Abstract
Psoriatic arthritis is an inflammatory arthritis that significantly impairs patient quality of life and affects multiple domains of life. Lifestyle modification has the potential to improve disease activity and quality of life. We review the cellular pathophysiology of psoriatic arthritis to better understand why lifestyle interventions can make a positive impact on disease, and we discuss the interventions that can be made to treat psoriatic arthritis, including weight loss and dietary changes, exercise, improvements in sleep, alcohol moderation, smoking cessation, and psychologic interventions.
“Patients with PsA who smoke are more likely to have fatigue, pain, and overall poorer quality of life.”
Introduction
Psoriatic arthritis (PsA) is a debilitating spondylarthritis associated with psoriasis. Moll and Wright first described this disease as an inflammatory arthritis involving peripheral and/or axial joints that is seronegative for rheumatoid factor. 1 Though there are 5 classic disease phenotypes, PsA commonly causes peripheral arthritis and frequently manifests as both dactylitis and enthesitis.2,3 It is also closely associated with iritis and inflammatory bowel disease.4,5 PsA usually arises after a patient is diagnosed with psoriasis, but it can occasionally precede the development of psoriatic skin lesions. 6 PsA is a relatively common rheumatologic condition, but its incidence varies significantly by population.7,8 Multiple risk factors have been correlated with the development of PsA, including nail involvement,9-12 heavy lifting, 13 psoriasis severity,6,9 and obesity.14-17 Obesity commonly afflicts patients with PsA, 18 and, as a modifiable risk factor, it poses a unique opportunity for clinical intervention. However, there are multiple other lifestyle domains that patients can address to improve their symptom burden, including exercise, dietary changes, sleep hygiene, reduction in alcohol intake, smoking cessation, and psychologic interventions. In this article, we review the pathophysiology of PsA and discuss lifestyle interventions to treat PsA
Cellular Pathophysiology of Psoriatic Arthritis
The cellular mechanisms of PsA involve both the innate and adaptive immune systems, with recent insights indicating the prominence of the interleukin-17/interleukin-23 axis (IL-17, IL-23). It is unclear exactly what incites PsA, but there is strong evidence for a genetic component. Genealogic evaluations have shown strong heritability in first-degree relatives of patients with PsA.19,20 Another theory for PsA induction involves the Deep Koebner phenomenon. The Koebner phenomenon is the development of psoriatic lesions at the site of a minor trauma in an area previously unafflicted by psoriasis. 21 The Deep Koebner phenomenon is similar in concept—minor injury in a patient with PsA causes microtrauma at the site of injury and activates the cellular mechanisms of disease. 22
Regardless of the exact etiology, an inciting factor stimulates resident synovial immune cells, including macrophages, dendritic cells, and innate lymphoid cells (Figure 1). Resident synovial macrophages constitute the major cell type in the PsA synovial milieu
23
and are broken into two subgroups, M1 and M2. M1 macrophages upregulate tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6),
24
which are critical cytokines for influencing naïve T-cell differentiation into TH-17 cells. M2 macrophages upregulate the expression of vascular endothelial growth factor (VEGF), which allows for neovascularization of the synovium
25
and, by extension, infiltration of the synovium by T cells.26,27 Though both M1 and M2 macrophages contribute to the disease process, PsA activity is most closely related to the presence of M2 macrophages.28,29 The third actors of the innate immune system, innate lymphoid cells (ILCs), are unique in function, as they produce cytokines like the adaptive immune system but are components of the innate immune system. It has been demonstrated that ILC-3s are upregulated in both the serum and synovium of PsA patients, and increased presence of ILC-3s is correlated strongly with disease activity.
30
In concert, these resident synovial cells facilitate the joint destruction mediated by the adaptive immune system. The cellular pathway of PsA. M1 and M2 macrophages are activated (1). M2 macrophages release VEGF, increasing synovial vascularity to ease leukocyte recruitment. M1 macrophages release TNF-α, IL-1β, and IL-6, which guides T cell differentiation into TH-17 cells. TH-17 cells and ILCs produce IL-17 and IL-22. These cytokines, in conjunction with IL-23 derived from macrophages, upregulate matrix metalloproteinases (MMPs) and RANK-L to induce joint damage (2). IL-17, particularly in the presence of TNF-α, stimulates fibroblast-like synoviocytes to contribute to joint damage. Lastly, dendritic cells are constitutively activated (3), allowing for perpetuation of the disease process.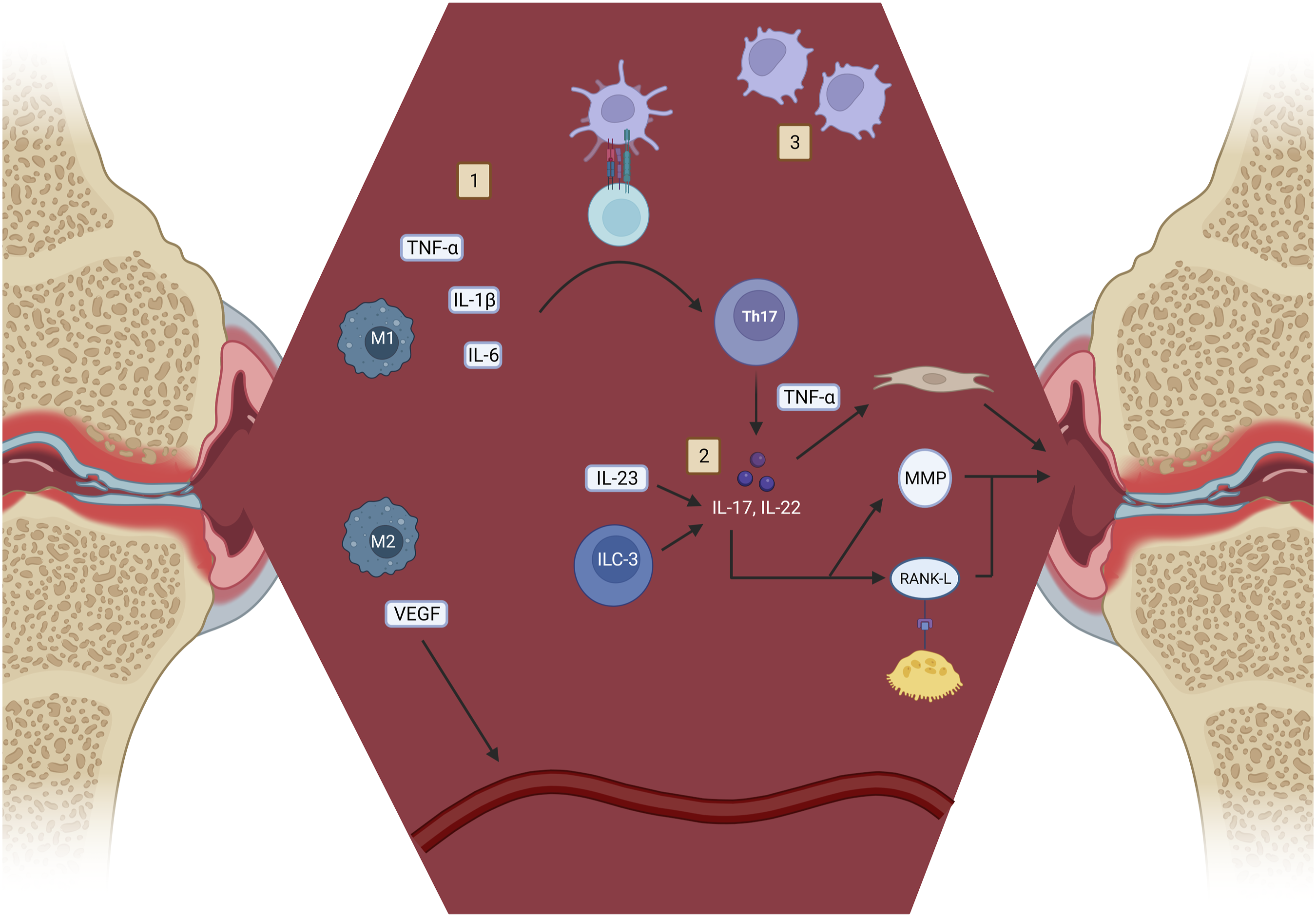
Dendritic cells connect the innate and adaptive immune systems by activating naïve T cells to induce differentiation. The majority of resident dendritic cells in PsA are young and display marked plasticity, 31 allowing for chronic immune responses. Guided by the inflammatory cytokines IL-1β and IL-6, 26 resident dendritic cells induce the development of TH-17 cells. TH-17 cells produce the inflammatory cytokines IL-17 and IL-22. These cytokines, in conjunction with the macrophage-derived cytokine IL-23, are the putative mediators of joint damage in PsA. 32 IL-17 upregulates matrix metalloproteinases and RANK-ligand (RANK-L), the central ligand that drives osteoclast differentiation. 33 IL-22 has been shown to upregulate fibroblast-like synoviocytes in PsA patients’ synovium, particularly in the presence of TNF-α. 34 IL-23 induces osteoclastogenesis indirectly by upregulating the expression of IL-17 and directly, likely through a pathway involving RANK-RANK-L, TNF-α, and IL-17.35,36 Evidence for the function and involvement of these cytokines in PsA has been demonstrated in animal models, 37 human studies,38-41 and clinical trials.42-48
Obesity, Weight Loss, and Diet
Obesity may be strongly correlated to PsA due to the cellular mechanisms of both diseases. Obesity is known to be a chronic inflammatory state, though the initiating events of the inflammatory cascade are incompletely understood.
49
Though both M1 and M2 macrophages are present in obese adipose tissue, M1 macrophages are predominantly involved in the pathogenesis of obesity-driven inflammation.50-55 The abundance of TNF-α in obesity activates dendritic cells, which allows for T cell activation
56
and adaptive immune system stimulation (Figure 2). The cellular pathway of obesity. Macrophages surround hypoxemic adipocytes and release cytokine mediators, stimulating dendritic cells to activate naïve T cells (1). Dendritic cells stimulate naïve T cell maturation, resulting in the formation of TH-1, TH-17, and TH-22 cells (2). These mature T cells release cytokines to maintain inflammation and cellular activity, allowing for the perpetuation of this cycle (3).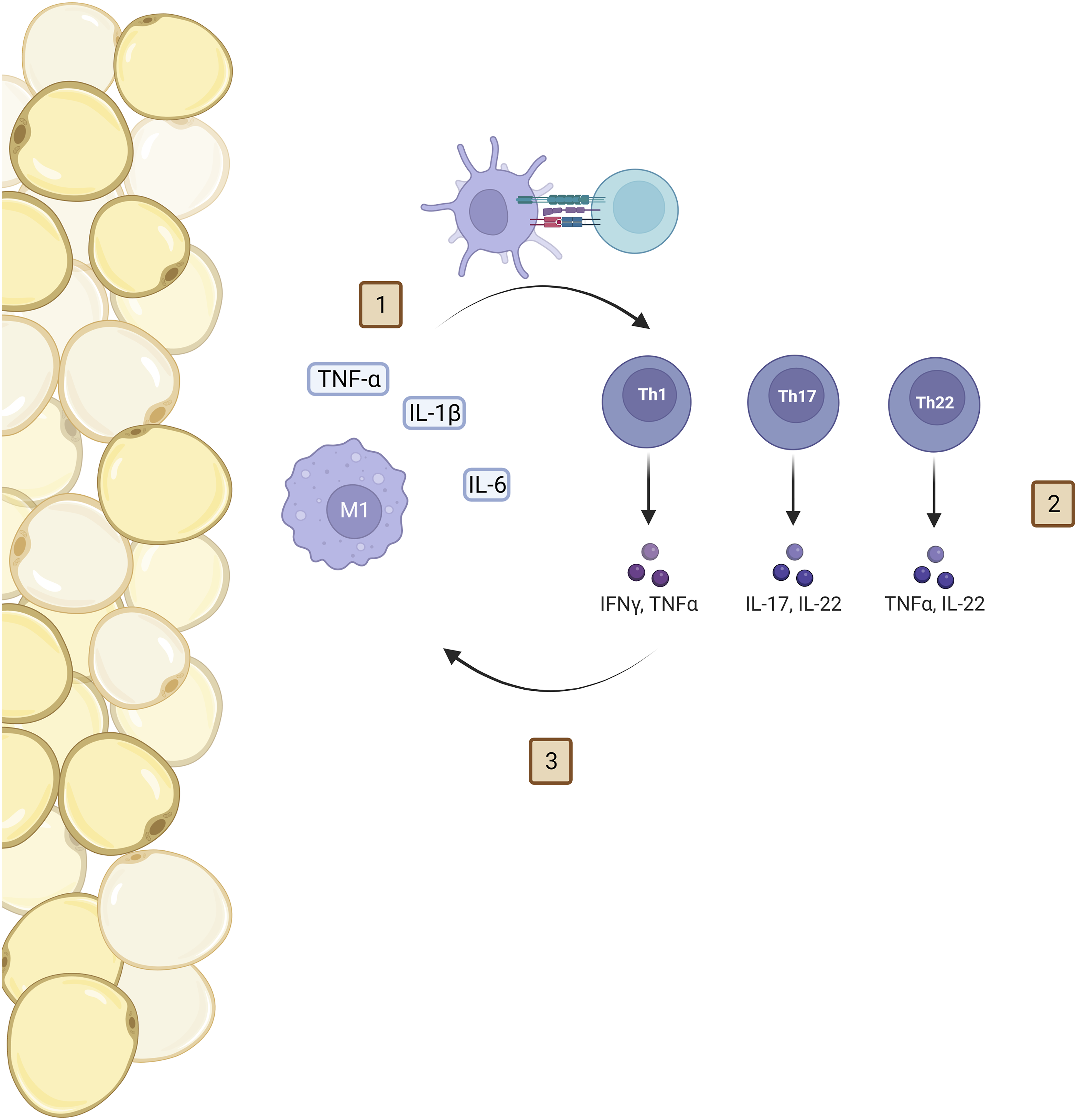
In obesity, dendritic cells are significantly increased, 57 and, once activated, they initiate T cell differentiation into TH-1, TH-17, and TH-22 cells. TH-1 cells produce interferon-gamma (IFN-γ) to induce further macrophage production of pro-inflammatory cytokines. 58 TH-1 cells have been demonstrated to be elevated in obese adipose tissue59,60 compared to nonobese adipose tissue, and TH-1 numbers correlate with markers of inflammation. 60 This is further supported by the upregulation of TH-1 produced cytokines IFN-γ and IL-1 in obesity.61,62 There is evidence for alterations in both TH-17 and TH-22 cells in obesity, but the role in pathogenesis these T cell subtypes play is still emerging. TH-17 and TH-22 cells have been found to be elevated in both the peripheral blood and adipose tissue of obese subjects compared to nonobese counterparts.62-64 TH-17 and TH-22 cells positively correlate with both TH-1 produced cytokines 65 and hyperglycemic states,62,66,67 both of which can be seen with obesity. In addition, IL-17 has been demonstrated to be elevated in overweight and obese people and has been correlated positively with body mass index (BMI).68,69
The cellular pathophysiology of PsA overlays that of obesity, with both relying on the innate and adaptive immune systems to mediate damage. Both rely in part on the cytokines produced by M1 macrophages to guide T cell differentiation, including TNF-α and IL-1β. PsA and obesity may also overlap in the cytokine milieu, with both conditions sharing upregulation of TH-17 and TH-22 cells. Again, though, the exact role of these cytokines in obesity is not well understood. Furthermore, the adaptive immune system is constitutively active in both obesity and PsA, allowing for chronic inflammation. This pathophysiologic overlap is projected on the clinical correlation of the two conditions.
Though some studies have yielded conflicting data, 10 most studies have demonstrated obesity to be a risk factor for the development of PsA, even after multivariate adjustments.14,16,17,70 The risk of obese patients developing PsA increases linearly with BMI,14,16 with this risk being strongest in patients with central adiposity. 17 The risk of developing PsA also increases linearly in PsO patients if they are currently obese 71 or were obese at a younger age. 10 In accord with an increased risk of developing PsA, patients with PsA are more likely to be overweight or obese than patients unafflicted by PsA.72,73 Patients with obesity are also more likely to have higher disease activity than nonobese patients.74,75
Obesity not only exacerbates PsA but also makes the condition more challenging to treat. Obese PsA patients are more likely to have arthritis that is refractory to TNF-α inhibitors than PsA patients with healthy BMIs.76,77 Patients with comorbid obesity and PsA are less likely to achieve minimal disease activity (MDA) on TNF-α inhibitors and more likely to relapse after reaching MDA than nonobese PsA patients. 76 Independent of disease-modifying antirheumatic drug (DMARD) and biologic use, obese patients with PsA are less likely to achieve minimal disease activity than their nonobese counterparts, and this correlation is dose-dependent. 78
Weight loss achieved through incretin therapy could potentially address both obesity and PsA. Incretins, such as glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP), have recently attracted attention for their weight-modulating properties. In clinical trials, GLP-1 agonists such as semaglutide and liraglutide have been found to cause clinically significant weight loss in patients with 79 and without 80 diabetes. Tirzepatide, a GIP and GLP-1 agonist, has been demonstrated to be even more potent inducers of weight loss in clinical trials. 81 Given the evidence for weight loss and its effects on PsA, these drugs may offer patients with PsA another way to achieve symptom control. There have not been any studies to date on PsA and GLP-1 agonists or GIP agonists, but there is some evidence from case reports that incretin-related medications can reduce disease activity in patients with PsO.82,83 A small clinical trial investigating liraglutide’s effects on PsO was unable to replicate these findings, 84 but this may be due to the power of the trial or the short investigation time compared to other reports. Interestingly, even at doses lower than that needed to modulate weight, patients had a significant reduction in psoriasis area and severity (PASI) scores and improvement in their quality of life. 85 This may be related to the immunomodulatory effects that incretins can induce.
Trials Evaluating Dietary Interventions in Patients With Psoriatic Arthritis.
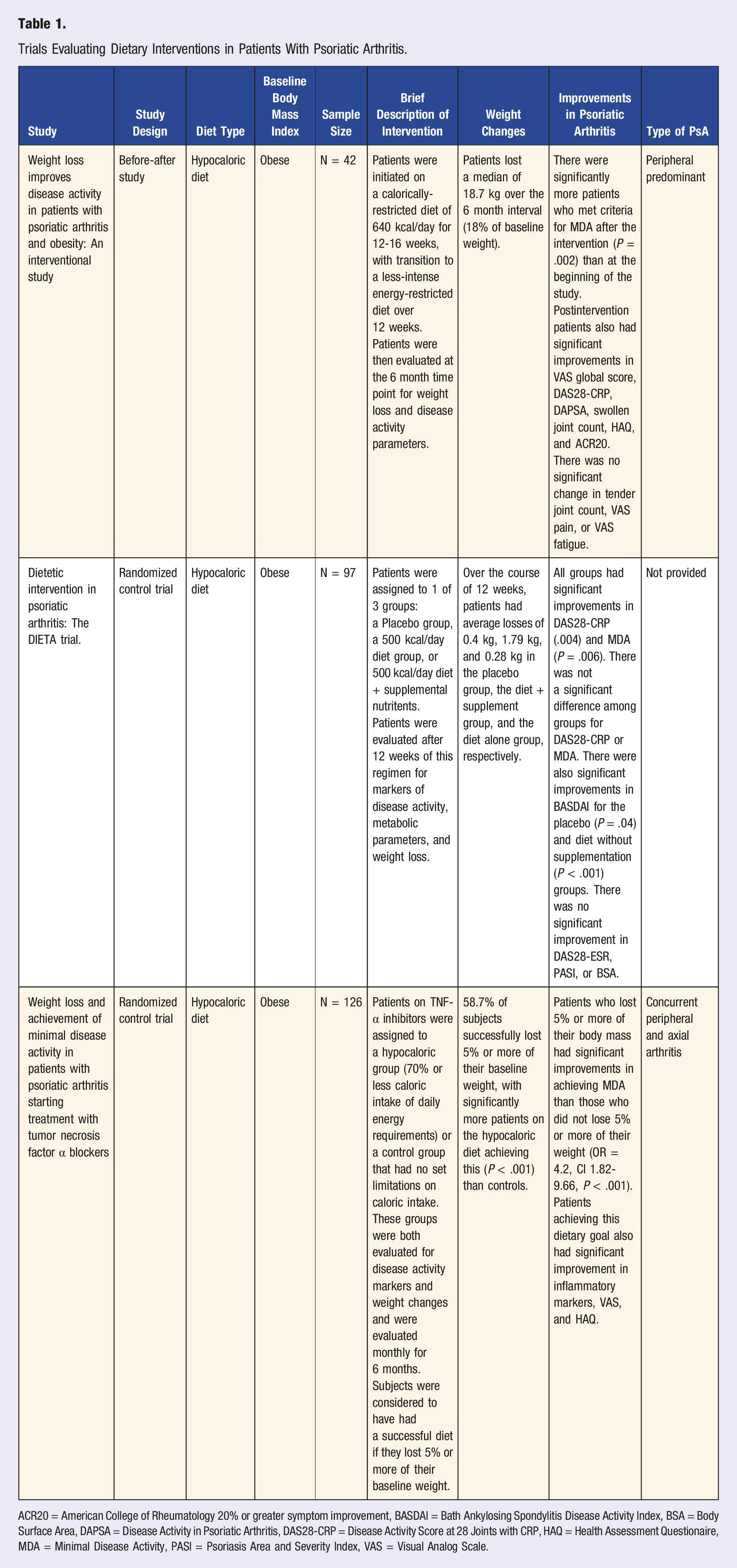
ACR20 = American College of Rheumatology 20% or greater symptom improvement, BASDAI = Bath Ankylosing Spondylitis Disease Activity Index, BSA = Body Surface Area, DAPSA = Disease Activity in Psoriatic Arthritis, DAS28-CRP = Disease Activity Score at 28 Joints with CRP, HAQ = Health Assessment Questionaire, MDA = Minimal Disease Activity, PASI = Psoriasis Area and Severity Index, VAS = Visual Analog Scale.
Dietary changes have the potential to impact disease activity independent of weight loss. Certain foods and diets are more inflammatory than others.89-92 Diets and foods that contain high amounts of saturated fats93-95 or certain omega-6 fatty acids (particularly relative to omega-3 fatty acids)96,97 have been tied to chronic inflammation and obesity. Diets that contain fewer inflammatory foods offer the potential to reduce chronic inflammation and potentially improve patient outcomes. One such diet, the Mediterranean diet, is based predominantly on legumes, fruits, vegetables, and whole grains. It incorporates a moderate intake of seafood, olive oil, and red wine, and it has minimal inclusion of red meat, saturated fats, and processed foods. Multitudes of studies of the Mediterranean diet have cemented its value in terms of all-cause mortality reduction, 98 improvement of cardiovascular outcomes,98-100 and improvement of markers of chronic inflammation.100-102 Other diets with similar macronutrient components, such as the DASH diet or the traditional Korean diet, have also demonstrated improved metabolic parameters 103 and reduction in inflammatory markers.104-106 Plant-based diets, such as vegan and vegetarian diets, have tended to reveal an inverse correlation with inflammatory markers,107-109 but this has not been consistently demonstrated.
Given the inflammatory nature of PsA, it follows that PsA patients could benefit from reduced dietary intake of inflammatory foods. This benefit could be amplified further in patients with concurrent obesity and PsA given their overlapping cellular mechanisms. Of the anti-inflammatory diets studied, the Mediterranean diet has attracted the most attention from PsO and PsA researchers. It has been shown that foods with higher inflammatory potential increase the risk both of developing PsO 110 and of increased PsO activity.110-112 In addition, poorer adherence to the Mediterranean diet in PsO patients is associated with more severe PsO. 113 Only one study to date has evaluated the Mediterranean diet in PsA patients, but it yielded similar data. Adherence to the Mediterranean diet was correlated with lower disease activity in patients with PsA. 114 In light of these data, the Medical Board of the National Psoriasis Foundation has suggested trialing the Mediterranean diet in patients with PsA in conjunction with pharmacotherapy. In addition, they recommend increasing dietary components of complex carbohydrates, fiber, omega-3 fatty acids, and monounsaturated fatty acids and reducing intake of omega-6 fatty acids, saturated fats, simple sugars, and the ratio of omega-6 to omega-3 fatty acids. 115
Other dietary modifications are less well-studied in PsA. No studies to date have evaluated the efficacy of vegetarian or vegan diets on PsA disease activity. However, there is one case report published on a patient who was able to significantly improve her PsA symptoms after adopting a whole-foods, vegan diet. 116 Conversely, consumption of red meat and processed meats could have the potential to worsen PsA given their saturated fat content. Some smaller studies indicate PsO patients eat more red meat red than their disease-free cohort, 111 and meat consumption has been significantly correlated with PsO disease severity. 117 A smaller study evaluated multiple inflammatory diseases, including PsA, and the potential relationship between biologic response and red meat and fiber consumption. However, there were no significant findings in the PsA group. 118 Further research is needed to clarify the importance of red meat and processed meat mitigation in PsA patients’ diets.
Exercise and PsA Management
Exercise has the capacity to improve PsA inflammation and disease severity. Exercise has been demonstrated to be effective in reducing PsA disease severity, alone or in conjunction with dietary modification.119,120 There has been one small trial that evaluated both PsA disease severity and inflammatory markers following a diet and exercise program, but evidence of a reduction in inflammatory biomarkers in the exercise-alone group was not forthcoming. 119 There is also some evidence correlating lower physical activity with a higher risk of progressing from PsO to PsA 17 ; however, further research is needed to substantiate this link.
Exercise has even more potential to help patients with comorbid PsA and obesity, particularly from a standpoint of inflammation. Studies have found a negative correlation between physical activity and inflammation in obesity, though this trend seems to be more pronounced in combined aerobic exercise and resistance training.121-123 From animal models, it is thought that exercise may improve inflammation in obesity by reducing levels of circulating TNF-α, decreasing the numbers of peripheral monocytes/macrophages, and lowering the ratio of M1:M2 macrophages. 53
The role of exercise in the management of PsA poses an interesting dilemma, particularly in light of the deep Koebner phenomenon. Multiple studies have linked a history of trauma,124,125 heavy lifting, 13 and high-impact activities 126 with the development of PsA. A recent study by Wervers et al investigated the relationship between sonographically-evident enthesitis in PsA patients and exposure to trauma. They discovered an inverse relationship between physical activity and the severity of enthesitis. 127 This places the clinician caring for PsA patients at a crossroads. Currently, the American College of Rheumatology weakly recommends patients with PsA engage in routine physical activity, with preference for low-impact exercise over higher-impact modalities. 128 The strength of this recommendation is likely due to the paucity of large-scale studies on this topic.
Psychologic Interventions: Meditation and Cognitive Behavioral Therapy
There may be a role for meditation and therapy in the management of PsA. These modalities of treatment have not been adequately studied in PsA, so much of the information must be extrapolated from related conditions. Meditation has been studied for chronic pain management, particularly mindfulness. Mindfulness is a type of meditation in which the meditator focuses on the present and how the body is feeling. One meta-analysis found that mindfulness can reduce patient pain perception, but there was no evidence of benefit in reduction of pain intensity or comorbid depression. 129 This meta-analysis was limited, though, due to the quality of the individual study designs. There is some data from PsO literature to suggest potential benefits from mindfulness in terms of skin disease activity, quality of life, and inflammatory marker reduction, though these studies had significant attrition or methodologic issues.130-132 No studies to date have evaluated PsA and mindfulness.
Cognitive behavioral therapy (CBT) is changing the framing in which one sees a problem to affect mood. There is data to suggest patients have significant improvements in their PASI score and a reduction in objective measures in anxiety/depression with cognitive behavioral therapy when compared with the standard of care, but this has not been consistently replicated for improvements in PASI score.133-135 CBT is a well-established option for managing anxiety, 136 which is common in patients with PsA. 137 So, although the data for psychologic interventions in patients with PsA is scant, it may behoove the clinician to suggest CBT to patients with PsA who also have anxiety.
Sleep
Sleep impairment is common in PsA, with 39.6%-84.1% of patients reporting sleep disturbances across studies.138-142 The domains of sleep most affected are subjective sleep quality, sleep onset latency, and daytime dysfunction.141,143 The etiology of sleep disturbances in patients with PsA appears to be multifactorial. Data from multiple studies indicate anxiety140,144 and pain138,140,145 play roles in impairment of sleep. There may be a role for disease activity in sleep degradation, as inflammatory markers139,144 and clinical metrics of disease, such as DAS-CRP scores and inflamed joint counts,138,139,141 are correlated with sleep quality. However, it is unclear if disease activity is contributing to poor sleep or if it is a result of poor sleep, as lower quality sleep is associated with upregulation of IL-17 and IL-23 in patients without PsA.146,147 Obstructive sleep apnea can also contribute to sleep impairment, as patients with PsA are at increased risk of developing it, even after controlling for body habitus. 148
Poor quality sleep greatly impacts patients’ lives, as sleep impairment in PsA is tied to patients’ ability to work, 140 fatigue,139,145 and poorer quality of life. 138 Adequate quality sleep is also critical for weight management, as sleep quality can affect appetite and metabolic rate. 149 Sleep quality improvement can make a positive difference for patients with PsA. Patients with PsA who have better quality sleep are more likely to have lower perceived levels of pain and have better reported quality of life. 139
Multiple lifestyle interventions can be implemented to improve PsA patients’ sleep. First, most patients can benefit from sleep hygiene interventions, such as maintaining regular sleep-wake intervals, minimizing alcohol and caffeine use, and improving patient sleeping environments. Sleep hygiene interventions have been demonstrated to improve sleep onset latency, sleep efficiency, and wake after sleep onset. 150 If there are minimal improvements in patient sleep quality with the implementation of sleep hygiene, CBT can be considered. CBT improves the same domains of sleep which sleep hygiene affects, but they do so more potently.150,151 Weight loss is another intervenable option, as reductions in weight have been correlated with improvement in sleep. 149 Lastly, PsA-related comorbidities such as obstructive sleep apnea or anxiety should be addressed to improve sleep quality.
Alcohol and Tobacco Use
Smoking has an interesting relationship with psoriatic disease. Smoking is correlated with the development of PsO and PsO activity.152,153 However, the relationship between smoking and the development of PsA has not consistently been demonstrated.13,71,154 It is also not clear if smoking worsens PsA disease. The discord between PsO and PsA patient data may be due to the design of the studies examining PsA and smoking. 155 Regardless, smoking cessation is an achievable goal for patients with PsA that can improve quality of life and potentially help with disease-related comorbidity. Patients with PsA who smoke are more likely to have fatigue, pain, and overall poorer quality of life.156-158 They are also more likely to have poor sleep.142,148 In addition to the effects on quality of life, smoking has the potential to compound cardiovascular disease. It is well known that smoking tobacco is associated with cardiovascular disease, atherosclerosis, and coronary artery disease. 159 Patients with PsA have an increased risk of atherosclerosis independent of smoking,160,161 with this risk increasing with PsA severity. 162 Smoking may also synergize with PsA to worsen CAD plaque burden. 161 Taken together, it is paramount that the physician encourages smoking cessation and aids patients with PsA in their journey towards quitting tobacco use.
Alcohol use in psoriatic disease is less well-studied than smoking. Patients with PsO have reported alcohol as a trigger for disease flares, with self-reported improvement in their skin lesions after cessation. 163 One epidemiologic study by Gerdes et al could support patient experience and suggests a relationship between PsO severity and alcohol consumption in women, 153 but the significance of this is unclear. Regarding PsA, observational studies have yielded inconsistent results on the relationship between disease development and alcohol consumption.9,13,164,165 PsA disease activity has not been correlated with alcohol use. 166 Further studies are needed to evaluate the effects of alcohol on disease activity. It is conceivable that patients with PsA could benefit from reducing alcohol intake. Many patients with PsA have metabolic comorbidities, such as cardiovascular disease 159 and insulin resistance, 167 and heavy alcohol use is correlated with both cardiovascular disease and incident diabetes. 168 In addition, alcohol also contributes to poor sleep and anxiety, 169 problems frequently encountered by patients with PsA. 170 Given this information, the clinician caring for a patient with PsA should advise the patient to moderate their alcohol intake.
Conclusions
Though weight management is the best-studied intervention for PsA, altering diet, improving sleep quality, reducing alcohol intake, exercising, quitting smoking, and engaging with therapy are evidence-based interventions that can improve the lives of patients afflicted with PsA. Further research is needed into most of these domains of lifestyle modification. Active PsA is deleterious to patient physical function, 171 community participation, mental health, 172 and overall quality of life. 170 Given the impact PsA has on patients’ quality of life, physicians and other health care providers should encourage lifestyle modification to help patients better treat their condition.
Footnotes
Acknowledgments
Permission has been obtained from those acknowledged to publish this work. Biorender was utilized to create Figures 1 and ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
