neuromuscular, and aerobic exercise may provide the most balanced benefits.
Tai Chi
Tai Chi, a traditional Chinese mind–body exercise integrating slow, controlled movement with breathing and mental focus, has gained increasing attention as a non-pharmacologic intervention for osteoarthritis (OA). A growing body of randomized controlled trials and systematic reviews demonstrates that Tai Chi improves pain, stiffness, physical function, and mental well-being in individuals with knee and spine-related OA. Importantly, Tai Chi is also shown to be safe, feasible, and acceptable in both supervised and unsupervised formats. We included 5 studies related to tai chi.
Multiple meta-analyses show that Tai Chi significantly improves pain, stiffness, and physical function in OA. A meta-analysis of 17 RCTs (n = 1122) by Qiao et al 70 (2024) evaluating mind–body exercises (including Tai Chi, Yoga, and Baduanjin) found that interventions lasting ≥8 weeks resulted in pain reduction: SMD = −0.65, stiffness reduction: SMD = −0.75, improved function: SMD = −0.82, reduction in depression symptoms: SMD = −0.32. These results suggest that Tai Chi offers comparable or greater improvements in joint symptoms than many pharmacologic interventions, particularly when performed consistently. This was supported by an earlier meta-analysis of 18 RCTs (n = 1260) by Kong et al 71 (2016) which found that Tai Chi improved OA-related pain with an SMD of −0.54 (95% CI: −0.77 to −0.30). Effect sizes remained significant when compared to waitlist (SMD: −0.42) and attention control groups (SMD: −0.60), suggesting a robust intervention effect beyond placebo or social support.
Even short-term Tai Chi interventions may yield substantial improvements. A pilot study by Yang et al 72 (2024) evaluated an “Optimized Tai Chi (OTC)” protocol in 15 patients with knee OA over 2 weeks. Participants experienced median VAS pain reduction from 5.0 to 1.0 (P < .001) and a 50% greater improvement in pain and function compared to traditional Tai Chi forms. These findings support the feasibility of abbreviated, high-efficiency Tai Chi formats, especially for patients with limited time or mobility.
Tai Chi also shows promise for axial symptoms and spinal mobility. In a 25-week intervention (50 sessions) involving 28 patients with osteoarthritis or inflammatory arthritis, Kalebota et al 73 (2024) observed significant improvements in spinal flexion, extension, rotation, and lateral flexion (P < .001), improvement in functional capacity for OA patients (P = .014), but no statistically significant change in resting VAS pain. These findings indicate that Tai Chi can enhance mobility and function even in the absence of large pain score changes, which may be particularly relevant for axial OA.
Recent work has explored the utility of Tai Chi in home-based, unsupervised formats. In developing the “My Joint Tai Chi” online platform, Zhu et al 74 (2024) engaged stakeholders and tested the feasibility of a 12-week unsupervised Tai Chi program using 10 Yang-style forms. Results from the usability phase showed high acceptability, safety, and perceived benefit. Participants reported improved confidence and satisfaction in practicing at home. An RCT evaluating its clinical effectiveness is ongoing, but early results suggest that digital Tai Chi interventions may offer scalable solutions for symptom management in OA.
Tai Chi Effects on Arthritis.
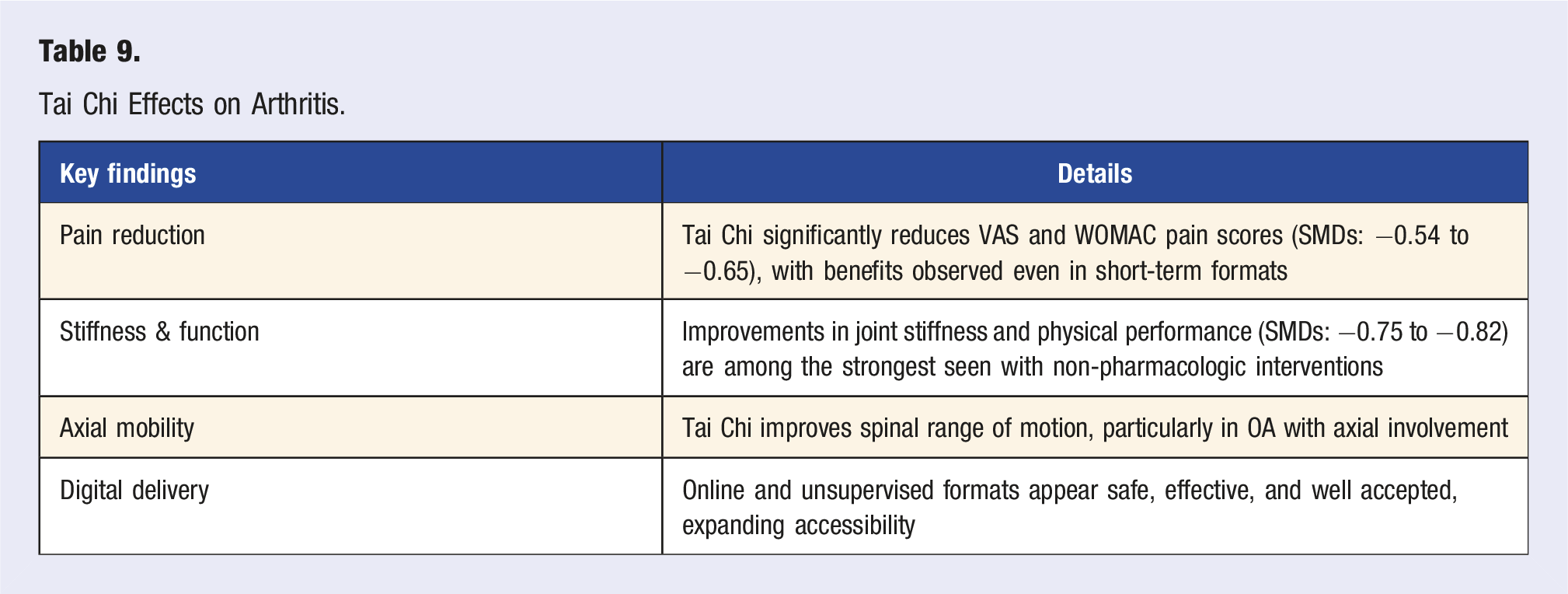
Conclusion
Tai Chi is a safe, feasible, and effective lifestyle intervention for individuals with osteoarthritis, producing moderate to large improvements in pain, function, stiffness, and mental health. Its adaptability to short-duration and digital formats makes it a promising adjunctive therapy, particularly for populations with limited access to supervised exercise programs. Future work should emphasize head-to-head comparisons with physical therapy and pharmacologic interventions and examine long-term adherence and structural outcomes.
Sleep
Sleep variability and quality are increasingly recognized as modifiable factors contributing to joint health and arthritis symptom severity. Emerging research suggests that both short sleep duration and poor sleep quality are associated with increased arthritis risk and greater pain burden, independent of inflammatory disease activity. Behavioral sleep therapies, including cognitive behavioral therapy for insomnia (CBT-I), offer evidence-based improvements in pain and function, positioning sleep optimization as a key target in lifestyle-based arthritis management. We included 9 studies related to sleep.
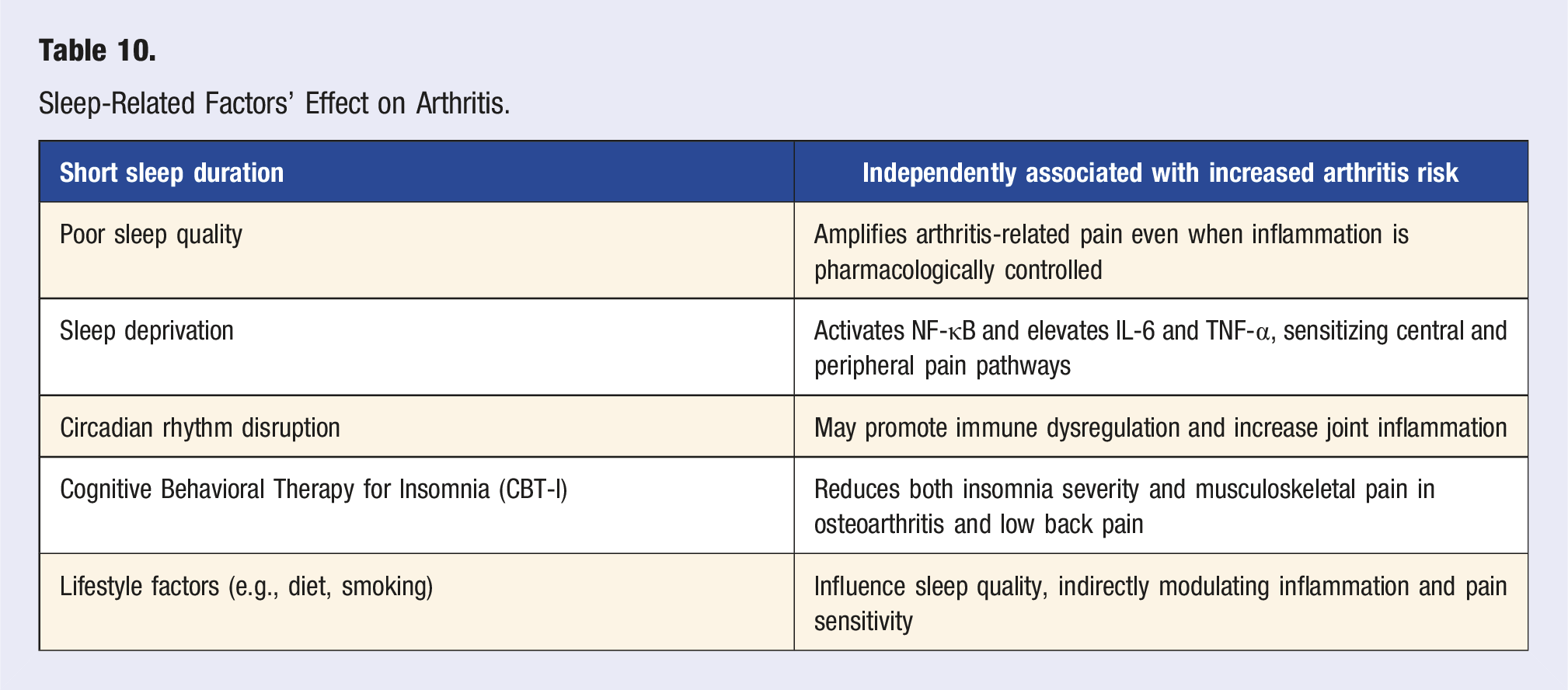
Large-scale cohort studies support a strong association between poor sleep and increased arthritis risk. In a 7-year longitudinal study of 6597 adults aged ≥45 years, Shang et al 75 (2024) found that each interquartile-range increase in sleep duration was associated with a 16% reduced risk of new-onset arthritis (OR: 0.864; 95% CI: 0.784-0.954), with BMI partially mediating the effect. Similarly, Wu et al 76 (2024) reported that individuals with poor sleep quality had a 38% increased risk of arthritis (HR = 1.38; 95% CI: 1.09-1.74), while sleep durations under 5 h were associated with a 56% increased risk (HR = 1.56; 95% CI: 1.27-1.91). Both of these studies assessed overall arthritis without distinguishing between OA and RA. These findings identify sleep as a meaningful lifestyle determinant in the prevention of arthritis, particularly in aging populations.
Sleep disruption has been shown to contribute directly to systemic inflammation, altered pain processing, and increased arthritis symptoms. Irwin et al 77 (2023) reported that fragmented or shortened sleep activates NF-κB signaling and elevates IL-6 and TNF-α levels, while reducing glucocorticoid receptor sensitivity, amplifying pro-inflammatory pathways in rheumatoid arthritis. In experimental models, Chang et al 78 (2022) found that sleep deprivation significantly decreased pain thresholds and increased spontaneous and evoked pain responses in healthy adults (Hedge’s g = 0.35-0.95), suggesting that even acute sleep loss alters nociceptive signaling. In arthritis populations, poor sleep quality is associated with worse pain outcomes. In the Nor-Hand cohort, Bordvik et al 79 (2025) found that individuals with hand osteoarthritis who reported severe sleep problems had significantly higher hand pain both at baseline and 3.5-year follow-up (NRS +1.68; 95% CI: 0.89-2.46), independent of sensitization effects. Similarly, Weman et al 80 (2024) reported that patients with rheumatoid or psoriatic arthritis who rated their sleep as “poor” had nearly 3-fold higher pain scores compared to those with “good” sleep, even in the absence of elevated disease activity. Behavioral interventions to improve sleep have been shown to reduce pain and improve function. A meta-analysis of 24 randomized controlled trials by Ho et al 81 (2019) demonstrated that cognitive behavioral therapy for insomnia (CBT-I) reduced the Insomnia Severity Index (ISI) by −6.78 in low back pain and −2.41 in osteoarthritis, and reduced pain intensity (VAS) by −12.77/100 in low back pain (95% CI: −17.57 to −7.97). These results support CBT-I as a non-pharmacologic intervention capable of reducing both insomnia and arthritis-related pain.
Sleep disturbances in arthritis frequently co-occur with other poor lifestyle habits. Gheisary et al 82 (2024) using Canadian national health data (n = 104 359), found that individuals with arthritis were significantly more likely to report short sleep duration, non-restorative sleep, and insomnia symptoms. These outcomes were associated with poor dietary intake, exposure to passive smoking, and sedentary behavior, suggesting that broader lifestyle changes may be necessary to address sleep dysfunction in this population. Emerging evidence also points to the role of circadian rhythm in regulating joint inflammation. Finsterbusch et al 83 (2018) identified leukocyte-platelet aggregates, early immune triggers of synovitis, sensitive to circadian misalignment. While direct human data is limited, maintaining consistent sleep-wake schedules may reduce systemic inflammation and complement existing arthritis interventions.
Sleep-Related Factors’ Effect on Arthritis.
Conclusion
Sleep plays a critical role in both the development and symptom progression of arthritis. Disturbed sleep heightens inflammation and pain, while interventions like CBT-I offer non-pharmacologic solutions for improving joint outcomes. Lifestyle-based approaches that target sleep, circadian health, and coexisting risk factors such as poor nutrition and smoking may provide additive benefits in arthritis prevention and management.
Discussion
Lifestyle and supplement interventions influence arthritis outcomes not through a single dominant mechanism, but by acting on multiple systems that intersect at the level of inflammation, joint mechanics, and pain perception. 2 This multimodal effect may help explain why even modest improvements in one domain, such as diet, are associated with measurable clinical benefits. While the impact of sleep and supplements aligns with evidence from mostly preclinical or observational research, the more consistent improvements seen with diet and exercise suggest that arthritis symptom expression is influenced by broader systemic processes, including metabolic function, inflammation, and mechanical stress.84,85 Notably, several studies led to improved symptoms in the absence of substantial changes in inflammatory biomarkers, suggesting that traditional clinical measures may not fully capture the effects of lifestyle change on patient-centered outcomes such as pain and function.9,14 Another consideration is the possibility of cross-domain synergy, for example, exercise improving sleep, or dietary improvements supporting greater physical activity, which may enhance outcomes beyond what each intervention could achieve alone. Taken together, these findings support an evolving model of arthritis care that moves beyond single-target pharmacologic treatment toward integrated, lifestyle-based strategies that reflect the complex, systemic nature of chronic joint disease.
The cumulative evidence warrants a fundamental shift in the approach to arthritis management, from reactive symptom control to proactive, systems-level intervention. For clinicians, this underscores the need to incorporate structured lifestyle assessment and evidence-based counseling into routine arthritis care. Addressing diet, exercise, sleep, and supplementation as discrete therapeutic targets may improve patient-reported outcomes, reduce medication burden, and mitigate comorbid disease risk. For patients, these interventions offer low-risk, scalable, and often underutilized options with the potential to improve both symptoms and long-term disease trajectory.
Despite promising results, the current literature is limited by variability in study quality, inconsistency in intervention fidelity, and heterogeneity in outcome measurement. Many trials are short in duration, lack structural or biomarker endpoints, and do not account for adherence or behavioral sustainability. Supplementation studies are often hindered by inconsistencies in dosing, bioavailability, and product standardization. Moreover, a few trials test lifestyle interventions in combination, limiting insight into additive or synergistic effects that reflect real-world applications. It is important to note that implementation of lifestyle interventions may vary across communities, as access to healthy foods, exercise resources, and healthcare support is often limited in socially deprived areas. Addressing these inequities through advocacy and community-based initiatives is essential to ensure equitable delivery and impact.
Future research should prioritize multi-domain intervention trials that reflect the complexities of arthritis care. Longitudinal studies with structural, inflammatory, and functional endpoints are needed to assess potential disease-modifying effects. Greater attention to individual response variability, mediated by factors such as metabolic phenotype, microbiome composition, and chronotype, may inform the development of personalized lifestyle prescriptions. Implementation science will be critical to scaling these interventions in diverse populations and care settings, with attention to equity, access, and integration into multidisciplinary models of care. Further analyses should also evaluate cost-effectiveness, behavioral sustainability, and the feasibility of incorporating lifestyle screening tools into electronic health records and clinical decision-making platforms.
Conclusion
Lifestyle interventions represent an evidence-informed approach to arthritis management that targets underlying contributors to disease beyond symptom control. Among the strategies reviewed, structured physical activity and anti-inflammatory dietary patterns show the most consistent benefits across pain, function, and quality-of-life outcomes. Sleep optimization and targeted supplementation serve as effective complements. Although not curative, these interventions are safe, accessible, and mechanistically relevant, supporting their integration as foundational elements of a comprehensive, patient-centered model for arthritis care.
Footnotes
Acknowledgments
The authors thank the Stanford Center for Lifestyle Medicine and the Department of Orthopedic Surgery Statisticians for their support and insights during the preparation of this manuscript.
Author Contributions
S.N. conceived and designed the review, conducted the literature search and analysis, and drafted the manuscript. M.K., M.S., C.R., A.F. reviewed, edited, and provided feedback on the manuscript. M.F. oversaw and provided guidance throughout the review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
