Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by both motor and non-motor symptoms that significantly impair quality of life. While clinical heterogeneity results from genetic and biological factors are nonmodifiable factors of PD, growing evidence highlights the impact of modifiable factors on symptom management, disease progression and quality of life. This review synthesizes current research on the role of sleep, diet, stress management, and physical activity in PD. Sleep disturbances, affecting nearly 88% of patients, exacerbate both motor and non-motor symptoms and may accelerate disease progression. Treatment strategies include sleep hygiene, cognitive behavioral therapy (CBT), and pharmacologic interventions customized for particular specific sleep disorders. Nutritional interventions, particularly Mediterranean or MIND diets, are linked to slower disease progression, while poor dietary habits may worsen outcomes. Stress and psychological distress, including depression and anxiety, are prevalent, and mindfulness practices provide substantial relief. Physical exercise, especially aerobic and strength training, helps improve motor function, cognitive performance, and quality of life, and programs lasting more than 12 weeks are more effective. These modifiable factors help improve PD treatments and quality of life.
“Managing sleep disorders is critical, as improved sleep quality can significantly enhance quality of life and potentially slow disease progression.”
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder characterized by progressive motor and non-motor symptoms. 1 It is considered the second most prevalent neurodegenerative disorder, with Alzheimer’s disease being by far the most common.2,3 There are more than 1 million people living with PD in US, and the number of PD patients worldwide is estimated to double to over 12 million by 2040.4,5 Neurological disorders have become the leading cause of disability worldwide, and PD is the fastest growing among them. 6 The hallmark features of PD include resting tremor, rigidity, bradykinesia, and postural instability which impair daily functioning.7,8 Non-motor symptoms include cognitive impairment, mood disorders, autonomic dysfunction, sensory abnormalities and more, involving other systems such as genitourinary, gastrointestinal, skin, eyes, etc.9-11
PD exhibits significant heterogeneity in clinical manifestations, outcomes, and responses to treatment.12-15 This is a critical factor in the management of disease.
16
This variability is mainly attributable to each patient’s unique genetic profile and disease pathogenesis (Figure 1).
17
PD is characterized by the loss of neurons in the specific regions of the substantia nigra and the accumulation of the protein alpha-synuclein, in the form of Lewy bodies and Lewy neurites, showing in the majority of the autopsies of the cases with clinical presentation of PD.
18
Modifiable and nonmodifiable factors in PD.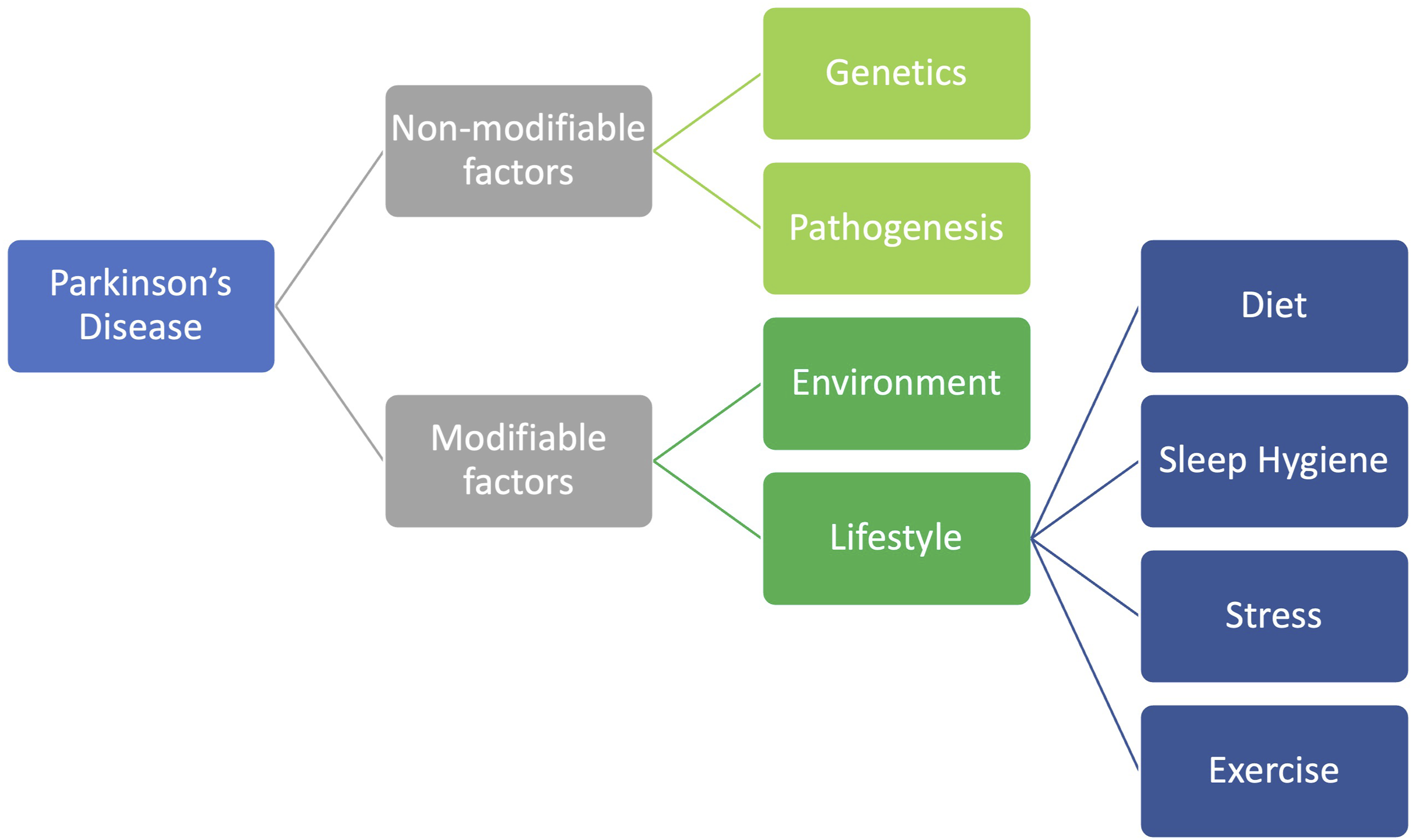
In addition to these nonmodifiable factors, modifiable factors, such as environmental and lifestyle influences, also play a role.19,20 Environmental exposures such as pesticides, heavy metals, solvents, and air pollution are strongly associated with an increased risk of PD development or trigger the initial etiopathogenesis of the disease. These factors contribute to neurodegeneration through oxidative stress and mitochondrial dysfunction, which are key mechanisms in PD pathogenesis.21-23
Substantial evidence supports the role of exercise, diet, sleep hygiene, and stress reduction techniques in alleviating PD symptoms, potentially modifying disease progression, and improving quality of life.20,24-27 Exercise is a cornerstone of PD management, with evidence suggesting it can improve both motor and non-motor symptoms. 24 Diet quality influences PD symptom severity and disease progression through multiple mechanisms such as its impact on gut microbiome composition. 28 Sleep disturbances are commonly observed in PD and affect up to 88% of patients. 29 Finally, mindfulness-based interventions have been shown to improve symptoms of PD, including reducing stress-related symptoms such as depression and anxiety. 26
This review aims to compile this evidence to assess how modifiable lifestyle factors may help patients manage their symptoms or potentially modify the disease progression.
Sleep and Parkinson’s Disease
Sleep disorders, a common non-motor symptom in PD, exacerbate motor and cognitive decline, increase fall risk, diminish quality of life, and may accelerate disease progression.27,30 About 88% of PD patients have sleep disturbances. 29
Sleep disorders in PD include insomnia, rapid eye movement (REM) sleep behavior disorder (RBD), restless legs syndrome (RLS), sleep-disordered breathing (SDB), and excessive daytime sleepiness (EDS). 31 Multiple sleep disorders can occur simultaneously. 27
Insomnia
Insomnia is a consistent difficulty with initiation, duration, or quality despite proper sleep conditions, leading to daytime impairment. Daytime issues include fatigue, cognitive impairment, irritability, and mood changes. Patients may experience physical symptoms such as headache, palpitations, and muscle tension. Both motor symptoms
(REM) Sleep Behavior Disorder
RBD is a form of parasomnia that takes place during REM sleep, marked by a loss of skeletal muscle paralysis, and patients show dream enactment behavior. 40 These behaviors can vary from mild muscle twitches and vocalizations to more intense and complex physical actions, which may result in falling out of bed, self or bed partner injury. 41 Polysomnography is necessary to establish a definitive diagnosis. 41 The loss of REM atonia is likely due to accumulation of alpha-synuclein in the pontine nuclei, which are responsible for sending inhibitory signals to motor neurons during REM sleep.42-45
For RBD establishing a safe sleeping environment and initial treatment with melatonin is recommended. 35 The most commonly used pharmacologic treatments for RBD are clonazepam and melatonin. 46 Melatonin can significantly improve REM atonia.47,48
RLS is characterized by an urge to move the legs due to unpleasant sensations. These uncomfortable feelings begin during periods of inactivity and may be alleviated by movement.
Restless Legs Syndrome
RLS can lead to insomnia, and may be associated with daytime fatigue and sleepiness. The RLS risk increases with iron deficiency, pregnancy, chronic renal failure, and the use of certain medications such as centrally acting dopamine blockers, sedating antihistamines, and most antidepressants. 34
RLS treatment starts with assessing iron levels and avoidance of alcohol and caffeine, followed by dopaminergic agents if necessary. 35
Sleep-Disordered Breathing
SDB includes obstructive sleep apnea, central sleep apnea, sleep-related hypoventilation, and sleep-related hypoxemia. 34 SDB can lead to chronic sleep deprivation and an increased risk of cardiovascular and cerebrovascular diseases. Sleep apnea may cause daytime sleepiness, poor concentration, fatigue, early morning headaches, etc. Treatment varies depending on the specific type of SDB. 34 Obstructive sleep apnea is typically managed with a continuous positive airway pressure (CPAP) machine. 35
Excessive Daytime Sleepiness
EDS can be related to disease progression and is not necessarily dependent on other sleep disorders. EDS may worsen with dopaminergic medications and is also associated with depression.49-53 Patients with PD and EDS show reduced uptake in the basal ganglia compared to PD patients without EDS. They also perform worse on motor, non-motor, cognitive, and autonomic assessments.50,54
Yoo et al.
4949
found that the EDS group across all domains of the
Sleep disorder management in PD should begin with sleep hygiene and non-pharmacological interventions such as exercise and cognitive behavior therapy (CBT). 12 Pharmacological treatments are tailored to each patient’s condition. 12
Yun et ai. 56 conducted a cross-sectional, questionnaire-based study in PD patients and found that sleep hygiene has a significant correlation with insomnia. They suggest that improving sleep hygiene may help in the management of insomnia. 56 Sleep hygiene involves behavioral and lifestyle interventions that affect the quality of sleep. These include maintaining a regular sleep routine (aiming for 7 to 9 h of sleep), limiting naps during the day, engaging in regular physical activity, limiting screen use near bedtime, avoiding caffeine in the afternoon and evening, avoiding alcohol, refraining from heavy meals close to bedtime, practicing mindfulness techniques, and creating a comfortable sleep environment (cool, dark, and quiet), using comfortable pillow and mattress. 56 Both insufficient and excessive total time spent in bed, were linked to a higher burden of non-motor symptoms. Moreover, non-motor symptoms were found to mediate the relationship between sleep quality and factors such as quality of life and PD progression. 57 The controlled daylight exposure can improve restorative sleep levels and promote overall patient well-being. This was accompanied by improvements in both motor and non-motor symptoms, as well as in quality of life. 58
Effective treatment of sleep disorders can improve quality of life and may influence PD progression. 31
However, the current evidence is limited and most studies are small, uncontrolled and rely on subjective outcome measures.
Nutrition and Dietary Intervention
About 45% of PD patients are at risk of malnutrition. 59 Both motor and non-motor symptoms can adversely affect nutritional status and quality of life, 25 highlighting the significance of regular nutritional assessments during follow-up. 60
Mischley et al., 61 in a cross-sectional study based on questionnaire and patient-reported data found that consumption of fresh vegetables, seeds and nuts, fresh fruit, olive oil, coconut oil, nonfried fish, wine, and fresh herbs and spices was associated with a reduced PD progression rate. Conversely, intake of fried foods, canned vegetables and fruits, beef, yogurt, cheese, ice cream, and soda were associated with an elevated progression rate. Additionally, iron supplements were associated with increased progression, whereas coenzyme Q10 and fish oil appeared to have protective effects. 61
A systematic review by Solch et al. 62 in 2022 indicated that adherence to a Mediterranean diet reduces the risk of developing PD. 62 Another review suggested that a protein-restricted diet may improve motor function in patients on levodopa therapy, while a ketogenic diet may benefit certain PD symptoms. However, due to small sample sizes and short follow-up periods, further research is needed on both ketogenic and protein-restricted diets in PD. Overall, the Mediterranean diet appears effective in both preventing or delaying PD and potentially slowing its progression. 25
Furthermore, a study on the MIND diet (Mediterranean-DASH Diet Intervention for Neurodegenerative Delay) reported a reduction in PD progression. 63
A meta-analysis involving 901,764 participants found a nonlinear dose-response relationship between caffeine/tea consumption and a decreased risk of PD. 64 Caffeine was also shown to reduce UPDRS scores and improve objective motor measures. 65 Another study reported that consuming more than 355 mL per day was associated with a lower hazard ratio of dyskinesia compared to consuming less than 4 ounces per day. 66 Additionally, Cho et al 67 found that caffeine drinkers exhibit lower Non-Motor Symptom Assessment Scale (NMSS) scores with improvement in mood and cognition compared to non-drinkers. 67
A randomized double-blind clinical trial found that omega-3 and omega-6 polyunsaturated fatty acids, based on the UPDRS, led to a delay in disease progression. 68
Recent Studies have referred the gut as the second brain due to the vagus nerve, which connects the gut to the nervous system and forms gut-brain axis. Inflammation in the gut can trigger the production of alpha-synuclein, a protein identified as an early-stage biomarker link to the PD. 69 Misfolded alpha-synuclein can travel to the brain via vagus nerve and damage substantia nigra. 70 Consumption of a prebiotic diet has been shown to reduce alpha-synuclein aggregation and microglial activation in the brain of mice. 71
The development and progression of neurodegenerative diseases are exacerbated by oxidative stress and inflammation, which lead to mitochondrial dysfunction, cellular damage and impairment of the DNA repair system. The consumption of antioxidants can influence the development of these disorders and improve quality of life of patients. Some polyphenols influence cognition, visual function, language, mood, and verbal memory function. Plant polyphenols enhance brain plasticity and improve cognitive function. 72 Curcumin is a member of the polyphenol subgroup that can protect mitochondria from oxidative stress. 73
Stress Management
Oxidative stress significantly contributes to the loss of dopamine-producing neurons in PD. Any disruption in the redox potential can affect other biological processes in the cell, eventually leading to cell death. 74 Reducing the early inflammatory response can decrease oxidative stress and microglial activation, which may slow neurons death in the substantia nigra pars compacta. 75
Psychological distress in PD can have significant detrimental effects. 76 Psychological stress may involve cognitive, behavioral, emotional, and biological responses to potentially threatening events. 77 Responses of immune and neuroendocrine systems may serve as biological mechanisms that help explain how psychological stress is connected to worse symptoms and health outcomes. These systems play a key role in regulating the body’s response to stressful psychological experiences. 78 The hypothalamic-pituitary-adrenal (HPA) axis triggers these reactions by releasing corticosterone in both rodents and humans, and cortisol specifically in humans. Glucocorticoids are key regulators of the immune system’s response to psychological stress. 79 Stress-induced disruption of the interactions between the immune and neuroendocrine systems can lead to increased oxidative stress, neuroinflammation, and degeneration of dopaminergic neurons in the central nervous system.80-85 Chronic psychological stress has been found to increase the sensitivity of the immune and neuroendocrine systems to future challenges, enhancing inflammation, oxidative damage, and neurodegeneration.81,86,87
Depression and anxiety are among the most common neuropsychiatric symptoms 88 yet are often underdiagnosed and undertreated. 89 Moreover, stressful situations can exacerbate both neuropsychiatric and motor symptoms such as tremors, thereby reducing the effectiveness of dopaminergic treatments. 76
Schrag et al. 90 identified depression as the factor most strongly correlated with quality of life in PD, reporting that 19.6% of patients suffer from moderate to severe depression. 90 Another study found depressive symptoms in 35% of patients. 91 Furthermore, a review indicated that mindfulness-based interventions can improve clinical outcomes and reduce psychological distress in PD. 76 Mindfulness is defined as maintaining awareness of the present moment with compassion and without judgment. 92 Dissanayaka et al. 93 observed that mindfulness intervention enhances both motor and cognitive functioning, while also reducing depression and anxiety. Additionally, a review demonstrated that various treatments including DBS, CBT, light therapy (LT), electroconvulsive therapy (ECT), and exercise, can effectively address depressive symptoms, with CBT also benefitting anxiety. 94
Treatment strategies that may help reduce stress include physical exercise (such as aerobic exercise, resistance training, and dance), Psychotherapy (both remote and in-person CBT), mind-body interventions (such as yoga, Tai Chi, and mindfulness-based interventions).95,96 Physical exercise interventions could improve symptoms of depression, anxiety, and overall quality of life. 97
Physical Activity and Exercise
Exercise induces molecular and cellular processes that lead to brain plasticity. Brain-derived neurotrophic factor (BDNF), which is associated with neuroplasticity, is increased by exercise. 98 Mellow et al 99 showed that acute aerobic exercise can enhance neuroplasticity. 99
Prag et al. 100 demonstrated that exercise in adult mice enhances cell proliferation and neural differentiation in the hippocampus. 100 A systematic review supports that exercise significantly benefits motor symptoms in PD while also improving quality of life, balance, and functional mobility. 101 Effective exercise mobilities include aqua-based training, mind-body training, dance, and strength/resistance training. 101 Given the minimal differences observed between exercise types, patient preferences, availability and compatibility should guide exercise selection. Furthermore, a recent systematic review and meta-analysis found that interventions lasting 12 weeks or more are more effective than shorter programs. 101 The World Health Organization (WHO) also recommends that adults, including those with disabilities, engage in a variety of exercise types, such as aerobic and muscle-strengthening activities. 102
Another review supports that aerobic exercise is safe for PD patients, with moderate to high intensity yielding greater improvements in motor symptoms, non-motor symptoms, and physical function while reducing disabilities. 103 Although resistance training is also safe, it is often accompanied by muscle soreness; however, no serious adverse effects have been reported. 103 This study noted that moderate to high intensity exercise emphasizing movement speed or muscle power reduced UPDRS motor scores and disabilities while enhancing physical function. 103 Additionally, gait training has proven effective in improving walking performance, particularly for aspects of gait impairment that are resistant to dopamine replacement therapy. Both moderate-intensity ground walking and treadmill training have been shown to improve gait speed, step length, and overall walking capacity. 103 Aerobic exercise further promotes structural and functional plasticity in cognitive control and corticostriatal sensorimotor networks, which benefiting both cognitive performance and motor function. 104
Feng et al. 105 conducted a single-blind, randomized, controlled trial with 28 patients to evaluate the effect of virtual reality (VR) technology on gait and balance compared to conventional physical therapy. Their findings indicated that 12 weeks of VR rehabilitation resulted in significantly greater improvements in gait and balance. 105
An umbrella review conducted by Padilha et al. 106 classified physical exercise for PD into five categories: strength exercise, aerobic exercise, combined exercise, sensorimotor activities, and other activity protocols. They found that all categories can improve balance and mobility, with aerobic exercise and sensorimotor activities specially benefiting motor symptoms, while strength exercise, combined exercise, and specific activities address both motor and non-motor symptoms. 106
Lee Silverman Voice Treatment-BIG (LSVT-BIG) therapy has been shown to improve gait speed and length. 107 Task-based LSVT-BIG can also enhance hand function, mental health, activity of daily living (ADL), and quality of life. 108 Moreover, another study demonstrated that both general exercise and LSVT-BIG positively affect motor and non-motor symptoms suggesting that general exercise may serve as effective alternative for PD patients without access to LSVT-BIG therapy. 109
Dual tasking (performing more than one task at the same time) is a common problem in PD patients. They tend to walk more slowly or take shorter steps when asked to perform another task while walking. 110 Dual-task training improves cognitive functions. 111 Fritz et al 112 found that PD patients who underwent dual-task training demonstrated improvements in single-task speed, step length, and step amplitude. 112
Tai Chi, a traditional Chinese material art practice that involves disciplined movements combined with breathing and medication to achieve dynamic balance, has been associated with both physical and mental well-being. Structured Tai Chi programs improve balance in PD patients and reduce risk of falls. It can also have a positive effect on motor symptoms, making Tai Chi a valuable component of a PD exercise routine. Additionally, Tai Chi may enhance quality of life. 113 Tai Chi and Qigong can improve both motor and non-motor symptoms. They can positively affect motor function, balance, depression and quality of life. 114 The Dance for PD based interventions positively impact motor symptoms. 115 Dance can be an alternative therapy for patients living with PD. It can enhance balance, functional mobility, and gait performance. McGill et al suggest using the World Health Organization’s International Classification of Functioning, Disability, and Health (ICF) as a framework for research on dance for PD. 116 Home-exercise tango can improve motor function in patients with PD. Dancing can be effective in controlling symptoms and improving gait and posture. Although home-based exercises cannot fully replace a live lesson, such as group interaction, they can still provide benefits. Tango can enhance confidence of patients in everyday activities, and improve their quality of life. 117 Group exercise may be more effective than individual exercise, but patients are encouraged to exercise regularly—whether in a group or individually—based on accessibility and personal preference 118 . King et al 119 compared three group interventions: (1) a home-exercise program, (2) individual physical therapy, and (3) a group class. They found that the home-based exercise program was the least effective for enhancing mobility; individually treated participants showed the greatest improvement in functional and balance scales, while the group class mainly improved gait. 119
Conclusions
While genetic and biological factors play essential roles, modifiable lifestyle factors like sleep, diet, stress management, and physical activity profoundly influence disease progression and symptom management.
Managing sleep disorders is critical, as improved sleep quality can significantly enhance quality of life and potentially slow disease progression. Approaches include sleep hygiene, CBT, and pharmacological interventions tailored to specific disorders such as insomnia, RLS, RBD, and sleep apnea.
Dietary interventions offer substantial benefits, with studies highlighting the Mediterranean diet, MIND diet, and caffeine consumption as effective strategies for slowing PD progression. Conversely, diets high in fried foods, processed items, and certain dairy products might accelerate symptom severity. Regular nutritional assessments and targeted dietary adjustments are important parts of overall PD care.
Psychological distress, notably depression and anxiety, significantly affects patient quality of life and symptom severity. Stress management techniques, including mindfulness practices, CBT, and various complementary therapies, effectively reduce psychological distress and improve both motor and non-motor symptoms.
Physical activity emerges as a potent lifestyle modification, consistently demonstrating improvements in motor function, gait, balance, cognitive abilities, and overall quality of life. Aerobic and strength exercises, as well as specialized programs like virtual reality rehabilitation and LSVT-BIG therapy, significantly benefit patients. Exercise regimens lasting at least 12 weeks appear highly effective.
Overall, lifestyle modifications offer significant, accessible, and complementary strategies to traditional pharmacological treatments, offering patients practical methods to enhance their health outcomes, daily functioning, and quality of life with PD.
Footnotes
Author Contributions
1. Study concept and design: A. B., HS. K., R. D., D. S., K. D.
2. Acquisition of data: Not applicable.
3. Analysis and interpretation of data: Not applicable.
4. Drafting of the manuscript: A. B., HS. K., R. D., D. S., K. D.
5. Critical revision of the manuscript for valuable intellectual content: A. B., HS. K., R. D., D. S., K. D.
6. Statistical analysis: Not applicable.
7. Administrative, technical, and material support: K. D.
8. Supervision: K. D.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Khashayar Dashtipour was investigator in the InfusON study and report fees for consultancy from Supernus Pharmaceuticals Inc and US WorldMeds, LLC. Dr Dashtipour has received compensation to serve as an advisor and speaker from Allergan, Acadia, Abbvie, Acorda, Amneal, Ipsen, Lundbeck, Neurocrine, Teva and US WorldMeds. None of the other authors had any personal or financial conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
