Abstract
A traumatic brain injury (TBI) is caused by an insult to the brain leading to the disruption of the brain’s normal cellular processes. This dysfunction can result in temporary vs permanent impairments, thus impacting ones’ quality of life and contributing to long-term disability. Lifestyle Medicine offers a complementary approach to TBI management with interventions that address diet, exercise, stress management, sleep, and other lifestyle factors. The goal of this article is to provide a review of Lifestyle Medicine interventions and their current findings in TBI management.
Keywords
“Alcohol misuse independently contributes to TBI risk, poor recovery outcomes, mood disorders, and recurrent TBIs.”
Objective of the Review
1. Overview of TBI and common sequelae. 2. Overview of Lifestyle Medicine and its current role in TBI management. 3. Discuss role of diet in TBI management, more specifically the dietary patterns and nutrients that have been suggested to improve TBI recovery. 4. Discuss the importance of physical activity in regards to recovery and identify most recent findings on initiation, type of exercise, and intensity within the different severities of TBI. 5. Discuss the relationship between sleep disorders and TBI. Identify specific interventions that have been shown to improve sleep quality in individuals with a TBI. 6. Discuss the role of stress, stress management, and psychological interventions on TBI recovery. 7. Overview of integrative and complementary approaches that have been shown to benefit individuals after a TBI. 8. Identify potential challenges/barriers with implementing lifestyle interventions in survivors of a TBI. 9. Identify current gaps in research and explore potential areas for future studies.
Key Takeaways
- Diet plays an important role in TBI, especially during the acute-phase, due to increased metabolic demands. There is strong evidence for early enteral nutrition and permissive glycemic control in severe TBI. - Individuals with a TBI have an increased risk in developing obesity, CVD, diabetes, hypertension, etc. Prescribing aerobic exercise can help mitigate these risks while also promoting cognitive and motor recovery. - After sustaining a mild TBI, it is recommended to undergo relative rest for 24 – 48 followed by the initiation of light physical activity. Prolonged rest is detrimental. Physical activity intensity is gradually increased but should be decreased if there is an exacerbation of symptoms. In moderate-severe TBI, further research is needed regarding physical activity in the acute-phase of TBI. - Cognitive Behavioral Therapy can be used to help significantly reduce symptoms of depression, anxiety, and anger in TBI patients. It can also be used as treatment for PTSD and/or insomnia in patients with concurrent TBI. - Appropriate sleep is crucial for TBI recovery as nearly 50% of survivors experience some form of sleep disturbance. More robust studies regarding lifestyle-based sleep interventions, specifically in the TBI population, are needed.
Target Audience
This review is intended as a resource for health care professionals (physicians, physician assistants, nurses, etc.) overseeing the care of individuals with a history of traumatic brain injury.
Background
According to the Centers for Disease Control and Prevention (CDC), a traumatic brain injury (TBI) is caused by a bump, blow, or jolt to the head, or a penetrating head injury that disrupts the normal function of the brain. In 2014, the CDC reported 2.87 million TBI-related hospital encounters—2.5 million Emergency Department visits, 288,000 hospitalizations and 56,800 deaths in the United States. 1 The annual TBI incidence is about 333 – 506.4 per 100,000. 2 Approximately 40% of survivors with moderate or severe TBI (msTBI) have long-term disability. This estimates 3.17 million US residents, or more than 1.1% of the US population, to be living with TBI-related disability.3-5 The cost of nonfatal TBI totaled approximately $40 billion dollars in 2016 in the United States alone. 6
Lifestyle Medicine is a medical specialty that employs evidence-based lifestyle interventions to help prevent, treat and even reverse chronic disease by addressing key health behaviors including physical activity, nutrition, sleep, and stress. 7 In the context of TBI, these interventions can be used to help improve TBI-associated symptoms, mitigate risks and improve outcomes through all stages and severity.
Traumatic Brain Injury: Pathophysiology, Grading and Prognostic Factors
TBI occurs when sudden trauma disrupts normal cellular processes within the brain through primary and secondary mechanisms. The primary injury includes direct, shearing and/or rotational forces that immediately damage the brain parenchyma. Secondary injury can exacerbate the primary injury with metabolic changes and swelling causing increased intracranial pressure (ICP), herniation and/or death without intervention. 8
Classification of Traumatic Brain Injuries.
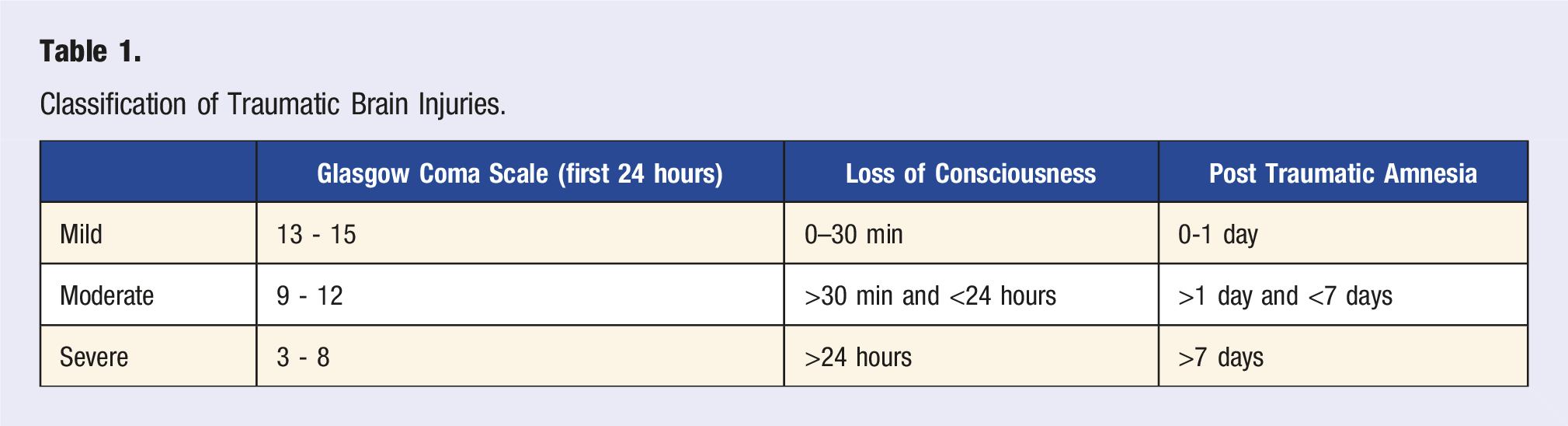
Outside of injury severity, premorbid factors including social history, comorbidities, lifestyle and genetics also influence outcomes. Social factors such as education and pre-injury employment are positively associated with functional outcomes, community re-integration and reemployment.5,10-13 Smoking pre- or post-TBI is associated with impaired motor and cognitive recovery as well as an increased risk in developing comorbid conditions.14,15 Chronic alcohol consumption is associated with impaired cognitive recovery. 16 A diagnosis of diabetes, psychiatric disorders, substance abuse, or pre-injury polypharmacy is associated with poor outcomes.10,13,17,18 Family dynamics and social support play a role in patient and caregiver outcomes. 19 Lifestyle habits that promote overall health, such as regular physical activity, may support better exercise adherence that promotes recovery. 20 Genetics can influence outcomes by mediating inflammatory responses and neuroplasticity. The most studied gene in regards to this is the e4 allele of Apolipoprotein E. Studies suggest the e4 allele confers a small risk of poorer outcomes in the acute-phase of severe TBI, although further research is needed for its effects on recovery.10,21,22 Understanding predictors of outcome can help anticipate a survivor’s needs, progression and prognosis.
Traumatic Brain Injury: Symptoms and Sequelae
In mild TBI (mTBI), individuals can experience post-concussive symptoms (PCS), which entails a constellation of symptoms including headache, dizziness, fatigue, photo- and/or phonophobia, sleep disturbances or impaired cognition. 23 The majority of PCS are transient, often resolving within 10 - 14 days. Some individuals may report persistence of symptoms for months to years after injury, known as Persistent Post Concussive Syndrome (PPCS). However, the etiology and diagnostic criteria are not agreed upon in current literature thus making it difficult to estimate prevalence or evaluate treatment efficacy. 24 Some studies estimate around 15% of individuals with mTBI develop PPCS.25,26 The validity of PPCS has also been debated, as the constellation of symptoms are not unique to mTBI and can be seen in other conditions, most commonly psychiatric disorders. 27
The sequelae of TBI include cognitive deficits, psychological symptoms, and physical symptoms. 28 Post-traumatic amnesia (PTA) is common, with its duration used as an indicator of severity. PTA is the period post-injury where memory formation and retrieval are impaired, often characterized by confusion, inattention and disorientation.29,30 Agitation is common in PTA, with its persistence contributing to poor recovery and morbidity. 8 Cognitive deficits include impairments in attention, memory, processing speed, emotional regulation, and executive functioning as well as pain, fatigue, and sleep disturbances.3,31 Physical deficits include impairments in movement, balance or coordination, bladder or bowel dysfunction and dysphagia. Both cognitive and physical deficits contribute to functional impairments such as limitations in self-care, toileting, transferring and ambulation.
There is also evidence that TBI survivors have an increased risk in developing comorbid health conditions such as diabetes, obesity, accelerated aging, cardiovascular disease, sleep disorders, chronic pain, and psychiatric disorders.32-35 These can develop secondary to the TBI itself or from new behaviors adopted post-TBI.
Lifestyle Medicine Overview
Lifestyle Medicine is a medical specialty focused on evidence-based interventions to prevent, treat, and reverse chronic diseases. 36 It emphasizes nutrition and activity modification, promotes healthy aging and encourages planetary health for global well-being.37-39 Behavioral change is central to lasting success, with health coaches playing a vital role in guiding patients toward better habits. 40 The six foundational pillars, as defined by the American College of Lifestyle Medicine (ACLM), are plant-based nutrition, regular physical activity, avoidance of risky substances, restorative sleep, positive social connections, and effective stress management. 41 Beth Frates, a pioneer in Lifestyle Medicine, emphasizes that “true health transformation occurs when we recognize each lifestyle medicine pillar’s interconnectedness and apply them holistically to patient care. Only then can we fully unlock the full potential of food as medicine and its ability to restore health. 42 ” This holistic approach has the potential to ameliorate outcomes and improve the quality of life (QOL) in TBI survivors.
Role of Diet in Traumatic Brain Injury Management
Nutritional Interventions.
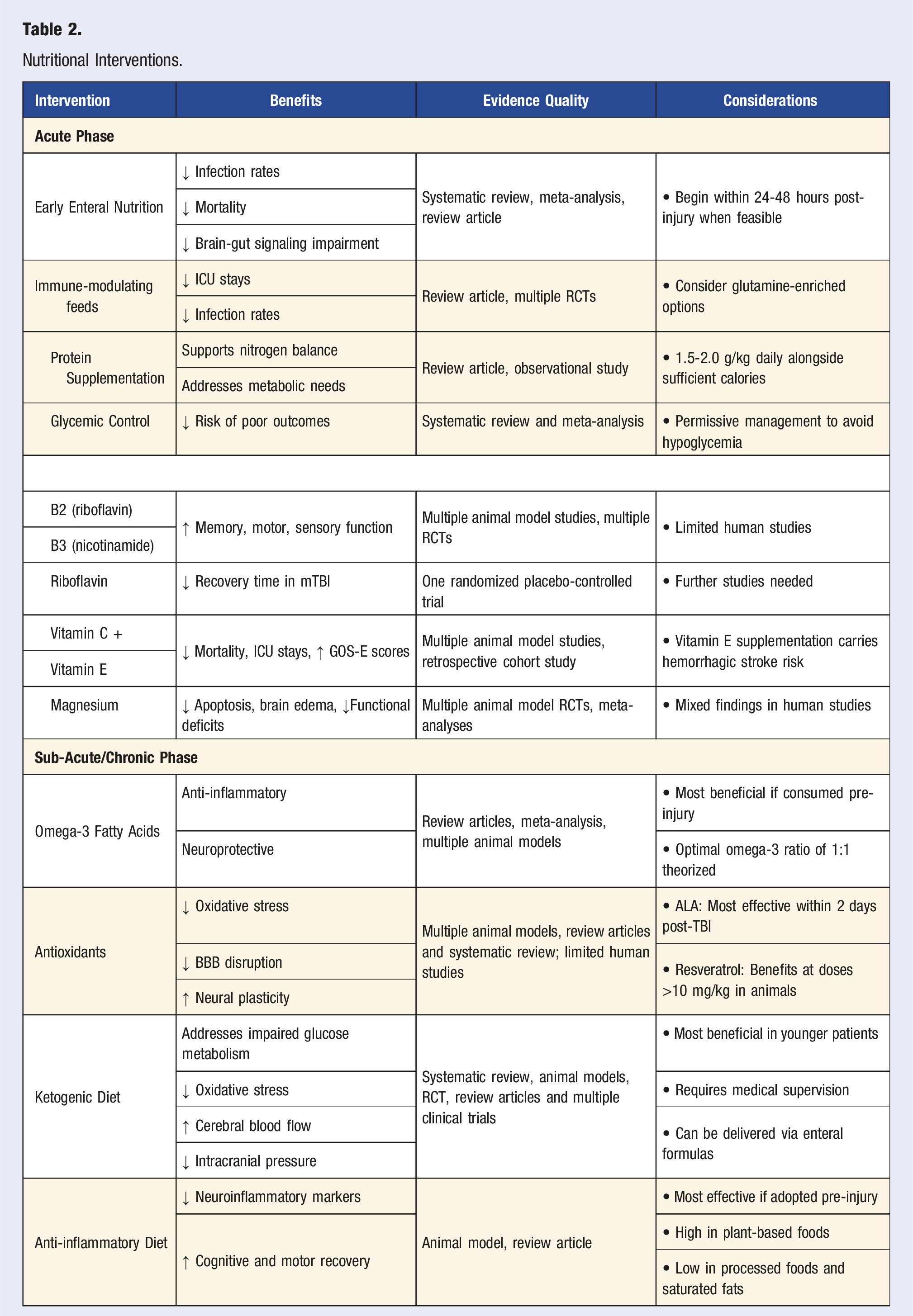
In the acute-phase of TBI, the Institute of Medicine: Nutrition in Clinical Practice Guidelines for TBI recommends prompt dietary assessment while hospitalized to provide optimal nutritional intake throughout treatment. 47 Currently, there is insufficient evidence to formulate diet specific guidance for mild and moderate TBI.47,48 In severe TBI, early initiation of nutritional support, preferably enteral nutrition, is recommended as this is associated with decreased rate of infection, mortality and sequelae associated with impaired brain-gut signaling.47,49-51 Immune-modulating enteral feedings, particularly glutamine-enriched options, can reduce Intensive Care Unit (ICU) stays and infections.50,52-54 Glycemic control is crucial, with permissive management preferred to avoid hypoglycemia which is associated with poorer outcomes. 55 Protein needs are high, with guidelines recommending 1.5-2.0 g/kg daily alongside sufficient caloric intake.56,57
Nutritional studies regarding the chronic-phase of TBI are limited. Western diets, rich in saturated fats and processed foods, are linked to mitochondrial dysfunction, insulin resistance and inflammation. Animal studies show that pre-injury consumption of these diets are associated with poorer outcomes following a TBI. 58 Dietary patterns like the Mediterranean, DASH and MIND may be beneficial as these emphasize plant-based whole foods and reduce cardiometabolic risk factors in the general population.59-61 In mTBI, emerging research suggests interventions like omega-3s, antioxidants, and ketogenic diets may improve cognition, memory, and sleep but further studies are needed at this time.48,62
Omega-3
Omega-3 fatty acids, particularly docosahexaenoic acid (DHA), offer neuroprotective and anti-inflammatory benefits for brain health. While animal studies demonstrate that high-dose DHA supplementation can reduce axonal damage, neuronal apoptosis, and glutamate cytotoxicity—leading to improved neurological function, cognitive outcomes, and inflammatory markers 63 —human clinical evidence remains limited.64,65 Animal studies suggest that premorbid omega-3 consumption may enhance brain resilience and mitigate inflammatory cascades, 66 although post-injury supplementation shows limited benefit.67,68 Human studies that report benefit in the chronic-phase of TBI often include other interventions, such as lifestyle education, thus confounding the isolated effect of omega-3’s.69,70 Although there is insufficient evidence regarding the efficacy and dosing of omega-3s for the treatment of TBI in humans, a recent meta-analysis suggests that dosing between 1000 to 2500 mg per day in the general population may improve cognitive function. 71
Antioxidants
Oxidative stress is a major contributor to TBI pathogenesis. Many antioxidants show promise in preclinical studies but lack sufficient evidence for its use in individuals with a TBI. In animal models, compounds such as α-lipoic acid (ALA), astaxanthin and epigallocatechin gallate decrease oxidative stress, reduce blood-brain barrier (BBB) disruption, and enhance neural plasticity markers.72,73 ALA administered in rats after a TBI has been found to decrease cerebral edema, increase glutathione levels and improve neurological function.74,75 Non-antioxidant compounds with free radical scavenging properties, such as vitamins and flavonoids, have also shown potential benefit. Resveratrol, a polyphenol found in grapes and berries, has demonstrated benefits in rodent studies such as a reduction in brain edema, memory loss, and inflammation at doses over 10 mg/kg.76-78
Vitamins & Minerals
In animal studies, vitamin B2 (riboflavin) and B3 (nicotinamide) have shown potential in improving memory, motor and sensory functions. 79 Nicotinamide has been shown to reduce cortical lesion size, decrease neuroinflammation and improve recovery time in rats,80-82 however evidence in human studies remains limited. In mTBI, a randomized controlled trial (RCT) showed that treatment with riboflavin (400 mg daily for 14 days) improved recovery time as compared to placebo. 83
Vitamin C (ascorbic acid) levels are often depleted after TBI. In animal models, its co-administration with vitamin E has been shown to increase superoxide dismutase levels.84,85 In individuals with severe TBI, this combination is associated with improved Glasgow Outcome Scale-Extended (GOS-E) scores, decreased mortality and reduced ICU stays. 86 However, vitamin E supplementation, in general, has been associated with an increased risk of hemorrhagic stroke.87-89 In animal studies, vitamin C alone demonstrated reduced vascular inflammation as well as improved learning and memory. 90 One human trial reported reduced brain edema in severe TBI with high-dose administration of vitamin C for 7 days. 91
Magnesium is an essential micronutrient involved in numerous physiological activities. Hypomagnesemia is associated with poor neurological outcome after severe TBI. 92 Magnesium can function as a NMDA-receptor antagonist which could play a role in attenuating glutamatergic NMDA-receptor excitotoxicity. 93 In animal models, magnesium in the acute-phase yields favorable outcomes by reducing apoptosis, 94 brain edema, 95 and functional deficits. 96 However, human studies lack consistent evidence to promote its routine use in TBI. A meta-analysis found that magnesium sulfate treatment in severe TBI had no significant effect on all-cause mortality or GOS score but yielded significant improvement in GCS. 97 In acute mTBI, treatment with oral magnesium oxide 400 mg twice daily for 5 days has been shown to decrease PCS severity in adolescents. 98 Riboflavin, magnesium and vitamin D3 (cholecalciferol) have also shown individual benefit with migraine prophylaxis.99-101 Although the preclinical data regarding the discussed vitamins and minerals appear promising, further human trials are needed to evaluate the efficacy, safety and optimal dosing of these supplements for the treatment of TBI.
Ketogenic Diets
Initially developed for pediatric epilepsy, the ketogenic diet (KD) has shown potential in mitigating secondary injuries following TBI. The benefit comes from shifting the brain’s energy source from glucose to beta-hydroxybutyrate, which is advantageous given the impaired cerebral glucose metabolism seen in TBI. 73 This reduces oxidative stress from mitochondrial dysfunction73,102,103 and may enhance cerebral blood flow through nitric oxide production and mTOR signaling.104,105
Studies on KD in rats report neurocognitive and behavioral improvements along with reduced cerebral edema in the acute and sub-acute phases of TBI. 106 Human trials indicate KD is effective in normalizing blood glucose, reducing lactate, 106 and does not impair key physiological parameters such as ICP, cerebral perfusion pressure, or acid-base balance.107-109 Rippee et al. demonstrated improvements in the visual memory domain of ImPACT after a 2 month KD in patients with mTBI and PPCS, 110 however more clinical trials measuring the effect of KD on functional and cognitive outcomes are still needed. To ensure proper carbohydrate-to-fat ratios and mitigation of long-term health risks, this diet requires medical guidance from a qualified healthcare professional 111
Anti-Inflammatory Diets
Anti-inflammatory diets have the potential to improve TBI recovery, especially if adopted pre-injury. One study involving mice revealed that a pre-injury diet enriched with fruits and vegetables reduced post-TBI neuroinflammatory markers, improved cognitive and motor functions, and accelerated recovery. 112 Another study found that supplementation with vitamin D3 and omega-3 fatty acids acutely post-injury resulted in reduced neurotrauma biomarkers (T-tau, GFAP, UCH-L1) in the chronic-phase of mTBI, which indicates reduced neuronal injury. 113 While promising, these findings are yet to be confirmed in human studies.
Physical Activity and Exercise in Traumatic Brain Injury
Physical Activity in TBI.
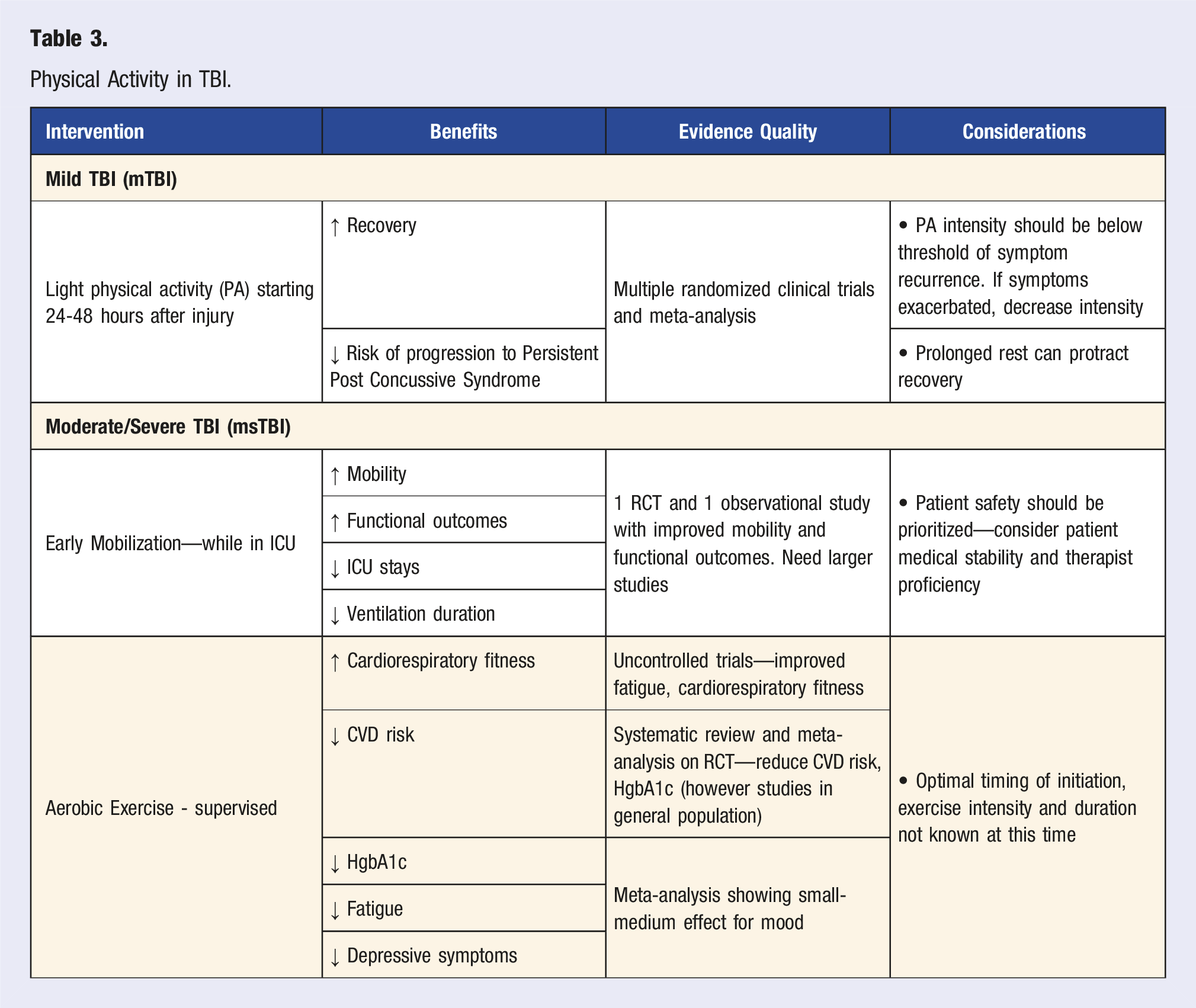
The appropriate initiation of PA post-TBI is crucial for optimal recovery. In mTBI, the International Conference on Concussion in Sport published evidence-based recommendations to guide management. Relative rest is recommended for the first 24-48 hours post-injury, followed by the initiation of light PA. During this time, it is important to evaluate for symptoms of PCS, as this will guide intensity progression or regression. 127 Evidence shows that prolonged rest should be avoided as this can prolong recovery. 128 Initiating sub-symptomatic aerobic activity according to guidelines has been shown to improve recovery, reduce the risk of progression to PPCS, as well as decrease symptoms in those with PPCS.128,129 For PPCS, another study revealed that ≥150 minutes a week of moderate-to-vigorous PA is associated with improved QOL and decreased functional impact of symptoms. 130
For the acute-phase of msTBIs, there are currently no guidelines regarding initiation of PA. Early mobilization in the ICU may provide benefit, although research is still ongoing. A recent RCT (n = 65) evaluated the effects of early out-of-bed mobilization (EM) therapy vs early in-bed upright positioning (control) and found that those who underwent EM experienced improved mobility and functional outcomes by hospital discharge, shortened ICU stays, and reduced ventilation duration with no adverse effects as compared to the control. 131 Other studies appreciated improved mobility and functional outcomes, but had varying results regarding ICU stay and ventilator duration.132,133 Due to the paucity of research at this time, patient safety should be prioritized, and an adequate evaluation of medical stability along with therapist proficiency should be completed before initiating EM as there can be a high risk of patient harm.
Not only can PA improve recovery, it can also mitigate the risk or progression of comorbid diseases. In all severities of TBI, individuals were found to have an increased risk of developing obesity, hypertension, diabetes, and cardiovascular disease (CVD) which can contribute to increased overall mortality.115,134-136 One study revealed that survivors of msTBI experience on average a 25%–35% reduction in maximal aerobic capacity by 6-8 months. 115 In order to combat this, prescribing exercise, predominantly aerobic exercise, is encouraged. Aerobic exercise has been shown to improve lipid profiles, lower HgA1c, improve cardiorespiratory fitness, and reduce CVD risk within the general population 137 with similar findings in those post-TBI.138-140 Mood may also improve with aerobic exercise, as shown in a meta-analysis of 9 studies, where the engagement in an exercise intervention resulted in reduced depressive symptoms. 141
The majority of research involving exercise for TBI survivors evaluated interventions that were supervised. Of the limited studies involving unsupervised interventions, one reported good adherence and proficiency in a community-based aerobic exercise program. 142 In this study, it is important to note that those enrolled underwent a review of medical records and neuropsychological screening to verify physical and cognitive safety.
A majority of the studies discussed, excluding studies on early mobilization, involved the chronic-phase of TBI; ≥6 months post-injury.138-142 Further research is needed regarding time to initiation, intensity and duration of exercise. It is important to note that even though aerobic exercises can improve multiple health factors, it may not be sufficient for weight loss if no dietary modifications are in place. 140
Stress Management and Psychological Interventions
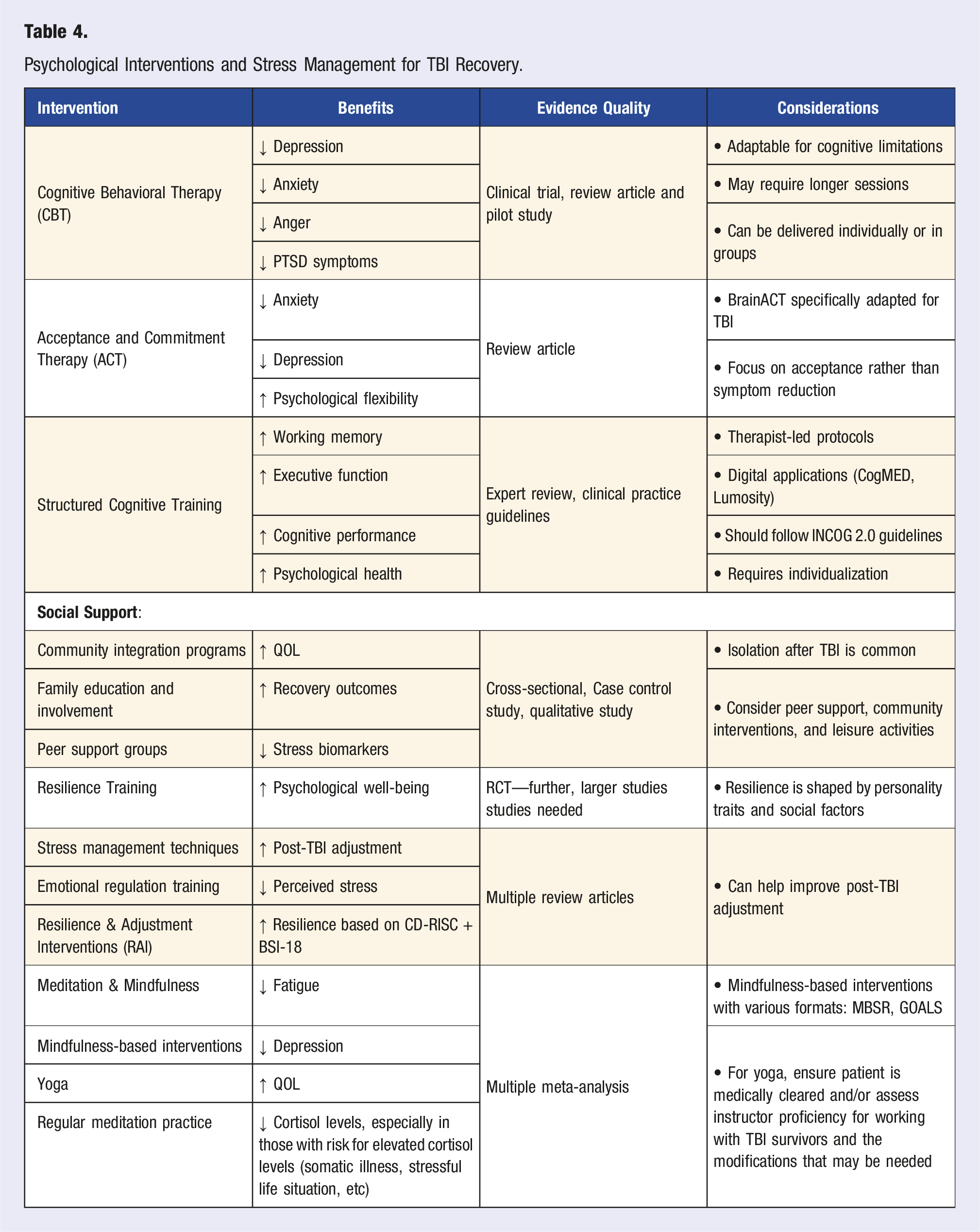
TBIs can disrupt physiological mechanisms such as the hypothalamic-pituitary-adrenal (HPA) axis, thus impairing the stress response and contributing to elevated glucocorticoids (Table 4 and 5). This disruption can worsen memory, hippocampal function, and be a precursor to the development of a mood disorder.143-147 In contrast, TBI-associated pituitary gland damage can lead to hypopituitarism, where hormones like adrenocorticotropic hormone (ACTH) are impaired, leading to hypocortisolism.148-150 Pituitary damage can lead to adrenal insufficiency, life-threatening hypotension, and hyponatremia. 151 Thus, routine screening for pituitary dysfunction, including cortisol levels, is recommended in both the acute and chronic phases following TBI. 152
At the cellular level, animal models reveal that stress can exacerbate neurodegeneration, autophagy and apoptosis subsequently impairing neuronal plasticity and increasing neurological deficits.153-155 Downstream effects include increased fatigue, depression, memory impairment, sleep disturbances and pain, thus making it crucial to manage stress in TBI survivors.156,157 Psychological interventions, including cognitive rehabilitation, have proven effective in mitigating the cognitive effects of TBI when applied under current practice guidelines. 158 CBT is a well-studied approach, significantly reducing symptoms of depression, anxiety, anger, and PTSD in TBI patients.159-161 Acceptance and commitment therapy (ACT), adapted for TBI as BrainACT, has also shown efficacy in alleviating anxiety and depressive symptoms.162,163 Studies show that meditation and mindfulness-based interventions can alleviate stress symptoms by promoting relaxation, reducing cortisol levels, 164 increasing cellular telomerase activity,165,166 and is associated with cognitive benefits.167,168 Mindfulness-based stress reduction (MBSR) and goal-oriented attention self-regulation (GOALS) reduce perceived stress, enhance coping skills, and improve mood and QOL.169-171
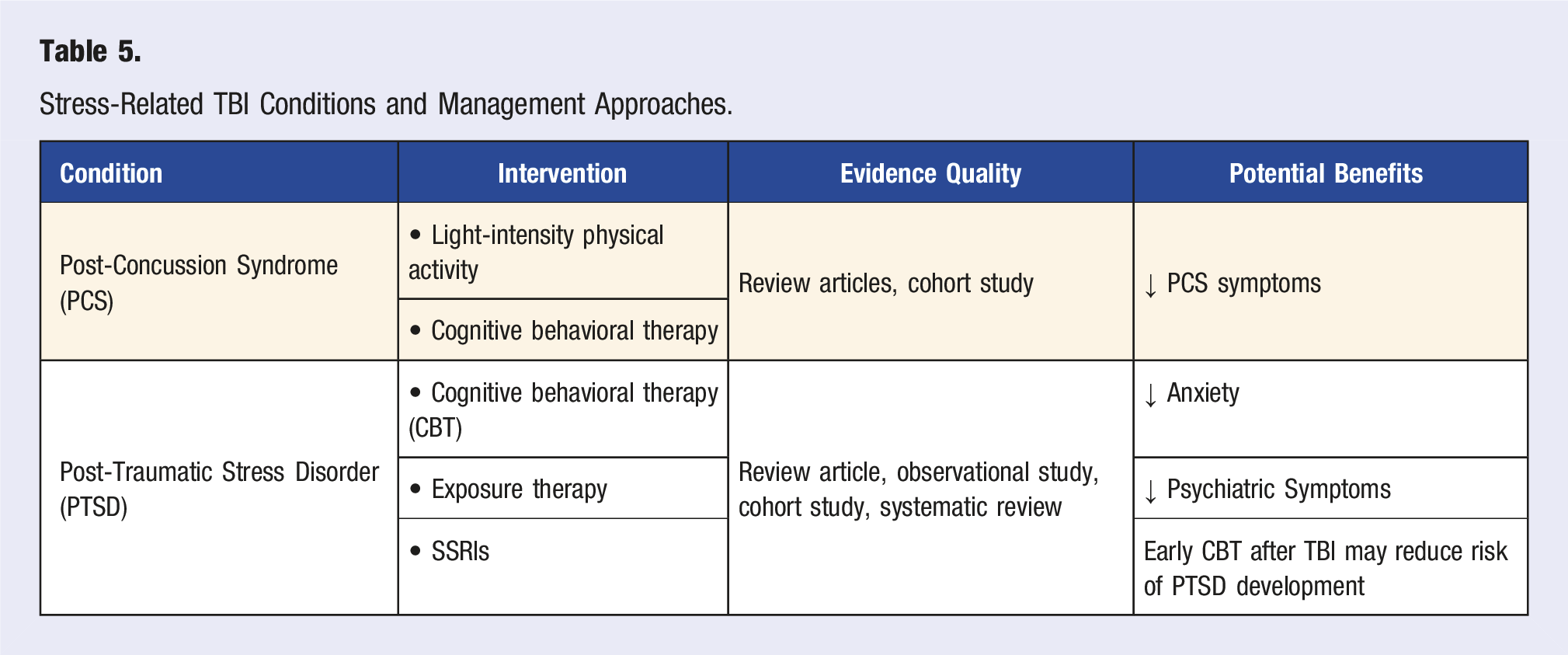
PCS and post-traumatic stress disorder (PTSD) are commonly seen after mTBI and are closely linked with stress. PCS is associated with HPA axis dysfunction and generally responds to treatment with light-intensity PA and CBT.145,172-174 Comorbid PTSD has been found to impede TBI recovery. It requires accurate diagnosis with treatment often involving CBT, exposure therapy, and potentially SSRIs.175-178 Some studies suggest that early treatment with CBT after injury can reduce the risk of developing PTSD, however findings are currently inconclusive. 178 Emerging therapies, such as with stem cells and hyperbaric oxygen, show promise but need further study. 179
Resilience—shaped by personality traits and social factors like community engagement—enables patients to better adapt to post-TBI stressors, emphasizing the value of fostering resilience skills during recovery. 180 Resilience and Adjustment Interventions (RAI), developed by Kreutzer and colleagues, combine stress management and emotional regulation techniques. RAI has been shown to increase resilience, improve psychological health and post-TBI adjustment, and produce more favorable scores on the Brief Symptom Inventory-18 (BSI-18).181,182 Social support is also crucial as isolation after TBI correlates with higher cortisol levels, impaired cognition, and increased depression, while strong social connections enhance QOL and recovery outcomes.183-185
Structured cognitive training has emerged as a powerful adjunct for promoting neuroplasticity and restoring executive function after TBI. Formal cognitive rehabilitation programs, including therapist-led protocols and commercially available digital applications such as CogMED and Lumosity, have shown promise in enhancing working memory, cognition, and improving psychological health. 186 The international group of cognitive researchers and clinicians (INCOG) developed the INCOG 2.0 guidelines that strongly support individualized, evidence-based cognitive rehabilitation strategies, emphasizing the role of adaptive tools and technologies to meet the diverse needs of TBI patients. 187
Sleep Disorders and Traumatic Brain Injury
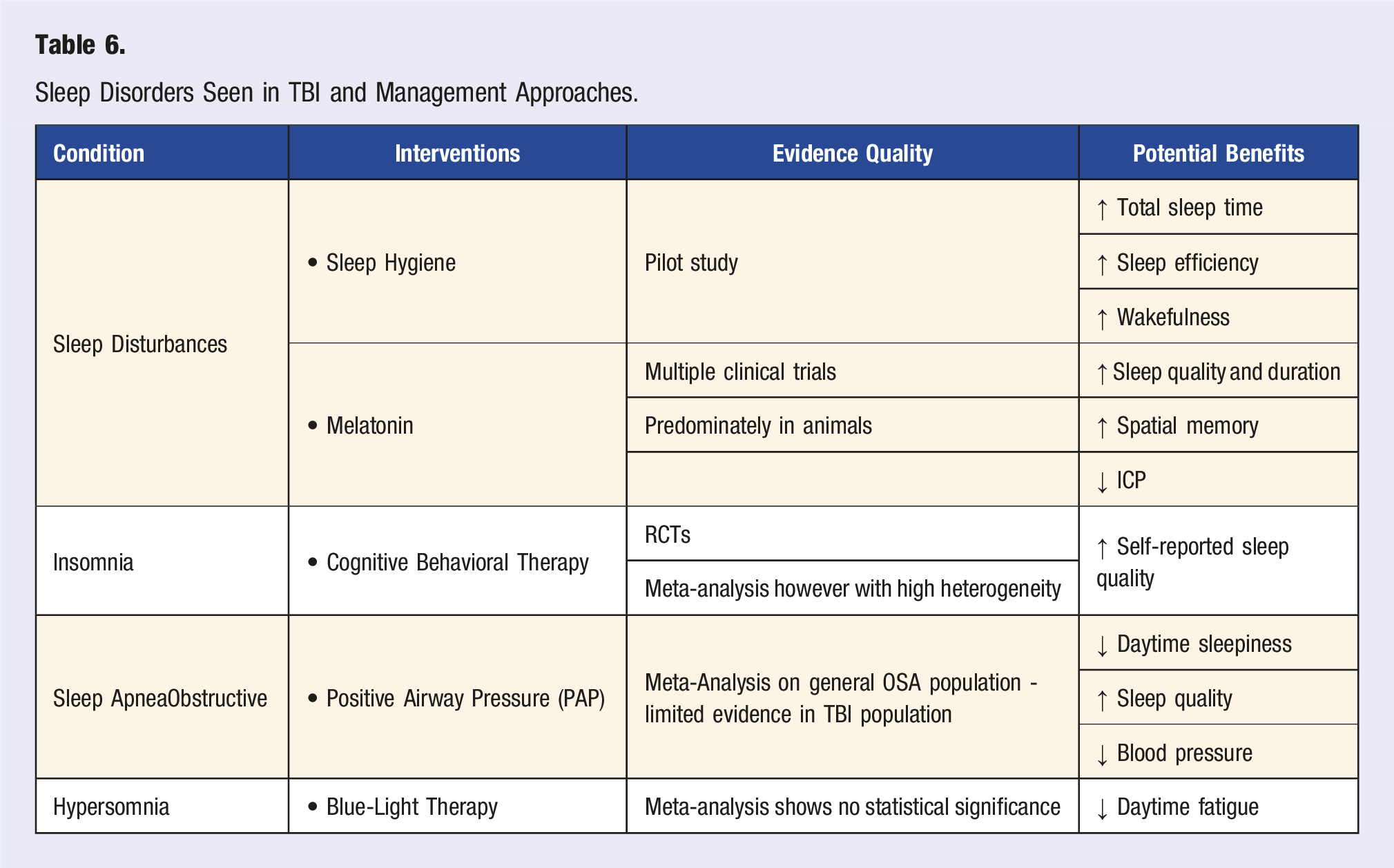
Appropriate sleep plays an important role in TBI recovery but unfortunately about 50% of survivors experience some form of sleep disturbance (Table 6). 188 These can arise from both internal factors, such as intrinsic physiology, 189 and external factors, such as sleep environment. 190 Sleep disturbances have been shown to reduce motivation, effort and attention, increase neuroinflammation, impair hippocampal function, contribute to HPA axis dysfunction, exacerbate comorbid conditions, as well as reduce recovery outcomes.129,144,191-200
One effective and cost-efficient intervention to improve sleep is sleep hygiene. 201 Sleep hygiene refers to the behavioral factors that affect sleep quality and duration. Literature shows that implementing a consistent sleep schedule, reducing evening light exposure and optimizing one’s sleep environment can help promote sleep efficiency. 201 Dietary considerations also play a role, such as limiting caffeine and alcohol consumption as well as eliminating food intake at least 2 hours before sleep. 201 Research regarding the effects of sleep hygiene on TBI is limited. In a 2020 inpatient TBI rehabilitation pilot study, the implementation of a sleep hygiene protocol appeared to improve total sleep time, sleep efficiency and wakefulness. However, this study also included 30 minutes of blue-light therapy which confounds the effect of sleep hygiene on the observed sleep improvement. 202
Outside of sleep disturbances, sleep disorder prevalence is elevated post-TBI, with insomnia at 30%–60%, hypersomnia at 10%–30%, and sleep apnea (both obstructive and central) at 23%–36%.189,203 The appropriate treatment is correlated with cognitive recovery as well as mitigation of adverse neurobehavioral outcomes including depression and anxiety.199,204,205
For the treatment of insomnia, cognitive behavioral therapy (CBT) for insomnia is the gold standard. However, in insomnia secondary to TBI, there is little research regarding its efficacy. A 2021 meta-analysis on 3 studies reported that CBT is associated with improved self-reported sleep quality with minimal adverse effects. 206 An alternative but similar option for CBT is Internet-guided CBT (CBT-I) which has demonstrated similar efficacy in improving insomnia symptoms in TBI.207-209 With this modality, it may be more practical for those who cannot access a local provider.
Sleep apnea, both obstructive (OSA) and central (CSA), has increased prevalence in individuals post-TBI. OSA in the general population is associated with impaired attention, memory, and executive function, 210 with similar findings in TBI survivors although evidence is limited. 211 There are minimal studies regarding the consequences of CSA, but current literature suggests an association with cognitive impairment, dementia and the development of cardiac abnormalities such as arrhythmia.212,213 Positive Airway Pressure (PAP) therapy is the standard treatment for both OSA 214 and CSA. 215 PAP therapy has been shown to reduce daytime sleepiness, improve sleep quality, reduce blood pressure, improve cognition, and reverse OSA-related white matter abnormalities in the general OSA population.216,217 However, adherence with PAP (defined as ≥4 hours/night) is relatively poor. The non-adherence rate is estimated to be 30%–40% in the general population, 218 with one study reporting 76% non-adherence in those following a TBI. 219 After prescribing PAP, follow up is encouraged to assess for and correct noncompliance.
Blue-light therapy (BLT) has been suggested to treat daytime fatigue, but studies have demonstrated mixed results. A 2022 meta-analysis shows BLT has a moderate effect in treating sleep disturbances post-TBI but lacked statistical significance in reducing symptoms of sleepiness or fatigue. 220
Short-term melatonin may benefit those unresponsive to lifestyle interventions by improving sleep quality.221-223 One RCT revealed improved sleep quality, increased sleep efficiency on actinograph, and decreased anxiety and fatigue. 224 Outside of improving sleep, rodent models have shown that melatonin may improve spatial memory, reduce neuronal death, and lower ICP.225-227 Additionally, a preliminary clinical trial with ramelteon, a melatonin receptor agonist, demonstrated improvement in total sleep time and cognitive functioning. 228 These findings suggest that targeting the melatonin system may offer benefits beyond sleep regulation, potentially aiding cognitive recovery. Use of other pharmacological sleep aids should be conservative due to the risk of side effects such as delirium. 189
Other Lifestyle Factors
Excessive alcohol consumption and smoking are linked to poorer outcomes after TBI, making abstinence and recidivism critical during recovery.14,229 Chronic smoking worsens neurocognitive outcomes by promoting BBB dysfunction, neuroinflammation, and oxidative stress which leads to neuronal damage.14,230,231 While preliminary studies suggest nicotine might benefit cognitive deficits by modulating nicotinic acetylcholine receptors and dopamine pathways, its risks, including cancer and chronic disease, outweigh potential benefits.15,232,233 Alcohol misuse independently contributes to TBI risk, poor recovery outcomes, mood disorders, and recurrent TBIs. Children and adolescents with TBI are particularly at risk of developing alcohol use disorders later in life.229,234-236 Assessing a patient’s pre-TBI alcohol use and providing guidance for cessation or moderation is essential. Comprehensive care should include education, referrals to addiction specialists, and connections to support groups.
Fluid resuscitation is a standard practice in acute TBI management, but excessive fluid administration can lead to complications such as cerebral swelling, elevated ICP, acute lung injury, and increased mortality.237,238 Conversely, inadequate hydration poses risks, including renal injury, highlighting the need for careful fluid balance. 239 In chronic TBI, proper hydration is essential to prevent symptoms such as headache and dizziness. Patients with cognitive impairment or disorders of consciousness may neglect hydration, increasing the risk of electrolyte imbalance, especially hypernatremia. 240
Exposure to toxins such as heavy metals and food additives may exacerbate TBI-related symptoms, as demonstrated in animal studies with substances such as pyridostigmine bromide and permethrin.241,242 This highlights the importance of a thorough history of toxic exposures, military service, and occupational risks. TBI also impairs the glymphatic pathway, reducing the brain’s ability to clear waste thus increasing vulnerability to neurotoxin accumulation. This could contribute to neurodegenerative diseases, such as Alzheimer’s disease, due to tau protein aggregation. 243 Educating patients on minimizing toxin exposure is a key component of care. 244
Integrative, Complementary and Modern Health Approaches
Integrative therapies such as acupuncture, yoga, meditation, and herbal supplements may support TBI recovery, although evidence remains limited. A meta-analysis reported significant improvements in fatigue and depressive symptoms with yoga and meditation-based interventions. 245 Programs such as LoveYourBrain Yoga, a specialized hatha yoga program for TBI patients, offer psychological benefits and aid in social re-integration. To ensure safety, instructors should receive training on physical modifications for head and neck movements as well as appropriate evaluation of the individual with a TBI.246-249
Acupuncture shows promise in alleviating symptoms such as anxiety and irritability by modulating the autonomic nervous system; with potential benefits in neurological recovery, consciousness, and working memory.250-255 However, larger, more robust studies are needed.
Herbal supplements may enhance recovery by leveraging neuroprotective properties. Examples include Panax ginseng, Carthamus tinctorius L., and Gastrodia elata, which have shown efficacy in improving symptoms when used alone or alongside conventional treatments.256,257 However, risks such as drug interactions and contamination must be considered. Patients should prioritize high-quality, third-party tested products to ensure safety and efficacy.
Digital health tools are emerging as adjuncts to the management of TBI recovery, particularly in tracking adherence to lifestyle interventions. Mobile applications and web-based platforms have been shown to improve self-monitoring, emotional regulation, and cognitive engagement in both survivors of TBI and caregivers. 258 For instance, applications such as MyFitnessPal assist with nutritional tracking, Sleepio offers CBT-based support for insomnia, and wearable devices like WHOOP or Oura monitor physiological parameters such as heart rate variability (HRV) and sleep cycles. These tools may facilitate early detection of stress or fatigue. Moreover, wearable fitness trackers have demonstrated feasibility and reliability in promoting physical activity among individuals with acquired brain injury, helping to personalize rehabilitation strategies and encourage long-term behavior change. 259 Integrating such technologies into care plans could enhance adherence, foster autonomy, and improve outcomes.
Clinical Applications
Research regarding the incorporation of lifestyle medicine into TBI management is still in the early stages. Driver et al conducted a 12-month RCT to assess the effects of a lifestyle intervention on weight loss and secondary health outcomes in TBI survivors who are overweight to obese (BMI ≥25 kg/m2). The study modified an evidence-based weight-loss program, Group LifeStyle Balance Program (GLB), to target the needs of TBI survivors (GLB-TBI). This intervention promotes weight loss by reducing calorie intake and increasing PA to at ≥150 minutes a week, which was maintained through education on topics regarding PA and food consumption. 260 The results demonstrated that the GLB-TBI group lost an average of 17.8 pounds (7.9% of body weight) while the control group lacked significant weight loss. For secondary outcomes, the GLB-TBI group had significant improvements in triglycerides, HDL cholesterol, diastolic blood pressure, waist size, 6-min walk test, satisfaction with life, and self-report habits. Compliance with the program was high at 89.6% attendance with the completion of weekly diet and activity self-monitoring logs at 68.8%. This demonstrates the GLB-TBI intervention can significantly reduce weight and metabolic risk factors in TBI survivors as well as increase self-reported habits for diet and exercise. 261
Smaller studies, such as case reports, indicate that combining modern medicine with traditional practices like acupuncture can be both safe and effective, though long-term safety remains unclear. 262 A review highlighted the need for personalized treatment plans which may or may not include complementary approaches. Given the complexity of TBI and limited robust evidence for alternative therapies, individualized care is essential. 263
Challenges and Considerations
Psychological Interventions and Stress Management for TBI Recovery.
Stress-Related TBI Conditions and Management Approaches.
Sleep Disorders Seen in TBI and Management Approaches.
Conclusion and Future Directions
Survivors of TBI are generally male, 268 are financially disadvantaged, 269 have a history of substance abuse, 270 a neuroendocrine disorder, 271 poor insight and impulse control. Future research into lifestyle modifications for those with TBI cannot be simple extrapolations of cognitive strategies that were applicable to the general population. In this review, a significant fraction of the evidence comes from animal models, and when the studies enrolled human subjects, many were not prospective with adequate controls. Designing blinded placebo controls for lifestyle modifications is far more challenging compared to drug trials, as sleep, food and exercise cannot be blinded. Therefore, the reliance on retrospective studies confounds our conclusions due to the inability to establish causality and instead settling for correlation alone. Finally, enrolling TBI survivors into studies is challenging as aphasia and insight impede the capacity to consent. Despite these challenges, our team believes that amongst all of the lifestyle interventions researched in this article, sleep hygiene may be the keystone modifier to prioritize—as sleep improves impulse control, 272 “will power,” which will be crucial to confronting substance recidivism, unhealthy foods, and physical inactivity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
