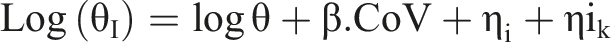Abstract
Background
Buprenorphine (BPN) is a widely used analgesic in the pediatric population, although there are few studies on the pharmacokinetics and pharmacodynamics of this drug.
Objective
The objective was to characterize the pharmacokinetics of BPN after intravenous administration and analyze the effect of age, gender, weight, height, body mass index (BMI), and drug-drug interactions as covariates.
Methods
Ninety-nine children (2-10 years), who underwent orthopedic surgery under regional, general, or combined anesthesia were included. Patients evaluated according to the American Society of Anesthesiologists Physical Status Classification, who received intravenous BPN 2 μg/kg were enrolled. Blood was collected from 1-240 min. Drug plasma concentrations were determined by LC-MS/MS. Population pharmacokinetic parameters were obtained with Monolix 2021R1 software. Pearson’s correlation and/or ANOVA were used for statistical analysis.
Results
Age was associated with changes in clearance and central compartment volume and the female gender was associated with lower intercompartmental clearance, while BMI modified clearance, central and peripheral compartment volume. Concomitant administration of BPN with fentanyl and dexamethasone produced decreases in clearance.
Conclusions
The covariates of sex, age, and BMI are directly related to the increase or decrease in BPN pharmacokinetic parameters.
Introduction
Buprenorphine (BPN) is a thebaine-derived semisynthetic opioid 1 that is a partial agonist of mu receptor, a partial or full agonist of opioid receptor like-1, and an antagonist of the kappa and delta receptors. 2 Due to its mechanism of action, it has a lower incidence of side effects than other opioids. It is also observed to have a long duration of action due to its slow dissociation from mu receptors,3–5 BPN can be 75 to 100 times more potent than morphine, with a ceiling effect on respiratory depression but not on analgesia. 6 Although BPN is not a first-line drug in pain treatment, it is a good analgesic with prolonged effects. 7
The risk of inducing ventilatory depression is one of the main reasons for the limited use of opioid analgesics; however, previous studies have demonstrated the suitability of BPN for postoperative pain in children.8,9 In pediatric populations receiving intravenous opioids, for safety reasons, it is recommended to observe them until they are fully responsive and ventilatory control has stabilized. 10
The pharmacokinetics of intravenous BPN has a bioavailability of 100%,1,11 has a high central compartment volume of distribution (Vd1), and is highly bound to plasma proteins.12,13 After oral administration, a maximum plasma concentration (Cmax) reached within 2-5 min,1,11 has a first-pass effect is metabolized in the liver by cytochrome P450, specifically CYP3A4, and forms an active metabolite, norbuprenorphine, with a potency of 25% relative to that of the parent drug. Fifteen percent of BPN is excreted unchanged in urine and its metabolite is excreted in bile after hepatic conjugation with glucuronide.12,13 Its elimination is carried out in 3 phases: an initial fast phase with a half-life time (t1/2 α) of 2-5 min, followed by a redistribution phase (t1/2 β) lasting 20-30 min and finally, a slow phase (t1/2δ) of 2-3 h.1,11
In children, information on its pharmacokinetic characteristics and the estimation of its population pharmacokinetic parameters is scarce. The lack of clinical studies on aspects of its pharmacokinetics, pharmacodynamics and safety support its use and forces us to limit the use of this opioid. Based on the above, our objective was to characterize the pharmacokinetics of BPN after intravenous administration (IV) and analyze the effect of age, gender, weight, height, body mass index (BMI) and drug-drug interactions as covariates in pediatric patients aged 2-10 years scheduled for orthopedic surgery.
Materials and Methods
Study Design
Descriptive, observational, longitudinal, prospective study. It was performed in the Laboratory of Pharmacology, National Institute of Pediatrics (INP) from November 2017 to December 2020. The protocol was approved by the Committees of the Research, Biosafety and Ethics (IRB 00013674). The project was registered with number INP 031/2016, all procedures were conducted following the Helsinki Declaration. The parents and/or guardians of the patients provided written informed consent.
Patient Population
This trial was randomized for the sampling times, a random number table was used, then the sealed envelope method was applied. The patient, anesthesiologist and the analyst were blinded during the study. The G power statistical program (version 3.1.9.2) was used to determine the sample size, input values: effect size of 0.2, error α = 0.05, 1-β = 0.95, for independent repeated samples and a loss of 20%, resulting in a total of 87 patients. During the study period ninety-nine patients of both genders, aged 2 to 10 years, who required orthopedic extremity surgery and who were administered BPN as analgesic after the surgical procedure, under regional, general or combined anesthesia were included. The patients were evaluated according to the American Society of Anesthesiologists (ASA) Physical Status Classification I and II14,15 after obtaining informed consent from their parents or guardians, and in the case of patients older than 7 years who agreed to participate in the study, the corresponding informed assent was also collected. Subjects were divided into 2 groups according to their age: preschoolers (2 to 5 years) and school children (5.1 to 10 years).
Patients who did not meet the inclusion criteria (severe uncontrolled systemic disease, non-orthopedic surgical procedure), those who did not complete the pharmacokinetic sampling of BPN (only one blood sample), those who, by medical decision, discontinued BPN administration, as well as the presence of any complication during the surgical procedure, or who decided to drop out of the study, were excluded.
Surgical Procedure and Perioperative Medication
For the planned surgical procedure, all of the patients underwent basic monitoring. Intravenous induction was performed; premedication was standardized with midazolam 0.05-0.1 mg/kg, balanced general anesthesia was administered with propofol 2-5 mg/kg, lidocaine 1-2 mg/kg, fentanyl 2-4 μg/kg, cisatracurium 0.05-0.1 mg/kg or rocuronium 0.3-0.6 mg/kg. If regional anesthesia was required, ropivacaine 0.2-0.75% or hyperbaric bupivacaine 0.5% plus sedation was used. Maintenance of anesthesia was performed under assisted or controlled ventilation with sevoflurane or desflurane in conjunction with an opioid such as fentanyl. Analgesics and adjuvant medications such as, atropine 0.002-0.01 mg/kg, paracetamol 10-15 mg/kg, ketorolac 0.5-1.0 mg/kg, ondansetron 100-150 μg/kg, dexamethasone 0.2-0.5 mg/kg and ibuprofen 5 mg/kg were allowed. The use of hydrocortisone, ketamine, tramadol or metamizole was allowed on the recommendation of the anesthesiologist. The administration of these drugs is shown as a CONSORT flowchart (Figure 1). Once the surgical procedure was completed, BPN was started at a dose of 2 μg/kg by IV bolus as an analgesic schedule; for sampling, a percutaneous catheter was inserted in each patient, still under anesthetic effect, exclusively for this procedure. In the immediate postoperative period, patients were monitored to ensure that they remained neurologically intact and hemodynamically stable, attending to and recording any possible adverse effects that might result from the administration of BPN. Study CONSORT flowchart. The design of the study included the follow-up of pediatric patients undergoing orthopedic surgery under anesthesia. BGA: Balanced General Anesthesia, MA: Mixed Anesthesia, RA: Regional Anesthesia, MDZ: Midazolam, LD: Lidocaine, PR: Propofol, FT: Fentanyl, CS: Cisatracurium, RC: Rocuronium, RP: Ropivacaine, BV: Bupivacaine, SV: Sevoflurane, DF: Desflurane, KTC: Ketorolac, KTM: Ketamine, AT: Atropine, DEX: Dexamethasone, OD: Ondansetron, HC: Hydrocortisone, PCM: Paracetamol, TM: Tramadol, IB: Ibuprofen, MT: Metamizole, BPN: Buprenorphine.
Sampling and Determination of the Plasma Concentrations of BPN
Two or 3 blood samples of 2.5 mL were taken from the patients at randomly assigned times according to optimal sampling theory 16 to construct the BPN pharmacokinetic curve (1, 3, 5, 7, 10, 15, 20, 30, 40, 45 min and 1, 2 or 4 h). Samples were collected in tubes with EDTA anticoagulant, centrifuged to obtain plasma and frozen at −80°C until analysis by Liquid Chromatography-Mass Spectrometry (LC-MS/MS). Buprenorphine hydrochloride standard (purity ≥98%) USP and BPN-d4 from Sigma‒Aldrich Co® (St. Louis, MO, USA) were used to determine the plasma concentrations. Acetonitrile and methanol (HPLC-grade) were obtained from EMD Millipore Co® (Billerica, MA, USA), while ethyl acetate, hexane and formic acid were purchased from Merck® (Darmstadt, Germany).
BPN calibrators were prepared at concentrations of 1000, 2000, 4000, 6000, 8000 and 10000 pg/mL and quality controls of 2500, 5000 and 7500 pg/mL. For processing, 1 mL of patient plasma was placed in a 15 mL screw-capped tube, and 100 μL of BPN-d4 (4000 pg/mL) and 3 mL of ethyl acetate: hexane (90:10 v/v) were added. They were placed in a water bath with ultrasound for 10 min. Subsequently, they were centrifuged at 4500 rpm for 10 min. A total of 2.4 mL of the organic phase was taken and evaporated at 40°C under a stream of nitrogen. It was reconstituted with 100 μL of acetonitrile: formic acid 0.15% mixture (65:35 v/v), and 10 μL was injected into the chromatographic system. The same procedure was performed for the calibrators and quality controls using 1 mL of healthy volunteer plasma.
Sample analysis was performed using an Acquity UPLC equipment from Waters® (Milford, MA, USA), which was coupled to a mass spectrometer Micromass Quattro Micro, Waters Micromass® (Manchester, UK) used in a positive electrospray interface mode. Chromatographic separation was performed on AcQuity BEH C18 column (2.1 x 50 mm, 1.7 μm) from Waters® (Milford, MA, USA) maintained at 40°C, while the autosampler was set at 15°C. The mobile phase consisted of 0.15% formic acid and acetonitrile at a 55:45 v/v ratio, and the flow rate was 0.15 mL/min. The analytes of interest were measured in Selected Reaction Monitoring mode, and the optimized ion transition was m/z1+ 468.2 > 395.9 for BPN and 472.3 > 400.2 for BPN-d4. The capillary voltage was kept at 1 Kv, while the cone voltage was kept at 50 V. The source and desolvation temperatures were 125 and 350°C, respectively. The cone gas flow rate was kept at 50 L/h, and the desolvation gas flow rate was 700 L/h. The collision energy was 35 V, while the dwell time was 0.1 sec. The data were processed with MassLynx® 4.1 software (Waters®, Milford, MA, USA).
Population Pharmacokinetics Analysis
Analysis of the population pharmacokinetics (PopPK) was performed using a method based on a nonlinear mixed effect. Data were analyzed using Monolix® software version 2021R1 (Lixoft, Antony, France).
Model Building
Structural models with variations in the number of compartments (one, 2 or 3) and/or distribution kinetics (first order, zero order or dual) were compared. In addition, residual error (RE) models were analyzed, but because it is difficult to obtain a large number of samples from pediatric patients. To determine the suitability of the structural model and the final model, the model with the lowest residual error values was sought by obtaining the values of the indeterminate parameters PopPK by least squares regression, in a process commonly referred to as curve fitting between predicted and observed values. Given that general modeling theory indicates that it is desirable to build mathematically simpler models, but it is a reality that mathematically more complex models usually result in lower residual error values, which can lead to a bias in the final choice of model, for this reason, we used the Akaike Information Criterion (AIC) and Bayesian Information Criterion (BIC) criteria, which incorporate a complexity penalty parameter in their calculation. 17 Thus, we selected the best final model as the one with the lowest value in the AIC and BIC criteria. The PK parameters were linearly scaled as a function of the body surface area (BSA), which in our population has a mean of 0.927 m2.
Analysis of Covariates
Using the basic model, the covariates of sex, weight, height, BSA and BMI were assessed. The presence/absence of drug interactions and adverse reactions due to low body weight were also evaluated.
BMI was calculated based on the Centers for Disease Control and Prevention growth charts for age. Patients were classified into 4 groups: BMI-A, underweight patients at risk of malnutrition (percentile below 5); BMI-B, normal or eutrophic weight (percentile from 5 to 85); BMI-C, overweight patients (percentile from 85 to less than 95); and BMI-D with obesity (percentile equal to greater than 95). 18
Covariates were added in a stepwise manner based on the implementation of the reduction value of the BIC and AIC reporting criteria. The selection and elimination of covariates were based on the P values of the Wald tests. Statistical significance was set at P < 0.01 for direct covariate inclusion and P < 0.001 for covariate elimination. Only significant covariates were included in the final model.
Estimation Parameters
For the estimation of the PopPK parameters, the Stochastic Approximation of the Expectation Maximization algorithm was used. Standard errors were calculated using the linearization method implemented in Monolix 2021R1.
Individual Distribution Parameter
A log-normal distribution was assumed for the parameters according to the following equation:
Model Selection and Evaluation
To build the pharmacokinetic model of BPN, the base model was determined, covariates were selected, and less statistically significant covariates were eliminated. Model selection in our work was based on the BIC and AIC, diagnostic goodness-of-fit test and relative standard errors (RSE) of the estimated parameters, 19 residual plots and Visual Predictive Check (VPC). Additionally, the population and individual pharmacokinetic parameters of the ASA patients and biological variables and drug-drug interactions were evaluated to establish possible factors that may modify them. Pearson’s correlation analysis (P < 0.05) and/or ANOVA (P < 0.05) were performed to determine possible alterations of these parameters using the statistical software SPSS v20.0 (Statistical Package for the Social Sciences, IBM Corp., Armonk, NY, USA).
Pharmacological Interactions
Information was searched in different databases such as UptoDate,™ Micromedex™ and Drugs.com™14,20,21 to review pharmacokinetic aspects and possible interactions of the drugs used in postoperative analgesia with BPN.
It is worth mentioning that although our study was done postoperatively, we performed an analysis of possible BPN interactions from premedication to postoperative period, according to the databases consulted.
Results
Patient Data
Clinical and Demographic Characteristics of the Patients.

Preschoolers (2-5 years), School children (5.1-10 years). American Society of Anesthesiologists (ASA) Physical Status Classification I and II. S.D. Standard deviation.
Model Evaluation
A tricompartmental distribution was selected as the best model describing the BPN concentrations based on the BIC and AIC results. Analysis of the relationship of both observed and individual predicted concentrations using individual and population parameters is shown in Figure 2. Analysis of the residual error distribution showed that the residuals were uniformly distributed around zero. The individual weighted residuals for the prediction were scattered around the horizontal line (zero) (Figure 3). Adjusted vs unadjusted individual concentrations obtained after analysis. Observed vs predicted unadjusted (A) and adjusted (B) individual concentrations for the final population model. Individual weighted residuals (IWRES) of the model as a function of time. The dots represent the weighted residuals for each observation, the solid lines represent the central tendency of the weighted residuals. and the dashed line at zero indicates the ideal value where the residuals would be zero. Circles represent confidence intervals for the weighted residuals at the points with the greatest amount of data available.

Predictive Model Verification
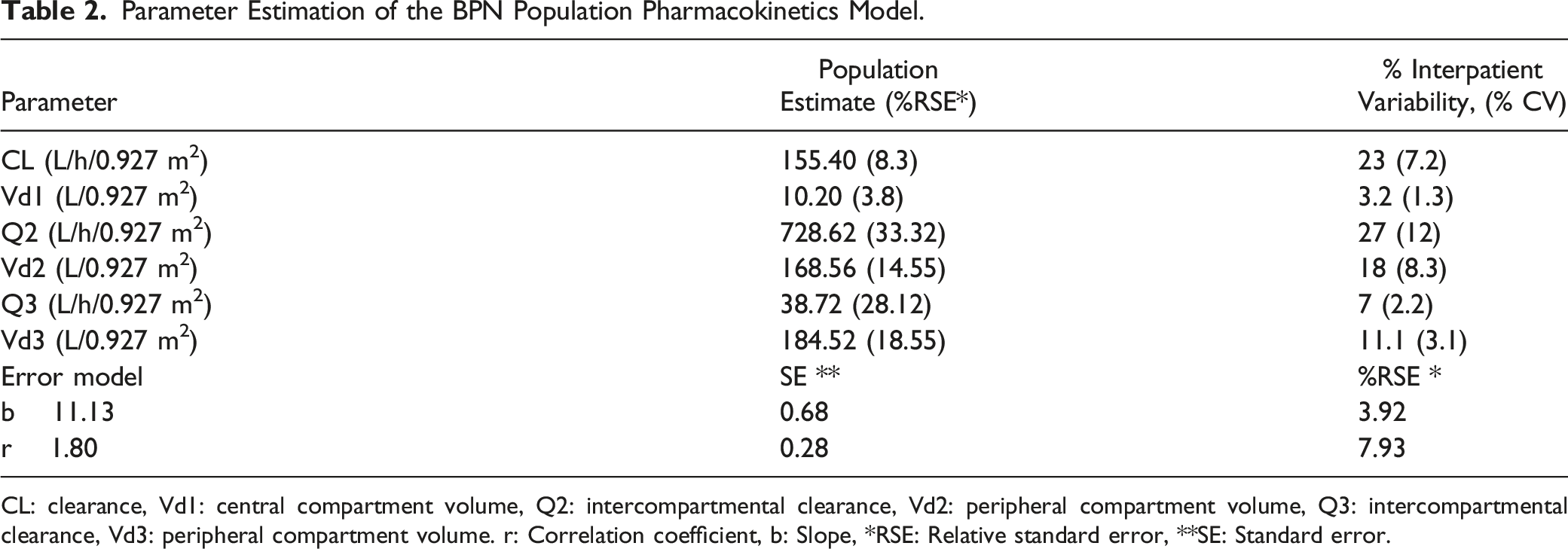
To explore whether the observed variability could be reproduced by the model, a VPC was applied as shown in Figure 4. The final parameter estimates of the BPN PopPK model are shown in Table 2. The values of % RSE were adequate for all estimated parameters. Distribution of visual predictive check. The solid blue lines represent the distribution of the observed concentrations. The colored areas represent the prediction intervals for each percentile. Blue shows the 10th and 90th percentiles, while pink shows the 50th percentile. The points marked with a red circle and the area in red show the outliers. Parameter Estimation of the BPN Population Pharmacokinetics Model. CL: clearance, Vd1: central compartment volume, Q2: intercompartmental clearance, Vd2: peripheral compartment volume, Q3: intercompartmental clearance, Vd3: peripheral compartment volume. r: Correlation coefficient, b: Slope, *RSE: Relative standard error, **SE: Standard error.
Effect of Covariates on BPN Pharmacokinetics
Variables Associated With Significant Changes in the BPN Pharmacokinetic Parameters.

Preschoolers (2-5 years), School children (5.1-10 years). Y: years, RSE: Relative standard error, Q2: Intercompartmental clearance, CL: Clearance, Vd1: Central compartment volume, Vd2: Peripheral compartment volume, Body mass index (BMI), BMI-A: Underweight with risk of malnutrition, BMI-D = Obesity, r: Correlation coefficient, P: Statistical significance with Pearson’s test P < 0.05.
Females had a decrease of 21% in the intercompartmental clearance. Age was associated with CL changes in preschoolers with an increase of 4.6%, while in school children the Vd1 was modified with an increase of 28.4%. In underweight children (BMI-A, n = 16), a significant increase in Vd2 of 7.4% was observed, whereas in the obese population (BMI-D, n = 14), both CL (11.62%) and Vd2 (5.1%) parameters decreased significantly (Table 3).
Effect of Concomitant Drugs on BPN Pharmacokinetics
The concomitant administration of some drugs can generate interactions that influence changes in pharmacokinetic behavior. In our study, it was observed that when fentanyl was administered concomitantly with BPN, there was a decrease in CL of 9.4% (155.40 vs 140.79 L/h/0.927 m2, P = 0.01), while dexamethasone was administered, the CL decreased by 8.1% (155.40 vs 142.5 L/h/0.927 m2, P = 0.01).
These results were obtained from the correlation analysis with Monolix 2021R1, as well as with Pearson’s test. These observations are important because a decrease in the CL of BPN could result in a longer analgesic effect of this drug or an increased risk of an adverse reaction due to the accumulation of BPN in the organism.
Buprenorphine Possible Drug Interactions
Pharmacological Interactions of BPN With Concomitant Drugs in the Perioperative.
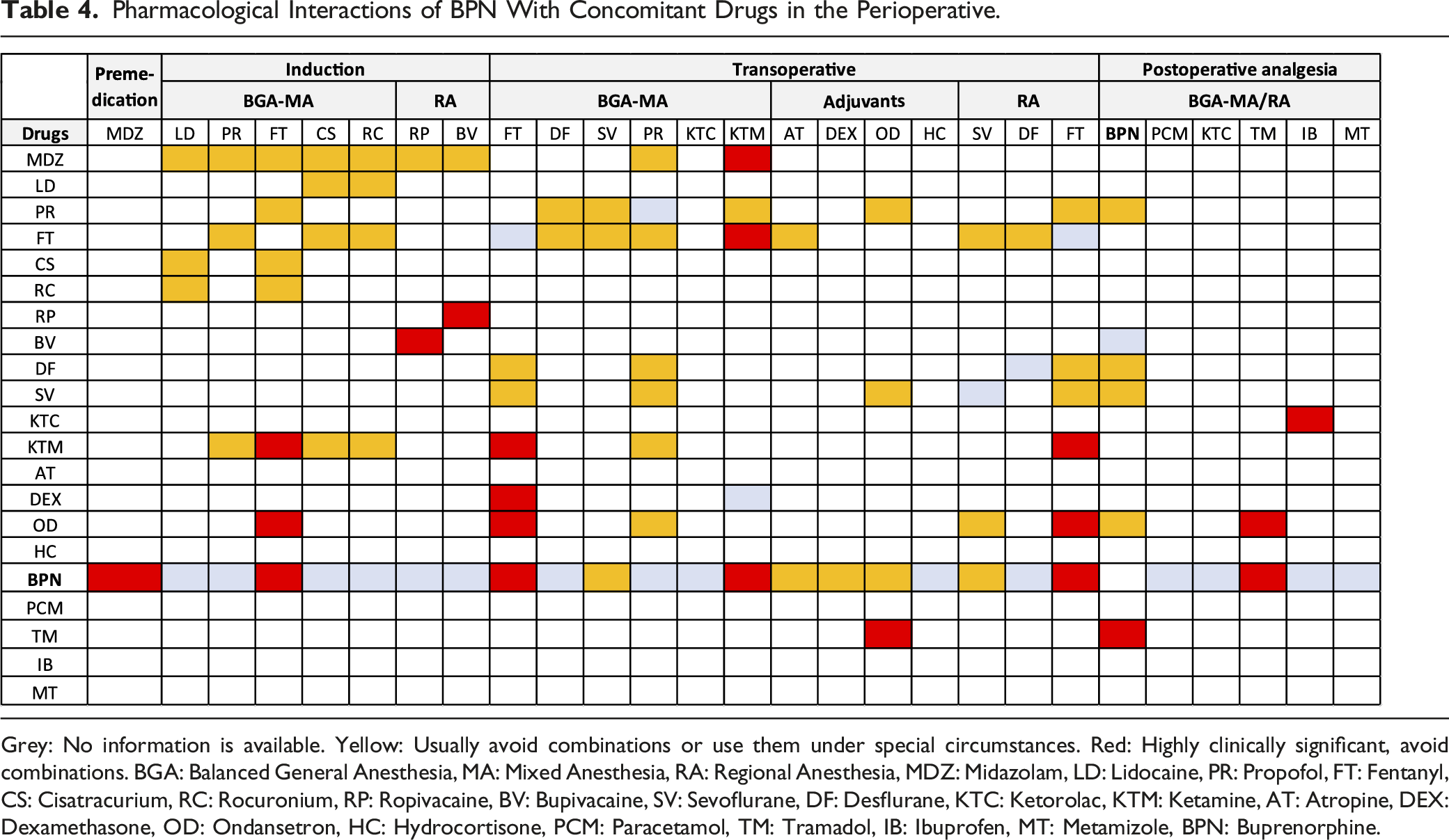
Grey: No information is available. Yellow: Usually avoid combinations or use them under special circumstances. Red: Highly clinically significant, avoid combinations. BGA: Balanced General Anesthesia, MA: Mixed Anesthesia, RA: Regional Anesthesia, MDZ: Midazolam, LD: Lidocaine, PR: Propofol, FT: Fentanyl, CS: Cisatracurium, RC: Rocuronium, RP: Ropivacaine, BV: Bupivacaine, SV: Sevoflurane, DF: Desflurane, KTC: Ketorolac, KTM: Ketamine, AT: Atropine, DEX: Dexamethasone, OD: Ondansetron, HC: Hydrocortisone, PCM: Paracetamol, TM: Tramadol, IB: Ibuprofen, MT: Metamizole, BPN: Buprenorphine.
Discussion
A tricompartmental model with first-order elimination kinetics was the best fit to our data, similar to previously reported results. 1 From the analysis of the individual concentrations determined in the model, we can see that there is a tendency for a fan-like dispersion as the BPN concentration increases. Since all patients received the same dose (2 μg/kg), this behavior could be due to interindividual variability or possible drug interactions. Additionally, there could be an accumulation of BPN in some patients due to metabolism, resulting in a longer mean residence time.
Since the pharmacogenomics of BPN in children of the ages within our study population has not been characterized, we cannot fully explain this behavior. The pharmacokinetic profile of a drug is influenced in different proportions by metabolizing enzymes, transporters, and genes, which play very important roles in drug metabolism. 22
In our study, age was a source of alterations in BPN pharmacokinetics with respect to clearance, as preschoolers have a clearance that is slower compared to school children, which may be due to lower cytochrome P450 enzyme activity in young children, as well as a small liver, limited hepatic blood flow and smaller amounts of circulating albumin, which is one of the most abundant proteins for drug binding.23,24
On the other hand, the influence of gender on anesthesia and analgesic therapy remains poorly understood, despite the physiological and pharmacological differences between men and women. It has been suggested that men wake up slower than women after general anesthesia and have less postoperative nausea and vomiting. Sex hormones seem to play a role in the occurrence of these differences. Women appear to be more sensitive than men to opioid receptor agonists. Women may experience respiratory depression and other adverse effects more readily if given the same doses as men. 25
CYP3A4 is known to be far more expressed in women than in men.26,27 In the case of opioid drugs, there is evidence to suggest that the pain response is a sexually dimorphic process. 28 Pain is a complex phenomenon regulated by a variety of physiological, cellular and hormonal modulations, including sexual ones, and there is evidence that sex may be a factor that could modify the response to analgesic drugs.29,30 It is also clear that sex is an important factor affecting the pharmacological activity of opioid drugs. 31 With respect to BPN, a mu receptor partial agonist, and its metabolites, the areas under the curve of plasma concentrations and peak plasma concentrations have been reported to be higher in women than in men due to differences in body composition, hepatic blood flow and CYP3A activity.32,33
In our study, we observed that clearance is slower in girls and in patients with obesity; this can be explained by the fact that BPN is highly lipophilic, 34 which means that it can enter and remain for longer in deep tissues, leading to changes in pharmacokinetics.
Regarding the alterations in the pharmacokinetic parameters of BPN after concomitant administration with other drugs, we can comment that fentanyl is highly lipophilic and is also an agonist of mu and kappa opioid receptors such as BPN. In addition, when there is a greater content of fatty tissue, as is the case in girls, both overweight and obese, both drugs penetrate this tissue and remain for longer, resulting in a decrease in elimination. Obese patients have increased extracellular water in relation to total body water. 35 In obese adults, volemia (an increase in circulating blood volume), increased cardiac output, and greater renal blood flow have been reported, 36 so these alterations may modify pharmacokinetic parameters, such as Vd and CL, as well as absorption.
In addition, in obese children, it has been observed that the lean mass is more hydrated, 37 which produces an increase in extracellular water, which modifies the Vd. This could explain part of our results, since Vd was the parameter most affected by obesity, as previously mentioned. 38 Additionally, the decrease in CL in patients with obesity may be because BPN is highly lipophilic and can be distributed in adipose tissue, resulting in changes in the Vd. Currently, both in the pediatric and adolescent population, an increase in cases of obesity has been observed, a factor that in this study was shown to be a source of changes in both CL and Vd, and the clinical management of patients with this condition requires close monitoring to optimize pharmacotherapy.
Dexamethasone is a moderate CYP3A4 inducer, while BPN is a strong CYP3A4 substrate, so we can speculate that there is a competitive interaction; therefore, there would be a higher concentration of BPN in blood with a reduction in CL; however, in the literature, it is specified that coadministration of BPN with dexamethasone decreases BPN levels and therefore this would imply that its clearance is higher. 14 In our study, the patients had a lower CL, which indicates that there is no competitive interaction between BPN and dexamethasone and that this decrease in CL is mainly due to factors such as age and interindividual metabolism, as dexamethasone was administered as an adjuvant during the patient’s recovery.
This is the first study to report changes in BPN pharmacokinetics due to covariates such as age, sex, and BMI in children aged 2 to 10 years. The inclusion of 99 patients increased the statistical power (1-β = 0.99) and ensures the reliability of the PK model and results. This study is limited by the 4-hour sampling period, as most of the patients were outpatients, had a short recovery stay and were quickly discharged, which hindered a longer pharmacokinetic characterization.
Conclusions
The covariates of sex, age, and BMI are directly related to the increase or decrease in BPN pharmacokinetic parameters. The coadministration of fentanyl and dexamethasone produces a longer residence time of BPN, increasing its analgesic effect and the risk of adverse reactions.
Footnotes
Authors’ contributions
Conceptualization, JLCP, EVR, LRE; Methodology, LRE, ESL; Recruitment and Anesthetic management, EVR, CFP; Samples analysis, LRE, ESL; Formal Analysis, JLCP, JFP, CFP, LRE; Pharmacokinetic analysis, ILA, JAGM; Writing, Original Draft Preparation, JLCP, JFP, LRE, CFP; Writing, Review and Editing, LRE, EVR, JFP, CFP, JLCP, ILA, JAGM. All authors have read an agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Program E022 of the Instituto Nacional de Pediatría to the project 031/2016.
IRB statement
The study was conducted following the Declaration of Helsinki and this study was approved by the Research, Biosafety and Ethics Committee (IRB 00013674) of the Instituto Nacional de Pediatría (protocol INP-031/2016).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.